A New Murine Model of Endovascular Aortic Aneurysm Repair
Summary
Histological and biochemical modifications after aneurysm endograft exclusion are still unclear. We describe a new model of endograft implantation on a murine aneurysm. Through thrombus analysis with and without persistant circulating blood stream interface, and new endovascular biomaterials evaluation, this model will help to better understand endovascular aneurysm exclusion pathobiology.
Abstract
Endovascular aneurysm exclusion is a validated technique to prevent aneurysm rupture. Long-term results highlight technique limitations and new aspects of Abdominal aortic aneurysm (AAA) pathophysiology. There is no abdominal aortic aneurysm endograft exclusion model cheap and reproducible, which would allow deep investigations of AAA before and after treatment. We hereby describe how to induce, and then to exclude with a covered coronary stentgraft an abdominal aortic aneurysm in a rat. The well known elastase induced AAA model was first reported in 19901 in a rat, then described in mice2. Elastin degradation leads to dilation of the aorta with inflammatory infiltration of the abdominal wall and intra luminal thrombus, matching with human AAA. Endovascular exclusion with small covered stentgraft is then performed, excluding any interactions between circulating blood and the aneurysm thrombus. Appropriate exclusion and stentgraft patency is confirmed before euthanasia by an angiography thought the left carotid artery. Partial control of elastase diffusion makes aneurysm shape different for each animal. It is difficult to create an aneurysm, which will allow an appropriate length of aorta below the aneurysm for an easy stentgraft introduction, and with adequate proximal and distal neck to prevent endoleaks. Lots of failure can result to stentgraft introduction which sometimes lead to aorta tear with pain and troubles to stitch it, and endothelial damage with post op aorta thrombosis. Giving aspirin to rats before stentgraft implantation decreases failure rate without major hemorrhage. Clamping time activates neutrophils, endothelium and platelets, and may interfere with biological analysis.
Introduction
Endovascular skills have progressed since the first endovascular aortic aneurysm repair (EVAR) by Parodi in 19913, almost half of all abdominal aneurysms are now treated by EVAR4. Unlike surgery, EVAR leaves the thrombus, excluding it from systemic bloodstream. Aneurysms are characterized by a hemoglobin rich multi layered thrombus, a thin media, and a fibrosed and inflamed adventitia. The crucial role of proteolysis5 in aneurysm progression has been demonstrated thirty years ago6, 7: aneurysm sac thrombus is associated with a thin aortic wall, lysis of elastin, low density of smooth muscle cells in the media, and a high level of immuno-inflammatory adventitial reaction8,9. These changes suggest that proteolytic enzymatic activity occurs within the thrombus, rather than directly in the aortic wall.
In addition, the luminal layer of the thrombus is rich in agglutinated red blood cells releasing hemoglobin. They are primarily involved in fibrin formation, platelet activation and induce thrombin formation. Finally, thrombus also induces t-PA and plasminogen retention, which are involved in fibrinolysis and recruiting leukocytes, mainly neutrophils. These are 12 times more important in the thrombus than in the blood stream10. Their presence is associated with a high rate of matrix metalloproteinase type 8 (MMP-8), MMP-9 and elastase: they release granular serine proteases and pro oxidative agents leading to degradation of the fibrillar matrix, and ultimately aortic wall rupture9,11-13.
Elective aneurysm intervention aims to prevent rupture. EVAR leaves both aneurysm wall and thrombus intact. Thus, aneurysm perfusion by aortic collaterals (lumbar arteries, inferior mesenteric artery…), is named endoleak, and is a specific complication of endovascular treatment14,15, sometimes leading to rupture even with low pressure endoleaks. Moreover, in certain patients, there is no increase in aneurysm diameter16. Interaction between circulating blood and aneurysmal wall maintains the mentioned intra luminal thrombus biological activity. Hence, Type V endoleaks, defined as expansion of the aneurysm sac without identified endoleak, could be explained by enzymatic activity inside the aneurysm thrombus.
