A Lateralized Odor Learning Model in Neonatal Rats for Dissecting Neural Circuitry Underpinning Memory Formation
Summary
This protocol introduces lateralized early odor preference learning in rats using acute single naris occlusion. Lateralized learning permits the examination of behavioral outcomes and underpinning biological mechanisms within the same animals, reducing variance induced by between-animal designs. This protocol can be used to investigate molecular mechanisms underpinning early odor learning.
Abstract
Rat pups during a critical postnatal period (≤ 10 days) readily form a preference for an odor that is associated with stimuli mimicking maternal care. Such a preference memory can last from hours, to days, even life-long, depending on training parameters. Early odor preference learning provides us with a model in which the critical changes for a natural form of learning occur in the olfactory circuitry. An additional feature that makes it a powerful tool for the analysis of memory processes is that early odor preference learning can be lateralized via single naris occlusion within the critical period. This is due to the lack of mature anterior commissural connections of the olfactory hemispheres at this early age. This work outlines behavioral protocols for lateralized odor learning using nose plugs. Acute, reversible naris occlusion minimizes tissue and neuronal damages associated with long-term occlusion and more aggressive methods such as cauterization. The lateralized odor learning model permits within-animal comparison, therefore greatly reducing variance compared to between-animal designs. This method has been used successfully to probe the circuit changes in the olfactory system produced by training. Future directions include exploring molecular underpinnings of odor memory using this lateralized learning model; and correlating physiological change with memory strength and durations.
Introduction
Olfaction is the primary sensory modality in rodents, without which they would not be able to successfully navigate or survive in their environment. It is especially critical for neonatal pups, which can neither see nor hear during the first post-natal week, to use olfaction in order to locate their mother to feed1. As a result, neonatal rat pups can be conditioned to prefer odors with simple experimental manipulations. A variety of stimuli have been used as the unconditioned stimulus (UCS) to induce conditioned responses to novel odors (conditioned stimulus, CS) in neonates, including the nesting environment2,3, milk suckling4-6, stroking or tactile stimulation7-12, tail pinch13, maternal saliva13, mild foot shock14-18, and intracranial brain stimulation19. The present study employs a well-established early odor preference paradigm wherein an odor, in this case peppermint, is combined with tactile stimulation in order to produce a preference for peppermint 24 hr later10,11,20. These odors memories are dependent on intact olfactory circuitry, primarily including the olfactory bulbs (OB)21-23 and the anterior piriform cortex (aPC)24,25.
Experimental investigations of the early odor preference learning have deepened and broadened our understanding of the molecular and physiological underpinnings of a mammalian memory. This mammalian model has several advantages in studying memory mechanisms. First, the neural sources of the UCS signal have been identified. Various stimuli as mentioned above stimulate locus coeruleus norepinephrine release26, which in turn activates multiple adrenoceptors in the OB and aPC, causing cellular and physiological effects that support learning22,27,28. Second, memory-supporting mechanisms take place in well-defined laminar neural structures. The simplicity of the olfactory circuitry in neonatal rats provides researchers with the ideal framework with which to uncover the intricate processes related to synaptic plasticity. Olfactory sensory neurons (OSN) in the olfactory epithelium project onto mitral/tufted cells in the OB and these mitral/tufted cells in turn project ipsilaterally to piriform cortex (PC) via the lateral olfactory tract (LOT), among other structures29. Both the OSN synapses in the OB30,31 and the LOT synapses24,25 in aPC have been identified as critical loci for synaptic changes that support learning and memory. Third, in an early age in rats, olfactory inputs can readily be lateralized. Each aPC has access to bilateral odor information via the anterior commissure once this white matter is fully formed at post-natal day 12 (PD12)32. Before PD 12, odor input can be isolated to ipisilateral OB and aPC through single naris occlusion24,25,31,33,34. Single naris occlusion permits the odor memory formation from the open naris, and prevents the same memory from the occluded naris prior to PD 1233. Odor memory is isolated to the ipsilateral hemisphere including both OB and aPC. Therefore, each rat pup can be its own control for learning and underpinning physiology.
