Isolation and Culture of Primary Endothelial Cells from Canine Arteries and Veins
Summary
Novel isolation methods of primary endothelial cells from blood vessels are needed. This protocol describes a new technique that completely inverts blood vessels of interest, exposing only the endothelial side to enzymatic digestion. The resulting pure endothelial cell culture can be used to study cardiovascular diseases, disease modelling, and angiogenesis.
Abstract
Cardiovascular disease is studied in both human and veterinary medicine. Endothelial cells have been used extensively as an in vitro model to study vasculogenesis, (tumor) angiogenesis, and atherosclerosis. The current standard for in vitro research on human endothelial cells (ECs) is the use of Human Umbilical Vein Endothelial Cells (HUVECs) and Human Umbilical Artery Endothelial Cells (HUAECs). For canine endothelial research, only one cell line (CnAOEC) is available, which is derived from canine aortic endothelium. Although currently not completely understood, there is a difference between ECs originating from either arteries or veins. For a more direct approach to in vitro functionality studies on ECs, we describe a new method for isolating Canine Primary Endothelial Cells (CaPECs) from a variety of vessels. This technique reduces the chance of contamination with fast-growing cells such as fibroblasts and smooth muscle cells, a problem that is common in standard isolation methods such as flushing the vessel with enzymatic solutions or mincing the vessel prior to digestion of the tissue containing all cells. The technique we describe was optimized for the canine model, but can easily be utilized in other species such as human.
Introduction
Dogs are used as large animal model for cardiovascular disease research and can also suffer from inborn (genetic) vascular abnormalities1, 2. To study these diseases commercial endothelial cell lines are often used to assess endothelial cell (EC) functionality. For dogs there is one commercial endothelial cell line available (CnAOEC), derived from canine aorta. This cell line is mostly used in studies as control normal ECs3-5. In human cardiovascular research the most commonly used endothelial cell lines are Human Umbilical Vein Endothelial Cells (HUVECs) and Human Umbilical Artery Endothelial Cells (HUAECs) derived from human umbilical cord vein and artery, respectively. HUVECs have been used as the golden standard in vascular research since the 1980s6. They are considered to be the classic model system to study endothelial function and disease adaptation. Endothelial cells isolated from different blood vessels vary in appearance and functionality due to genetic background and exposure to the microenvironment7. In addition, HUVECs and HUAECs are derived from umbilical cord, a developmental vascular structure that might not fully mimic adult blood vessels with respect to the conditions they are exposed to and response to disease. Hence, translating results found in HUVECs and HUAECs to cardiovascular disease in general is inadequate.
When studying adaptation and behavior of adult ECs, primary ECs from the vessel of interest should be used as a more direct approach. To isolate these cells, several methods have been reported. A widely described method, which is also used for HUVECs, is flushing the vessel with an enzymatic digestion solution8. This often results in contamination with non-ECs such as smooth muscle cells and fibroblasts9. Another frequently used method for isolation is enzymatic digestion of minced vessel tissue followed by fluorescence-activated cell sorting (FACS) based on endothelial cell marker Cluster of Differentiation (CD)317, 8. FACS sorting and subsequent cell culture requires relatively large amounts of cells and is therefore not suitable for the isolation of endothelium from small blood vessels. We therefore aimed at developing a new robust method for isolating a pure endothelial cell population from various canine blood vessels with high purity. To test the efficiency of the new isolation method, we isolated and obtained pure Canine Primary Endothelial Cell (CaPEC) cultures from different canine arteries and veins, both large and small. This method also enables the culture of endothelial cells originating from diseased and/or aberrant vessels such as inborn intra- or extra-hepatic portosystemic shunts, a common disease in dogs2. The method allows the isolation of additional relevant cell types of the same vessel such as vascular smooth muscle cells since most of the vessel remains intact during the procedure.
Protocol
Ethics statement: Blood vessels used in this study were harvested as surplus material obtained from fresh canine cadavers (n= 4) from healthy dogs euthanized for other unrelated research (University 3R policy). Aberrant blood vessels (intra- and extrahepatic portosystemic shunts, n= 1 each) were harvested post-mortem after informed consent of the owners from dogs presented to the University Clinic for Companion Animals of Utrecht University.