Neither radiological imaging (18FDG-PET scan, platelet activation scintigraphy, iron oxyde contrast MRI17) nor peripheral blood sampling (MMP-9, platelet derived microparticles, plasmin/anti-plasmin complex) used to explore the endovascular impact on aortic aneurysms in humans directly evaluate biochemical pathways.
In our knowledge, there is no any reproducible and cheap experimental aneurysm exclusion animal models. This one allows pre and post aneurysm exclusion biological and histological modifications to be explored.
Protocol
Methods
The protocol has been approved by the Bichat-Debré Ethic committee (N° 2012-15/698-0074).
The elastase perfusion model has been first described by Anidjar in 19901: abdominal aortic aneurysm is induced by elastase perfusion in male Wistar rats, aged 8 to 9 weeks, 250-300 g. Two to 4 weeks later, a re-laparotomy is performed, and a 3 mm diameter covered coronary stentgraft is used to exclude the aneurysm. The graft, inserted through a distal aortotomy, is deployed under magnified vision. Two weeks later, stentgraft patency is confirmed with an aortography by the left common carotid artery before sacrifice. The whole stented aorta and surrounding tissues are then harvested and conditioned for analysis.
Protocol
1. Step 1 : Abdominal Aortic Aneurysm Induction (Day 0)
- Anesthetize the animal with isoflurane, and perform an intra peritoneal penthobarbital injection. 0.1 ml/100 g of penthobarbitol is diluted in 0.1 ml of saline/100 g of animal weight. Anesthesia is adequate for the procedure.
- Shave the abdomen, and clean the skin with alcohol.
- Make Xypho pubic skin incision with a blade. Abdominal muscles layer then peritoneum, avoiding internal organs, are open with scissors. Place the spreaders and wrap the intestines in a wet compress on the left side of the animal.
- Open the posterior peritoneum directly above the aorta, without tearing it. The goal is to prevent future bowel adhesions by economic dissection.
- Ligate all collaterals from the left renal vein to the iliac bifurcation with only one ligature, at the origin of the aorta. If the aorta is stuck to the bowels or the vena cava, there’s a high risk of bleeding during the second intervention; this is often fatal for the rat.
- Pass a 4-0 silk twice around the aorta, 15 mm below the left renal vein. It’s important to entirely dissect the aorta, approx. 15 mm below the left renal vein and just proximal to the iliac bifurcation to allow insertion of the Gilbert approximator clamps.
- After clamping the aorta proximally just below the left renal vein, and distally just above the iliac bifurcation, remove the adventitia, and perform a small aortotomy as proximal as possible to the distal clamp. The aorta has to be carefully flushed with saline and an angulated canula and all remaining blood washed to avoid clot embolization when declamping.
- Introduce heat-tapered polyethylene tubing (PE 10) smoothly inside the aorta, and pass the 4-0 silk around the aorta (at step 1.6) and tie up to secure the tube and avoid pressure expulsion.
- Inject elastase into a 10 to 13 mm length of aorta (from the proximal clamp to the silk).
- Infuse 725 μl of the elastase solution is d at constant speed-rate in 30 min. It is important to carefully survey the injection: during infusion, the aorta should expand by 140-150% of its normal diameter.
- At the end of the infusion, remove the catheter, flush the elastase with saline, cut the silk and close the aortotomy with about 3 stitches of 10-0 Prolene. Remove the clamps: distal then proximal. Care has to be taken to avoid trans-aortic suture. With a few practice and considering the small length of the aortotomy, aorta thrombosis is extremely rare.
- Close abdominal wall with a continuous absorbable suture, and the skin with interrupted non absorbable sutures. Inject a 5 mg/kg dose of Carprofen subcutaneously to prevent pain. Return the rats to their cage and give the standard rat laboratory chow and water ad libidum.
2. Step 2 : Abdominal Aortic Aneurysm Exclusion with a Coronary PTFE (Polytetrafluoroethylene) Covered Stentgraft (Day 15-30)
- Prior to the operation, dilute Aspirin 1,000 mg in 1 L of water. Daily water intake was estimated of 10-12 ml/100 g/ of body weight a day.