In the present study, the lateralized early odor preference learning protocol is introduced. This method serves as a powerful tool for studying neural mechanisms underpinning odor learning by providing an intra-animal control24,25,31 , thereby reducing both the number of animals required and the general variation. Naris occlusion is reversible in that the grease or nose plug can be applied and removed with minimal stress or damage to the animal. Here, first, detailed procedures of early odor preference training and testing are described, with a focus on the lateralized protocol using single naris occlusion with a nose plug. Then results are presented to demonstrate the effectiveness of single naris occlusion in isolating odor input and producing lateralized odor memory. Finally, the potentials of using this lateralized learning model to study physiological changes in the olfactory system that both generate learning and support memory expression are discussed.
Protocol
Sprague Dawley rat (Charles River) pups of both genders are used. Litters are culled to 12 on PD1 (birth being PD0). The dams are maintained on a 12 hr light/dark cycle with ad libitum access to food and water. Experimental procedures have been approved by Memorial University’s Institutional Animal Care Committee.
1. Nose Plug Construction
NOTE: This procedure was adapted and modified from Cummings et al. (1997)35.
- Aquire polyethylene-20 tubing and 3-0 silk suture thread.
- Cut a small piece of polyethylene-20 tubing to approximately 0.8 mm.
- Thread silk suture through the prepared tubing such that there is thread on either side of the section of tubing.
- On one end of the thread outside of the plug, tie a knot in the thread.
- Pull the section of tubing down over the knot in the thread. The knot should lodge inside the tubing.
- Trim both ends of the thread such that ~2 mm of thread is protruding from one end of the tubing (see Figure 1A).
2. Naris Occlusion before Training
- Remove pup from dam and place in a secure dish covered with regular bedding.
- Use a cotton tip application to dab a local anesthetic jelly, 2% Xylocaine, on the naris to be occluded.
- Allow the pup to rest in the dish for ~3 min.
- Hold the pup gently but securely in the non-dominant hand.
- Using the dominant hand, pick up a nose plug and dab the same local anesthetic jelly around the tip from which the thread is not protruding. This will act as both an anesthetic for any minor pains associated with plug insertion and will act as a lubricant inside the naris. NOTE: The effect of Xylocaine starts within a couple of minutes and lasts 20-30 min. Pups in general show good tolerance for plug insertion after xylocaine jelly application (minimum struggling and vocalization).
- Gently insert the nose plug by firmly holding the pup and slowly rotating the plug with very gentle pushes until the plug is fully inserted and only the 2 mm thread is protruding from the naris (see Figure 1B). There should be no bleeding from either naris during this process. Pups with bleeding during nose plug insertion are excluded and returned to dams
- Allow the animal to rest in this dish for 5 min in order to habituate to the plug.
- Remove the pup from the habituation dish and begin the conditioning paradigm24.
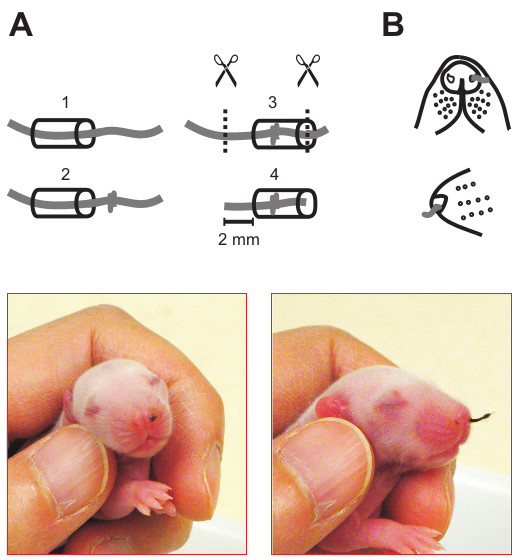
Figure 1. Construction of a nose plug. A) Schematics showing the steps of making a nose plug. A thread is pulled through polyethylene tubing; a knot is made and pulled into the middle of the tubing to block it; two ends are cut with a 2 mm residue in one end out of the tubing. B) Front and lateral view of a rat with a nose plug in one naris.
3. Scented Bedding Preparation
- Wearing new gloves and in a fume hood to prevent odor contamination, place 500 ml of woodchip bedding into a plastic bag.
