Pre-clinical Orthotopic Murine Model of Human Prostate Cancer
Summary
Prostate cancer is the second most common cause of cancer-related deaths in the United States. An orthotopic cancer model provides a useful approach to understand the biology of prostate cancer and to evaluate the efficacy of therapeutic regimens. This protocol describes detailed steps necessary to establish an orthotopic prostate cancer mouse model.
Abstract
To study the multifaceted biology of prostate cancer, pre-clinical in vivo models offer a range of options to uncover critical biological information about this disease. The human orthotopic prostate cancer xenograft mouse model provides a useful alternative approach for understanding the specific interactions between genetically and molecularly altered tumor cells, their organ microenvironment, and for evaluation of efficacy of therapeutic regimens. This is a well characterized model designed to study the molecular events of primary tumor development and it recapitulates the early events in the metastatic cascade prior to embolism and entry of tumor cells into the circulation. Thus it allows elucidation of molecular mechanisms underlying the initial phase of metastatic disease. In addition, this model can annotate drug targets of clinical relevance and is a valuable tool to study prostate cancer progression. In this manuscript we describe a detailed procedure to establish a human orthotopic prostate cancer xenograft mouse model.
Introduction
Prostate cancer is the second most prevalent cause of cancer deaths (9%) among males in the United States, next to cancer of the lung and bronchus (28%)1. According to recent data, it is estimated that 220, 800 newly diagnosed prostate cancer cases and 27, 540 deaths will occur in 20151. The five year relative survival rate of early stage prostate cancer is >99% while that of advanced metastatic disease is only 28%1. A major challenge for treatment of advanced metastatic disease is the lack of understanding of molecular mechanisms underlying the propensity of this disease to metastasize to other organs, particularly to the bone, which is a frequent site for prostate cancer. Hence, there is a clear need to study the molecular makeup of these prostate tumors in order to develop effective therapeutic regimens against progression to advanced metastatic disease2,3.
Prostate tumors exhibit high biological heterogeneity without a well-defined pathway to progression. Metastases often occur with no prior indication of tumor invasiveness4. This clinical heterogeneity is attributed to the molecular diversity of prostate cancer. Understanding the molecular makeup of these lethal tumors is the key to design better diagnostic and therapeutic strategies for this disease. Consequently, prostate cancer research is currently focused on understanding and preventing metastasis.
Pre-clinical in vivo mouse models offer a variety of options to understand the molecular mechanisms of prostate cancer progression to advanced metastatic disease. In addition, these models are important for preclinical evaluations of new therapeutic strategies against this disease. The most commonly used animal models include transgenic mouse models, tail-vein injection, intra-cardiac implantation and human orthotopic mouse models. Transgenic studies are time consuming and correlation of prostate cancer development in mice with that of humans have shown variability11. In spontaneous metastatic mouse models, cells are injected directly into the circulation and though, they have rapid turnaround time, they cannot be used to study the primary tumor or the initial steps in the metastatic cascade5. Orthotopic xenograft models have the limitation of developing bone metastatic lesions, the common site of prostate cancer metastasis. Nonetheless, the human orthotopic prostate cancer xenograft mouse model is well characterized and widely used to study the molecular events of primary tumor development, cross-talk between tumor and organ microenvironment, initial phase of the metastatic disease and use of experimental drugs for therapeutic intervention6,7,8-11.
Protocol
Protocols for all procedures involving animals must be reviewed and approved by an Institutional Animal Care and Use Committee (IACUC). Follow officially approved procedures for the care and use of laboratory animals. Intra-prostatic injection requires open-abdominal surgery and animals should be kept in a pathogen-free environment with a designated surgery room where proper surgical aseptic techniques are used during the entire procedure.
1. Preparation of Cells for Implantation
NOTE: Based on research need, any prostate cancer cell line can be used. Cell lines are cultured according to the supplier's instructions.
- For the PC3M-Luc-C6 cell line that stably expresses the firefly luciferase gene, culture cells in Minimum Essential Medium (MEM) supplemented with 10% Fetal Bovine Serum (FBS), 1x Non-essential aminoacids, 1x Phleomycin D1 and 1mM sodium Pyruvate. Maintain cells in an incubator with a humidified atmosphere of 95% air and 5% CO2 at 37 °C. PC3M-Luc-C6 cells were procured from UCSF core facility.
