Full-root Aortic Valve Replacement by Stentless Aortic Xenografts in Patients with Small Aortic Roots
Summary
Full-root aortic valve replacement by stentless aortic xenograft is a viable option in patients with small aortic roots. We describe, a technique for the full-root implantation of stentless aortic xenografts, with emphasis on the management of the proximal suture line and coronary anastomoses, and discuss its limitations and alternative options.
Abstract
In patients with small aortic roots who need an aortic valve replacement with biological valve substitutes, the implantation of the stented pericardial valve might not meet the functional needs. The implantation of a too-small stented pericardial valve, leading to an effective orifice area indexed to a body surface area less than 0.85 cm2/m2, is regarded as prosthesis-patient mismatch (PPM). A PPM negatively affects the regression of left ventricular hypertrophy and thus the normalization of left ventricular function and the alleviation of symptoms. Persistent left ventricular hypertrophy is associated with an increased risk of arrhythmias and sudden cardiac death. In the case of predictable PPM, there are three options: 1) accept the PPM resulting from the implantation of a stented pericardial valve when comorbidities of the patient forbid the more technically demanding operative technique of implanting a larger prosthesis, 2) enlarge the aortic root to accommodate a larger stented valve substitute, or 3) implant a stentless biological valve or a homograft. Compared to classical aortic valve replacement with stented pericardial valves, the full-root implantation of stentless aortic xenografts offers the possibility of implanting a 3-4 mm larger valve in a given patient, thus allowing significant reduction in transvalvular gradients. However, a number of cardiac surgeons are reluctant to transform a classical aortic valve replacement with stented pericardial valves into the more technically challenging full-root implantation of stentless aortic xenografts. Given the potential hemodynamic advantages of stentless aortic xenografts, we have adopted full-root implantation to avoid PPM in patients with small aortic roots necessitating an aortic valve replacement. Here, we describe in detail a technique for the full-root implantation of stentless aortic xenografts, with emphasis on the management of the proximal suture line and coronary anastomoses. Limitations of this technique and alternative options are discussed.
Introduction
Biological aortic valve replacement is recommended for patients older than 65 years1. In patients with small aortic roots, the implantation of a stented biological valve substitute based on the labelled size given by the manufacturer might not meet the functional needs. In this situation, Rahimtoola first described the prosthesis-patient mismatch (PPM) as follows: "mismatch can be considered to be present when the effective prosthetic valve area, after insertion into the patient, is less than that of a normal human valve"2. The effective orifice area of the valve prosthesis is to be related to the patient's body size and, more commonly, to the patient's body surface area. The hemodynamic consequence of a too-small prosthetic valve is an abnormally high transvalvular gradient3. It has been shown that the relationship between the transvalvular gradient and the effective orifice area indexed to the body surface area (EOAI) is curvilinear and that gradients increase exponentially when the indexed EOA is less than 0.8 to 0.9 cm2/m2. On the basis of this relation, an EOAI less than 0.85 cm2/m2 is generally regarded as the threshold for PPM in the aortic position4. The impact of the PPM on early and late clinical outcomes is controversial. However, it has been reported that PPM negatively affects the regression of left ventricular hypertrophy and thus the normalization of left ventricular function and the alleviation of symptoms4. Persistent left ventricular hypertrophy is associated with an increased risk of arrhythmias and sudden cardiac death5.
It is therefore advisable to avoid PPM as much as possible4. In the case of a predictable PPM for a planned aortic valve replacement with a biological valve substitute, the options are: 1) to accept the PPM resulting from the implantation of a stented pericardial valve when comorbidities of the patient forbid a more technically demanding operative technique to implant a larger prosthesis, 2) to enlarge the aortic root to accommodate a larger stented valve substitute6, or 3) to implant a stentless biological valve7 or homograft8.
Aortic root enlargement has been reported to enhance perioperative bleeding, necessitating a re-sternotomy and increasing early mortality9. Aortic homografts may have excellent hemodynamic profiles and good long-term outcomes when implanted by experienced surgeons8. However, their limited availability and the accelerated rate of calcification make aortic homografts less suitable biological valve substitutes than their counterpart, porcine stentless aortic xenografts10.
The shortage and drawbacks of homografts have prompted the conception and development of alternative biological valve substitutes. To this purpose, stentless aortic xenografts were introduced into clinical practice11. On one hand, thanks to the elimination of the cumbersome sewing ring, stentless aortic xenografts can reproduce the hemodynamic advantages of homografts. On the other hand, as a result of the application of anti-calcification technology, the durability of stentless aortic xenografts has been optimized to match and even exceed the longevity of homografts11. Hemodynamic advantages of stentless aortic xenografts are entirely attained by full-root implantation12. In contrast to subcoronary and root inclusion techniques, full-root implantation places the stentless aortic xenograft on top of the aortic annulus, and not inside it. This fact underlies the rationale to opt for the full-root implantation technique, which grants the implementation of the largest internal functional diameter of the stentless valve substitute. In addition, the preservation of the Valsalva sinuses en-bloc with the valve leaflets favors more physiological opening and closing movements and hence a longer life expectancy of the leaflets. This advantage further contributes to the amelioration of long-term results12.
However, concerns regarding the increased potential for bleeding and for the possible distortion of coronary ostia anastomoses prevent a number of cardiac surgeons from shifting from a classical aortic valve replacement with a stented biological valve to the more technically demanding procedure represented by full-root replacement with stentless aortic xenografts.