- Use a syringe to draw up 0.3 ml peppermint extract, and spray this over the bedding in the plastic bag.
- Tie the bag shut, shake the bag vigorously, and allow the bedding to rest in the bag for 5 min.
- Place the scented bedding in a clear, shallow, acrylic training box (20 x 20 x 5 cm3, Figure 2A) uncovered in a fume hood for 5 min before use. Once the bedding is prepared, discard these gloves, and do not allow these gloves to come in contact with the animals.
- Place the unscented bedding in an identical clear plastic box, and ensure that it does not come into contact with the scented bedding or used gloves.
4. Odor Conditioning Paradigm (See Picture in Figure 2A)
Pups undergo either a single conditioning session, on PD 6, or multiple trial sessions (one session per day, PD 3-6).
- Place the habituated pup on scented bedding. For control odor only (O/S–) pups, leave these pups on the bedding for 10 min, then skip to step 4.5. For experimental odor + stroke (O/S+) pups, continue to the following steps in this section.
- Stroke the pup for 30 sec using a small paintbrush. Use rapid circular motions primarily around the hind region of the pup.
- Allow the pup to rest for 30 sec.
- Repeat steps 4.2 & 4.3 for a total of 10 min (i.e. 10 pairings of stroking + odor).
- Remove pup from the conditioning box, remove the nose plug and return the pup to the dam.
5. Lateralized Odor Preference Testing (See Picture in Figure 2B)
Testing occurs at various time points (e.g., 24 or 48 hr) following the final training session. Testing is carried out in a stainless steel testing chamber (30 x 20 x 18 cm3), which is placed on top of two training boxes (training box is described in 3.4), separated by a 2 cm neutral zone. One training box contains peppermint-scented bedding while the other box contains clean, unscented bedding. The floor of the testing chamber is a metal grid, which is then covered by a removable sheet of plastic mesh (Figure 2B).
- Prepare one peppermint and one unscented bedding as per Section 3, and place each box under opposite sides of the testing chamber, 2 cm apart. Place the plastic mesh on the metal-grid floor of the testing chamber.
- Remove the pup from the dam and place a firm dab of odorless silicone grease on the naris that is occluded during training. Re-apply the grease throughout the first testing procedure as needed. NOTE: Random naris occlusion during training and testing may be considered to avoid bias.
- Place pup in the neutral zone of the testing chamber.
- Allow the pup to explore the chamber for 1 min, recording how long the pup spent over the two sides of the chamber (i.e. over peppermint or neutrally-scented bedding).
- Allow the pup to rest for 1 min in a covered plastic holding chamber.
- Repeat steps 5.2 & 5.3 for a total of 10 min (i.e. 5 test trials separated by 5 rest trials) switching the initial orientation of the pup in the chamber in order to control for direction preferences.
- Immediately following testing, wipe away the grease from the naris.
- Insert a polyethylene noseplug into the opposite naris as per section 2 and allow the animal to rest for 10 min.
- Test the pup once again as in 5.3 -5.6, remove the plug, and return the pup to the dam. Remove and clean the plastic mesh of the testing chamber with 95% ethanol and allow the liquid to evaporate. Place the mesh back before testing the next pup.
NOTE: Applying silicone grease at the first naris occlusion during testing prevents the chance of bleeding and stress associated with nose plug insertion.

Figure 2. Early odor preference training and testing. A) Early odor preference training using odor + stroking paradigm. B) Two choice odor preference testing with peppermint bedding on one side, control unscented bedding on the opposite side. A 2 cm neutral zone is placed in between.
6. Testing the Effectiveness of Single Naris Occlusion
This experiment is performed to determine whether single naris occlusion leads to lateralized activation of the olfactory system.
- Perform unilateral naris occlusions on PD 6 or 7 pups as described in section 2.
- After ~5 min habituation, place the pup in a covered plastic container and expose it to 30 µl pure peppermint oil soaked in a piece of tissue for 10 min.
- Immediately after the peppermint odor exposure, inject the pup intraperitoneally (i.p.) with chloral hydrate (400 mg/kg) as a general anesthetic, or pentobarbitol, (150 mg/kg).