1. Isolation and Culture of Primary Canine Endothelial Cells
- Pre-coat 6-well plates with 2 ml/well 0.5% w/v gelatin and leave for 2 hr in an incubator with a humidified atmosphere of 5% CO2 at 37 °C. Remove excess gelatin solution prior to seeding the primary cells.
- Aseptically remove the blood vessel(s) of interest (e.g., aorta, vena cava, vena porta) from a fresh canine cadaver (Figure 1A). Maintain the anatomy of the vessel system by placing straight forceps on both ends of the vessel of interest. Avoid unnecessary manipulation of the tissue with forceps to prevent damage to the endothelial cells.
- Cut the clamped vessel on both ends with surgical scissors and remove from the cadaver. Transport in Hank's Balanced Salt Solution (HBSS) on ice.
NOTE: A vessel length of approximately 5 cm is preferred for this procedure, but those that measure 1 cm will also provide enough cells to culture.
- Cut the clamped vessel on both ends with surgical scissors and remove from the cadaver. Transport in Hank's Balanced Salt Solution (HBSS) on ice.
- Transfer the blood vessel to a Petri dish filled with ice-cold HBSS. Using surgical scissors, remove any adherent tissue and fat from the outside of the vessel, keeping the vessel itself intact (Figure 1B). Make sure the vessel is clearly visible at all times when cutting: pull aside the surrounding tissue with a clamp or forceps for optimal view.
- Close any branches of the vessel with ligatures (e.g., polyglactin 3-0) and subsequently remove them with surgical scissors or a scalpel.
- Carefully enter a vessel end with a curved Halsted mosquito forceps, clamp the tip of the forceps at the other end of the vessel and then retract, slowly inverting the vessel until it is completely inside out (Figure 1C-G). The outside now consists of the endothelial cell layer. If any difficulty is encountered upon inverting the vessel, submerge in HBSS again to reduce friction.
- Place purse-string sutures at both ends of the vessel to close it completely and to prevent exposure of any non-endothelial vascular tissue to the digestion procedure (Figure 1H). Use the ligature ends to manipulate the inverted vessel.
- If digestion and culture is performed later, cryopreserve the inverted vessel prior to the digestion protocol (step 1.7).
- Place the inverted vessel in a cryovial and fill with cell culture freezing medium. Freeze down to -80 °C using a freezing container. Store at -80 °C if vessels are to be used within one week. For long term storage, place cryovials at -180 °C. When performing the EC isolation, thaw the cryovials rapidly in a water bath (37 °C) and immediately place in ice-cold HBSS. Proceed as indicated in 1.7.
NOTE: Take into account that the total yield of ECs after isolation will be lower due to loss of viability during the freezing process.
- Place the inverted vessel in a cryovial and fill with cell culture freezing medium. Freeze down to -80 °C using a freezing container. Store at -80 °C if vessels are to be used within one week. For long term storage, place cryovials at -180 °C. When performing the EC isolation, thaw the cryovials rapidly in a water bath (37 °C) and immediately place in ice-cold HBSS. Proceed as indicated in 1.7.
- Transfer the vessel to a 50 ml tube and rinse it twice in HBSS to remove erythrocytes (or residual freezing medium in case of thawing) (Figure 1I).
- Digest the vessel in a 50 ml tube with a solution of 30 ml collagenase type II (0.15 U/ml) and dispase (0.15 U/ml) in Canine Endothelial Cells Growth Medium (CECGM) for 1 hr at 37 °C with intermittent gentle agitation.
- Remove the vessel from the tube and centrifuge the cell suspension for 5 min at 250 x g.
- Resuspend the cell pellet in CECGM and seed 2 ml cell suspension per well (1 – 3 wells depending on vessel size). Culture the cells at 37 °C in a humidified atmosphere with 5% CO2 in air and change the culture medium twice a week.
- Passage the cells when a confluency of 70 – 80% is reached.