Given the potential hemodynamic advantages of stentless aortic xenografts, we have adopted full-root implantation to avoid PPM in patients with small aortic roots necessitating an aortic valve replacement (Table 1). In these patients, the aim is to attain a projected EOAI of greater than 0.85 cm2/m2 for the newly implanted aortic valve. This intention is based on the reports of Pibarot and co-workers showing an unacceptably high transvalvular gradients for valve substitutes having a projected EOAI of less than 0.85 cm2/m2, with the subsequent incomplete relief of symptoms and the persistent risk of adverse outcomes3,4. Following the initial identification of adult patients with an aortic annulus diameter of less than 20 mm on their pre-operative echocardiography, patients are further selected to have a body surface area of greater than 1.6 m2. In this subgroup of patients, the implantation of a 19-mm stented pericardial aortic valve (EOA: 1.28 cm2) would result in a projected EOAI of less than 0.85 cm2/m2. In this protocol, these patients are candidates for the full-root implantation of stentless aortic xenografts. The final decision is made intra-operatively after the removal of the aortic valve. If a 19-mm valve sizer for the stented pericardial aortic valve passes too-tightly through the aortic annulus and the patient is hemodynamically stable and can tolerate a longer operation, the full-root implantation of a stentless aortic xenograft is performed.
For the stentless aortic xenografts, we use two commercially available valve substitutes interchangeably (for details, see the Table of Materials). Both valves are procured from the porcine aortic root bearing the aortic valve. They are prepared using a low-pressure (0-2 mmHg) fixation process, with anti-calcification (e.g., XenoLogiX) treatment for one valve and alpha amino oleic acid (AOA) anti-calcification treatment for the other. In those patients for whom the 19 mm sizer for the stented pericardial valves passes too-tightly through the aortic annulus, the 23 mm sizer for the stentless aortic xenograft fitting well in the aortic annulus denotes that the stentless aortic xenograft size of 23 mm is to be chosen. This protocol describes in detail the technique of full-root implantation of stentless aortic xenografts, with emphasis on the management of the proximal suture line and coronary anastomoses. Limitations of this technique and alternative options are discussed.
Protocol
The protocol follows the institutional guidelines of the human research ethics committee.
1. Pre-selection of the Patient
- Using the preoperative echocardiography, identify patients with an estimated aortic annulus diameter of less than 20 mm. Be aware that in heavily calcified aortic valves and annuli, the preoperative echocardiography can sometimes be misleading.
- Select from these patients a subgroup with a body surface area greater than 1.6 m2.
- Verify on the chart provided by the valve manufacturer that a stented pericardial aortic valve substitute labelled 19 mm has an effective orifice area of 1.28 cm2.
- Make sure that the implantation of a stented pericardial aortic valve substitute labelled 19 mm into the above-mentioned subgroup of patients would result in an EOAI of less than 0.85 cm2/m2 (1.28 cm2 divided by 1.6 m2 = 0.8 cm2/m2).
- Inform the staff of the possibility of a full-root stentless aortic xenograft implantation.
- Make the final decision intraoperatively after the aortic valve is removed. Check that the coronary ostia are not heavily calcified and that a 19 mm valve sizer for the stented pericardial aortic valve substitute passes too-tightly through the debrided aortic annulus.
2. Preparation for Surgery
Note: Preparation for surgery follows institutional guidelines and recommendations for adult cardiac surgery patients.
- Clean and prepare the surgical suite in a typical manner. In order to facilitate the communication between the surgeon and the perfusionist, place the heart lung machine to the left of the patient, opposite to the surgeon.
- Pre-medicate the patient by the oral administration of 5 mg of midazolam, 30-60 min prior to the induction of anesthesia.
- Monitor the patient according to standard guidelines with direct arterial and central venous pressure access by the anesthesiologists. Induce anesthesia using an initial intravenous injection of 0.5-1.5 mg/kg propofol, 1-2 µg/kg fentanyl, and 0.6 mg/kg rocuronium.
- Perform a tracheal intubation and then maintain the anesthesia by the intravenous infusion of propofol 100-150 µg/kg/min, fentanyl 0.015 – 0.03 µg/kg/min, and rocuronium 0.6 – 1.2 µg/kg/min, as needed.
- Install the patient in a supine position and drape her/him in a sterile fashion, leaving the chest, the abdomen, and the groin free in the operative field.
3. Surgery
- Access the heart through a median sternotomy.
- Incise the skin longitudinally over 15 cm with an 18 blade knife, starting 1 cm below the suprasternal notch. Take care to stay in the middle of the sternal width.
- Saw the sternum with an oscillatory saw. Take care to stay in the middle of the sternal width.
- Grab the pericardium with Carpentier dissection forceps and cut it with Metzenbaum scissors. Continue cutting the pericardium up to its reflection line over the ascending aorta using electrocautery.
- Give 300 I.U. of heparin/kg (concentration: 5,000 U/mL) through the IV line.
- Cannulate the distal ascending aorta using commercially available arterial cannulas13.
- Cannulate the right atrium using a commercially available atrial caval venous cannula13.
- Start the cardiopulmonary bypass. Cool down the patient to 32 °C.
- Place a commercially available vent through the right upper pulmonary vein and the mitral valve in the left ventricle to unload the left heart13. Secure it with a braided polyester 2/0 ligature with polybutylene coating.
- Insert a commercially available retrograde cardioplegic cannula through the coronary sinus. Secure it with a braided polyester 2/0 ligature with polybutylene coating.
- Insert a commercially available antegrade cardioplegic cannula into the aortic root at the sino-tubular junction. Secure it with a braided polyester 2/0 ligature with polybutylene coating.
- Clamp the distal ascending aorta below the arterial cannulation site.
- Deliver the antegrade cold blood cardioplegia through the antegrade cannula in the aortic root and repeat it retrogradely every 20 min. After the completion of the coronary ostial anastomoses, repeat the antegrade cold blood cardioplegia directly into the coronary ostia using a commercially available coronary artery cardioplegic cannula.
