Studying Cell Death Initiation Using a Digital Microscope
Summary
Here we present a protocol for studying the rate of programmed cell death initiation by continuously imaging inoculated leaves following cell death induction.
Abstract
Hypersensitive response (HR)-conferred resistance is an effective defense response that can be determined by the N resistance genes. HR is manifested as the formation of cell death zones on inoculated leaves. Here, a protocol for studying the rate of cell death initiation by imaging inoculated leaves in the time between the cell death initiation and the cell death appearance using a digital microscope is presented. The digital microscope enables a continuous imaging process in desired intervals, which allows an accurate determination of cell death initiation rate up to minutes exactly, as opposed to hours in traditional methods. Imaging with the digital microscope is also independent of light and can therefore be used during day and night without disturbing the circadian rhythm of the plant. Different pathosystems resulting in programmed cell death development could be studied using this protocol with minor modifications. Overall, the protocol thus allows simple, accurate, and inexpensive identification of cell death initiation rate.
Introduction
Potato is one of the world's most widely grown food crops, fourth place behind rice, wheat, and corn. However, potato production can be severely affected by potato virus Y (PVY), which is currently considered its most important virus pathogen1,2. In potato plants cv. Rywal, several strains of PVY (including PVY strain N-Wilga) trigger hypersensitive response (HR)-conferred resistance, where the pathogen's restriction to the infection site manifests as necrotic lesions on inoculated leaves3. In this pathosystem, HR is mediated by Ny-1 resistance gene, which is temperature-dependent, as plants grown at lower temperatures efficiently develop necrotic lesions, whereas in plants grown constitutively at elevated (28 °C) temperature, abortion of resistance is demonstrated as lack of lesion formation and systemic virus spread3,4. When the plants are transferred to a lower temperature (22 °C), cell death is initiated, which can be exploited to follow the rate of cell death initiation by imaging inoculated leaves in the time between the cell death initiation and the cell death appearance.
This protocol demonstrates a simple method for the determination of cell death initiation rate using a digital microscope. By imaging the inoculated leaves after transferring the plant from 28 °C to 22 °C, a digital microscope enables continuous observation of the leaf in desired intervals. Unlike the use of other methods (for example, confocal microscopy or observation of lesion formation with the naked eye), this allows determination of the exact time of lesion formation and therefore, the cell death initiation rate up to minutes exactly, as opposed to hours in aforementioned methods5,6. Digital microscope use is also independent of light and can therefore be used during day and night. This protocol can also be used to identify components involved in cell death initiation or to determine the effects of different components on cell death initiation rate if used plants are transgenic and have altered levels of components of interest.
Protocol
NOTE: Sections 1 and 2 describe a modified protocol for plant material preparation based on the methods outlined by Lukan et al.7. Specifically, some modifications to controlled environmental conditions and inoculum preparation were made.
1. Growing potato plants
- Grow healthy potato cv. Rywal plants in stem node tissue culture8.
- After 6-8 weeks, cut ten 1 cm-long explants containing nodes from potato plants on sterile paper under sterile conditions using sterile tweezers and a scalpel knife.
- Transfer them to plastic boxes with Murashige and Skoog (MS30) medium (Figure 1A) and grow them under controlled environmental conditions (22 °C in the light and 19 °C in the dark with 250 µE or µmol/m2/s radiation and a 16-h photoperiod, at a relative humidity of 55% ± 5%).
- Transfer them to the soil 2 weeks after micropropagation.
- Fill the pots (r = 10 cm) with soil. Use a finger to create a 3-4 cm deep hole in the soil in the middle of a pot, fill the hole with water, and wait for it to absorb.
- Place one plant per pot into the hole, leaving the leaves above the surface. Gently cover the roots with soil (Figure 1B).
NOTE: Some plants might not develop roots. Exclude those plants from the analysis.
- Grow the plants in soil in a growth chamber under controlled environmental conditions (22 °C in the light and 19 °C in the dark, at a relative humidity of 55% ± 5%, with 250 µmol/m2/s radiation and a 16-h photoperiod).
- After 3-4 weeks, plants are ready. Use these plants to be inoculated.