- Wash the cells with pre-warmed HBSS to remove dead or non-attached cells. Add 200 µl recombinant cell-dissociation enzyme (1x) to each well and place back in the incubator for 5 min or until all cells are detached.
- Transfer the cells to a 15 ml tube and stop trypsinization by adding 10 ml of culture media (CEGM) including 10% Fetal Calf Serum (FCS). Centrifuge for 5 min at 250 x g. Continue the culture in a gelatin pre-coated plate or flask.
2. Characterization
- Culture Canine Aortic Endothelial Cells (CnAOECs) and compare the morphology of the primary and commercial cell lines twice weekly with a microscope.
NOTE: The typical growing pattern of ECs in patches can best be observed when a confluency of > 70% is reached.- Culture CnAOECs in CECGM on a 0.5% w/v gelatin pre-coated T75 flask. Passage cells once a week when the confluency is 70 – 80%.
- For passaging, wash the cells once with pre-warmed HBSS and add 1 ml recombinant cell-dissociation enzyme (1x). Place the flask back in the incubator for 5 min or until all cells are detached. Inactivate trypsin by adding 10 ml culture medium (CEGM) including 10% FCS. Centrifuge the cell suspension for 5 min at 250 x g, discard the supernatant and resuspend the cells in 1 ml culture medium.
- Take a 10 µl aliquot of the cell suspension and dilute 1:1 with 0.4% trypan blue. Count the cells using an automated cell counter, and plate out 4.0 x 105 viable cells in a new T75 flask. Add 10 ml of pre-warmed CECGM to the flask and culture at 37 °C in a humidified atmosphere with 5% CO2.
- Culture CnAOECs in CECGM on a 0.5% w/v gelatin pre-coated T75 flask. Passage cells once a week when the confluency is 70 – 80%.
- Isolate RNA from passage 1 of the cultured CaPECs and CnAOECs.
- Collect at least 1 x 103 cells as a pellet, add 20 µl of sample preparation reagent (SPR) and incubate for 1 min to lyse the cells. After incubation, carefully collect the cell lysate containing total RNA and store at -70 °C.
- Convert mRNA to cDNA using a cDNA synthesis kit (e.g., iScript) following the instructions of the manufacturer. Store cDNA at 4 °C up to one week, or at -20 °C for long term until ready to perform the qPCR.
- Measure gene expression of the endothelial marker CD31 to confirm endothelial cell origin. Perform experimental setup and quantification using the MIQE précis guidelines10. For normalization, measure expression of reference genes GAPDH, RPS19, and B2MG11.
- Prepare a 10-fold dilution of the obtained cDNA in nuclease free water.
- Prepare a 4-fold dilution from pooled cDNA samples for the standard line and use nuclease free water as a non-template control. Dilute the samples five times to reach a 50-fold total dilution for qPCR reactions. Pipette 10 µl reactions in duplicate in a 384-well format using 4 µl cDNA and 6 µl of the fluorophore mixed with 20 pmol of reverse and forward primer (Table 1).
- Set the program to 95 °C for 5 min for Taq polymerase activation, followed by 39 cycles of 10 sec at 95 °C for denaturation, and 30 sec at Tm for annealing and elongation. The Tm for each primer set is shown in Table 1.
- Perform melting curve analyses following every run to ensure only one product is amplified. Normalize the expression levels of the samples using the average relative amount of the reference genes, and calculate the ΔCt if reaction efficiency is between 95% and 105%.
- Assess EC functionality with an angiogenesis assay.
- Add 10 µl of extracellular matrix to each well of a pre-cooled angiogenesis slide and spread with a pipette tip to cover the surface of the well. Keep the extracellular matrix on ice and avoid the introduction of air bubbles into the gel. Solidify the gel by placing the slide in a Petri dish with water soaked paper towels in an incubator at 37 °C with 5% CO2 for 30 min.
- Add 1.0 x 104 primary ECs in 50 µl Endothelial Growth Medium per well. Incubate the slide for 6 hr at 37 °C with 5% CO2.
- After 6 hr take photographs with a 20X magnification, making sure to include the whole well in the image.
