Neonatal Cardiac Scaffolds: Novel Matrices for Regenerative Studies
Summary
In these studies, we provide methodology for novel, neonatal, murine cardiac scaffolds for use in regenerative studies.
Abstract
The only definitive therapy for end stage heart failure is orthotopic heart transplantation. Each year, it is estimated that more than 100,000 donor hearts are needed for cardiac transplantation procedures in the United States1-2. Due to the limited numbers of donors, only approximately 2,400 transplants are performed each year in the U.S.2. Numerous approaches, from cell therapy studies to implantation of mechanical assist devices, have been undertaken, either alone or in combination, in an attempt to coax the heart to repair itself or to rest the failing heart3. In spite of these efforts, ventricular assist devices are still largely used for the purpose of bridging to transplantation and the utility of cell therapies, while they hold some curative promise, is still limited to clinical trials. Additionally, direct xenotransplantation has been attempted but success has been limited due to immune rejection. Clearly, another strategy is required to produce additional organs for transplantation and, ideally, these organs would be autologous so as to avoid the complications associated with rejection and lifetime immunosuppression. Decellularization is a process of removing resident cells from tissues to expose the native extracellular matrix (ECM) or scaffold. Perfusion decellularization offers complete preservation of the three dimensional structure of the tissue, while leaving the bulk of the mechanical properties of the tissue intact4. These scaffolds can be utilized for repopulation with healthy cells to generate research models and, possibly, much needed organs for transplantation. We have exposed the scaffolds from neonatal mice (P3), known to retain remarkable cardiac regenerative capabilities,5-8 to detergent mediated decellularization and we repopulated these scaffolds with murine cardiac cells. These studies support the feasibility of engineering a neonatal heart construct. They further allow for the investigation as to whether the ECM of early postnatal hearts may harbor cues that will result in improved recellularization strategies.
Introduction
Heart failure is common and deadly. It is a progressive disease that results in decreased contractility of the heart, which impairs blood flow to organs and leaves the metabolic demands of the body unmet. It is estimated that 5.7 million Americans have heart failure and it is the primary cause of hospitalization in the United States9. The collective cost of treating patients in heart failure in the United States exceeds $300 billion dollars per year 9-10. The only definitive therapy for end stage heart failure is orthotopic heart transplantation. Each year, it is estimated that more than 100,000 donor hearts are needed for cardiac transplantation procedures in the United States1-2. Due to the limited numbers of donors, only approximately 2,400 transplants are performed each year in the U.S.2. Clearly, this organ shortage needs to be addressed as other strategies are required to produce additional organs for transplantation and, ideally, these organs would be autologous so as to avoid the complications associated with rejection and lifetime immunosuppression.
Mammalian adult cardiomyocytes demonstrate a limited regenerative capacity upon injury but recent evidence suggests that mammalian neonatal hearts maintain a remarkable regenerative capacity following injury5-8. Specifically, following partial surgical resection, a regenerative window has been discovered between birth and postnatal day 7. This regenerative period is characterized by a lack of fibrotic scar, formation of neovascularization, release of angiogenic factors from the epicardium, and cardiomyocyte proliferation 5-8,11. This regenerative window of time provides the potential for using the neonatal heart as a novel source of material for the development of a bioartificial heart.
The extracellular matrix is known to provide important cues to promote cardiomyocyte proliferation and growth. Distinct differences in the availability of molecules in the neonatal and adult matrices12 and their ability to promote regeneration have been explored13. Decellularized adult matrices have been used in several studies to provide an ECM scaffold for cellular repopulation and the generation of a bioartificial heart. While these studies, and new discoveries in stem cell technologies, are advancing rapidly, several hurdles have yet to be met. For example, limitations in preserving native structure of the matrix, cellular integration into the matrix wall, and ability to support proliferation and growth all limit the success of this approach. While superior regenerative attributes have been ascribed to the neonatal heart, the practical aspects of using such a tissue have limited its exploration.
Based on the demonstrated regenerative capacity of the neonatal heart, we have developed novel matrices by developing a technique of decellularization for the P3 mouse heart. The P3 heart was chosen for these studies as it is within the window of cardiac regeneration as previously determined6 but the heart is large enough to harvest, decellularize and recellularize. The goal of this study is to demonstrate the feasibility of creating a matrix from a neonatal mouse heart. Our studies provide evidence for the feasibility of decellularizing a minute, neonatal heart while maintaining the structural and proteinaceous integrity of the ECM. We also demonstrate the ability to recellularize this cardiac ECM with mCherry expressing cardiomyocytes and we have examined these cardiomyocytes for expression of various cardiac markers following recellularization. This technology will allow for the testing of the superiority of a neonatal matrix for the development of a bioartificial heart.