NOTE: Plants should have at least 3-4 fully developed leaves with visible leaflets (Figure 1C). They should look healthy, with no visible symptoms (yellow or brown leaves), which could be mistaken for lesions caused by PVY. To achieve comparable results, plants should be inoculated at the same time (e.g., at 9 am) in all experiments to avoid possible effects of circadian rhythm on plant immune response and lesion formation9,10,11.
2. Inoculum preparation and potato inoculation
- Inoculate the plant with PVY strain N-Wilga (PVYN-Wi; accession no. EF558545) inoculum as stated below.
NOTE: More plants can be inoculated in parallel if more than one digital microscope is available. Plants should have at least three fully developed leaves before inoculation (Figure 1C). PVYN-Wi is multiplied and maintained in potato cv. Pentland.- Prepare phosphate buffer, supplemented with sodium diethyldithiocarbamate (DIECA), for inoculation: Mix 1.3 mL of 0.2 M NaH2PO4, 8.7 mL of 0.2 M Na2HPO4, and 0.225 g of DIECA. Make up the volume to 100 mL using ddH20. Adjust pH to 7.6 by adding 1 M NaOH or 1 M HCl solution.
- Harvest 6-8 weeks old PVYN-Wi infected potato cv. Pentland plants from tissue culture in extraction bags with filter net (0.5 g per 6 plants) and add phosphate buffer supplemented with DIECA (4x mass of plant material, mass: volume ratio). Use a hand homogenizer for 1-2 min to obtain a homogeneous solution.
- Lightly dust the first three bottom fully developed leaves of 3-4 weeks old potato plants (from step 1.6) with carborundum powder.
NOTE: Adjust the amount of carborundum powder. Too much may cause damage, while too little might result in ineffective infection. Optimally, 0.1 mg/cm of carborundum powder is used, which is approximately 1.5 mg of powder per leaf of average size (approximately 15 cm2). - Gently rub the leaves with the inoculum (~100 µL per leaf). After 10 min, thoroughly wash the leaves with tap water.
- Be careful not to harm the leaves. Do not exceed the incubation time. Adjust the amount of the inoculum according to leaf size – 6.5 µL/cm, which is approximately 100 µL per leaf of average size (approximately 15 cm2).
- Transfer the plants to a growth chamber and grow them under controlled environmental conditions (28 °C in the light and 28 °C in the dark at a relative humidity of 55% ± 5%, with 250 µmol/m2/s radiation and a 16 h photoperiod) for 3 days.
3. Plant preparation and using the digital microscope for recording lesion development
- Transfer the inoculated plants from the growth chamber maintained at 28 °C to a growth chamber at 22 °C 3 days after inoculation, with other conditions as described in step 1.5.
- Select a plant for observation. Immobilize the second inoculated leaf using tape (Figure 1D). Make sure that the desired leaf is fully immobilized before starting with imaging (Figure 1D).
- Install a software application for image capture on the laptop computer. Connect the digital microscope to the computer. Open the software.
NOTE: Make sure that the plant, digital microscope, and laptop are positioned on the shelf near the socket to plug in the computer. The decision on the selection of leaves to be monitored might depend on the biological question studied, e.g., the type of process that leads to the formation of limited programmed cell death. - Adjust the microscope above the immobilized leaf. Focus using the dial on the digital microscope (shown in Figure 1E).
- Ensure that the line of sight is as wide as possible to increase the chances of capturing the formation of the lesion but still magnified enough to spot the lesion's appearance (typically, 25x magnification is used).
- Set the camera settings. Click on the Settings button in the top right corner of the image (Figure 2A, circled part 1) and adjust camera settings: Brightness (60-70), Contrast (10-15), Hue (0), White Balance, Saturation (45-50), Sharpness (0), Gamma (5) (Figure 2B). The settings used in this study were as follows: Brightness (64), Contrast (14), Hue (0), Saturation (47), Sharpness (0), Gamma (5).
NOTE: Camera settings should be adjusted to outside light to ensure the best image quality. Settings could be adapted according to the user's conditions in the growth chamber. - Set the image capture settings by clicking on the icon for Image Capture (Figure 2C, circled part 2). Click on the button Time-Lapsed Video and set Image Capture every 15 min for 24 h (Figure 2C).
NOTE: The interval between the images taken and imaging duration are fully flexible and could be adapted according to the needs of the experiment. In the time between image capture, the LED light is switched off. The LED light is automatically switched on during image capture. - Start image capture by clicking on the START button (Figure 2C).