Representative Results
Different blood vessels were successfully subjected to the described isolation protocol (Figure 2). It was possible to dissect and invert aorta, vena cava, vena porta, and coronary artery from healthy dogs (all vessels from each dog, n= 4). With the same approach ECs were isolated from two congenital portosystemic shunts (extrahepatic and intrahepatic, n= 1 each). Although aorta was easily inverted, thoracic aorta segments were more challenging than abdominal aorta. In thoracic segments the aorta has many intercostal arteries branching from it, which need to be ligated individually to ensure strictly endothelial exposure to the digestion solution. The dissected segment of caudal vena cava included the branching point of the renal veins, which needed to be ligated before inversion. For the portal vein the contributing branch of the vena gastroduodenalis was ligated before inversion of the blood vessel. The coronary artery in dogs is a much smaller blood vessel (approximately 1 – 2 mm diameter in a medium sized dog), from which we excised a segment of the circumflex branch. Because it has a small diameter, it proved to be easier to invert a rather short segment of 1 cm because the Mosquito forceps could not be inserted much further.
One day post isolation adhered cells were visible in the culture plate. In culture, CaPECs had a polygonal shape and displayed a tendency to grow in patches as shown in Figure 3. Numerous colonies of endothelial cells could be observed 3 – 6 days after isolation. After approximately 10 days in culture a confluency of 80% was reached and cells could be surpassed. On average the primary endothelial cultures could be maintained for a maximum of 8 passages (once weekly at a split rate of 1:4) at which point they stopped growing.
Isolated endothelial cells expressed endothelial cell marker CD31 as indicated by qPCR (Figure 4). The expression in endothelial cells derived from aorta, vena cava, and vena porta from four dogs was compared with a control culture of CnAOECs. The cultured primary cells had a comparable CD31 expression with the control ECs (Kruskall-Wallis, p=0.856).
CaPECs derived from aorta, vena cava and vena porta showed branching after 6 hr incubation on the angiogenesis slide as shown in Figure 5.
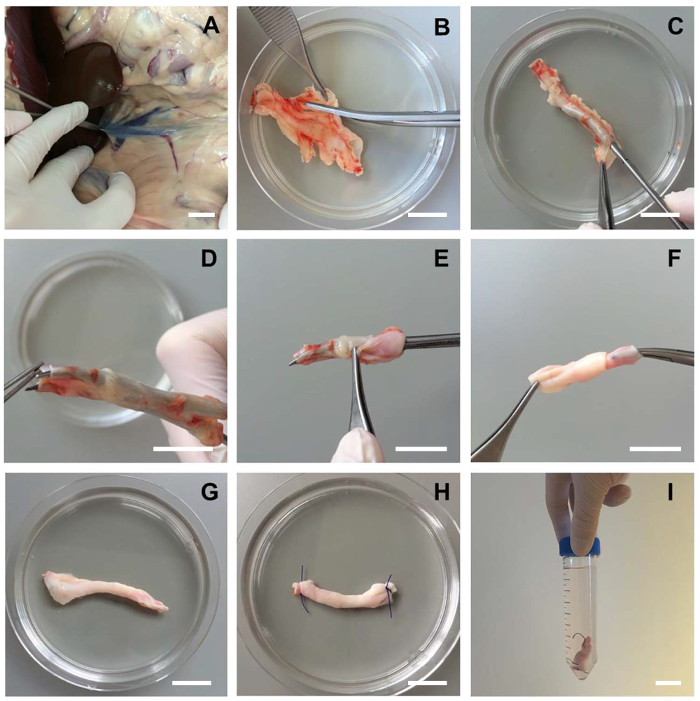
Figure 1: Dissecting and Inverting Blood Vessels for Endothelial Cell Isolation. A) The blood vessel of interest is aseptically removed from a fresh canine cadaver. B) Adherent tissue and/or fat surrounding the vessel should be removed carefully with surgical scissors without damaging the vessel itself (canine vena cava, abdominal segment of 5 cm). C) With a curved Halsted Mosquito forceps, the vessel can be entered without perforating the endothelial layer. D-G) Secure the forceps on the other end of the blood vessel and gently retract, thereby completely inverting the blood vessel. The endothelial layer is now on the outside of the vessel. H) Purse-string sutures are placed at the ends closing off the non-endothelial surface of the inverted vessel. I) The blood vessel is transferred to a 50 ml tube for washing and subsequent digestion. Scale bars indicate 2 cm. Please click here to view a larger version of this figure.
