Analyses of Mitochondrial Calcium Influx in Isolated Mitochondria and Cultured Cells
Summary
Here, we present two protocols for the measurement of mitochondrial Ca2+ influx in isolated mitochondria and cultured cells. For isolated mitochondria, we detail a plate reader-based Ca2+ import assay using the Ca2+ sensitive dye calcium green-5N. For cultured cells, we describe a confocal microscopy method using the Ca2+ dye Rhod-2/AM.
Abstract
Ca2+ handling by mitochondria is a critical function regulating both physiological and pathophysiological processes in a broad spectrum of cells. The ability to accurately measure the influx and efflux of Ca2+ from mitochondria is important for determining the role of mitochondrial Ca2+ handling in these processes. In this report, we present two methods for the measurement of mitochondrial Ca2+ handling in both isolated mitochondria and cultured cells. We first detail a plate reader-based platform for measuring mitochondrial Ca2+ uptake using the Ca2+ sensitive dye calcium green-5N. The plate reader-based format circumvents the need for specialized equipment, and the calcium green-5N dye is ideally suited for measuring Ca2+ from isolated tissue mitochondria. For our application, we describe the measurement of mitochondrial Ca2+ uptake in mitochondria isolated from mouse heart tissue; however, this procedure can be applied to measure mitochondrial Ca2+ uptake in mitochondria isolated from other tissues such as liver, skeletal muscle, and brain. Secondly, we describe a confocal microscopy-based assay for measurement of mitochondrial Ca2+ in permeabilized cells using the Ca2+ sensitive dye Rhod-2/AM and imaging using 2-dimensional laser-scanning microscopy. This permeabilization protocol eliminates cytosolic dye contamination, allowing for specific recording of changes in mitochondrial Ca2+. Moreover, laser-scanning microscopy allows for high frame rates to capture rapid changes in mitochondrial Ca2+ in response to various drugs or reagents applied in the external solution. This protocol can be applied to measure mitochondrial Ca2+ uptake in many cell types including primary cells such as cardiac myocytes and neurons, and immortalized cell lines.
Introduction
Mitochondria are critical sites of intracellular Ca2+ storage and signaling. Decades of research have demonstrated that mitochondria have the ability to import and sequester Ca2+ 1,2. Mitochondria, however, are not merely passive sites of Ca2+ storage. Ca2+ at the mitochondrial compartment performs fundamental signaling functions including regulation of metabolic output and activation of mitochondrial-mediated cell death pathways, which has been reviewed previously3. For metabolic regulation, Ca2+ enhances the activity of three matrix-localized dehydrogenases of the tricarboxylic acid cycle as well as respiratory chain complexes, to increase mitochondrial energy production4,5. With mitochondrial Ca2+ overload and dysregulated mitochondrial Ca2+ handling, Ca2+ triggers mitochondrial permeability transition pore (MPTP) opening, leading to mitochondrial inner membrane permeabilization, membrane potential loss, mitochondrial dysfunction, swelling, rupture and ultimately, cell death6,7,8,9. Thus, mitochondrial Ca2+ signaling directly impacts both cellular life and death pathways through metabolic control and MPTP-death axis.
In recent years, there has been rapidly expanding interest in the study of mitochondrial Ca2+ dynamics due in large part to the identification of the molecular constituents of the mitochondrial Ca2+ uniporter complex, a mitochondrial inner membrane transporter that is a primary mode of Ca2+ import into the mitochondrial matrix 10,11,12. Identification of these structural and regulatory subunits of the uniporter has brought forth the possibility of genetically targeting mitochondrial Ca2+ influx to modulate mitochondrial function and dysfunction and facilitated the study of the contribution of the uniporter complex and mitochondrial Ca2+ influx to disease13,14,15. Indeed, mitochondrial Ca2+ signaling has been implicated in the pathologies of a diverse array of diseases ranging from cardiac disease to neurodegeneration, and cancer16,17,18,19,20.