- Harvest cells by trypsinization. Wash culture plates once with phosphate buffered saline (PBS). Add 2 ml 0.05% Trypsin to a 10 cm dish and incubate for 3-5 min in an incubator with a humidified atmosphere of 95% air and 5% CO2 at 37 °C till cells are detached.
- To avoid clumping, do not agitate the cells by hitting or shaking the dish while waiting for cells to detach. Collect the cells in 5 ml of complete media and spin down for 5 minutes at 200 x g. Wash the cell pellet with PBS to remove trypsin.
- Enumerate live cells by trypan blue exclusion assay. Mix 10 µl of the cell suspension in PBS with 10 µl of 0.4% (wt/v) trypan blue solution. Load the mixture into a hemocytometer or counting chamber slide and count cells immediately under a microscope or read the chamber slide in a cell counter.
- Prepare a cell suspension containing 2.5 x 105 cells in 10 µl media. Mix the cell suspension with 10 μl basement membrane-like extracellular matrix extract and place the cells on ice. Add luciferin to the cell suspension (1:200 µl; stock 30 mg/ml concentration) before injecting into mice.
NOTE: This allows immediate bio-imaging of animals to check the consistency of cell injections among different experimental groups. Inject cells as quickly as possible, preferably within 30 min after trypsinization since cell viability decreases rapidly after detachment.
- Prepare a cell suspension containing 2.5 x 105 cells in 10 µl media. Mix the cell suspension with 10 μl basement membrane-like extracellular matrix extract and place the cells on ice. Add luciferin to the cell suspension (1:200 µl; stock 30 mg/ml concentration) before injecting into mice.
2. Preparation of the Surgical Area
- Perform surgery in an uncluttered, disinfected area that promotes asepsis.
- Sanitize the counter/lab bench with bleach solution before surgery.
NOTE: The use of alcohol is discouraged due to long contact time required to take effect (15 min). - Use sterile drapes, clean absorbable pads or towels, and replace these materials after each surgical session. Sterilize all instruments prior to use. Preferred methods are a steam autoclave, glass bead sterilizer, ethylene oxide gas, or hydrogen peroxide vapor sterilization.
- Use an aseptically cleaned dissecting microscope to perform the surgical procedure or experienced researchers can perform it without a microscope.
3. Implantation of Tumor Cells
- Use male 6-8 week old immunocompromised Balb/c or NOD/SCID mice.
NOTE: It is more difficult to operate on smaller animals and larger animals tend to have slower kinetics of tumor growth and metastasis. - Inject pre-surgery pain medication according to the animal facility's instructions. For example, intra-peritoneal Buprenorphine at a dose of 0.1 mg/kg body weight can be used.
- Anesthetize animals by placing them into an isoflurane chamber with 1-3% isoflurane in oxygen and wait until animals are fully anesthetized. Ensure that there is no toe reflex of muscle tone at this point. Use proper veterinary ophthalmic ointment lubricant to prevent blindness due to xerophthalmia during general anesthesia.
NOTE: Anesthetize the animals by the investigator's preferred method e.g., for Pentobarbital Sodium, 0.05 mg per gram body weight is administered intra-peritoneal or Ketamine/Xylazine solution (concentration: 17.16 mg/ml) at a dose of 65 mg/kg body weight is used subcutaneously. Isoflurane inhalation is a preferred method of anesthetization. The eye ointment should be gently applied without rubbing against the cornea. - Remove the animal from the isoflurane chamber and place into a nose cone apparatus with continuous flow of 1-2% isoflurane in oxygen to ensure that the animal is under full anesthesia before proceeding.
- Remove hair from mice by shaving or use a hair removing cream before beginning the procedure.
NOTE: It would be preferable to place the mouse on a sterile heating pad during surgery.
- Remove hair from mice by shaving or use a hair removing cream before beginning the procedure.
- Place mouse in a supine position. Clean the lower abdomen with 10% w/w povidone-iodine solution followed by 70% ethanol swabs.