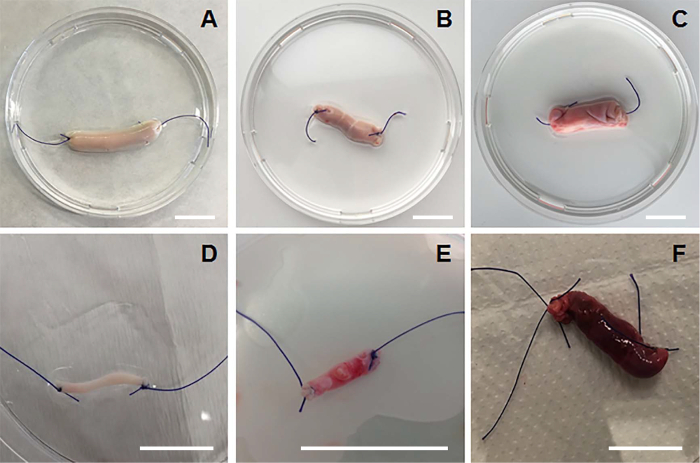
Figure 2: Inverted Vessels of Different Origin. A) An inverted aorta segment (healthy dog). B) An inverted vena porta (healthy dog). C) An inverted vena cava segment (healthy dog). D) An inverted coronary artery segment (healthy dog). E) An inverted extrahepatic portosystemic shunt derived from a Cairn terrier (age: 6 weeks old). F) An inverted intrahepatic portosystemic shunt derived from an Irish wolfhound (age: 8 weeks old). Scale bars indicate 2 cm. Please click here to view a larger version of this figure.

Figure 3: Cell Morphology. Pictures were taken two weeks after digestion of the vessels. A) ECs derived from canine aorta in passage 2. B) ECs derived from canine vena cava in passage 2. C) ECs derived from canine vena porta in passage 2. All pictures are taken with 4X original magnification. Scale bars indicate 500 µm. Insert shows 10X magnification. Please click here to view a larger version of this figure.
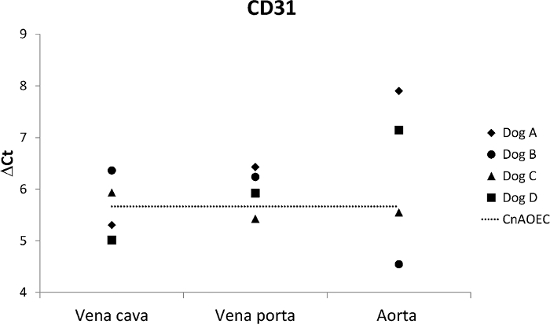
Figure 4: Gene Expression of CD31. Expression of CD31 in endothelial cells in passage 1 derived from aorta, vena cava, and vena porta (n= 4 dogs). No significant expression differences were observed between the CaPECs and the CnAOECs. Please click here to view a larger version of this figure.