Given the fundamental importance of mitochondrial Ca2+ signaling in metabolism and cell death, and combined with the broad reach of biological systems that mitochondrial Ca2+ signaling impacts, methods to assess mitochondrial Ca2+ influx are of great interest. Not surprisingly, a variety of techniques and tools to measure mitochondrial Ca2+ have been developed. These include methods that utilize tools such as fluorescent Ca2+-sensitive dyes21,22 and genetically-encoded Ca2+ sensors targeted to the mitochondria, such as cameleon and aequorin23,24. The goal of this article is to highlight different methods and model systems in which mitochondrial Ca2+ uptake can be measured. We present two experimental methods to assess mitochondrial Ca2+ influx capacity. Using cardiac mitochondria as an example, we detail a plate reader-based platform for measuring mitochondrial Ca2+ uptake using the Ca2+ sensitive dye calcium green-5N that is ideally suited for isolated tissue mitochondria14. Using cultured NIH 3T3 cells, we also describe a confocal microscopy imaging-based assay for measurement of mitochondrial Ca2+ in permeabilized cells using the Ca2+ sensitive dye Rhod-2/AM25.
Protocol
All methods described in this protocol have been approved by the Institutional Animal Care and Use Committee of Emory University.
NOTE: The first part is the experimental procedure for measuring mitochondrial Ca2+ influx in isolated cardiac mitochondria using a plate reader.
1. Reagents and Solutions
- Make 500 mL of MS-EGTA buffer for mitochondrial isolation: 225 mM mannitol, 75 mM sucrose, 5 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), 1 mM ethylene glycol-bis(β-aminoethyl ether)-N,N,N',N'-tetraacetic acid (EGTA), pH adjusted to 7.4 with KOH. Sterilize it through a 0.22 μm filter and store it at 4 °C. Ensure that the MS-EGTA buffer is pre-chilled to 4 °C before use.
- Prepare 100 mL of KCl Buffer: 125 mM KCl, 20 mM HEPES, 1 mM KH2PO4, 2 mM MgCl2, 40 μM EGTA and pH adjusted to 7.2 with KOH. Store it at room temperature.
- Prepare 1 mM calcium green-5N stock in dimethyl sulfoxide (DMSO). The calcium green-5N stock can be aliquotted and stored at -20 °C.
- Prepare substrates for Ca2+ uptake.
- Prepare 1 M sodium pyruvate, pH 7.4. Store it in aliquots at -20 °C.
- Prepare 500 mM malate, pH 7.4. Store it in aliquots at -20 °C.
2. Isolation of Cardiac Mitochondria
- Euthanize the mouse according to institutional standards.
NOTE: For our institutionally approved method of euthanasia, mice were anesthetized by isofluorane inhalation and sacrificed by cervical dislocation. - Collect the heart by opening chest cavity, cutting along either side of the ribs, flanking the heart, cutting away the diaphragm, and excising the heart tissue.
NOTE: In this protocol, mitochondrial isolation is performed from a whole heart collected from an adult mouse (approximately 120 mg), which should be sufficient for 3-4 experiments. For mitochondrial isolation from other mitochondria-rich tissues such as the liver, up to 200 mg of tissue can be used following the protocol described. - Rinse the tissue thoroughly in 25 mL of ice-cold 1x phosphate buffered saline (PBS) ensuring that all blood is squeezed out of the ventricles.
NOTE: The heart will be sufficiently rinsed when the liquid squeezed out of the heart runs clear. - Mince the heart into small pieces in 5 mL of ice-cold 1x PBS using a pair of sharp scissors.
- Discard the PBS and transfer minced heart tissue to a pre-chilled 7 mL glass-teflon dounce homogenizer with a 0.10 to 0.15 mm clearance.
- Add 5 mL of ice-cold MS-EGTA buffer, and homogenize the sample until tissue pieces are no longer visible (approximately 11 strokes).
NOTE: Do not over-homogenize the tissue as the goal is to obtain intact and functional mitochondria. - Transfer the homogenate to a 15 mL tube.
- Centrifuge the homogenate at 600 x g at 4 °C for 5 min to pellet nuclei and unbroken cells.
- Transfer the supernatant to a fresh 15 mL tube and centrifuge it at 10,000 x g at 4 °C for 10 min to pellet mitochondria.
- Discard the supernatant and keep the mitochondrial pellet on ice.
- Wash the mitochondrial pellet twice using ice-cold MS-EGTA buffer. For each wash, resuspend the mitochondrial pellet in 5 mL of MS-EGTA buffer, centrifuge it at 10,000 x g at 4 °C for 10 min, and discard the supernatant.
- After the final wash, discard the supernatant and resuspend the mitochondria in 100 μL of ice cold MS-EGTA buffer. Keep the mitochondria on ice.