Protocol
All mouse experiments were performed in accordance with US Animal Welfare Act and were approved by the Institutional Animal Care and Use Committee at the University of Minnesota.
1. Method for Mouse Heart Isolation
- Euthanize a neonatal mouse by decapitation with a single use blade.
- Swab the thorax with 70% ethanol.
- Dissect the skin from the chest by cutting it away from the chest wall with standard scissors while pulling the skin laterally with a pair of #5 forceps.
- Perforate the abdomen just inferior to the sternum with the scissors by cutting through the abdominal wall. Grasping the xiphoid process with #5 forceps, retract the sternum rostrally from the body while cutting though the ribs on either side of the chest with the scissors. The rib cage is reflected superiorly with the forceps to reveal the heart.
- Bluntly dissect the two main lobes of the thymus by pulling laterally with #5 forceps, exposing the arch of the aorta, as well as the caval and pulmonary veins.
- Transect the major arteries from the aortic arch, and the aorta itself, with the 10 cm spring scissors. Retain the aorta between the base of the heart and the brachiocephalic artery. Grasp the ends of the severed vessels with the #5 forceps to reflect the heart forward, separating it from the trachea and esophagus.
- Transect the pulmonary vasculature and other major veins, by cutting between the lungs and the heart on both sides of the heart with the 10 cm spring scissors. The veins remain open to provide drainage.
- Remove the heart from the mediastinum with the #5 forceps, grasping the severed ends of the major vessels. Place the heart in a 60 mm culture dish containing sterile phosphate buffered saline (PBS) for catheterization.
2. Method for Decellularization by Langendorff Perfusion
- Prepare the catheter assembly in advance. Draw a 4 cm section of PE 50 tubing in a small alcohol flame to create a smaller, thinner catheter. Trim the ends with a blade to meet the dimensions (Approx. 300 µm outside diameter O.D. with a flange of approx. 500 µm O.D.) shown in Figure 1. This will provide two symmetrical catheters from each side of the pull.
- Point the cut end of the tubing into the flame briefly, to melt a flange onto the opening at the thin end.
- Fill the 12 ml syringe with PBS and assemble the catheter by placing a 3-way stopcock on the syringe. Add a 22 G x 1 needle to the stopcock and drive the needle through the septum. Slide the drawn PE tubing catheter onto the needle (Fig. 1).
- Irrigate the assembled parts with PBS, assuring that all air bubbles have been removed.
- Insert the catheter, prepared as outlined above, into the aorta of the isolated heart, not extending past the aortic valve and ligate with one tie of 7-0 suture. Rest the flange of the catheter proximally against the tie, making a tight seal and preventing the heart from coming off the catheter.
- Observe the catheterization under magnification, while gently perfusing the heart using the syringe containing PBS. Ensure that there are no leaks in the system and the tissue homogeneously blanches as latent blood is removed.
- Place the septum into the neck of the inlet adaptor and seal it by folding the sides over the glass.
- Attach the reservoir filled with 60 ml of 1% sodium dodecyl sulfate (SDS) in distilled water (dH2O) via a line of sufficient length to produce a column that generates 20 mm Hg pressure as shown in Fig. 1. Calculate the pressure from the liquid column height based on the relationship of 1 mm Hg is equal to 1.3595 cm H2O.
Caution: SDS is a highly flocculent powder with irritant properties. Contact should be avoided. Handle the powder using appropriate protective clothing. - Perfuse the heart with 1% SDS for 14 hr. The heart will be translucent in appearance with no observable remaining tissue.
- Flush the system up to the stopcock of the remaining SDS solution and replace it with 10 ml dH2O. Perfuse the heart with 10 ml 1% Triton X-100 (diluted in distilled water), followed by 10 ml dH2O, followed by 60 ml PBS containing 1x penicillin streptomycin (Pen-Strep).
- Store the heart in PBS with 1x Pen-Strep at 4 °C. If the intended post decellularization application requires perfusion then the catheter in the aorta should be maintained, otherwise cut the suture tie and remove the catheter.
3. DNA Determination
- Prepare a digest of the decellularized ECM or control heart using 200 μg/ml proteinase K in 50 mM KCl, 2.5 mM MgCl2, 0.45% Tween 20 in 10 mM Tris (pH 8.3).
- Incubate the tissue at 55 °C with agitation until the tissue is dissolved, typically 4-5 hr.