- With a pair of fine forceps, lift an area of skin 2 mm above the preputial gland, about 1-2 cm above the penis sheath, and about 2-3 cm below the bottom of the rib cage.
- Make a midline incision 1cm in length, first through the skin with a scalpel and then through the muscle layer with a scissor (Figure 1).
- Locate the bladder in the body cavity. It is a yellow-light brown spherical organ, located directly under the incision (Figure 1).
- With a pair of fine forceps grip the bladder and lift upward then downward out of the body cavity towards the penis sheath. This will expose the two seminal vesicles which are a pair of white saclike organs and clearly distinct.
- With a cotton swab in each hand, externalize the seminal vesicles, one by one, and pull them out of the body cavity and lay them face down on the outer surface of the abdomen with the bladder in the middle (Figure 2).
- Using the cotton swabs, gently tilt back the seminal vesicles at the point of insertion near the bladder neck, towards the penis sheath, so that the two dorsal prostate lobes are clearly visible. Use wet cotton swabs to avoid tissue damage (Figure 3).
- Agitate the cell suspension with a micropipette before loading in the syringe.
- While placing the seminal vesicles in position with a cotton swab, insert the syringe needle into the dorsal prostatic lobe under the microscope (Figure 4). Slowly inject 20 µl of cell suspension until a bulla formation is identified. A bulging bulla indicates that the injection is correct.
- While retracting the needle, press lightly on the injection site with a cotton swab and hold for a few seconds to prevent leakage.
- Carefully lift the seminal vesicles with cotton swabs and insert them back in to the body cavity one by one followed by the bladder. Avoid 'twirling' the internal organs while performing this procedure.
- After placing the organs back into the body cavity, suture the muscle layer first in an interrupted pattern with absorbable 4-0 chromic catgut sutures followed by the skin closure with non-absorbable 4-0 nylon surgical suture. The skin can also be pulled together and closed with surgical clamps to close the incision completely.
NOTE: Mice have a habit of scratching and biting at their wound, which may lead to re-opening of the wound, hence use of tissue glue is also recommended along with sutures. Image animals immediately to assure that there is equal bioluminescent intensity among all experimental groups. - Return the animals to clean cages and keep them under a warming lamp or heating pad. Monitor the animals constantly until they completely recover from anesthesia and maintain sternal recumbency.
4. Monitoring of Animals
- Monitor the animals regularly until the end of the experiment, according to institutional protocols. If using surgical metal clips, remove after one to two weeks. Duration of the experiment depends on the specific research need.
NOTE: This experiment was conducted for 21 days to examine the successful establishment of the implanted cancer cells in the mouse prostate. - Administer pain medication based on the animal facility's instructions. For example, intra-peritoneal Buprenorphine at a dose of 0.1 mg/kg body weight can be used at the time of the procedure with a second dose after 6 hr and additional doses every 8-12 hr as needed.
- Monitor animal weight, food consumption, skin color and texture, activity and frequency of urination and defecation. Euthanize animals immediately if there is a significant loss of body weight greater than 15%.
- For euthanasia, deliver CO2 from a pressurized tank into an un-crowded cage at a flow rate to displace 10-30% of the chamber or cage volume/minute, allowing CO2 to enter the chamber slowly so that unconsciousness and complete narcotization occur prior to death.
- Maintain CO2 flow for at least one minute after respiratory arrest and leave animals in the chamber for a sufficient time so that death occurs prior to performing a physical method.
- Perform decapitation, cervical dislocation or any other IACUC approved physical method after euthanizing the animals chemically.
NOTE: Significantly reduced body weight often indicates a lethargic condition. Tumor bearing animals should be in good health except for the presence of tumors until the end of the experiment. Euthanasia should be consistent with the AVMA Guidelines on Euthanasia and must be listed in the approved IACUC protocol.
5. Non-invasive Bio-imaging of Animals
- Monitor the animals weekly using a non-invasive imaging technique to track colonization of cancer cells, tumor growth and any distant metastasis.