NOTE: Mitochondria should be used for experimentation within 1 h. - Measure mitochondrial protein concentration using a Bradford Protein Assay26.
3. Plate Reader-based Measurement of Mitochondrial Calcium Uptake
NOTE: Here, it is described the protocol for analyzing mitochondrial Ca2+ uptake using a multimode plate reader fitted with injectors. Any plate reader with the capability of reading calcium green-5N fluorescence (excitation/emission of 506/532 nm) in a kinetic mode with automated reagent injectors to keep the reaction protected from light can be used.
- Program the plate reader to perform a kinetic read of calcium green-5N fluorescence with measurements taken every second for a total assay time of 1,000 s. Additionally, program the reagent injectors to dispense 5 μL of CaCl2 solution at the 30 s, 150 s, 300 s, 480 s, and 690 s time points.
NOTE: The timing of the CaCl2 injections are user defined, and can be adjusted for according to experimental needs. - Prime the reagent injectors with the CaCl2 solution to be used.
NOTE: The concentration of the CaCl2 solution is user defined, and can be adjusted in subsequent runs to titrate the amount of Ca2+ required to trigger MPTP opening. - Add 200 μg of mitochondria to an individual well of a 96 well plate.
- Add the appropriate volume of KCl buffer to the well such that the total volume of mitochondria and KCl buffer comes to 197 μL.
- Add 1 μL of 1 M pyruvate, and 1 μL of 500 mM malate to the mitochondria mixture. Pipet gently to mix, and incubate the mitochondria with the substrates for 2 min at room temperature to allow mitochondria to become energized.
- Add 1 μL of 1 mM calcium green-5N stock. Mix gently by pipetting.
NOTE: Protect the reaction from light following the addition of the calcium green-5N dye. - Start the pre-programmed kinetic protocol and monitor calcium green-5N fluorescence.
4. Reagents and Solutions
NOTE: The second part is experimental procedure for confocal imaging of mitochondrial Ca2+ in cultured cells
- Make Tyrode's solution (130 mM NaCl, 4 mM KCl, 2 mM CaCl2, 1 mM MgCl2, 10 mM glucose, and 10 mM HEPES, pH 7.2 with KOH). Store it at 4 °C. Warm it to room temperature before use.
- Prepare Wash Solution (100 mM potassium acetate, 15 mM KCl, 5 mM KH2PO4, 5 mM Mg-ATP, 0.35 mM EGTA, 0.12 mM CaCl2, 0.75 mM MgCl2, 10 mM phosphocreatine, 10 mM HEPES, pH 7.2 with KOH). Store it at 4 °C. Warm it to room temperature before use.
- Prepare Permeabilization Solution, which contains 0.005% saponin in Wash Solution. Prepare it fresh daily.
- Prepare 0 Ca2+ Internal Solution (100 mM potassium acetate, 15 mM KCl, 0.35 mM EGTA, 0.75 mM MgCl2, 10 mM HEPES, pH adjusted to 7.2 with KOH). Store it at 4 °C. Warm it to room temperature before use.
- Prepare internal solution containing Ca2+. Add CaCl2 to the internal solution above to reach the desired concentration of free Ca2+. The amount of CaCl2 to add is calculated using the MaxChelator program (maxchelator.stanford.edu).
- Prepare 1 mM Rhod-2/AM stock in DMSO and store it at -20 °C until use.
- Prepare 1 mM MitoTracker green stock in DMSO and store it at -20 °C until use.
5. Plating Cells for Imaging
- Wash 22 x 22 cm2 glass coverslips with 100% ethanol and allow the coverslips to air dry.
- Place the glass coverslips into individual wells of a 6-well tissue culture dish.
NOTE: Coverslips can be coated with laminin, poly-L-lysine, or similar matrix to promote cell attachment. - Trypsinize and plate the cells onto the coverslips aiming for approximately 70% confluence on the day of imaging.
6. Loading Cells with the Rhod-2/AM and MitoTracker Green
- Prepare Rhod-2/AM-MitoTracker green working solution. Add 20 μL of Rhod-2/AM 1 mM stock, 0.2 μL of MitoTracker green 1 mM stock, and 2.5 μL of 20% pluronic F-127 to 1 mL of Tyrode's solution. The final concentration of Rhod-2 in the working solution is 20 µM and the final concentration of MitoTracker green is 200 nM.