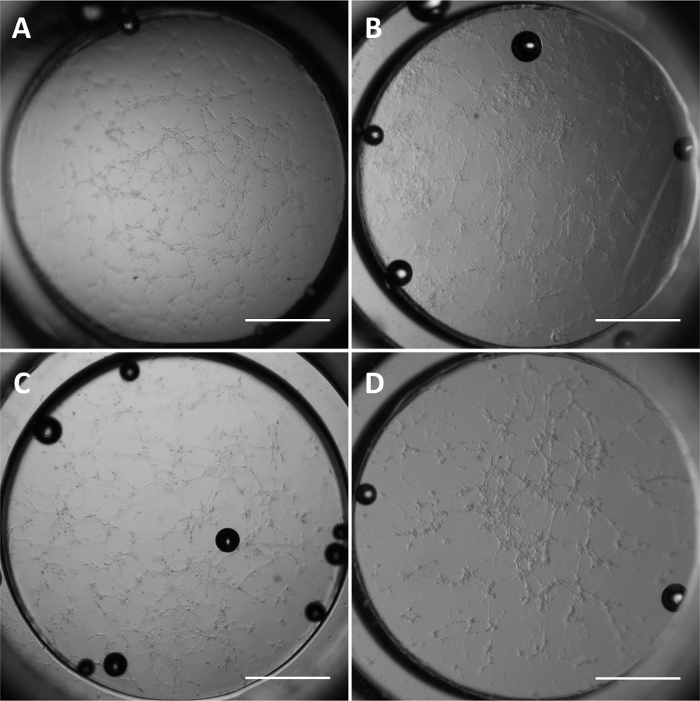
Figure 5: Angiogenesis. Photographs of CnAOECs and CaPECs from aorta, vena cava and vena porta after 6 hr incubation on an angiogenesis slide (20X magnification). Branch formation is visible after 6 hr in culture. A) CnAOECs. B) CaPECs derived from aorta. C) CaPECs derived from vena cava. D) CaPECs derived from vena porta. All cells were in passage 3. Scale bars indicate 1 mm. Please click here to view a larger version of this figure.
| GOI | Direction | 5’-Sequence-3’ | Tm annealing | Product size (bp) | Genebank number | |
| CD31 | Forward | GTTCTGCGTGTCAAGGTG | 59 °C | 85 | XM_005624261.1 | |
| Reverse | TGTCCTTCCCAAACTCCA | |||||
| beta-actin | Forward | GATATCGCTGCGCTTGTGGTC | 58 °C | 384 | NM_001195845 | |
| Reverse | GGCTGGGGTGTTGAAAGTCTC | |||||
| RPS19 | Forward | CCTTCCTCAAAAAGTCTGGG | 63 °C | 95 | XM_005616513 | |
| Reverse | GTTCTCATCGTAGGGAGCAAG | |||||
| B2MG | Forward | TCCTCATCCTCCTCGCT | 63 °C | 85 | AB745507 | |
| Reverse | TTCTCTGCTGGGTGTCG | |||||
Table 1: qPCR Primer Sets. qPCR primers for canine reference genes and CD31.
Discussion
In studies focusing on canine ECs the CnAOEC primary line is used to model the endothelial lineages of the dog3, 12, 13. In human studies, the HUVEC culture is still considered the gold standard. Clearly, focusing merely on ECs derived from umbilical cord is a firm restriction in cardiovascular research. Endothelial cells have a specific gene expression pattern determining arteriovenous specification. In order to account for these differences in postnatal vessels we present this novel isolation method based on the specific anatomical location of the endothelial cells. The methods commonly used for primary EC isolation are flushing the vessel with an enzyme solution or mincing the vessel prior to digestion, two methods that both have a risk of contamination with non-ECs. We established a technique for the isolation of Canine Primary Endothelial Cells (CaPECs) with a lesser chance of contamination that can also be used on small vessels. This isolation method of endothelial cells is based on the inversion of the vessel, which avoids digestion of all other vessel cell types. It is important to remove the vessels aseptically, as illustrated in Figure 1A, to prevent bacterial or fungal contamination of the endothelial cultures. In case of bacterial growth an additional antimicrobial agent (e.g., gentamycin) can be added to the culture medium. In case of fungal infection treatment is often not successful in our experience.
In order for the inversion to succeed, it is critical to obtain a vessel that is approximately 5 cm in length. A second important step to facilitate inversion of the blood vessel is the removal of any adherent tissue and fat with surgical scissors. Inverting the vessel is done carefully by clamping the forceps at the opposite end and slowly inverting the vessel. When placing the purse-string sutures make sure to touch the endothelial layer as little as possible. Damage of the endothelium can result in poor yield of viable endothelial cells and access of the digestion media to underlying tissue. This can also happen if the vessel is not closed completely. The vessel can easily be handled by picking it up at the end of a ligature.