- Once fully anesthetized (showing no response to tail or foot pinch), transcardially perfuse the pup with ice-cold solutions of saline (0.9%) for ~1 min, followed by paraformaldehyde (4%, dissolved in 0.1 M phosphate buffer solution, PBS).
- After 10 min of paraformaldehyde perfusion, collect the brain and place it in paraformaldehyde overnight at 4 °C, then transfer the brain to a sucrose solution (20% in PBS) for an additional 24 hr.
- Cut coronal brain slices at 30 μm thickness with a cryostat. Collect OB and PC slices and mount onto gelatin-coated slides, followed by standard immunohistochemistry staining for pCREB antibody21,25,30.
7. Testing the Reversibility of Single Naris Occlusion
This experiment tests whether the blocking effect is reversible at 24 hr following the removal of the nose plug.
- Perform unilateral naris occlusions on PD 6 or 7 pups as described in section 2.
- After 15 min (equivalent to the duration of the naris occlusion during training – 5 min habituation + 10 min training), remove the nose plug, and return the pup to the dam.
- 24 hr later, expose the pup to the peppermint odor in a covered plastic container for 10 min as described in 6.2.
- Follow same steps in sections 6.3-6.6.
Representative Results
Here we review some of the previously established results24 to demonstrate the effectiveness of the naris occlusion in isolating odor input and learning to one hemisphere, and the reversibility of this method.
Single naris occlusion during early odor preference training leads to a lateralized odor memory24. The memory is confined to the spared naris (Figure 3). When pups are tested for odor preference with the same naris occluded as during training, they show preference for the conditioned odor (e.g., peppermint). When pups are tested with the opposite naris occluded, they show no preference for the conditioned odor. Together, these results suggest that an odor preference memory is only formed and expressed through the spared naris that underwent the odor + stroking associative conditioning.
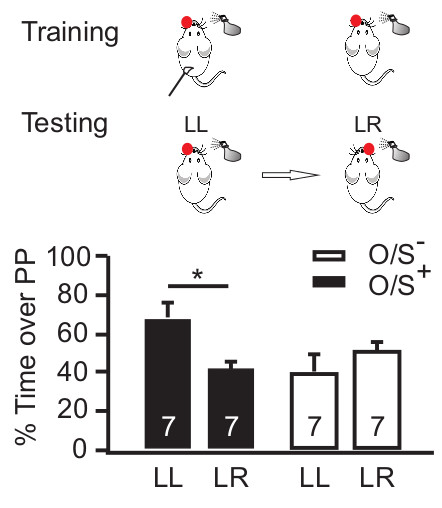
Figure 3. Single naris occlusion induces lateralized odor learning. The behavioral protocol is shown in the upper panel. Odor + stroking (O/S+) or odor only (O/S–) animals with single naris occluded during training, underwent odor preference testing first with the same naris occluded then with the opposite naris occluded. The lower panel shows the percentage of time spent over peppermint-scented bedding among different groups in a two choice odor test. *p < 0.05. Error bars, mean ± SEM. Reproduced from Fontaine et al. J. Neuroscience (2013) with permission.
The lateralized odor training results in lateralized activation of the olfactory system during odor exposure (Figure 4)24. Single naris occlusion prevents activation of the OB and the PC of the ipsilateral hemisphere during odor exposure. This is demonstrated by monitoring CREB phosphorylation in the OB, and the PC. As shown in Figure 4, using immunohistochemistry, phosphorylated CREB (pCREB) is significantly less in the occluded hemisphere following peppermint odor exposure, compared to the contralateral spared hemisphere. Nissl staining (Figure 4A) demonstrates comparable cell bodies in the mitral cell layer of the OB, and in the pyramidal cell layer of the PC of both hemispheres. However, pCREB is significantly less in both cell layers in the hemisphere ipsilateral to the occluded naris (Figure 4B).