- Quantitate the DNA content of the homogenate with a DNA binding assay4,11.
4. Fixation and Sectioning of Tissue
- Fix decellularized, recellularized or control P3 hearts in 4% paraformaldehyde in PBS for 30 min at room temperature.
- Wash the tissue in PBS 3x.
- Place the tissue in a solution of 7.5% sucrose in 0.1 M phosphate buffer at 4 °C until it sinks.
- Change the solution to 15% sucrose in 0.1 M phosphate buffer at 4 °C, again, until it sinks.
- Warm the tissue to 37 °C, replace half of the volume with gelatin solution in 15% sucrose & 0.1 M phosphate buffer and equilibrate overnight. Concentrations of gelatin are increased stepwise through 1%, 2.5%, 5% and finally 7.5% twice.
- Place the samples in cryomolds using fresh 7.5% gelatin. To restore the shape of the decellularized samples, liquid gelatin can be perfused into the chambers as the hearts are molded. Freeze by floating the cryomolds on liquid nitrogen. Store at -80 °C.
- Cut (10 µm thick) sections with a cryostat. Bring the slide close to the section surface and observe the section move off from the knife onto the surface due to the charge on the electrostatically treated slides. Dry the slides overnight and store at -80 °C for future analysis.
- Remove the slides from the freezer, equilibrate to room temperature and then place in a Coplin jar containing PBS for 20 min at 37 °C to dissolve the gelatin.
- Stain the cut sections from Step 4.7 with Hematoxylin and Eosin. Place slides in a Coplin jar. Expose the slides hydrated in Step 4.8 to Hematoxylin for 45 sec, tap water for 1.5 min, buffer for 3 min, tap water for 1 min, bluing agent for 1.5 min, 80% ethanol for 1 min, Alcoholic Eosin Y for 8 sec, 80% ethanol for 1 min, 100% ethanol for 1 min 2x, clearing agent (xylene substitute) for 1 min 3x, coverslip with resin based mounting medium. Examine the slides microscopically.
- To confirm the retention of ECM protein reactivity of the heart ECM, stain for ECM structural proteins such as collagen IV by placing the hydrated slides in a humidified chamber and treat with 10% normal donkey serum (NDS) in phosphate buffered saline with 0.1% Triton-X100 (PBST) for 1 hr at room temperature (RT). Use a sufficient volume to cover the tissue sections.
- Replace the solution with the primary antibody of choice at an empirically determined dilution in 5% NDS / PBST. For example, the Collagen IV antibody was diluted at 1:150. Incubate overnight at 4 °C.
- Wash the slides with PBST 3x and apply a secondary antibody of choice conjugated with a fluorescent dye. Dilute the antibody by an empirically determined amount. Incubate for 1 hr at RT. Wash with PBS 3x.
- Stain the slides with a DNA binding dye such as DAPI to verify absence of cell nuclei by using a DAPI containing mounting medium when cover slipping the slides.
- Examine the slides with a fluorescent microscope at 50 to 400X.
5. P1 Neonatal Murine Ventricular Cardiomyocytes for Recellularization
- Spray each pup with 70% ethanol and decapitate with a single use blade.
- Hold each pup between the thumb and forefinger while the thorax is split at the midline with small sterile scissors to expose the thoracic cavity. Apply pressure to allow the heart to protrude freely from the chest while it is cut free of the major vessels and the atria leaving only the ventricular tissue.
- Place each heart directly into a 50 ml tube containing 20 ml ice-cold Calcium, Bicarbonate free Hank's buffered saline solution with 20 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (CBFHH) until all the hearts are collected.
- Aspirate the CBFHH and add 10-15 ml of fresh CBFHH (ice-cold) to the tube. Wash the tissue by swirling the tissue in the tube several times.
- Decant the solution containing the hearts into a 60 mm plastic Petri dish.
- Dissect any latent atrial tissue from the hearts under magnification on ice by trimming it away from the hearts with the spring scissors. Mince up the ventricles into homogenously sized small pieces. Remove any separated non-ventricular tissue from the dish with #5 forceps.
- Pipet as much of the CBFHH solution as is needed to transfer the tissue fragments into a small sterile vial that contains a sterile micro stir bar.
- Allow the tissue to sediment to the bottom of the vial and remove the CBFHH solution.
- Make up 50 ml of an enzyme solution 1.75 mg/ml trypsin, 20 µg/ml DNase II in CBFHH. Pipet 5 ml of this enzyme solution and add it to a tube containing minced ventricular tissue and place on a magnetic stirrer. Stir at a constant rate (340 rpm) at room temperature for 8 min.