- Gently remove the growth media from the coverslip.
NOTE: A wash step is not necessary prior to dye solution addition. - Add the Rhod-2/AM-MitoTracker green solution in a dropwise fashion onto the coverslip until the coverslip is just covered (approximately 3-4 drops per coverslip).
- Incubate it for 30 min at room temperature protected from light to allow the cells to load with the dyes.
- De-esterify the Rhod-2/AM. Gently remove the Rhod-2/AM-MitoTracker green solution, replace it with fresh room temperature Tyrode's solution, and incubate it for 30 min at room temperature protected from light.
7. Confocal Imaging of the Mitochondrial Rhod-2/AM and MitoTracker Green Fluorescence
- Transfer the coverslip to the microscope imaging chamber and fill the chamber with wash solution.
- Under settings for observing cells in phase contrast at 40X, adjust focus until cells are visible and in focus.
- Permeabilize the plasma membrane of Rhod-2/AM-MitoTracker green loaded cells to remove cytosol-localized Rhod-2, while retaining mitochondria-localized dye.
- Remove the Wash Solution from the coverslip.
- Replace it with Permeabilization Solution for approximately 1 min.
- Visually monitor the plasma membrane morphology throughout the permeabilization process. Permeabilized cells will develop a roughened surface.
- When permeabilization is complete, immediately remove the Permeabilization Solution and replace it with 0 Ca2+ Internal Solution.
- Simultaneously image Rhod-2 fluorescence (excitation using the 559 nm laser line and emission collected at wavelengths between 575-675 nm) and MitoTracker green fluorescence (excitation using the 488nm laser line and emission collected at wavelengths between 505-525 nm). Focus on permeabilized cells displaying a clear colocalization of Rhod-2 and MitoTracker green.
- Decrease the microscope laser and gain settings such that the mitochondrial Rhod-2 fluorescence is dim and just visible.
- Select microscope settings to acquire 2-dimensional scans at an appropriate frame rate and time course for the specific application. A frame rate of at least 30 frames/s is recommended to accurately capture the kinetics of changes in mitochondrial Ca2+.
- Remove the 0 Ca2+ Internal Solution without disturbing the cells or microscope focus.
- Start image acquisition.
- Add Ca2+-replete Internal Solution at the 10 s time point either manually or with a perfusion system.
- Select regions of interest (ROIs) to encompass regions of colocalization between mitochondrial Rhod-2 and MitoTracker green signal in the image acquisition software.
NOTE: Arbitrary fluorescence (F) values for each time point of the recording are obtained for each ROI. These values are background subtracted and normalized to the initial fluorescence (prior to Ca2+ addition; F0) and plotted as F/F0 over time for data presentation and quantification of the amplitude of the change of mitochondrial Ca2+.
Representative Results
Figure 1 shows mitochondrial Ca2+ uptake measurements in isolated cardiac mitochondria using the plate reader-based platform and the Ca2+ dye calcium green-5N. Under control conditions (Figure 1A), cardiac mitochondria were suspended in KCl buffer containing calcium green-5N and then challenged with sequential pulses of CaCl2 (5 μL of a 0.6 mM CaCl2 solution) added at the 30 s, 150 s, 300 s, 480 s, and 690 s time points. The timing and number of CaCl2 additions can be adjusted by user to allow for complete mitochondrial uptake of added Ca2+ prior to subsequent additions. In this assay, increases in calcium green-5N signal reflect elevated buffer Ca2+ levels. As mitochondria import Ca2+, Ca2+ is removed from the buffer and the calcium green-5N fluorescence decreases. Importantly, at the third addition of Ca2+, the calcium green-5N fluorescence curve undergoes a sudden and sharp inflection upwards, instead of continuing to remove Ca2+ from the buffer and is indicated by the red arrow in Figure 1A. This sudden increase in fluorescence reflects mitochondrial Ca2+ overload and MPTP opening. The total amount of Ca2+ taken up by mitochondria prior to MPTP activation reflects the mitochondrial Ca2+ capacity, and can be expressed as μmol Ca2+/mg protein. By making adjustments to the timing and concentrations of calcium added, the amount of Ca2+ required to trigger permeability transition can be determined. In Figure 1B, mitochondria were pre-treated for 10 min at room temperature with 10 μM Ru360, a well-characterized inhibitor of the mitochondrial Ca2+ uniporter complex27. Ru360 inhibits uniporter-dependent mitochondrial Ca2+ uptake, and this is evidenced by the step-wise increases in calcium green-5N fluorescence following each Ca2+ addition.