- After anesthesia as previously described, perform a re-laparotomy, carefully avoiding potential internal organs adherent to the abdominal wall. Aneurysm was defined by an increase of 50% at least of the aorta compared to proximal aorta diameter measured under direct vision. Based on our experience, the success rate is 90%, including early deaths, mostly due to aorta burst (unpublished data)
- Dissect the aorta at the aortic bifurcation and also at the proximal neck. The aneurysm should arise a few millimeters below the left renal vein; this allows a satisfactory length between the distal neck and the distal clamp to insert the catheter, and suture the aorta.
Stentgraft length depends on the aneurysm size. Both proximal and distal necks should be covered by at least 2 millimeters. - Wrap a double loop silk around the proximal neck. Clamp the proximal aorta above the aneurysm and below the left renal artery, and distally above the aortic bifurcation.
- Perform a large aortotomy just above the previous aortotomy to avoid tearing the aortic wall when introducing the stentgraft. Do not rinse the aorta to preserve integrity of the thrombus. Push the stentgraft towards the proximal clamp. Remove the clamp is and control the tension of the silk to allow graft progression. Adapt the tension around the delivering catheter to allow stentgraft progression until the proximal edge of the stentgraft reaches the proximal neck, but also this will prevent massive hemorrhage after proximal clamp removal. The balloon and the proximal end of the delivering catheter may cover the renal arteries, but not the stentgraft which has to be deployed below the renal arteries.
- Deploy the stentgraft under direct visual control using an inflator syringe at 8 atm pressure. Deflate the balloon whilst tightening the silk and remove the balloon shaft.
- Wash the stentgraft with saline. Place a clamp just below the distal part of the aneurysm, and close aorta with interrupted 10-0 Prolene sutures.
- Close the wound, inject Carprofen subcutaneously and let the animals return to their cage. Give the standard laboratory chow and water ad libidum.
3. Step 3 : Control of Stentgraft Patency (Day 30)
15 days after stentgraft insertion, perform an arteriography to control graft patency
- After anesthesia, perform a transverse elbow to elbow incision. The left common carotid artery is located beneath the submandibular and sublingual glands and beneath the sternomastoid muscle. 2 cm length exposure is recommended.
- Ligate the artery distally with a silk 4-0. After proximal clamping, insert an 18G catheter retrogradely and secure with a silk 4-0 passed twice around the carotid artery and tied. A sufficient length allows insertion and securing the catheter before proximal clamp is removed. If it’s hard to expose 2cm of artery, the tube, clamped after filled with saline can be pushed proximally whilst removing the proximal clamp.
- Inject contrast through the catheter under direct fluoroscopy. Perform a stentgraft angiographic control to both confirm the absence of endoleak which could result of an inappropriate deployment of the stentgraft, and confirm stentgraft patency. This step is important because the high numbers of collaterals vessels allow adequate leg perfusion even with a thrombosed stentgraft. We didn’t try to perform Duplex ultrasonography: we postulate that stentgraft steel structure prevents correct assessment of stentgraft patency and endoleaks.
- Sacrifice the rat by exsanguination according to the recommendations18, and the aneurysm, stentgraft, thrombus and aortic wall are harvested for histological or biochemical studies.
Representative Results
Aneurysm is fixed in a paraformaldehyde 4% solution during 24 hr, then into a phosphate buffered saline solution until fixation in a resin bloc. We use a HPS (Hematoxylin Phloxin Safran) (Figure 1) staining, showing healing proliferative cells around the PTFE (Figure 2). Mesenchymal cells are then characterized by α-actin staining (Figure 3).