- Prepare the aortic root for implantation.
- Remove the antegrade cardioplegic cannula. Grab the ascending aorta with Carpentier dissection forceps on each side of the opening left following removal of the cardioplegic cannula.
- Enlarge the opening with an 18-blade knife. Finish the transection of the aorta with Metzenbaum scissors. Verify the presence of the right coronary ostium and the left coronary ostium.
- Grab the top of the left sinus of Valsalva with Carpentier dissection forceps and dissect free the left coronary ostium and the proximal main left coronary artery of the patient from the aortic wall, with a surrounding patch of 5 mm, using Metzenbaum scissors. Put a 5/0 polypropylene stay suture through the top of the patch around the left coronary ostium.
- Grab the top of the right sinus of Valsalva with Carpentier dissection forceps and dissect free the right coronary ostium and the proximal right coronary artery of the patient from the aortic wall, with a surrounding patch of 5 mm, using Metzenbaum scissors. Put a 5/0 polypropylene stay suture through the top of the patch around the right coronary ostium.
- Confirm the pathological changes of the aortic valve leaflets.
- Grab the right coronary leaflet with Carpentier dissection forceps and excise it with Metzenbaum scissors. Grab the non-coronary leaflet with Carpentier dissection forceps and excise it with Metzenbaum scissors.
- Grab the left coronary leaflet with Carpentier dissection forceps and excise it with Metzenbaum scissors. Grab the non-coronary Valsalva sinus wall with Carpentier dissection forceps and make a perpendicular incision from the top to the base of the sinus. Stay 1 cm away from the aortic annulus.
- Calibrate the aortic annulus with a commercially available valve sizer. Make sure that the 19-mm sizer for the stented pericardial valves passes too-tightly through the aortic annulus and that the 23-mm sizer for stentless aortic xenografts fits well on the top of the aortic annulus.
- Unpack the valve as soon as its size is determined.
- Start rinsing the valve by placing it in a minimum of 750 mL of sterile, physiological saline solution. Ensure that the saline solution completely covers the valve. Gently swirl the valve back and forth for a minimum of 1 min. Discard the rinse solution. Repeat this process at least two additional times, using new saline solution, for a total of three rinses, 1 min each.
- Create the left coronary neo-ostium in the stentless aortic xenograft by extending the native left coronary ostium or the ligated left coronary artery of the stentless aortic xenograft towards the commissure between the left and non-coronary sinuses. Adapt the size of this neo-ostium to the size of the patient's left coronary ostium with its surrounding patch.
- Let an assistant hold the stentless aortic xenograft wall between the thumb and index finger so that the left neo-coronary ostium of the graft is oriented upwards.
- Begin the implantation of the stentless aortic xenograft with the proximal suture.
- Place a 4/0 polypropylene stay suture above each aortic commissure. Pull up these traction sutures to better expose the aortic annulus of the patient.
- Start the first suture of the proximal anastomosis at the middle of the left coronary sewing ring of the stentless aortic xenograft, below the left coronary neo-ostium, using a 4/0 polypropylene suture. Pass this first stich from the outer aspect of the xenograft to the inner aspect.
- Pass the first stich in the middle of the left coronary sinus into the aortic annulus of the patient, from ventricle outward, using a Ryder needle holder and Carpentier dissection forceps. Make sure that the left coronary button hole of the xenograft exactly faces the left coronary ostium of the patient.
- Cautiously grab the ridge of the sewing ring of the stentless aortic xenograft with Carpentier dissection forceps. Pass the second stich 2 mm to the left of the first one, from outer aspect of the xenograft to the inner aspect. Pull the suture gently through the graft to get full length of it.
- Grab the aortic wall on the top of the commissure between the left and non-coronary sinuses (with Carpentier dissection forceps) and pull it up to stretch out the left sinus part of the aortic annulus of the patient.
- Pass the second stich in the left coronary sinus of the aortic annulus of the patient 2 mm to the left of the first one, from the ventricle outward. Use a Ryder needle holder and Carpentier dissection forceps.
- Slide down the stentless aortic xenograft by gently pulling the two ends of the first suture. Put the right end of the first running suture on traction.
- Grab the aortic wall on the top of the commissure between the left and right coronary sinuses of the stentless aortic xenograft with Carpentier dissection forceps and pull the graft gently towards the right to expose the left half of the left coronary sinus of the patient.
- Pass the third stich 2 mm to the left of the second one, from the outer aspect to the inner aspect of the stentless aortic xenograft.
- Let the first assistant tightly hold the left end of the first suture and grab the aortic wall on the top of the commissure between the left and right coronary sinuses of the patient using Carpentier dissection forceps. Let a second assistant gently unfold the right coronary sinus of the patient using a lung retractor.
- Pass the third stich in the left coronary sinus of the aortic annulus of the patient 2 mm to the left of the second one, from ventricle outward. Use a Ryder needle holder and Carpentier dissection forceps.
- Run the first suture up to the commissure between the left and right coronary sinuses of the patient. Put the left end of the first suture on traction.
- Begin the second 4/0 polypropylene running suture by placing the first stitch 2 mm to the side of the last stitch in the sewing ring of the stentless aortic xenograft, from outside to inside.
- Pass the first stich of the second running suture in the right coronary sinus of the aortic annulus of the patient 2 mm to the right of the last one, from the ventricle outward, using a Ryder needle holder and Carpentier dissection forceps.
- Place the second stitch of the second 4/0 polypropylene running suture in the sewing ring of the stentless aortic xenograft, 2 mm to the side of the first one. Let the first assistant loosely hold the running end of the suture.