For confocal imaging of mitochondrial calcium using Rhod-2/AM, we show the representative results from NIH 3T3 cells. MitoTracker green is a mitochondria-selective dye that preferentially stains the mitochondrial network (Figure 2A). Following saponin-mediated permeabilization of the plasma membrane, cytosolic Rhod-2 is washed away, leaving a Rhod-2 stained mitochondrial network which co-localizes with the MitoTracker green (Figure 2A). Rhod-2 may also accumulate in non-mitochondrial structures, so ROI's to be analyzed should focus of regions of co-localization between the MitoTracker green and Rhod-2. In control cells, the addition of a Ca2+-replete internal solution containing 2 μM free Ca2+ causes a rapid rise in Rhod-2 fluorescence, which is inhibited when uniporter complex is inhibited with 10 μM Ru360 (Figure 2B).
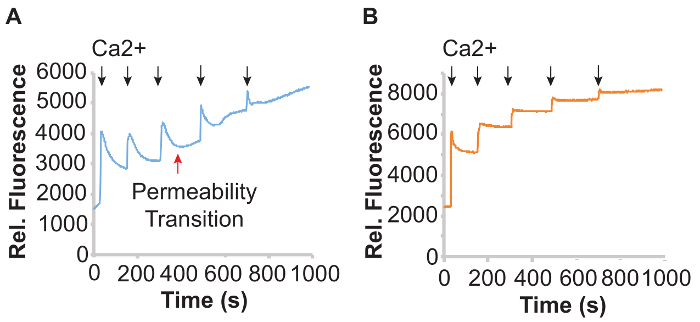
Figure 1: Mitochondrial Ca2+ uptake in isolated cardiac mitochondria. (A) Graphs of relative calcium green-5N fluorescence of control heart mitochondria and (B) mitochondria pre-treated with 10 μM Ru360 for 10 min at room temperature, challenged with 5 μL of 0.6 mM CaCl2 (black arrows). Mitochondrial Ca2+ overload-induced permeability transition is indicated with the red arrow. Please click here to view a larger version of this figure.
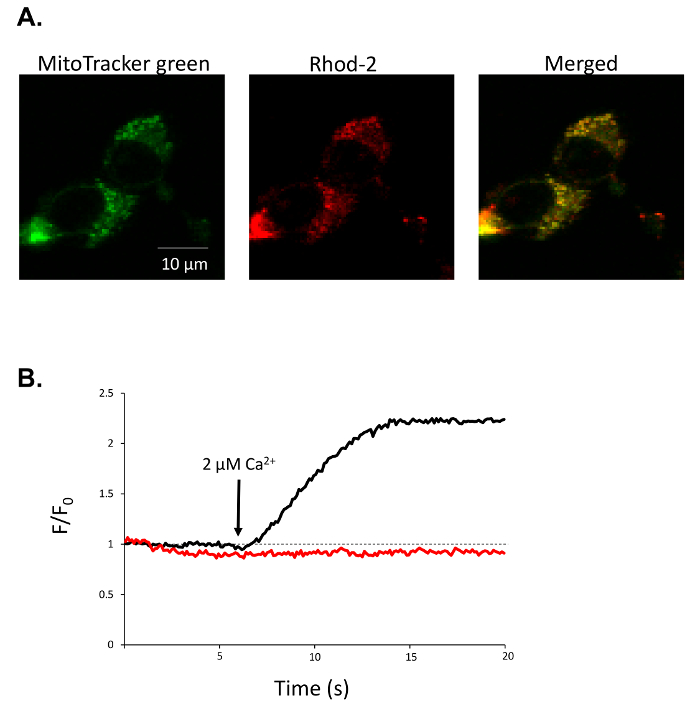
Figure 2: Representative analysis of mitochondrial Ca2+ in permeabilized cells. (A) Representative fluorescence image of 3T3 cells loaded with rhod-2 (red) and MitoTracker green (green) and merged images of rhod-2 and MitoTracker green (yellow) after permeabilization with saponin. (B) Rhod-2 fluorescent trace from a single 3T3 cell during application of 2 µM free Ca2+ internal solution under control conditions (black trace) or after 10 µM Ru360 pre-treatment (red trace). Please click here to view a larger version of this figure.