NOTE: After being transferred from 28 °C to 22 °C, plants should start to develop lesions within 24 h. If not, this could be a sign of ineffective inoculation. Adjust the amount of carborundum powder, ensure that the time of inoculum incubation on leaves was correct, and determine virus abundance in the inoculum by qPCR. - To save images, select all images and click the Save icon (Figure 2B, circled part 3). Set Export Options. Set DPI at maximum (300) (Figure 2D). After saving, delete all pictures in the program.
- For image analysis, any program for image viewing/editing is sufficient. The usage of free software for image editing, ImageJ, is explained below.
- Import the time sequence of images from a single field of vision (on the top left corner, click File, select Import, and then Image Sequence). Paste the path of the directory of saved pictures and press the OK button to start the conversion.
- After the conversion, the software automatically opens an internal video player showing the final video. Export the video file by clicking on File > Save as option and selecting AVI Format. A small window opens. Set the Frame Rate as 0.3 fps and press OK to save the video as an AVI video file.
NOTE: The time of lesion development can also be determined by manually checking all of the images and finding an image where the lesion appears.
Representative Results
This study demonstrates a step-by-step protocol for studying cell death initiation through lesion occurrence on potato cv. Rywal, with a digital microscope. This enables determining the exact time of programmed cell death initiation.
Plants that have developed roots were put in soil 2 weeks after potato cv. Rywal micropropagation (Figure 1A,B). After 3-4 weeks of growth under described conditions, plants with at least 3-4 fully developed leaves with visible leaflets that looked healthy, with no signs of abscission, were used for further analysis (Figure 1C). Using a digital microscope as described in this protocol, we observed the same area on the inoculated leaf at 15-min-intervals and determined the lesion occurrence and expansion in time (Figure 3). The lesion occurred at 15 h 30 min (Figure 3).

Figure 1: Plant preparation for analysis with a digital microscope. (A) A plastic box with MS 30 medium and potato cv. Rywal plant explants containing nodes. (B) Potato cv. Rywal plant in soil (2 weeks after micropropagation). (C) Potato cv. Rywal plant, ready for inoculation (4 weeks after being put in soil), having at least three fully developed leaves. (D) Second inoculated leaf (arrow) of potato cv. Rywal plant positioned and immobilized (arrow) with tape. (E) Plant positioned under the digital microscope with the arrow pointing to the dial used for focusing. Please click here to view a larger version of this figure.
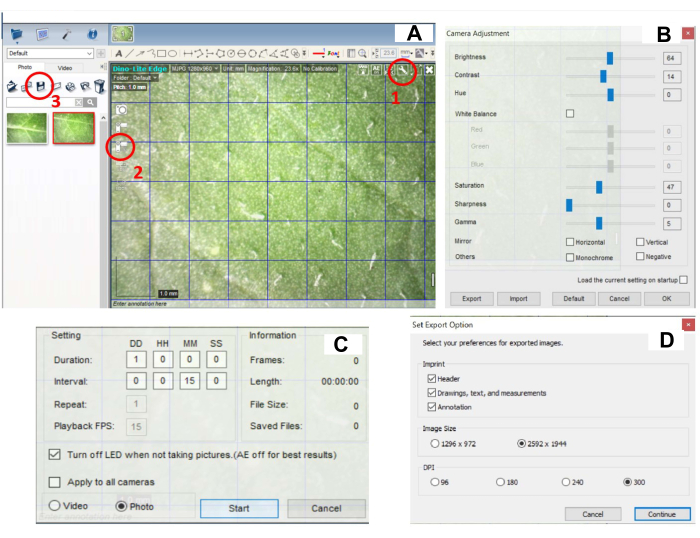
Figure 2: Digital software setting for recording lesion development. (A) Software interface – circled with red are options for the button for (1) camera settings, (2) image capture settings, and (3) saving images. (B) Window with camera settings, which opens with a click on (1) in panel A. Brightness, Contrast, Saturation, Sharpness, and Gamma should be properly adjusted. (C) Window with image capture settings, which opens with a click on (2) denoted in panel A. (D) Window with image saving settings, which opens with a click on (3) denoted in panel A. Please click here to view a larger version of this figure.