- Pass the second stitch of the second 4/0 polypropylene running suture into the right coronary sinus of the aortic annulus of the patient, 2 mm to the right of the first one, from ventricle outward. Use a Ryder needle holder and Carpentier dissection forceps.
- Place the third stitch of the second 4/0 polypropylene running suture in the sewing ring of the stentless aortic xenograft, 2 mm to the side of the second one. Let the first assistant loosely hold the running end of the suture.
- Pass the third stitch of the second 4/0 polypropylene running suture into the right coronary sinus of the aortic annulus of the patient, 2 mm to the right of the second one, from ventricle outward. Use a Ryder needle holder and Carpentier dissection forceps.
- Run the second suture to the middle of the right coronary sinus of the aortic annulus of the patient. Slowly pull the two ends of the second running suture to bring the graft and the aortic annulus tightly together. Put the two ends of the second running suture on traction.
- Place the first stitch of the third 4/0 polypropylene running suture 2 mm to the side of the last stitch in the sewing ring of the stentless aortic xenograft, from the outside to the inside. Let the first assistant loosely hold the running end of the suture.
- Pass the first stitch of the third 4/0 polypropylene running suture 2 mm to the side of the last stitch in the right coronary sinus of the aortic annulus of the patient.
- Run the third 4/0 polypropylene running suture to the commissure between the right and non-coronary sinuses of the patient. Put both ends of the third suture on traction.
- Place the fourth 4/0 polypropylene running suture, with 2 mm of space on both the graft and the aortic annulus sides, starting at the commissure between the right and non-coronary sinuses and running to the middle of the non-coronary sinus. Initially, let the first assistant hold the suture loosely.
- Slowly pull the two ends of the fourth running suture to tightly bring together the graft and the aortic annulus of the patient. Let the first assistant grab the graft with Carpentier dissection forceps and bring it towards the aortic annulus of the patient. Put the two ends of the fourth running suture on traction.
- Place the fifth 4/0 polypropylene running suture, with 2 mm of space on both the graft and the aortic annulus sides, starting at the middle of the non-coronary sinus and running to the commissure between the non-coronary and left coronary sinuses. Initially, let the first assistant hold the suture loosely.
- Slowly pull the two ends of the fifth running suture to tightly bring together the graft and the aortic annulus of the patient. Let the first assistant grab the graft with Carpentier dissection forceps and bring it towards the aortic annulus of the patient. Put the two ends of the fifth running suture on traction.
- Place the sixth 4/0 polypropylene running suture, with 2 mm of space on both the graft and aortic annulus sides, starting at the commissure between the non-coronary and left coronary sinuses and running to the middle of the left coronary sinuses to meet the right end of the first 4/0 polypropylene running suture. Initially, let the first assistant hold the suture loosely.
- Slowly pull the two ends of the sixth running suture to tightly bring together the graft and the aortic annulus of the patient. Let the first assistant grab the graft with Carpentier dissection forceps and bring it towards the aortic annulus of the patient. Put the two ends of the sixth running suture on traction.
- Tie the two ends of the adjacent running sutures together. Verify the absence of any gap between the bites after having suctioned around the proximal suture line and in the coronary ostia.
- Reconnect the left coronary ostium of the patient to the left coronary neo-ostium of the graft.
- Place the first stitch of a 6/0 polypropylene running suture at the deepest point of the left coronary neo-ostium of the graft, from inside to outside. Use a microneedle holder and DeBakey dissection forceps.
- Pass the first stitch of the 6/0 polypropylene running suture to the deepest point of the left coronary ostium of the patient, from outside to inside. Slowly pull the two ends of the 6/0 polypropylene running suture to tightly bring together the left coronary neo-ostium of the graft and the left coronary ostium of the patient.
- Place the second stitch of the suture to the right of the first one, from the inside to the outside of the graft.
- Bring the suture up to the middle of the right ridge of the left coronary anastomosis. Put this end of the suture on traction.
- Continue by passing the left end of the suture into the left coronary neo-ostium of the graft, from the outside to the inside, and then into the left coronary ostium of the patient, from the inside to the outside.
- Gently grab the graft and the ridge of the left coronary ostium in succession to bring them together each time the suture is pulled through.
- Bring the suture to meet the other end. Then, tie the two ends together.
- Reconnect the right coronary ostium of the patient to the right coronary neo-ostium of the graft.
- Grab the ligated right coronary ostium of the graft with DeBakey dissection forceps. Create a right coronary neo-ostium in the graft by excising the ligated right coronary of the graft and enlarging the opening horizontally, towards the commissure between the right and non-coronary sinuses of the graft, using a scalpel (e.g., sharpoint).
- Place the first stitch of a 6/0 polypropylene running suture at the left end of the lower ridge of the right coronary ostium of the patient, from the inside to the outside. Use a microneedle holder and DeBakey dissection forceps.
- Pass the first stitch of the 6/0 polypropylene running suture at the left end of the lower ridge of the right coronary neo-ostium of the graft, from the outside to the inside. Slowly pull the two ends of the 6/0 polypropylene running suture to tightly bring the right coronary neo-ostium of the graft and the right coronary ostium of the patient together.
- Place the second stitch of the suture to the right of the first one, from the inside to the outside of the right coronary ostium of the patient, and then from the outside to the inside of the right coronary neo-ostium of the graft.
- Bring the suture to the middle of the right ridge of the right coronary anastomosis. Put this end of the suture on traction.
- Continue with the left end of the suture by placing the stitches from the outside to the inside of the right coronary artery of the patient and from the inside to the outside of the right coronary neo-ostium of the graft.