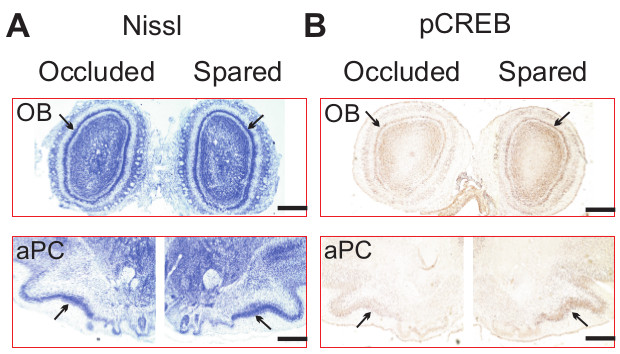
Figure 4. Single naris occlusion results in lateralized activation of the olfactory system during odor exposure. A) Nissl staining of the olfactory bulb (OB) and anterior piriform cortex (aPC). B) PCREB expressions in the occluded and spared hemispheres following peppermint exposure in a single naris occluded pup. Arrows indicate mitral cell layer in the OB and pyramidal cell layer in the aPC. Scale bars, 500 μm. Reproduced from Fontaine et al. J. Neuroscience (2013) with permission.
The effect of a single trial (15-20 min) naris occlusion is transient and reversible, and does not result in visible longer-term neuronal damage that could lead to altered odor perception and reduced neuronal activation to odors during testing24. As indexed by pCREB staining in the OB and the PC (Figure 5), pCREB expressions in mitral cells of the OB, and the pyramidal cells in the PC to odor exposure are comparable between the occluded and spared hemispheres, 24 hr following the removal of the nose plug – the same time point that odor preference testing is carried out following early odor preference training.
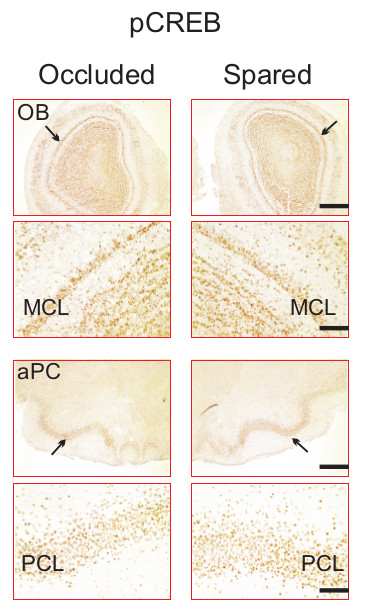
Figure 5. Assessment of neuronal reactivity following reversible naris occlusion. PCREB staining of the OB and the aPC, 24 hr following the removal of a nose plug in one pup. Arrows indicate mitral cell layer in the OB and pyramidal cell layer in the aPC. MCL, mitral cell layer. PCL, pyramidal cell layer. Scale bars, 500 µm for lower magnification and 100 μm for higher magnification.
Discussion
The lateralized odor learning and memory model in rat pups within a critical time window was first established by Hall and colleagues. In a series of studies33,34,36, they showed that an odor preference memory could be lateralized by odor + milk pairings to one naris at PD 6 in rat pups. Preference memory was robust when the same naris was open during training and testing, but not observed when the occluded naris was unblocked and tested. However, at PD 12, when anterior commissural connections from the anterior olfactory cortices become functional32, the untrained naris alone could support the expression of the odor preference acquired at PD 6. Lesion of the anterior commissure restored the lateralized memory so it was no longer accessible from the untrained naris33. This lateralized learning model has been successfully replicated with an odor + stroking paradigm24,25,31. Odor + stroking training with single naris occlusion on PD 6 leads to a lateralized memory on PD 731. Repeated single naris occlusions on PD 3-6 leads to a longer lateralized memory that lasts at least 48 hr24.
The lateralized odor learning protocol results in “split brain” in individual animals. This model has great advantages for studying behavior and the underpinning bio-physiological changes. For instance, comparing behavioral outputs using a within-subject model sufficiently reduces variations in between-animal designs. Pups at this early age vary considerably in their activity and responsiveness. Intra-animal control removes the inherent individual variability in performance and responsiveness as well as intrinsic differences in biology from the assessment of molecular and physiological changes. Additionally, this powerful infant model of lateralized learning lends us an opportunity to relate individual memory performance to individual physiology, and to assess the underpinnings of memories of different durations24,25,31. Using this lateralized learning model in combination with ex vivo experiments comparing the physiology of the two hemispheres within the same animals, It has been recently demonstrated that early odor learning induces synaptic plasticity such as increased AMPA receptor responses at synapses in both the OB31, and the aPC24,25. Enhanced synaptic transmission following early odor learning translates into an enhanced output in olfactory network representations24.