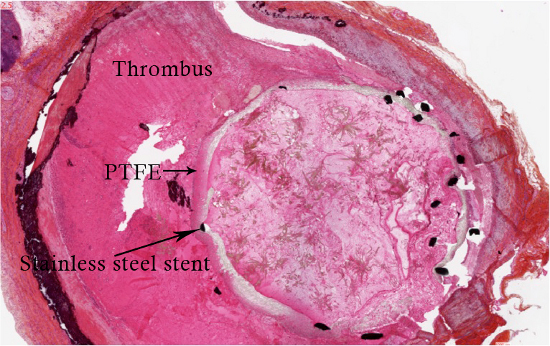
Figure 1. HPS coloration of the aneurysm after exclusion by a 3 mm diameter covered stentgraft.
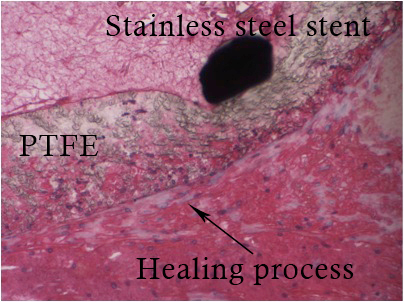
Figure 2. Zoom in on Figure 1 showing a healing process.
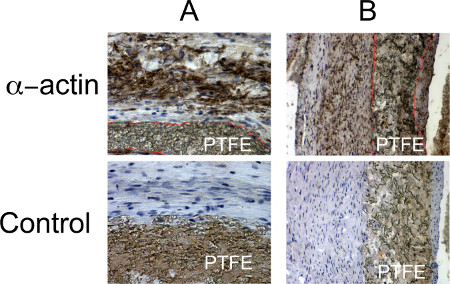
Figure 3. Histological characterization of the healing process. Mesenchymal cells are brown colored by α-actin staining. PTFE is delimitated by the red line. A: x40, B: x20.
Discussion
Two models of AAA have been already described in our lab: xenograft19 and elastase induced models.1 Elastase model is the most similar to human aortic aneurysms: a large circumferential intraluminal thrombus is then excluded by endovascular aneurysm repair.
Endoleaks are a usual complication of EVAR14,15. Lots of questions remain unsolved: AAA rupture have been described with low endoleaks, and sometimes without aneurysm sac enlargement16. Endoleak is a very usual complication after EVAR, maintaining an interface between blood circulating cells and aneurysm thrombus. Type V endoleaks, also named endotension, cannot only explain aneurysm growth by themselves20. Enzymatic activity could be part of this complication. Animal experimentations, with covered and uncovered stentgraft: maintaining an interface between thrombus and circulating blood, is necessary to elucidate the pathobiology of these complications.
Actually, only covered stentgrafts have been deployed on our model. Many ways of research are planned to be explored: implanting non covered endografts, testing the flow divertion materials, enriching the thrombus activity by porphyromonas gingivalis injections21. This model of endovascular exclusion of an aortic aneurysm is the first described in the literature in the small animal. We believe it to be of great help for a better understanding of excluded aneurysm remodeling22. Aneurysm stentgraft exclusions have already be published on bigger animals, but with a lower reproducibility, hard to realize, and time consuming4,5. Rat elastase aneurysm model is cheap and feasible with a few practice, and with a low mortality23.
The main limitation of this model was the high frequency of vessel occlusion. The stentgraft, or the aorta easily thrombose, due to possible intima injuries during stentgraft insertion and clots inside the stentgraft after deployment. Washing carefully the stentgraft is really important before stitching the aorta. Preventive oral aspirin administration, started 5 days before the second operation to the end of the protocol significantly decreased thrombosis rate, without significantly per and post operative hemorrhages.
Clamping induced blood stase activates platelets, endothelium activators and neutrophils24. After 30 minutes25, neutrophils and monocyte are already activated, crawling and binding to the normal endothelium, which in turn causes injuries of the endothelium. For an experienced operator, total clamping duration range from 15 to 20 min. We didn’t explore consequences of ischemia on the thrombus. However, it is impossible to deploy the stentgraft without aorta clamping. Neutrophil and platelets activation induces systemic modifications, but short time clamping limited, and unfortunately impossible to prevent. Nonetheless, we believe these modifications having a limited impact on biological analysis. Considering all models and their limitations26, 27, this one is the first to open animal experimentations after AAA exclusion by stentgrafts.