NOTE: The stirring action should be gentle and yet allow for the suspension of the slurry. - Remove the vial from the stir plate and allow the slurry to sediment for 3 min. Pipet and discard the initial digest supernatant, as it primarily contains blood cells and tissue debris.
- Pipet a second 5 ml aliquot of the enzyme solution and add it to the digesting ventricle. Stir for 8 min and allow it to sediment for another 3 min. Prior to beginning the digests, prepare two 50 ml tubes (labeled 1 and 2) each containing 12 ml ice-cold fetal bovine serum (FBS). Place a 40 µm cell strainer atop each tube. Pipet this supernatant through the filter on tube 1.
- Repeat the digestion process an additional 8 times, alternating the placement of the digest supernatant between the two tubes containing FBS. Split the last supernatant equally between tubes 1 and 2 to ensure equal volumes in each tube.
NOTE: Each collection tube will now contain 32.5 ml total volume of cell suspension. - Centrifuge the tubes containing the supernatant 150 x g for 6 min at 4 °C.
- While collecting the digestion aliquots, prepare five 100 mm tissue culture plates with 6 ml of media (Dulbecco's Modified Eagle's Media (DMEM) with 10% FBS, 1x Pen-Strep, 1x L-glutamine) per plate. Incubate these aliquots at 37 °C, 5% CO2 for 30 min to equilibrate the media.
- Aspirate the CBFHH enzyme mixture and resuspend the cells into the 30 ml of culture media prepared above, combining the cells from both collection tubes.
- Return 6 ml of cell suspension to each of the 100 mm plates and incubate for 45 min at 37 °C.
- At the end of the incubation, transfer the non-adherent cells to 5 new dishes and re-incubate for 45 min (the fibroblast fraction will have begun to adhere to the dishes and can be grown out separately if desired).
- At the end of the second round of pre-plating, collect the media and non-adherent cells from the dishes and spin the media at 150 x g for 6 min.
- Draw off excess media to bring the cells to the approximate desired concentration, (4.0 x 105 cells per construct in 100 µl of perfusate). Remove an aliquot for counting and or viability testing14.
6. Bioreactor Recellularization of P3 Heart Matrix
- Pretreat the isolated heart matrix with culture media (see Step 5.15) overnight before adding the cells.
- Assemble the elements of a Langendorff system as shown in Fig. 2. Autoclave the glass parts and ethylene oxide sterilize the plastic parts to assure sterility. Various sub units of the system can be assembled in a laminar flow hood before being mounted on the support stand. Circulating water bath and heated water flow path has been omitted for clarity.
- Fill the assembled system with 120 ml of tissue culture media (see Step 5.15).
- Place the catheterized heart matrix from Step 2.14 in a 60 mm culture dish under magnification and connect a 1 ml syringe with 22 G x 1 needle loaded with cell suspension from Step 5.20 to the catheter. Ensure that this assembly is free of air bubbles to avoid embolizing the heart.
- Gently perfuse the 100 µl of cell suspension into the heart matrix via the coronary arteries. Once complete, detach the syringe/needle combination.
NOTE: A slow perfusion of approx. 20 µl per min is required to prevent the cells from flowing out of the veins, transiting the heart. - With a 22g blunt stub secured to the Luer fitting on the underside of the bioreactor heart jar lid, push the catheter onto the stub.
- Start the peristaltic pump and observe that the flow proceeds as outlined in Figure 2. Flow proceeds from the reservoir via the pump through the bubble trap to the catheter placed in the aorta in Step 2.5 (red path in Fig. 2). Observe the flow of the cardiac circulation out of the veins and drip from the apex, recirculating to the media reservoir.
NOTE: The perfusion flow rate is dependent on individual factors such as the vascular resistance of the construct, and the size of the heart, but a rate of 50 to 100 µl/min is a good starting point. At intermediate time points, functional assessments can be executed. The size scale of the neonatal recellularized heart limits the assessments, that can be performed but we have determined that optically based systems can be used to quantify beating behavior just as they have been used in adult rat hearts.4 The construct can be perfused for extended periods of time (up to 23 hr).
Representative Results
Decellularization
On average, the time to decellularization of a P3 heart using this protocol is approximately 14 hr. given an average heart weight of 23 mg for the P3 neonate.
Acellularity
Figure 3a demonstrates a fully intact P3 neonatal heart (whole mount). Figure 3b shows the same heart following decellularization. Figures 4a and 4b show the hematoxylin and eosin staining of intact and decellularized hearts, respectively. Note the absence of hematoxylin positive nuclei and the diminution of eosinophilic structures in the decellularized heart. In addition, the DNA content of the decellularized heart is significantly reduced from 68.08 ± 2.25 µg in the intact heart (n=6) to 4.73 ± 2.27 µg in the decellularized heart (n=5).