Future studies should explore the molecular underpinnings of odor memory using this lateralized learning model. This includes correlational studies looking at proteins and genes activated following learning, and causal studies looking at the effects of gain-of-function and loss-of-function of proteins and genes of particular interest. Another exciting and important possibility is to be able to relate physiological and molecular changes to the strength of behavioral memory. For each pup, it is possible to first derive a preference memory measure for the open and occluded nares. Subsequent ex vivo experiments on the corresponding trained and untrained cortices would provide correlative physiological changes. It is possible, however, that odor re-exposure during preference testing itself will alter synaptic strength, or that other brain regions contribute significantly to memory expression. In our present studies, physiology and behavior are carried out on separate cohorts. This removes concerns about behavioral testing influencing the very parameters of interest.
One caveat using nose plug is potential neural tissue damage that is associated with plug insertion and removal. For this reason, care should been taken in insertion of the nose plug and pups with bleeding during insertion should be excluded, to avoid any potential longer-term effects associated with bleeding such as inflammation. Prolonged blockage (hours, days to months) of naris or permanent ablation of olfactory epithelium leads to long-term olfactory deprivation, neural damage, and reduced neuronal activities in the OB and PC37-41, even though some of these effects due to prolonged naris occlusion are fully reversible39,40. Tissue integrity following acute naris occlusion (~15 min) has been validated by immunohistochemistry staining of pCREB, an activity-dependent neuronal marker, which has been shown to be reduced in the PC of young adult mice following 5-day naris occlusion39. pCREB levels in the ipsilateral OB and PC of the occluded naris were significantly less during peppermint odor exposure, confirming the successful lateralization of olfactory throughput in rat pups during naris occlusion. However, 24 hr following the removal of the nose plug, at the time of odor preference testing, the pCREB levels are comparable in both hemispheres, suggesting the effect of acute occlusion is fully reversible24. Therefore, the lack of preference to peppermint tested with the previously occluded naris is due to lack of memory, but not due to an alteration or lack of odor perception associated with tissue damage during testing. Additionally, electrophysiological recordings of control O/S– animals (with one naris occluded during odor exposure without stroking) showed no differences in the fEPSPs or number of activated pyramidal cells seen with calcium imaging – also confirming there is no functional change in the piriform cortex due to these short-term reversible naris occlusions24,25.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work was supported by a CIHR operating grant (MOP-102624) to Q. Y. We thank Dr Carolyn Harley for helpful discussions throughout the study, Dr. Qinlong Hou, Amin Shakhawat, and Andrea Darby-King for technical support.
Materials
| Polythylene 20 tubing | Intramedic | 427406 | Non radiopaque, Non toxic |
| 3-0 silk suture thread | Syneture | Sofsilk | Non absorbant |
| Silicone grease | Warner Instrument | 64-0378 | Odorless |
| 2% xylocaine gel | AstraZeneca | Prod. No 061 | Lidocaine hydrochloride jelly, purchased at local pharmacy |
| Paint brush | Dynasty | 206R | Similar size/other brands work too |
| Peppermint extract | Sigma-Aldrich | W284807 | Other brand should be okay too |
| Training box | Custom-made | N/A | Acrylic box (20x20x5cm3), see Figure 2A. Parameters and material for the box are not critical and can be modified. Material used should be odorless and does not absorb odors |
| Testing chamber | Custom-made | N/A | Stainless steel (30x20x18cm3), see Figure 2B. Parameters and material for the chamber are not critical and can be modified. For example, an acrylic chamber instead of a stainless steel one can be used |
| pCREB antibody | Cell Signaling | 9198 | Ser 133 (87G3) Rabbit mAb |
| Chloral hydrate | Sigma-Aldrich | C8383 | N/A |
| Paraformaldehype | Sigma-Aldrich | P6148 | N/A |
| Sucrose | Sigma-Aldrich | S9378 | N/A |
Riferimenti
- Gregory, E. H., Pfaff, D. W. Development of olfactory-guided behavior in infant rats. Physiol Behav. 6, 573-576 (1971).