- Let the first assistant gently retract the right ventricular outflow tract with a lung retractor to improve the exposition of the operative field.
- Bring the left end of the suture towards the right end and tie them together.
- Upon completion of the coronary anastomoses, put a coronary artery ostial cannula 45° in the left coronary ostium and a coronary artery ostial cannula 90° in the right coronary ostium of the patient. Repeat the antegrade cold blood cardioplegia directly into the coronary arteries.
- Complete the distal anastomosis.
- Place the first stitch of a 5/0 polypropylene running suture at the left end of the lower rim of the graft, from the inside to the outside, and then into the facing distal ascending aorta, from the outside to the inside.
- Leave a pump sucker beside the right flank of the anastomosis for a relatively blood-free operative field.
- Put the next stitch to the right of the previous one. Continue the suture from the inside to the outside in the graft and from the outside to the inside of the distal ascending aorta using a microneedle holder and Carpentier dissection forceps.
- With Carpentier dissection forceps, grab the adventitia on the right end of the lower rim of the distal ascending aorta and pull it horizontally to the right to unfold the lower aspect of the anastomosis. Repeat this maneuver for each passage from the outside to the inside of the aorta.
- Follow and place each stitch on the posterior rim of the anastomosis from the inside so that all stitches sit regularly, at distances of 2 mm from each other. Bring the suture to the middle of the right rim of the anastomosis. Put the end of the suture on traction.
- Continue with the left end of the suture. At the same time, start delivering retrograde warm blood cardioplegia. Place the stitches equidistant on the anterior aspect of the anastomosis, from the outside to the inside on the stentless graft and from the inside to the outside on the distal ascending aorta.
- Bring the left end of the suture towards the right end and tie them together.
- Let the operating table tilt in the Trendelenburg position. Let the pump flow reduce to 50% of the full flow. Slowly remove the aortic cross-clamp under gentle aspiration of the left ventricular vent.
- Resume the full flow of the cardio-pulmonary bypass. Remove the lung retractor on the outflow tract of the right ventricle. Check the operative field for undue surgical bleeding.
- Rewarm the patient to 37 °C and separate the patient from the cardio-pulmonary bypass. By the time a stable blood pressure is reached, neutralize the heparin with a protamin-infused IV in a 1:1 ratio (3 mg/kg corresponding to 300 U/kg of heparin). Check for hemostasis and use chest drainage as needed. Close the chest in a standard fashion.
4. Postoperative Patient Care
- Transfer the patient to the intensive care unit.
- Leave the patient under anesthesia with the intravenous infusion of propofol 100-150 µg/kg/min, fentanyl 0.015-0.03 µg/kg/min, and rocuronium 0.6-1.2 µg/kg/min, as needed.
- Taper down the anesthesia when the core temperature of the patient reaches 37 °C.
- Extubate the patient when the arterial partial pressure of oxygen is greater than 9 kPa and the arterial partial pressure of CO2 is less than 5 kPa under an inspired oxygen fraction of 0.3.
- Make sure of the absence of excessive bleeding by checking the chest tube drainage hourly for the first 3 h (less than 100 mL/h) and 6, 12, and 24 h afterwards (global drainage of less than 30 mL/h).
- Make sure of the absence of myocardial ischemia by checking the ECG and myocardial enzymes upon arrival in the intensive care unit and 6, 12 and 24 h afterwards.
- Mobilize the extubated patient as soon as the absence of bleeding and myocardial ischemia are documented.
- Transfer the patient to the ward by the time stable hemodynamics are achieved.
Representative Results
Statistical analysis
The values of projected effective orifice area indexed to the body surface area (EOAI, cm2/m2) for implanted stentless aortic xenografts sized 23 mm are expressed as means ± SD and compared to the calculated EOAI of stented pericardial valves 4 mm smaller (i.e., 19 mm) using the non-parametric Mann Whitney test. In Table 2, continuous variables are compared using the non-parametric Mann Whitney test and categorical variables by Chi-square test. Statistical analysis is performed using commercially available software, with the statistical significance set at p<0.05.
Effective orifice area indexed
In patients with small aortic roots (Figure 1), the implantation of a 19 mm-sized stented pericardial valve would have resulted in moderate to severe PPM4 by an EOAI of 0.7 ± 0.09 (range 0.55 – 0.84) cm2/m2 (Figure 1). With this technique, the implantation of a stentless aortic xenograft in these patients yielded a significantly higher EOAI of 1.09 ± 0.14 cm2/m2 (range 0.87-1.31 cm2/m2, p <0.0001), thus eliminating any PPM (Figure 2).
Intraoperative and early postoperative data
As expected, the cross-clamp, cardio-pulmonary bypass, and operative times for the full-root implantation of stentless aortic xenografts in our patients were longer than those reported for isolated aortic valve replacement with stented valves13,14. Nevertheless, perioperative morbidity and mortality were very low and not adversely affected by the prolongation of the operative times (Table 2)13.
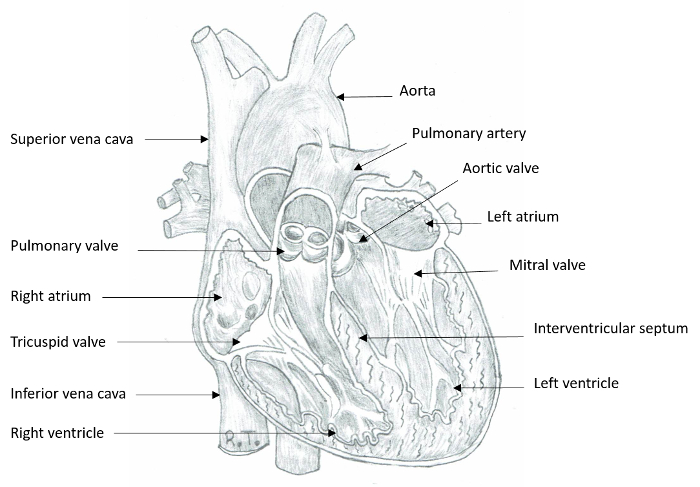
Figure 1: Internal Anatomy of the Heart. The anterior aspect of the heart is partially removed to depict the four cardiac chambers and valves. The anterior leaflet of the mitral valve is in continuity with the left and non-coronary cusps of the aortic valve. Please click here to view a larger version of this figure.