Collagen Immunoreactivity
The maintenance of the extracellular matrix (ECM) following decellularization is essential to repopulation with exogenous cells and to the functionality of the matrix. To evaluate the content of the neonatal ECM in intact and decellularized ECM, immuno-staining for collagen IV was performed. Figure 4c and d demonstrate that collagen IV is robustly expressed in both the intact and decellularized heart and that the localization of this protein is maintained following cell removal, while DAPI positive nuclei are effectively removed (Figure 4e-h).
DNA Content
The presence of DNA is used as an additional indication of cellularity. In Figure 5, DNA is demonstrated to be decreased by 93% in neonatal hearts following detergent based decellularization. This degree of DNA reduction is consistent with reports in the literature using detergent based decellularization in other tissues15-16.
Recellularization
We have performed immunohistochemistry to determine the expression of various cardiomyocyte markers in the recellularized heart. Figure 6 illustrates cells that have migrated into the wall of the left ventricle (Figure 6A and 6B). Figure 6C demonstrates the DAPI labeling of the recellularized heart. Figure 6D-G illustrates cells that are positive for NKX 2.5, mCherry, α-actinin and DAPI, respectively. NKX2.5 is known to label cardiac progenitor cells, whereas α-actinin is a sarcomeric protein that marks differentiated cardiomyocytes. We have observed a majority of cells which express all of these markers (pink; Figure 6H), indicating that these cells continue to express cardiomyocyte markers even after 23 days of perfusion.

Figure 1. Schematic of decellularization hardware. A. 60 cc syringe barrel reservoir for detergent solution. B. Catheter assembly with irrigation syringe as detailed. C. Detail of the drawn PE tubing catheter tip. D. Decellularization chamber and septum with drain. Please click here to view a larger version of this figure.

Figure 2. Schematic representation of the heart bioreactor. 1. Humidification of carbogen gas (Green lines represent gas flow). 2. Peristaltic pump drive for media perfusion and oxygenation (Purple lines represent media flow through oxygenator). 3. Thin wall oxygenator. 4. Sheet oxygenator and media reservoir. 5. Preload chamber and bubble trap. 6. Heart chamber (Red lines represent media flow to and from the heart). Please click here to view a larger version of this figure.
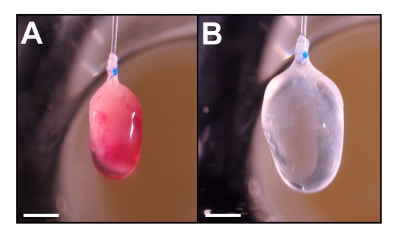
Figure 3. Whole mount of P3 neonatal mouse heart before (A) and after decellularization (B). This heart is decellularized using the Langendorff perfusion method as described in Method 2. Note that the heart becomes translucent and slightly enlarged following perfusion (B). Scale = 2mm. Please click here to view a larger version of this figure.

Figure 4. Histology of native and decellularized P3 neonatal mouse heart. Cryostat section (10 µm) were stained with H&E (A, B), Collagen IV (C, D, G, H) and DAPI (E, F, G, H). Merged images are represented in G and H. H&E staining shows an absence of cell nuclei and cytoplasm in decellularized tissue (B) when compared to the native heart (A). While the Collagen IV content remains following decellularization (D), DAPI staining (a marker of nuclei) is abolished. The merged images demonstrate the colocalization of Collagen IV and DAPI in the naïve heart (G) and the absence of this colocalization in the decellularized heart (H). These data indicate that cells no longer populate the collagen matrix of the heart. Scale = 500 µm. Please click here to view a larger version of this figure.

Figure 5. Evaluation of DNA content. Control (n=6) and decellularized (n=5) hearts assayed for DNA content by the pico-green method. Quantitation expressed as µg of DNA per heart ± standard deviation. Asterisk indicates p<0.01 compared to control. These data indicate that decellularization reduces DNA content significantly in the P3 neonatal heart. Please click here to view a larger version of this figure.