NOTE: Imaging modalities such as GFP-imaging, Luciferase imaging, X-rays or 3D micro-computed tomography (uCT) etc. can be used based upon the specific research need12-15.
Representative Results
Following orthotopic implantation of PC3M-Luc-C6 cells into the posterior prostatic lobe, mice were weekly imaged by using a live animal bioluminescence imaging system to monitor the colonization of cells and tumor growth over the course of experiment (Figure 5A–B). Quantification of the bioluminescent signal indicated that PC3M-Luc-C6 cells successfully colonized the prostate lobes. Increased bioluminescence is indicative of increased primary tumor growth over the course of the experiment (Figure 5B). Based on the research goal, mice can be monitored weekly non-invasively by radiography, fluorescence or luminescence imaging to monitor tumor growth and any distant metastatic lesions. Other parameters that can be achieved with this model are: changes in body weight and food consumption over the course of experiment; effect of drug treatment on tumor size and weight; quantification of tumor size and weight at the termination of experiment; extraction of DNA/RNA/protein to determine molecular changes occurring inside the primary tumor after the termination of the experiment.
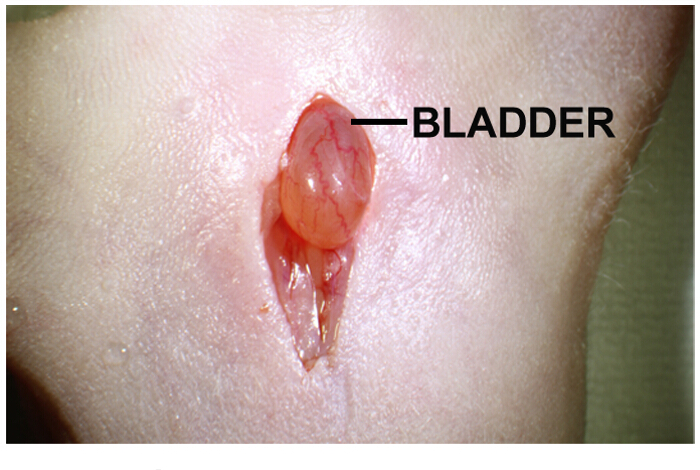
Figure 1: Abdominal midline incision for intra-prostatic implantation of tumor cells. Abdominal midline incision is approximately 1-2 cm long. Urinary bladder is directly under the incision. Gentle pressing on both sides of the incision helps to protrude the urinary bladder. Please click here to view a larger version of this figure.
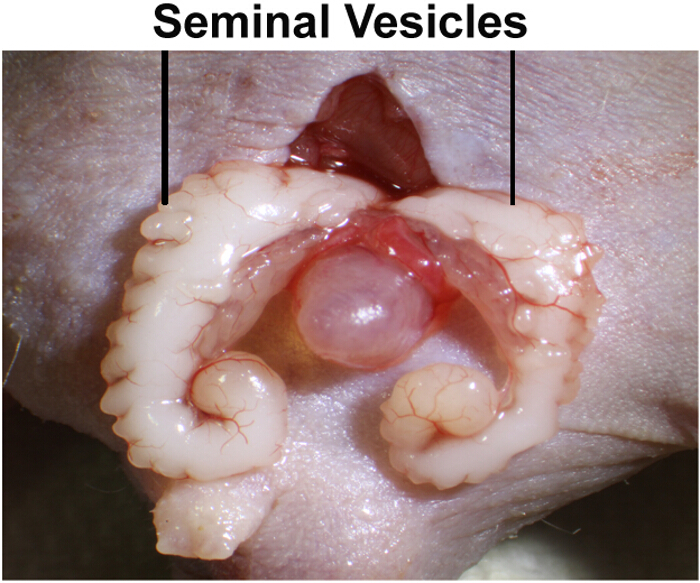
Figure 2: Arrangement of seminal vesicles for intra-prostatic implantation of tumor cells. Seminal vesicles are white sac-like organs and are located directly adjacent to the bladder. Seminal vesicles are exteriorized with cotton swabs and arranged left and right with the bladder in the center. Please click here to view a larger version of this figure.