Recent studies show a significant aneurysm growth percentage after endovascular repair in humans28. A precise analysis of the different stages of thrombus retraction, hypothesis validations and endograft material evaluation can be performed on this model.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
The authors would like to thank Christine Guesdon (Abbott) for the gifts of the Stentgrafts.
Materials
| Animals | |||
| Wistar rats | aged from 8 to 9 weeks. Weight range from 350 to 400 g | ||
| Instruments | |||
| Isoflurane anesthesic system | 4,5% at the beginning, then 2% | ||
| Penthobarbital | Ceva sante animale, Libourne, France | ||
| Dissection stereomicroscope | |||
| Steriles gloves | |||
| Microsurgical Steriles instruments | Moria, Antony, France | Needle holder, Forceps, Scissors,Gilbert approximator o 3 Micro clamps | |
| N°40 Silk string | |||
| Prolen 9-0 and 10-0 | Ethicon, Johnson & Johnson, Auneau, France | ||
| Heat-tapered polyethylene tubing | PE 10 | ||
| Syringe infusion pump | |||
| Porcine pancreatic elastase | Sigma, St. Louis, Mo., USA | 550 μl of salin is mixed with 175 μl of elastase | |
| Indeflator | |||
| 9 to 16 mm length, 3 mm diameter coronary PTFE covered stentgraft | Abbott, Abbott Park, Illinois, USA | ||
| C-Arm OEC 9800 | GE Medical Systems, Milwaukee, WI | ||
| Aspegic 1000 mg | Aspirin, Sanofi-Aventis, Paris, France | ||
| Reagent | |||
| Aerane isoflurane 100ML | Baxter | Aerane | |
| Penthobarbital | Centravet | 053pen203 | |
| Silk Suture 4-0 | Fine Science | 18020-40 | |
| Microsurgical steriles instruments | Moria | 9980-9983-9987-204/A-204/D-8148-4877A-4878A | |
| Prolen 9-0 and 10-0 | Ethicon | W2829 – NS2850 | |
| Heat tapered Polyethylene tubing : PE 10 | Bioseb | MRE-010 | |
| Infusion pump | World precision instrument | AL-1000 | |
| Porcine pancreatic elastase | Sigma | E1250-100MG | |
| Indeflator | Longreal | KY025 25 100 | |
| Aspegic 1000mg (Aspirin) | Sanofi-Aventis | 3400931898191 | |
| C-Arm OEC 9800 | General electric. | OEC 9800 | |
| Stentgraft : Jostent | Abbott | 210CG1230 210CG1630 |
Riferimenti
- Anidjar, S., et al. Elastase-induced experimental aneurysms in rats. Circulation. 82, 973-981 (1990).
- Azuma, J., Asagami, T., Dalman, R., Tsao, P. S. Creation of murine experimental abdominal aortic aneurysms with elastase. J. Vis. Exp. (29), e1280 (2009).
- Parodi, J. C., Palmaz, J. C., Barone, H. D. Transfemoral intraluminal graft implantation for abdominal aortic aneurysms. Ann. Vasc. Surg. 5, 491-499 (1991).
- Lees, T., Stansby, G. . The nationalvascular database report, 2009. , (2012).
- Sakalihasan, N., Limet, R., Defawe, O. D. Abdominal aortic aneurysm. Lancet. 365, 1577-1589 (2005).
- Busuttil, R. W., Abou-Zamzam, A. M., Machleder, H. I. Collagenase activity of the human aorta. A comparison of patients with and without abdominal aortic aneurysms. Arch. Surg. 115, 1373-1378 (1980).
- Michel, J. B. Contrasting outcomes of atheroma evolution: intimal accumulation versus medial destruction. Arterioscler. Thromb. Vasc. Biol. 21, 1389-1392 (2001).
- Vorp, D. A., et al. Association of intraluminal thrombus in abdominal aortic aneurysm with local hypoxia and wall weakening. J. Vasc. Surg. 34, 291-299 (2001).