Figure 6. Histology of P3 heart matrix 23 days following recellularization with P1 mCherry expressing cardiomyocytes. Stained with H&E (A, scale = 250 µm, B, scale = 50 µm), DAPI (C, scale = 250 µm), NKX 2.5, mCherry, α-actinin, DAPI, and merged (D, E, F, G, H, scale = 50 µm). We have demonstrated the effective repopulation of the collagen matrix with P1 cardiomyocytes (A-C). Additionally, we observed that m-cherry positive cardiomyocytes express Nkx2.5 and α-actinin 23 days following introduction into the collagen matrix. These data indicate that these cells maintain their cardiomyocyte identity for extended periods of time. Please click here to view a larger version of this figure.
Discussion
The dependence of this technique on repeated perfusions of the heart makes the avoidance of an embolism a critical component of a successful outcome. From the initial catheterization of the heart in Steps 2.2-2.6, to the changes of solution between Steps 2.8-2.14, there are manipulations that can allow introduction of air bubbles which compromise the flow of perfusate into the myocardium. Due to the diminutive size of the neonatal heart, even minute bubbles in the vasculature can cause a technical infarct, thus rendering the decellularization incomplete. Additionally, in the later wash steps, incomplete perfusion can result in detergent residue that negatively impacts biocompatibility. Moreover, rapid changes in temperature, such as when removing the matrix from 4 °C storage as suggested in Step 2.14, should be approached with care as this can also be a source of air bubble formation when dissolved gas moves out of solution with the change in temperature. When proceeding to recellularization, additional care should be applied, when preparing the syringe with the cell suspension, to ensure the infusion is bubble free (Step 6.4).
It may be necessary to manipulate the specifics of this protocol to accommodate other tissues types. There are a number of decellularization protocols reported4,9,11 which could provide guidance. The end goal(s) of any decellularization protocol should include the maintenance of the ECM protein structure and related biochemistry, and adequate removal of native cellular components as exemplified by residual DNA. In this setting, the application of excessive perfusion pressure leads to disruption of the ECM ultrastructure, which can be visualized histologically.
The size of the P3 mouse heart puts some constraints on cell administration compared to adult tissue. While adult hearts present a thick enough ventricular wall that makes transmural injection a possible cell delivery modality, the P3 heart is small enough that a needle, the size of which will not lyse a cell suspension, does substantial damage to the heart. Perfusion is a viable delivery strategy, but depends on cells of a certain size and shape to be delivered effectively. Neonatal murine myocytes work well in this regard. Other small circular cells have also been shown to serve this purpose11. The size scale of these hearts precludes the use of conventional physiologic functional analysis such as pressure volume catheters. Other approaches based on video capture may have to be considered.
These data support the feasibility of decellularization and recellularization of the neonatal mouse heart. Previous studies have demonstrated the use of adult hearts for the purpose of decellularization/recellularization studies. The matrices produced from these adult hearts have been repopulated with neonatal cardiomyocytes4 and human induced pluripotent stem cells (hiPSCs)17. In the case of the hiPSCs, the repopulated hearts displayed a decrease in pluripotency markers such as NANOG, SOX2, and OCT4 and formed muscle-like structures; all suggestive of maturation. The extracellular cues of the ECM, however, have been shown to play a critical role in the development of cells, tissues, and organs which prompted us to attempt to generate matrices from tissues known to harbor regenerative capacity. In our studies, we demonstrate that recellularized hearts still express cardiomyocyte markers, even after prolonged culture. These data indicate that, using novel matrices, cardiomyocytes can be maintained for extended periods of time without losing their identity as cardiomyocytes. Our data support the technique and feasibility for decellularizing neonatal mouse heart. The neonatal matrices hold the potential for providing novel constructs for repopulation studies as well as for the production of gels that have superior regenerative capacity. Using these neonatal ECMs, we are now addressing the superiority of these scaffolds to form functional tissues with a variety of cell types, including hiPSCs.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
The authors gratefully acknowledge Ms. Cynthia DeKay for the preparation of the figures.