- Alberts, J. R., May, B. Nonnutritive, thermotactile induction of filial huddling in rat pups. Dev Psychobiol. 17, 161-181 (1984).
- Galef, B. G., Kaner, H. C. Establishment and maintenance of preference for natural and artificial olfactory stimuli in juvenile rats. J Comp Physiol Psychol. 94, 588-595 (1980).
- Johanson, I. B., Hall, W. G. Appetitive learning in 1-day-old rat pups. Science. 205, 419-421 (1979).
- Johanson, I. B., Hall, W. G. Appetitive conditioning in neonatal rats: conditioned orientation to a novel odor. Dev Psychobiol. 15, 379-397 (1982).
- Johanson, I. B., Teicher, M. H. Classical conditioning of an odor preference in 3-day-old rats. Behav Neural Biol. 29, 132-136 (1980).
- McLean, J. H., Darby-King, A., Sullivan, R. M., King, S. R. Serotonergic influence on olfactory learning in the neonate rat. Behav Neural Biol. 60, 152-162 (1993).
- Moore, C. L., Power, K. L. Variation in maternal care and individual differences in play, exploration, and grooming of juvenile Norway rat offspring. Dev Psychobiol. 25, 165-182 (1992).
- Pedersen, P. E., Williams, C. L., Blass, E. M. Activation and odor conditioning of suckling behavior in 3-day-old albino rats. J Exp Psychol Anim Behav Process. 8, 329-341 (1982).
- Sullivan, R. M., Hall, W. G. Reinforcers in infancy: classical conditioning using stroking or intra-oral infusions of milk as UCS. Dev Psychobiol. 21, 215-223 (1988).
- Sullivan, R. M., Leon, M. Early olfactory learning induces an enhanced olfactory bulb response in young rats. Brain Res. 392, 278-282 (1986).
- Weldon, D. A., Travis, M. L., Kennedy, D. A. Posttraining D1 receptor blockade impairs odor conditioning in neonatal rats. Behav Neurosci. 105, 450-458 (1991).
- Sullivan, R. M., Hofer, M. A., Brake, S. C. Olfactory-guided orientation in neonatal rats is enhanced by a conditioned change in behavioral state. Dev Psychobiol. 19, 615-623 (1986).
- Camp, L. L., Rudy, J. W. Changes in the categorization of appetitive and aversive events during postnatal development of the rat. Dev Psychobiol. 21, 25-42 (1988).
- Moriceau, S., Wilson, D. A., Levine, S., Sullivan, R. M. Dual circuitry for odor-shock conditioning during infancy: corticosterone switches between fear and attraction via amygdala. J Neurosci. 26, 6737-6748 (2006).
- Roth, T. L., Sullivan, R. M. Endogenous opioids and their role in odor preference acquisition and consolidation following odor-shock conditioning in infant rats. Dev Psychobiol. 39, 188-198 (2001).
- Roth, T. L., Sullivan, R. M. Consolidation and expression of a shock-induced odor preference in rat pups is facilitated by opioids. Physiol Behav. 78, 135-142 (2003).
- Sullivan, R. M. Developing a sense of safety: the neurobiology of neonatal attachment. Ann N Y Acad Sci. 1008, 122-131 (2003).
- Wilson, D. A., Sullivan, R. M. Olfactory associative conditioning in infant rats with brain stimulation as reward. I. Neurobehavioral consequences. Brain Res Dev Brain Res. 53, 215-221 (1990).
- Sullivan, R. M., Wilson, D. A., Leon, M. Associative Processes in Early Olfactory Preference Acquisition: Neural and Behavioral Consequences. Psychobiology. , 29-33 (1989).
- McLean, J. H., Harley, C. W., Darby-King, A., Yuan, Q. pCREB in the neonate rat olfactory bulb is selectively and transiently increased by odor preference-conditioned training. Learn Mem. 6, 608-618 (1999).