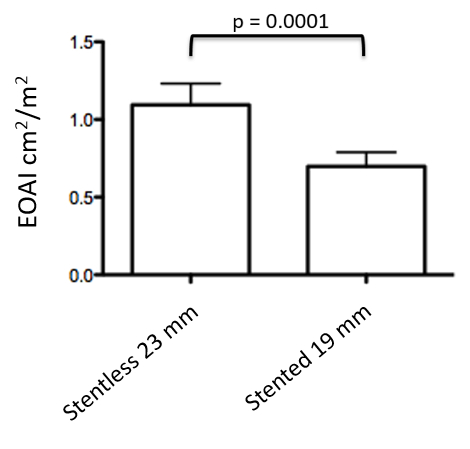
Figure 2: Effective Orifice Area Indexed. In patients with small aortic roots (N = 22), the projected effective orifice area indexed to the body surface area (EOAI, cm2/m2) for implanted stentless aortic xenografts sized 23 mm is significantly higher than the calculated EOAI if they had received a stented pericardial valve 4 mm smaller (i.e., 19 mm). The projected EOAI is calculated by dividing the effective orifice area (cm2) of the valve substitute provided by the manufacturer by the body surface area of the patient (m2). The values are expressed as means ± standard deviations.
| Full root Stentless Xenografts | |
| N | 22 |
| Age, years (mean±SD) | 63±10 |
| Female gender | 18 (82%) |
| Body surface area, m2 (mean±SD) | 1.85±0.24 |
| Ejection Fraction (%) (mean±SD) | 53±11 |
| Aortic regurgitation | 5 (22%) |
Table 1: Patients' Characteristics. Patients' characteristics are depicted in this table. The decision to implant a full-root stentless aortic xenograft is based on the body surface area to avoid a projected effective orifice area of less than <0.85 cm2/m2, considered a prosthesis-patient mismatch.
| Full-root Stentless Xenografts 23 mm | Isolated aortic valve replacement with stented pericardial valves1 | p | |
| N | 22 | 36 | |
| Cross-clamp time (min) | 83 ±9 | 62.3 ±9.4 | 0.0001 |
| CPB time (min) | 134 ±32 | 101 ±27.2 | 0.0001 |
| OP time (min) | 242±48 | 191.7 ±53.2 | 0.0001 |
| Re-exploration for bleeding | 0 | 1(3%) | ns |
| Pace maker | 0 | 0 | ns |
| Stroke | 1(4.5%) | 1(3%) | ns |
| Sternal infection | 0 | 0 | ns |
| Early mortality | 0 | 1(3%) | ns |
| 1 Adapted with permission from Biomed Central from reference 13 | |||
| ns = not significant |
Table 2: Comparison of Intraoperative Data and 30-day Morbidity and Mortality to Previously Reported Data. Cross-clamp, cardio-pulmonary bypass (CPB), and operative times are reported (mean values ± standard deviation), along with 30-day morbidity and mortality in the presented group of patients, and are compared with previously reported data for stented pericardial aortic valves. Adapted with permission from Biomed Central from reference13.
Discussion
This study reports a detailed description of the surgical technique of full-root aortic valve replacement using stentless aortic xenografts in patients with small aortic roots. Early morbidity and mortality are very low and compare favorably with other reports7. Heavily calcified coronary ostia constitute an anatomical limitation to this technique. Another drawback to this technique is represented by patients with a poor general condition who would not tolerate longer operative times. In these cases, a standard stented pericardial aortic valve replacement, with or without aortic root enlargement, should be preferred to the more demanding full-root stentless aortic xenograft implantation6.
This protocol calls for six running 4/0 polypropylene sutures for the proximal anastomosis. Similarly to multiple interrupted sutures used by other authors14,15, this technique for the proximal anastomosis leads to a better distribution of tension on the proximal anastomosis and thus avoids pleating of the suture line. Compared to multiple interrupted sutures, the use of six semi-continuous running sutures is slightly time-saving. Moreover, it makes the use of reinforcement of the suture line by pericardial or polytetrafluoroethylene strips for the control of postoperative bleeding7,12,14 unnecessary. In these patients, no resternotomy was necessary for bleeding. A potential disadvantage of a single running suture for the proximal anastomosis advocated by some authors7,12 is the risk of pleating of the valve substitute and the left ventricular outflow tract.
Besides the potential of increased postoperative bleeding, coronary anastomotic problems constitute another major concern of cardiac surgeons who are not willing to use the full-root technique for the implantation of stentless aortic xenografts. Possible coronary anastomotic problems can arise, from kinking of patient's coronaries after re-implantation to tissue tear related to excessive tension on the anastomosis. To adapt the placement of the coronary neo-ostia of the graft to the coronary ostia of the patient, the left coronary neo-ostium is first created in the graft by extending the existing left coronary hole or ligated artery towards the commissure between the left and non-coronary sinus of the stentless graft. Upon completion of the proximal suture line and re-connection of the left coronary anastomosis, the place of re-connection of the right coronary ostium of the patient is adjusted according to the right coronary sinus of the graft. To do so, the right coronary neo-ostium in the graft is created by extending the existing right coronary hole or ligated artery towards the commissure between the right and non-coronary sinus of the stentless graft. Proximal mobilization over 1-2 cm of the main coronary arteries further helps in eliminating excessive tension on the coronary anastomoses and thus the potential for tissue tear and bleeding. This technique avoids the rotation of the stentless graft suggested by other authors12.