Materials
| 1. Materials For Mouse Heart Isolation | |||
| P1 mouse pups (as shown; B6;D2-Tg(Myh6*-mCherry)2Mik/J) | Jackson Laboratories | 21577 | or equivalent |
| 60 mm Culture dish | BD Falcon | 353004 | or equivalent |
| Phosphate buffered saline pH 7.4 (sterile) | Hyclone | SH30256.01 | or equivalent |
| Single Use Blade | Stanley | 28-510 | or equivalent |
| Standard Scissors | Moria Bonn (Fine Science Tools) | 14381-43 | or equivalent |
| Spring Scissors 10 cm | Fine Science Tools | 15024-10 | or equivalent |
| Vannas Spring Scissors – 3mm Cutting Edge | Fine Science Tools | 15000-00 | or equivalent |
| #5 Forceps | Dumnot (Fine Science Tools) | 11295-00 | or equivalent |
| 2. Materials For Decellularization | |||
| Inlet adaptor | Chemglass | CG-1013 | autoclavable |
| Septum | Chemglass | CG-3022-99 | autoclavable |
| 1/8 in. ID x 3/8 OD C-Flex tubing | Cole-Parmer | EW-06422-10 | autoclavable |
| Male luer to 1/8" hose barb adaptor | McMaster-Carr | 51525K33 | autoclavable |
| Female luer to 1/8" hose barb adaptor | McMaster-Carr | 51525K26 | autoclavable |
| Prolene 7-0 surgical suture | Ethicon | 8648G | or equivalent |
| Ring stand | Fisher Scientific | S47807 | or equivalent |
| Clamp | Fisher Scientific | 05-769-6Q | or equivalent |
| Clamp regular holder | Fisher Scientific | 05-754Q | or equivalent |
| 60 cc syringe barrel | Coviden | 1186000777T | or equivalent |
| Beaker | Kimble | 14000250 | or equivalent |
| 22g x 1 Syringe Needle | BD | 305155 | or equivalent |
| 12 cc syringe | Coviden | 8881512878 | or equivalent |
| 3-way stop cock | Smith Medical | MX5311L | or equivalent |
| 22 x 1 g needle | BD | 305155 | or equivalent |
| PE50 tubing | BD Clay Adams Intramedic | 427411 | Must be formable by heat. Polyethylene recommended |
| 1% SDS | Invitrogen | 15525-017 | Ultrapure grade recommended. Make up fresh solution and filter sterilize before use. |
| 1% Triton X-100 | Sigma-Aldrich | T8787 | Make up fresh solution from a 10% stock and filter sterilize before use. |
| Sterile dH2O | Hyclone | SH30538.02 | Or MilliQ system purified water. |
| 1X Pen/Strep | Corning CellGro | 30-001-Cl | or equivalent |
| 3. Materials For DNA Quantitation | |||
| Proteinase K | Fisher | BP1700 | >30U/mg activity |
| KCl | Sigma-Aldrich | P9333 | or equivalent |
| MgCl*6H2O | Mallinckrodt | 5958-04 | or equivalent |
| Tween 20 | Sigma-Aldrich | P1379 | or equivalent |
| Tris base/hydrochloride | Sigma-Aldrich | T1503/T5941 | or equivalent |
| Pico-Green dsDNA assay kit | Life Technologies | P7589 | requires fluorimeter to read |
| 4. Method for fixation and sectioning of tissue. | |||
| Paraformaldehyde | Sigma-Aldrich | P6148 | or equivalent |
| Gelatin Type A from porcine skin | Sigma-Aldrich | G2500 | must be 300 bloom or greater |
| 5. Method for tissue histology | |||
| Cryomolds 10 x 10 x 5mm | Tissue-Tek | 4565 | or equivalent |
| Cryostat | Hacker/Bright | Model OTF | or equivalent |
| Microscope Slides 25 x 75 x 1 mm | Fisher Scientific | 12-550-19 | or equivalent |
| Hematoxylin 560 | Surgipath/Leica Selectech | 3801570 | or equivalent |
| Ethanol | Decon Laboratories | 2701 | or equivalent |
| Define | Surgipath/Leica Selectech | 3803590 | or equivalent |
| Blue buffer | Surgipath/Leica Selectech | 3802915 | or equivalent |
| Alcoholic Eosin Y 515 | Surgipath/Leica Selectech | 3801615 | or equivalent |
| Formula 83 Xylene substitute | CBG Biotech | CH0104B | or equivalent |
| Permount Mounting Medium | Fisher Chemical | SP15-500 | or equivalent |
| Collagen IV Antibody | Rockland | 600-401-106.1 | or equivalent |
| α-Actinin Antibody | Abcam | AB9465 | or equivalent |
| mCherry Antibody | Abcam | AB205402 | or equivalent |
| NKX2.5 Antibody | Santa Cruz Biotechnology | SC-8697 | or equivalent |
| Donkey anti-mouse AF488 Antibody | Life Technology | A21202 | or equivalent |
| Donkey anti-chicken AF594 Antibody | Jackson Immunoresearch | 703-585-155 | or equivalent |
| Donkey anti-goat CY5 Antibody | Jackson Immunoresearch | 705-175-147 | or equivalent |