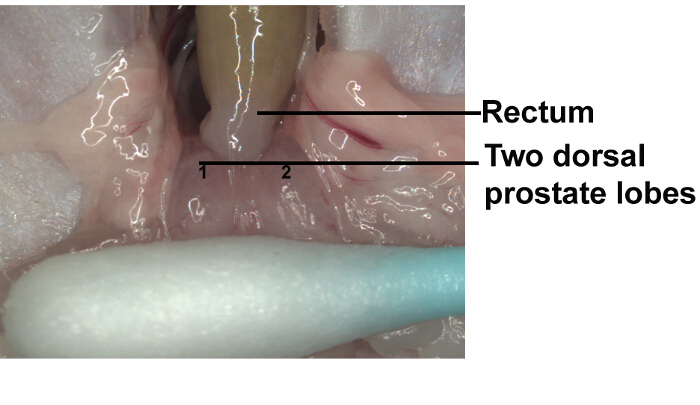
Figure 3: Dorsum of prostate. At the point of insertion, gently tilt back the seminal vesicles towards the penis sheath so that the two dorsal prostate lobes are clearly visible. Use wet cotton swabs to avoid tissue damage. Please click here to view a larger version of this figure.
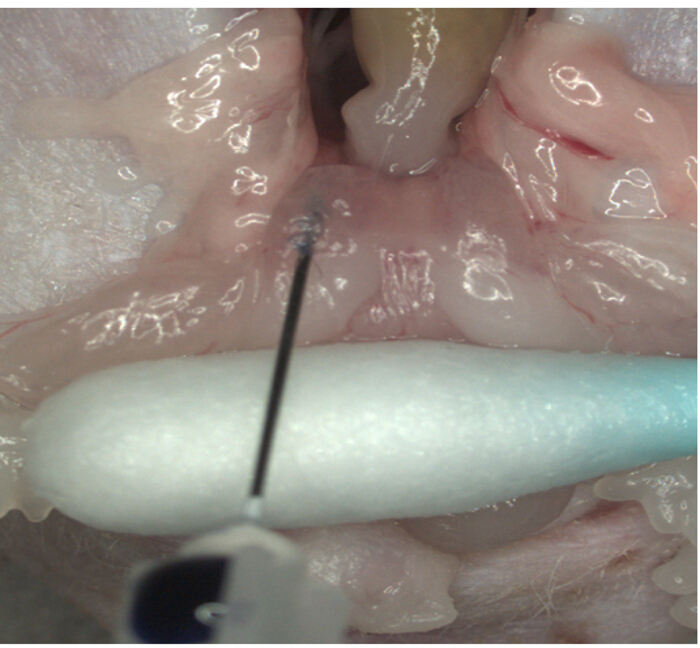
Figure 4: Intra-prostatic implantation of tumor cells. Tumor cells are injected into the dorsal lobe of prostate. Please click here to view a larger version of this figure.
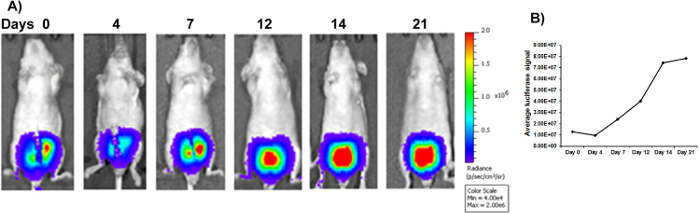
Figure 5: In vivo bioluminescence imaging of intra-prostatic implantation model. (A) In vivo bioluminescence images of mice over the experimental time course after luciferase labeled PC3M-Luc-C6 cells were implanted into the dorsal prostatic lobe of nude mice. (B) Quantification of the bioluminescence signal shows that PC3M-Luc-C6 cells successfully colonized the prostate gland with increased orthotopic tumor growth over the course of the experiment. Please click here to view a larger version of this figure.
Discussion
This manuscript describes a detailed procedure for establishing a human orthotopic prostate cancer xenograft mouse model. This model was established by direct implantation of the human prostate cancer cell line PC3M-Luc-C6 into the dorsal prostatic lobes of immunocompromised mice. Tumors were allowed to develop over the course of the experiment. Tumor growth was monitored weekly by a non-invasive bioluminescence imaging system during the experiment.