The cross-clamp, cardio-pulmonary bypass, and operative times for this technique are expectedly longer than those for standard aortic valve replacements with stented valves16. However, they compare favorably with those reported by Kunihara et al.7 for the full-root implantation of stentless aortic xenografts. Despite the prolongation of the operative times in our patients, the perioperative morbidity and mortality are not adversely affected, as compared to those reported in the STS Adult Cardiac Surgery database17 for isolated aortic valve replacement. In particular, the incidence of re-exploration for bleeding, complete heart block requiring definitive pace maker implantation, stroke, deep sternal infection, or early mortality are very low and similar to those reported by Kunihara et al.7 for the full-root implantation of stentless aortic xenografts and by ourselves for the isolated aortic valve replacement by stented pericardial valves13.
The labelled size of all commercially available mechanical and biological valve substitutes indicates the global (external) diameter of the valve. However, for stented valve substitutes, including mechanical and biological stented pericardial valves, the sewing ring of the valve occupies between 3 and 4 mm of the global (external) diameter of the valve and correspondingly reduces the functionally useful internal diameter. Biological stentless aortic xenografts are devoid of similar obstructive sewing rings. Hence, the functionally useful internal diameter of these valve substitutes is very close to their global (external) diameter as commercially labelled. As a result, for a given labelled size, biological stentless valves offer a functionally useful internal diameter larger than biological stented pericardial valves. A larger functionally useful internal diameter allows for a greater opening area of the valve, known as the effective orifice area of the valve. A better opening area of the valve, adjusted for the body surface area, known as the indexed effective orifice area, provides for superior hemodynamics and potentially for better functional relief of the patient. In this regard, a comparison between 23 mm stented and stentless valves implanted by the full-root technique showed better hemodynamics in the latter in terms of transvalvular gradients. As a consequence, better patient outcomes were observed in terms of regression of left ventricular hypertrophy18.
The selection of adult patients with small aortic roots for full-root stentless aortic xenograft implantation was based on the projected (i.e., preoperatively expected) EOAI. In concrete terms, in patients with an aortic annulus diameter of less than 20 mm, estimated on preoperative echocardiography, a subgroup with a body surface area of greater than 1.6 m2 was identified. These patients are at risk of PPM if they receive a stented pericardial valve offering an effective orifice area of 1.28 cm2, with a resulting indexed orifice area of 0.8 cm2/m2. It has been shown that projected EOAI of less than 0.85 cm2/m2 predicts PPM, with adverse effects on postoperative, long-term transvalvular gradients and patient outcomes3,4.
Another option to accommodate a larger aortic valve prosthesis into a small aortic root is represented by patch enlargement of the aortic annulus6. However, this technique is not devoid of drawbacks, even in experienced hands9. Increased postoperative bleeding requiring resternotomy and enhanced early mortality following patch enlargement of the aortic annulus have been reported by Sommers and David9.
In conclusion, in patients with small aortic roots, we recommend the full-root implantation of stentless aortic xenografts to avoid PPM. This technique can be performed without adversely affecting the early morbidity or mortality. The additional time necessary for the full-root implantation of stentless valves compared to the implantation of pericardial stented valves is therefore not detrimental to the early clinical outcomes and could be rewarded by better mid- and long-term outcomes. Limitations to this technique are represented by heavily calcified coronary ostia and patients in poor general condition who would not tolerate longer operative times.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work was supported by a grant from the Swiss Cardiovascular Foundation to RT.
Materials
| Heart surgery infrastructure: | |||
| Heart Lung Machine | Stockert | SIII | |
| EOPA 24Fr. arterial cannula | Medtronic | 77624 | |
| Atrial caval venous cannula 34/48Fr. | Medtronic | 93448 | |
| LV vent catheter 17Fr. | Edwards | E061 | |
| Antegrade 9Fr. cardioplegia cannula | Edwards | AR012V | |
| Retrograde 14Fr. cardioplegia cannula | Edwards | NPC014 | |
| Coronary artery ostial cannula 90° | Medtronic | 30155 | |
| Coronary artery ostial cannula 45° | Medtronic | 30255 | |
| Name | Company | Catalog Number | Comments |
| Valve subsitutes: | |||
| Stentless aortic xenograft Prima Plus 23mm | Edwards | 2500P-23 | anti-calcification XenoLogiX treatment |
| Stentless aortic xenograft Sizer 23mm | Edwards | 1170 | |
| Stentless aortic xenograft Freestyle 23 mm | Medtronic | FR995-23 | alpha amino oleic acid (AOA) anti-calcification treatment |