- Sullivan, R. M., Stackenwalt, G., Nasr, F., Lemon, C., Wilson, D. A. Association of an odor with activation of olfactory bulb noradrenergic beta-receptors or locus coeruleus stimulation is sufficient to produce learned approach responses to that odor in neonatal rats. Behav Neurosci. 114, 957-962 (2000).
- Yuan, Q., Harley, C. W., McLean, J. H. Mitral cell beta1 and 5-HT2A receptor colocalization and cAMP coregulation: a new model of norepinephrine-induced learning in the olfactory bulb. Learn Mem. 10, 5-15 (2003).
- Fontaine, C. J., Harley, C. W., Yuan, Q. Lateralized odor preference training in rat pups reveals an enhanced network response in anterior piriform cortex to olfactory input that parallels extended memory. J Neurosci. 33, 15126-15131 (2013).
- Morrison, G. L., Fontaine, C. J., Harley, C. W., Yuan, Q. A role for the anterior piriform cortex in early odor preference learning: evidence for multiple olfactory learning structures in the rat pup. J Neurophysiol. 110, 141-152 (2013).
- Nakamura, S., Kimura, F., Sakaguchi, T. Postnatal development of electrical activity in the locus ceruleus. J Neurophysiol. 58, 510-524 (1987).
- Harley, C. W., Darby-King, A., McCann, J., McLean, J. H. Beta1-adrenoceptor or alpha1-adrenoceptor activation initiates early odor preference learning in rat pups: support for the mitral cell/cAMP model of odor preference learning. Learn Mem. 13, 8-13 (2006).
- Shakhawat, A. M., Harley, C. W., Yuan, Q. Olfactory bulb alpha2-adrenoceptor activation promotes rat pup odor-preference learning via a cAMP-independent mechanism. Learn Mem. 19, 499-502 (2012).
- Isaacson, J. S. Odor representations in mammalian cortical circuits. Curr Opin Neurobiol. 20, 328-331 (2010).
- Lethbridge, R., Hou, Q., Harley, C. W., Yuan, Q. Olfactory bulb glomerular NMDA receptors mediate olfactory nerve potentiation and odor preference learning in the neonate rat. PLoS One. 7, e35024 (2012).
- Yuan, Q., Harley, C. W. What a nostril knows: olfactory nerve-evoked AMPA responses increase while NMDA responses decrease at 24-h post-training for lateralized odor preference memory in neonate rat. Learn Mem. 19, 50-53 (2012).
- Schwob, J. E., Price, J. L. The development of axonal connections in the central olfactory system of rats. J Comp Neurol. 223, 177-202 (1984).
- Kucharski, D., Hall, W. G. New routes to early memories. Science. 238, 786-788 (1987).
- Kucharski, D., Johanson, I. B., Hall, W. G. Unilateral olfactory conditioning in 6-day-old rat pups. Behav Neural Biol. 46, 472-490 (1986).
- Cummings, D. M., Henning, H. E., Brunjes, P. C. Olfactory bulb recovery after early sensory deprivation. J Neurosci. 17, 7433-7440 (1997).
- Kucharski, D., Hall, W. G. Developmental change in the access to olfactory memories. Behav Neurosci. 102, 340-348 (1988).
- Brunjes, P. C. Unilateral odor deprivation: time course of changes in laminar volume. Brain Res Bull. 14, 233-237 (1985).
- Kass, M. D., Pottackal, J., Turkel, D. J., McGann, J. P. Changes in the neural representation of odorants after olfactory deprivation in the adult mouse olfactory bulb. Chem Senses. 38, 77-89 (2013).
- Kim, H. H., Puche, A. C., Margolis, F. L. Odorant deprivation reversibly modulates transsynaptic changes in the NR2B-mediated CREB pathway in mouse piriform cortex. J Neurosci. 26, 9548-9559 (2006).
- Korol, D. L., Brunjes, P. C. Rapid changes in 2-deoxyglucose uptake and amino acid incorporation following unilateral odor deprivation: a laminar analysis. Brain Res Dev Brain Res. 52, 75-84 (1990).
- Leung, C. H., Wilson, D. A. Trans-neuronal regulation of cortical apoptosis in the adult rat olfactory system. Brain Res. 984, 182-188 (2003).