- Kazi, M., et al. Influence of intraluminal thrombus on structural and cellular composition of abdominal aortic aneurysm wall. J. Vasc. Surg. 38, 1283-1292 (2003).
- Kuijper, P. H., et al. Neutrophil adhesion to fibrinogen and fibrin under flow conditions is diminished by activation and L-selectin shedding. Blood. 89, 2131-2138 (1997).
- Touat, Z., et al. Renewal of mural thrombus releases plasma markers and is involved in aortic abdominal aneurysm evolution. Am. J. Pathol. 168, 1022-1030 (2006).
- Fontaine, V., et al. Role of leukocyte elastase in preventing cellular re-colonization of the mural thrombus. Am. J. Pathol. 164, 2077-2087 (2004).
- Sakalihasan, N., Delvenne, P., Nusgens, B. V., Limet, R., Lapiere, C. M. Activated forms of MMP2 and MMP9 in abdominal aortic aneurysms. J. Vasc. Surg. 24, 127-133 (1996).
- Alsac, J. M., et al. The significance of endoleaks in thoracic endovascular aneurysm repair. Ann. Vasc. Surg. 25, 345-351 (2011).
- Buth, J., Harris, P. L., van Marrewijk, C., Fransen, G. The significance and management of different types of endoleaks. Semin. Vasc. Surg. 16, 95-102 (2003).
- Fransen, G. A., et al. Rupture of infra-renal aortic aneurysm after endovascular repair: a series from EUROSTAR registry. Eur. J. Vasc. Endovasc. Surg. 26, 487-493 (2003).
- van der Vaart, M. G., et al. Application of PET/SPECT imaging in vascular disease. Eur. J. Vasc. Endovasc. Surg. 35, 507-513 (2008).
- Close, B., et al. Recommendations for euthanasia of experimental animals: Part 2. DGXT of the European Commission. Lab. Anim. 31, 1-32 (1997).
- Allaire, E., Guettier, C., Bruneval, P., Plissonnier, D., Michel, J. B. Cell-free arterial grafts: morphologic characteristics of aortic isografts, allografts, and xenografts in rats. J. Vasc. Surg. 19, 446-456 (1994).
- Gawenda, M., Jaschke, G., Winter, S., Wassmer, G., Brunkwall, J. Endotension as a result of pressure transmission through the graft following endovascular aneurysm repair–an in vitro study. Eur. J. Vasc. Endovasc. Surg. 26, 501-505 (2003).
- Delbosc, S., et al. Porphyromonas gingivalis Participates in Pathogenesis of Human Abdominal Aortic Aneurysm by Neutrophil Activation. Proof of Concept in Rats. PLoS One. 6, e18679 (2011).
- Trollope, A., Moxon, J. V., Moran, C. S., Golledge, J. Animal models of abdominal aortic aneurysm and their role in furthering management of human disease. Cardiovasc Pathol. 20, 114-123 (2011).
- Yamaguchi, T., et al. Factors influencing mortality in the rat elastase-induced-aneurysm model. J. Surg. Res. 94, 81-83 (2000).
- von Bruhl, M. L., et al. Monocytes, neutrophils, and platelets cooperate to initiate and propagate venous thrombosis in mice in vivo. J. Exp. Med. 209, 819-835 (2012).
- Smith, P. D. Neutrophil activation and mediators of inflammation in chronic venous insufficiency. J. Vasc. Res. 36, 24-36 (1999).
- Zaragoza, C., et al. Animal models of cardiovascular diseases. J. Biomed. Biotechnol. 2011, 497841 (2011).
- Kuivaniemi, H., Elmore, J. R. Opportunities in abdominal aortic aneurysm research: epidemiology, genetics, and pathophysiology. Ann. Vasc. Surg. 26 (12), 862-870 (2012).
- Schanzer, A., et al. Predictors of abdominal aortic aneurysm sac enlargement after endovascular repair. Circulation. 123, 2848-2855 (2011).