In specific cases a modification of the technique can be applied. In blood vessels with a small diameter it is sometimes impossible to insert a Mosquito forceps in order to invert the vessel. A solution is to place stay sutures on one end of the vessel and push their ligatures through the blood vessel using the forceps. At the other end they can then be secured with a Mosquito forceps and pulled, thereby inverting the vessel. Sometimes the vessel will tear as a consequence of the extra sutures, so this modification is not the preferred approach. Another problem that can arise is when many erythrocytes are present in the culture plates upon seeding of the primary cells. This can be the result of insufficient washing of the blood vessel prior to digestion. When this occurs, washing the wells on day 2 with warm HBSS is often sufficient to remove the majority of the erythrocytes. In any case, the erythrocytes will not persist in the culture and are lost upon passaging of the CaPECs.
The isolated CaPECs expressed CD31, indicating that the population of cells that was digested from the blood vessel is indeed of endothelial origin. As recently published, this marker is also expressed in endothelial cells from the canine mitral valve14. The formation of colonies in culture indicates the cultures start from either single cells or from small cell clusters. Rapid growth on endothelial specific media is also indicative of correct cell type. The primary cells can be cultured for eight passages, after which the cultures cease expansion. This is an indication of senescence and makes it less likely that stem/progenitor cells are cultured with this protocol. In an angiogenesis assay, CaPECs from aorta, vena cava and vena porta showed branching within 6 hr. This endothelial functionality provides solid evidence for their origin.
Based on availability only canine material was studied, but the isolation method has possible applications for isolation of human primary endothelial cells or endothelial cells from other organisms. The technique is also possible for very small vessels (1 cm length), but will yield less isolated cells. For this reason CaPECs obtained from small vessels will require an extra passage before they have reached a sufficient number for experiments.
The ability to perform the isolation method on small vessels is a great advantage for studies in vascular disease. ECs could be isolated from diseased or aberrant vessels like portosystemic shunts. These are vessels connecting the portal vein and the systemic circulation which causes blood to bypass the liver2. The shunt itself can be as small as 1 cm. Comparing the ECs from a shunt vessel with cells originating from a healthy caval vein and portal vein could give new information relevant for the shunt-development since the pathogenesis is still not completely understood. In addition, new angiogenic genes could be investigated on a functional level by performing genetic modifications such as siRNA mediated gene silencing. In conclusion, this novel isolation method can be a powerful model to study (congenital) cardiovascular diseases, disease modelling, and (tumor) angiogenesis15, 16.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
The authors would like to acknowledge Hans de Graaf and Tomas Veenendaal for their technical assistance in culturing the ECs.
Materials
| Collagenase type II | Life Technologies | 17101-015 | |
| Dispase | Life Technologies | 17105-041 | |
| DMEM (1X) + GlutaMAX | Life Technologies | 31966-021 | |
| Hank's Balanced Salt Solution | Life Technologies | 14025-050 | |
| Canine Endothelial Cells Growth Medium | Cell Applications | Cn211-500 | |
| CnAOECs | Cell Applications | Cn304-05 | |
| Fetal Calf Serum (FCS) | GE Healthcare | 16000-044 | |
| TrypLE Express | Life Technologies | 12604-013 | |
| SPR | Bio-Rad | 170-8898 | |
| iScript synthesis kit | Bio-Rad | 170-8891 | |
| SYBR green super mix | Bio-Rad | 170-8886 | |
| Recovery Cell Freezing Medium | Gibco/Life Technologies | 12648-010 | Keep on ice prior to use |
| Freezing container, Nalgene Mr. Frosty | Sigma-Aldrich | C1562 | |
| Gelatin | Sigma-Aldrich | G1890 | |
| Surgical scissors (Mayo or Metzenbaum) | B. Braun Medical | BC555R | |
| Mosquito forceps | B. Braun Medical | FB440R | |
| Mosquito forceps curved | B. Braun Medical | FB441R | |
| polyglactin 3-0 | Ethicon | VCP311H | |
| Trypan blue | Bio-Rad | 145-0013 | |
| Automated counting chamber | Bio-Rad | 145-0102 | |
| Counting Slides, Dual Chamber | Bio-Rad | 145-0011 | |
| Matrigel | BD Biosciences | BD356231 | Slowly thaw on ice |
| µ-Slide Angiogenesis | Ibidi | 81501 | |
| Endothelial Growth Medium | Lonza | CC-3156 | |
| EGM-2 SingleQuot Kit | Lonza | CC-4176 |
Riferimenti
- Haidara, M. A., Assiri, A. S., Yassin, H. Z., Ammar, H. I., Obradovic, M. M., Isenovic, E. R. Heart Failure Models: Traditional and Novel Therapy. Curr. Vasc. Pharmacol. 13 (5), 658-669 (2015).