The most important factor in establishing xenograft tumor models is to achieve consistency throughout the implantation of tumor cells. To obtain statistically significant results, each experimental group should contain 5-10 mice and tumor size variation should not exceed more than 10% of average tumor size. To achieve this goal, some critical steps within the protocol are important, such as: i) conducting surgery in area that promotes asepsis during surgery; ii) cells should be transplanted as soon as possible after detachment from culture; iii) injection volume should be consistent; iv) careful lifting of internal organs in and out of body cavity during implantation of cells; v) all animals should be injected using the same technique and by one investigator; vi) animals should be randomized into experimental groups after tumor cell implantation.
Some problems that may occur are: i) tumor does not develop at all or tumor nodules develop in the mesentery or body cavity; ii) uneven tumor size is observed among the same experimental group; iii) there may be high surgery-related mortality. These problems can be overcome by taking simple measures such as: i) testing the cell culture for any contamination with mycoplasma etc.; ii) preventing leakage of the tumor cell suspension into the mesentery and abdominal cavity during injection; iii) agitating the cell suspension before each syringe loading; iv) proper anesthesia dosage should be followed and heating pads should be used to maintain body temperature during the procedure.
A wide variety of data can be collected utilizing this model depending on a particular research goal including mouse weight, food consumption, tumor size and weight, genetic and molecular changes in the tumor cells that contribute to tumor growth as well as regional lymph node metastasis10,16. Hoffman and his group developed the technique of surgical orthotopic implantation (SOI) and have extensively used this technique to transplant histologically-intact fragments of major types of human cancers including prostate, bladder and kidney cancers in the rodents17. These orthotopic models have advantage over the transgenic or subcutaneous mouse models as they accurately represent the clinical cancer18,19. These models were also used to transplant the tumors taken directly from the patients to the corresponding organ of the immunodeficient rodents. Orthotopic models are also well suited to examine the effects of drug treatment on tumor growth and lymph node metastasis10. They are also useful for examining the effects of altered gene expression ex vivo, and determining its effect on tumor incidence as well as intra-prostatic growth and metastasis20. However, a limitation of orthotopic prostate cancer model is that no such models have been reported to lead to spontaneous metastasis to the bone which is the most frequent site for prostate cancer metastasis12.
Failure to achieve bone metastases may be due to mice dying of urinary obstruction before any bone metastatic lesions can develop, or because the microenvironment of the mouse fails to recapitulate the human microenvironment, thus failing to develop bone metastases12. Nonetheless, this model does recapitulate the early events in the metastatic cascade prior to embolism and entry of tumor cells into the circulation and therefore is a valuable tool to study the primary tumor, early process of metastatic transformation and for preclinical evaluations of new therapeutic strategies10,12.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We thank Dr. Roger Erickson for his support and assistance with the preparation of the manuscript. This work was supported by the National Cancer Institute at the National Institutes of Health through grant numbers RO1CA160079, RO1CA138642, UO1CA184966 and VA funded program project number 1P1 BX001604.
Materials
| PC3 prostate cancer cell line | ATCC | CRL-1435 | |
| Minimum Essential Medium (MEM) | GIBCO,Life Technology | 11095-080 | |
| PBS | GIBCO,Life Technology | 10010-023 | |
| FBS | GIBCO,Life Technology | 10437-028 | |
| Zeocin | Invitrogen,Life Technology | R250-01 | |
| Trypsin | GIBCO,Life Technology | 25300-54 | |
| IVIS | Xenogen-Caliper | ||
| Insulin Syringes (300ul, 28.5g) | Becton Dickinson | 309300 | |
| Mice | Charles River Laboratories, Inc | ||
| Alcohol Swabs | MEDEquip Depot | 326895 BD | |
| PVP Iodine Prep Pad | MEDEquip Depot | C12400PDI | |
| Surgical CatGut Chromic Suture | Demetech | CC224017F0P | |
| Matrigel | Corning | 354248 |
Riferimenti
- Siegel, R. L., Miller, K. D., Jemal, A. Cancer statistics, 2015. CA Cancer J Clin. 65 (1), 5-29 (2015).