| Stentless aortic xenograft Sizer 23mm | Medtronic | 7900 | |
| Electrocautery | Covidien | Force FXTM | |
| Name | Company | Catalog Number | Comments |
| Sutures: | |||
| Polypropylene 4/0 | Ethicon | 8871H | |
| Polypropylene 5/0 | Ethicon | 8870H | |
| Polypropylene 6/0 | Ethicon | EH7400H | |
| Braided polyesther 2/0 ligature with polybutylate coating | Ethicon | X305H | |
| Micro knife Sharpoint | TYCO Healthcare PTY | 78-6900 | |
| Name | Company | Catalog Number | Comments |
| Drugs: | |||
| Midazolam | Roche Pharma | N05CD08 | |
| Rocuronium | MSD Merck Sharp & Dohme | M03AC09 | |
| Propofol | Fresenius Kabi | N01AX10 | |
| Fentanil | Actavis | N01AH01 | |
| Heparin | Braun | B01AB01 | |
| Protamin | MEDA Pharmaceutical | V03AB14 | |
| Name | Company | Catalog Number | Comments |
| Instruments: | |||
| Cooley vascular aortic clamp | Delacroix-Chevalier | DC40810-16 | |
| Dissection forceps Carpentier | Delacroix-Chevalier | DC13110-28 | |
| Scissors Metzenbaum | Delacroix-Chevalier | B351751 | |
| Needle holder Ryder | Delacroix-Chevalier | DC51130-20 | |
| Dissection forceps DeBakey | Delacroix-Chevalier | DC12000-21 | |
| Micro needle holder Jacobson | Delacroix-Chevalier | DC50002-21 | |
| Micro scisors Jacobson | Delacroix-Chevalier | DC20057-21 | |
| Lung retractor | Delacroix-Chevalier | B803990 | |
| Allis clamp | Delacroix-Chevalier | DC45907-25 | |
| O’Shaugnessy Dissector | Delacroix-Chevalier | B60650 | |
| 18 blade knife | Delacroix-Chevalier | B130180 | |
| Leriche haemostatic clamp | Delacroix-Chevalier | B86555 | |
| Name | Company | Catalog Number | Comments |
| Data analysis: | |||
| Mann-Whitney and Chi-square tests | GraphPad | Prism 7 |
Riferimenti
- Bonow, R. O., et al. Focused update incorporated into the ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1998 Guidelines for the Management of Patients With Valvular Heart Disease): endorsed by the Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Circulation. 118 (15), e523-e661 (2008).
- Rahimtoola, S. H. The problem of valve prosthesis-patient mismatch. Circulation. 58 (1), 20-24 (1978).
- Pibarot, P., Dumesnil, J. G. Prosthesis-patient mismatch: definition, clinical impact, and prevention. Heart. 92 (8), 1022-1029 (2006).
- Pibarot, P., Dumesnil, J. G. Hemodynamic and clinical impact of prosthesis-patient mismatch in the aortic valve position and its prevention. J. Am. Coll. Cardiol. 36 (4), 1131-1141 (2000).
- Kang, Y. J. Cardiac hypertrophy: a risk factor for QT-prolongation and cardiac sudden death. Toxicol. Pathol. 34 (1), 58-66 (2006).
- Castro, L. J., Arcidi, J. M., Fisher, A. L., Gaudiani, V. A. Routine enlargement of the small aortic Root: a preventive strategy to minimize mismatch. Ann. Thorac. Surg. 74 (1), 31-36 (2002).
- Kunihara, T., Schmidt, K., Glombitza, P., Dzindzibadze, V., Lausberg, H., Schäfers, H. J. Root replacement using stentless valves in the small aortic root: A propensity score analysis. Ann. Thorac. Surg. 82 (4), 1379-1384 (2006).
- Yacoub, M., et al. Fourteen-year experience with homovital homografts for aortic valve replacement. J. Thorac. Cardiovac. Surg. 110 (1), 186-194 (1995).
- Sommers, K. E., David, T. E. Aortic valve replacement with patch enlargement of the aortic annulus. Ann. Thorac. Surg. 63, 1608-1612 (1997).
- El-Hamamsy, I., et al. Rate of progression and functional significance of aortic root calcification after homograft versus freestyle aortic root replacement. Circulation. 120 (Suppl 1), S269-S275 (2009).
- Bach, D. S., Kon, N. D., Dumesnil, J. G., Sintek, C. F., Doty, D. B. Ten-year outcome after aortic valve replacement with the Freestyle stentless bioprosthesis. Ann. Thorac. Surg. 80 (2), 480-487 (2005).
- Ennker, I. C., Albert, A., Dalladaku, F., Rosendahl, U., Ennker, J., Florath, I. Midterm outcome after aortic root replacement with stentless porcine bioprostheses. Eur. J Cardio-thorac. Surg. 40 (2), 429-434 (2011).
- Tavakoli, R., et al. Full-root aortic valve replacement with stentless xenograft achieves superior regression of left ventricular hypertrophy compared to pericardial stented aortic valves. J. Cardiothorac. Surg. 10, 15 (2015).
- Kon, N. D., Cordell, A. R., Adair, S. M., Dobbins, J. E., Kitzman, D. W. Aortic root replacement with the Freestyle stentless porcine aortic root bioprosthesis. Ann. Thorac. Surg. 67 (6), 1609-1616 (1999).
- Dapunt, O. E., et al. Stentless full-root bioprosthesis in surgery for complex aortic valve-ascending aortic disease: a single center experience of over 300 patients. Eur. J. Cardiothorac. Surg. 33 (4), 554-559 (2008).
- Saxena, A., Dinh, D., Smith, J. A., Reid, C. M., Shardey, G., Newcomb, A. E. Training surgeon status is not associated with an increased risk of early or late mortality after isolated aortic valve replacement surgery. Cardiol. J. 21 (2), 183-190 (2014).
- D’Agostino, R. S., et al. The Society of Thoracic Surgeons Adult Cardiac Surgery Database: 2016 Update on Outcomes and Quality. Ann. Thorac. Surg. 101 (1), 24-32 (2016).
- Fries, R., Wendler, O., Schieffer, H., Schäfers, H. J. Comparative rest and exercise hemodynamics of 23-mm stentless versus 23-mm stented aortic bioprostheses. Ann. Thorac. Surg. 69 (3), 817-822 (2000).