Figure 3: Lesion formation on the inoculated leaf observed under the digital microscope. Images of the central part of PVY-inoculated potato leaf at 23.6x magnification as seen under the digital microscope, taken at intervals of 5 min. Inoculated plants were put at 28 °C for 3 days, and on the third day, the observation with a digital microscope at 22 °C started at 7:00. (A) At 21:02, the lesion is not yet visible, (B) 90 min later, at 22:32, the lesion is visible. (C) The lesion expansion was observed at 01:02 and (D) 07:32 the next morning. Experiment was repeated two times, and the lesions occurred 8 h 15 min, and 12 h after cell death initiation, respectively. Please click here to view a larger version of this figure.
Discussion
The demonstrated protocol allows the user to accurately determine the cell death initiation rate by continuously imaging inoculated leaves in the time between the cell death initiation and the cell death appearance using a digital microscope. Even though there are numerous ways of monitoring lesion and plant disease occurrence12,13,14,15, this protocol presents the advantage of light-independent measuring without disturbing the plant's circadian rhythm, as the light is switched off between measurements.
After the inoculation, plants should grow at 28 °C for 3 days. Ny-1 resistance gene, which induces a hypersensitive response, is temperature-dependent, and in plants grown at higher temperatures, leads to abortion of resistance, which is manifested as lack of lesion formation and systemic virus spread3. After plants are transferred to 22 °C, cell death is initiated, so for accurate results, observation with a digital microscope should begin as soon as possible after this transfer. Another crucial step in the preparation of the plant for imaging is the immobilization of the leaf (Figure 1D), as the plant will continue to grow during imaging, which could move the observed leaf out of focus, or such set-up will not give desired results.
If the described protocol is used on transgenic plants with altered components of interest, hypothesized to be involved in cell death initiation, the protocol enables the user to determine if the decreased level of a studied component affects the rate of cell death initiation. By that, components involved in cell death initiation can be identified in pathosystems, where programmed cell death occurs, using this protocol. Other methods for identifying these components are, for example, transcriptomic analysis such as RNA-seq or various forms of microscopy, which can be expensive and time-consuming16. The method described in this protocol allows for easy and inexpensive identification of components involved in cell death initiation by observing differences in cell death initiation rates between transgenic and control plants. Optimally, in such a set-up, two digital cameras must be used, as a transgenic plant should be analyzed in parallel with a control plant within the same experiment.
In this protocol, PVY strain N-Wilga was used; however, other strains of this virus, for example, GFP-tagged PVY (PVY-N605(123)-GFP)7, could also be used. Moreover, other pathosystems, which result in programmed cell death development could be studied using this protocol with minor modification.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank Barbara Jaklič for technical assistance. This research was financially supported by the Slovenian Research and Innovation Agency (research core funding no. P4-0165 and project Z4-3217: Deciphering redox-related signaling interconnectedness in potato resistance against viruses).
Materials
| Alcohol burner | Mikro+Polo | SH-234002455 | For tweezers and scalpel sterilization |
| Autoclave A-21 CAV | Kambi |
N/A | |
| Bacto Agar | Becton, Dickinson and Company | 214010 | |
| Carborundum powder | VWR Chemicals | 22505297 | |
| DinoCapture 2.0 | Dino-Lite | Version 2.0 | software for digital microscope |
| Dino-Lite Edge AM7915MZTL digital microscope | AnMo Electronics Corporation | AM7915MZTL | |
| Ethanol, 70% | Stella Tech | P94000 | For tweezers and scalpel sterilization |
| Extraction bags | Bioreba | 420100 | |
| Growth chamber FS-WI | Photon Systems Insturments | N/A | |
| Hand homogenizer | Bioreba | 400010 | |
| Hawita Special Substrate | HAWITA Gruppe | 2000000071701 | Ready to use substrate, made using peat (H4-H6 and H6-H8) |
| Hydrochloric acid (HCl) | Merck | 109057 | |
| Label tape | Sigma | L8144-5EA | |
| Laptop computer with installed DinoCapture 2.0 | HP | Z2V77EA#BED | Computer needs to be transferable as experiment takes part in a growth chamber |
| Murashige and Skoog medium | Duchefa Biochemie | M02220100 | |
| Na2HPO4 | Emsure | 1065860500 | |
| NaH2PO4 | Emsure | 1064700250 | |
| Pasteur pipette 0.5 mL | Brand | 21500209 | |
| pH-meter | Mettler Toledo | ML1601 | |
| Plastic boxes | Cvetlice Dornig | VCG10.5 | Radius = 10.5 cm |
| Plastic pots | Lab Associates | DIS40003 | Radius = 11.5 cm (top), Radius = 9.8 cm (bottom) |
| Saccharose | Kemika d.d. | 1800408 | |
| Sodium Diethyldithiocarbamate (DIECA) | Sigma-Aldeich | 228680 | Sodium diethyldithiocarbamate trihydrate, ACS reagent |
| Sodium hydroxide (NaOH) | Merck | 106462 | |
| Sterile surgical blades | Braun | 4511733633 | |
| Tweezers | Braun | BD033R |
References
- Karasev, A. V., Gray, S. M. Continuous and emerging challenges of potato virus y in potato. Annual Review of Phytopathology. 51, 571-586 (2013).