- Andrieu, C., et al. Heat shock protein 27 confers resistance to androgen ablation and chemotherapy in prostate cancer cells through eIF4E. Oncogene. 29 (13), 1883-1896 (2010).
- Fusi, A., et al. Treatment options in hormone-refractory metastatic prostate carcinoma. Tumori. 90 (6), 535-546 (2004).
- Hughes, C., Murphy, A., Martin, C., Sheils, O., O’Leary, J. Molecular pathology of prostate cancer. J Clin Pathol. 58 (7), 673-684 (2005).
- Pavese, J., Ogden, I. M., Bergan, R. C. An orthotopic murine model of human prostate cancer metastasis. J Vis Exp. (79), e50873 (2013).
- Pettaway, C. A., et al. Selection of highly metastatic variants of different human prostatic carcinomas using orthotopic implantation in nude mice. Clin Cancer Res. 2 (9), 1627-1636 (1996).
- Rembrink, K., Romijn, J. C., van der Kwast, T. H., Rubben, H., Schroder, F. H. Orthotopic implantation of human prostate cancer cell lines: a clinically relevant animal model for metastatic prostate cancer. Prostate. 31 (3), 168-174 (1997).
- Kim, S. J., et al. Blockade of epidermal growth factor receptor signaling in tumor cells and tumor-associated endothelial cells for therapy of androgen-independent human prostate cancer growing in the bone of nude mice. Clin Cancer Res. 9 (3), 1200-1210 (2003).
- Kim, S. J., et al. Targeting platelet-derived growth factor receptor on endothelial cells of multidrug-resistant prostate cancer. J Natl Cancer Inst. 98 (11), 783-793 (2006).
- Park, S. I., et al. Targeting SRC family kinases inhibits growth and lymph node metastases of prostate cancer in an orthotopic nude mouse model. Cancer Res. 68 (9), 3323-3333 (2008).
- Zhang, J., et al. AFAP-110 is overexpressed in prostate cancer and contributes to tumorigenic growth by regulating focal contacts. J Clin Invest. 117 (10), 2962-2973 (2007).
- Park, S. I., Kim, S. J., McCauley, L. K., Gallick, G. E. Pre-clinical mouse models of human prostate cancer and their utility in drug discovery. Curr Protoc Pharmacol. Chapter 14, Unit 14.15 (2010).
- Johnson, L. C., et al. Longitudinal live animal micro-CT allows for quantitative analysis of tumor-induced bone destruction. Bone. 48 (1), 141-151 (2011).
- Steinbauer, M., et al. GFP-transfected tumor cells are useful in examining early metastasis in vivo, but immune reaction precludes long-term tumor development studies in immunocompetent mice. Clin Exp Metastasis. 20 (2), 135-141 (2003).
- Yang, M., et al. A fluorescent orthotopic bone metastasis model of human prostate cancer. Cancer Res. 59 (4), 781-786 (1999).
- Stephenson, R. A., et al. Metastatic model for human prostate cancer using orthotopic implantation in nude mice. J Natl Cancer Inst. 84 (12), 951-957 (1992).
- Hoffman, R. M. Orthotopic metastatic mouse models for anticancer drug discovery and evaluation: a bridge to the clinic. Invest New Drugs. 17 (4), 343-359 (1999).
- Wang, X., An, Z., Geller, J., Hoffman, R. M. High-malignancy orthotopic nude mouse model of human prostate cancer LNCaP. Prostate. 39 (3), 182-186 (1999).
- An, Z., Wang, X., Geller, J., Moossa, A. R., Hoffman, R. M. Surgical orthotopic implantation allows high lung and lymph node metastatic expression of human prostate carcinoma cell line PC-3 in nude mice. Prostate. 34 (3), 169-174 (1998).
- Kim, S. J., et al. Reduced c-Met expression by an adenovirus expressing a c-Met ribozyme inhibits tumorigenic growth and lymph node metastases of PC3-LN4 prostate tumor cells in an orthotopic nude mouse model. Clin Cancer Res. 9 (14), 5161-5170 (2003).