- van Steenbeek, F. G., van den Bossche, L., Leegwater, P. A., Rothuizen, J. Inherited liver shunts in dogs elucidate pathways regulating embryonic development and clinical disorders of the portal vein. Mamm. Genome. 23 (1-2), 76-84 (2012).
- Murai, A., Asa, S. A., Kodama, A., Hirata, A., Yanai, T., Sakai, H. Constitutive phosphorylation of the mTORC2/Akt/4E-BP1 pathway in newly derived canine hemangiosarcoma cell lines. BMC Vet. Res. 8 (1), 128 (2012).
- Boilson, B. A., et al. Regulation of circulating progenitor cells in left ventricular dysfunction. Circ. Heart Fail. 3 (5), 635-642 (2010).
- Gonzalez-Miguel, J., Morchon, R., Siles-Lucas, M., Simon, F. Fibrinolysis and proliferative endarteritis: two related processes in chronic infections? The model of the blood-borne pathogen Dirofilaria immitis. PLoS One. 10 (4), e0124445 (2015).
- Sacks, T., Moldow, C. F., Craddock, P. R., Bowers, T. K., Jacob, H. S. Oxygen radicals mediate endothelial cell damage by complement-stimulated granulocytes. An in vitro model of immune vascular damage. J. Clin. Invest. 61 (5), 1161-1167 (1978).
- Aranguren, X. L., et al. Unraveling a novel transcription factor code determining the human arterial-specific endothelial cell signature. Blood. 122 (24), 3982-3992 (2013).
- van Balkom, B. W., et al. Endothelial cells require miR-214 to secrete exosomes that suppress senescence and induce angiogenesis in human and mouse endothelial cells. Blood. 121 (19), 3997-4006 (2013).
- Crampton, S. P., Davis, J., Hughes, C. C. Isolation of human umbilical vein endothelial cells (HUVEC). J. Vis. Exp. (3), e183 (2007).
- Bustin, S. A., et al. MIQE precis: Practical implementation of minimum standard guidelines for fluorescence-based quantitative real-time PCR experiments. BMC Mol. Biol. 11, 74 (2010).
- Brinkhof, B., Spee, B., Rothuizen, J., Penning, L. C. Development and evaluation of canine reference genes for accurate quantification of gene expression. Anal. Biochem. 356 (1), 36-43 (2006).
- Heishima, K., et al. MicroRNA-214 and MicroRNA-126 Are Potential Biomarkers for Malignant Endothelial Proliferative Diseases. Int. J. Mol. Sci. 16 (10), 25377-25391 (2015).
- Liu, M. M., Flanagan, T. C., Lu, C. C., French, A. T., Argyle, D. J., Corcoran, B. M. Culture and characterisation of canine mitral valve interstitial and endothelial cells. Vet. J. 204 (1), 32-39 (2015).
- van den Bossche, L., van Steenbeek, F. G. Canine congenital portosystemic shunts: disconnections dissected. The Veterinary Journal. 211, 14-20 (2015).
- Sobczynska-Rak, A., Polkowska, I., Silmanowicz, P. Elevated Vascular Endothelial Growth Factor (VEGF) levels in the blood serum of dogs with malignant neoplasms of the oral cavity. Acta Vet. Hung. 62 (3), 362-371 (2014).
- Zhang, Q., et al. In vitro and in vivo study of hydralazine, a potential anti-angiogenic agent. Eur. J. Pharmacol. 779, 138-146 (2016).