- Quenouille, J., Vassilakos, N., Moury, B. Potato virus Y: A major crop pathogen that has provided major insights into the evolution of viral pathogenicity. Molecular Plant Pathology. 14 (5), 439-452 (2013).
- Szajko, K., et al. The novel gene Ny-1 on potato chromosome IX confers hypersensitive resistance to Potato virus Y and is an alternative to Ry genes in potato breeding for PVY resistance. Theoretical and Applied Genetics. 116 (2), 297-303 (2008).
- Szajko, K., Strzelczyk-Żyta, D., Marczewski, W. Ny-1 and Ny-2 genes conferring hypersensitive response to potato virus Y (PVY) in cultivated potatoes: Mapping and marker-assisted selection validation for PVY resistance in potato breeding. Molecular Breeding. 34 (1), 267-271 (2014).
- Lukan, T., et al. Cell death is not sufficient for the restriction of potato virus Y spread in hypersensitive response-conferred resistance in potato. Frontiers in Plant Science. 9, 168 (2018).
- Baebler, &. #. 3. 5. 2. ;., et al. Salicylic acid is an indispensable component of the Ny-1 resistance-gene-mediated response against Potato virus y infection in potato. Journal of Experimental Botany. 65 (4), 1095-1109 (2014).
- Lukan, T., Coll, A., Baebler, &. #. 3. 5. 2. ;., Gruden, K. Analysis of virus spread around the cell death zone at spatiotemporal resolution using confocal microscopy. Methods in Molecular Biology. 2447, 261-270 (2022).
- Vinterhalter, D., Dragiüeviü, I., Vinterhalter, B. Potato in vitro culture techniques and biotechnology. Fruit, Vegetable and Cereal Science and Biotechnology. 2, 16-45 (2008).
- Wang, W., et al. Timing of plant immune responses by a central circadian regulator). Nature. 470, 110-115 (2011).
- Roden, L. C., Ingle, R. A. Lights, rhythms, infection: The role of light and the circadian clock in determining the outcome of plant-pathogen interactions. Plant Cell. 21 (9), 2546-2552 (2009).
- Srivastava, D., et al. Role of circadian rhythm in plant system: An update from development to stress response. Environmental and Experimental Botany. 162, 256-271 (2019).
- Mulaosmanovic, E., et al. High-throughput method for detection and quantification of lesions on leaf scale based on trypan blue staining and digital image analysis. Plant Methods. 16, 62 (2020).
- Martinelli, F., et al. Advanced methods of plant disease detection. A review. Agronomy for Sustainable Development. 35, 1-25 (2015).
- Ali, M., Bachik, N., Muhadi, N. A., Tuan Yusof, T. N., Gomes, C. Non-destructive techniques of detecting plant diseases: A review. Physiological and Molecular Plant Pathology. 108, 101426 (2019).
- Sankaran, S., Mishra, A., Ehsani, R., Davis, C. A review of advanced techniques for detecting plant diseases. Computers and Electronics in Agriculture. 72 (1), 1-13 (2010).
- Rowarth, N. M., et al. RNA-Seq analysis reveals potential regulators of programmed cell death and leaf remodelling in lace plant (Aponogeton madagascariensis). BMC Plant Biology. 21 (1), 375 (2021).