| Fab Fragment Goat Anti-Rabbit IgG (H+L) AF594 | Jackson Immunoresearch | 111-587-003 | or equivalent |
| Prolong Gold Antifade Mountant with DAPI | ThermoFisher | P36930 | or equivalent |
| 6. Isolation of neonatal ventricular cardiomyocytes using pre-plating. | |||
| HBSS (Ca, Mg Free) | Hyclone | SH30031.02 | or equivalent |
| HEPES (1M) | Corning CellGro | 25-060-Cl | or equivalent |
| Cell Strainer | BD Falcon | 352340 | or equivalent |
| 50 mL tube | BD Falcon | 352070 | or equivalent |
| Primeria 100 mm plates | Corning | 353803 | Primeria surface enhances fibroblast attachment promoting a higher myocyte purity |
| Trypsin | Difco | 215240 | or equivalent |
| DNase II | Sigma-Aldrich | D8764 | or equivalent |
| DMEM (Delbecco's Minimal Essential Media) | Hyclone | SH30022.01 | or equivalent |
| Vitamin B12 | Sigma-Aldrich | V6629 | or equivalent |
| Fibronectin coated plates | BD Bioscience | 354501 | or equivalent |
| Fetal bovine serum | Hyclone | SH30910.03 | or equivalent |
| Heart bioreactor glassware | Radnoti Glass Technology | 120101BEZ | Must be sterilizable by autoclaving or gas. |
Riferimenti
- Yusen, R. D., et al. Registry of the International Society for Heart and Lung Transplantation: Thirty-second official adult lung and heart-lung transplantation report–2015. J Heart Lung Transplant. 34 (10), 1264-1277 (2015).
- Go, A. S., et al. Heart disease and stroke statistics–2014 update: A report from the american heart association. Circulation. 129 (3), e28-e292 (2014).
- Kapelios, C. J., Nanas, J. N., Malliaras, K. Allogeneic cardiosphere-derived cells for myocardial regeneration: current progress and recent results. Future Cardiol. 12 (1), 87-100 (2016).
- Ott, H. C., et al. Perfusion decellularized matrix: Using nature’s platform to engineer a bioartificial heart. Nat Med. 14 (2), 213-221 (2008).
- Porrello, E. R., Olson, E. O. A neonatal blueprint for cardiac regeneration. Stem Cell Research. 13 (3 Pt B), 556-570 (2014).
- Porrello, E. R., et al. Transient regenerative potential of the neonatal mouse heart. Science. 331 (6020), 1078-1080 (2011).
- Polizzotti, B. D., et al. Neuregulin stimulation of cardiomyocyte regeneration in mice and human myocardium reveals a therapeutic window. Sci Transl Med. 7 (281), 281ra45 (2015).
- Jesty, S. A., et al. c-kit+ precursors support postinfarction myogenesis in the neonatal, but not adult, heart. Proc Natl Acad Sci U S A. 109 (33), 13380-13385 (2012).
- Ambrosy, A. P., et al. The Global Health and Economic Burden of Hospitalizations for Heart Failure: Lessons Learned From Hospitalized Heart Failure Registries. J Am Coll Cardiol. 63 (12), 1123-1133 (2014).
- Roger, V. L., et al. Executive Summary: Heart Disease and Stroke Statistics-2012 Update A Report From the American Heart Association. Circulation. 125 (22), 188-197 (2012).
- Kennedy-Lydon, T., Rosenthal, N. Cardiac regeneration: epicardial mediated repair. Proc R Soc B. 282 (1821), 2147-2172 (2015).
- Williams, C., Sullivan, K., Black, L. D. Partially Digested Adult Cardiac Extracellular Matrix Promotes Cardiomyocyte Proliferation In Vitro. Adv Healthcare Mat. 4 (10), 1545-1554 (2015).
- Borg, T. K., et al. Recognition of extracellular matrix components by neonatal and adult cardiac myocytes. Dev Biol. 104 (1), 86-96 (1984).
- Strober, W. Trypan blue exclusion test of cell viability. Curr Protoc Immnol. 111, A3-B1-3 (2015).
- Gilbert, T. W., Freund, J. M., Badylak, S. F. Quantification of DNA in biologic scaffold materials. J Surg Res. 152 (1), 135-139 (2009).
- Akhyari, P., et al. The quest for an optimized protocol for whole-heart decellularization: a comparison of three popular and a novel decellularization technique and their diverse effects on crucial extracellular matrix qualities. Tissue Eng Part C Methods. 17 (9), 915-926 (2011).
- Lu, T. Y., et al. Repopulation of decellularized mouse heart with human induced pluripotent stem cell-derived cardiovascular progenitor cells. Nat Commun. 4 (2307), 1-11 (2013).