Discussion
Here, we describe two different approaches to measure mitochondrial Ca2+ influx. The plate reader-based calcium green-5N method monitors extramitochondrial Ca2+ levels and is a Ca2+ uptake assay that is well suited for measurements in isolated mitochondria. While we have shown representative results from isolated murine cardiac mitochondria, this assay can be readily adapted for mitochondria isolated from tissues with high mitochondrial abundance including the liver, skeletal muscle, and brain. Moreover, the plate reader system may be an ideal option for laboratories where specialized equipment traditionally used for Ca2+ measurements, like fluorometers, may not be immediately available. The limited maximal time span that a plate reader can scan and the need to pre-program reagent additions may be a weakness of this technique compared to fluorometers, however, the cost and availability of fluorometers may outweigh this benefit. By varying the concentration of Ca2+ in the reagent injectors, the amount of Ca2+ that mitochondria are able to sequester before triggering MPTP opening (the mitochondrial Ca2+ capacity) can be determined.
The confocal imaging protocol that we describe for Rhod-2/AM measurements of mitochondrial Ca2+ is ideal for cultured cells and can also be adapted for primary cells. In this method, the plasma membrane is permeabilized with saponin, which allows for the washout of cytosolic dye. Only dye entrapped in organelles remains after this procedure, allowing accurate and specific measurement of mitochondrial fluorescence. Moreover, this permeabilized cell assay allows for experimental control over extramitochondrial Ca2+ levels as well as user-defined conditions under which the mitochondria are exposed. Rhod-2/AM confocal imaging of mitochondrial Ca2+ may be used in intact, non-permeabilized cells but this requires the optimization of dye loading and de-esterification to ensure a mitochondria-specific Rhod-2 localization28. The strengths of the Rhod-2/AM method are that Rhod-2/AM is commercially available and the dye can be readily applied to a wide variety of cell types. The localization of the Rhod-2 dye, however, is a limitation to be considered. To ensure the measurement of mitochondria-specific Rhod-2 fluorescence, a second spectrally-distinct dye, such as MitoTracker green, may be used to stain mitochondria prior to imaging. Genetically-encoded Ca2+ sensors targeted to the mitochondria may circumvent this issue, however, the cells need to be transfected/transduced with the sensor construct and have enough time to express the protein. This is not always ideal for primary cells with limited survival time, and dye loading may be a timesaving process.
In both protocols, we used the uniporter complex inhibitor Ru360 to illustrate the effect mitochondrial Ca2+ influx inhibition. Ru360 is the gold standard for uniporter inhibition and to date, it is the only known specific inhibitor of this tranporter29. Alternatively, ruthenium red may also be used, however, ruthenium red is a nonspecific uniporter inhibitor that has been shown to also inhibit Ca2+ release from the sarcoplasmic reticulum30. Impaired mitochondrial Ca2+ handling can be a sign of mitochondrial dysfunction, as uniporter complex-dependent Ca2+ transport is mitochondrial membrane potential dependent, and by extension, this is reliant on respiratory chain function. Thus, mitochondrial uncouplers, such as carbonyl cyanide-4-(trifluoromethoxy)phenylhydrazone (FCCP) and carbonyl cyanide m-chlorophenyl hydrazone (CCCP) which dissipate mitochondrial membrane potential, may also be used as control compounds to inhibit mitochondrial Ca2+ influx. Since mitochondria are sites of intracellular Ca2+ storage, mitochondria may function as Ca2+ buffers within the intracellular space. Thus, alterations in mitochondrial Ca2+ handling may impact the shape of the cytosolic Ca2+ landscape. Considering the growing number of disease conditions where mitochondrial Ca2+ dynamics may play a role, the methods illustrated here could be applied to the study of mitochondrial Ca2+ influx in animal models and cellular models of the disease.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work was supported by grant funding from the American Heart Association (J.Q.K.).
Materials
| Olympus FV1000 Laser Scanning confocal microscope | Olympus | FV1000 | |
| Synergy Neo2 Multimode microplate reader with injectors | Biotek | ||
| Tissue Homogenizer | Kimble | 886000-0022 | |
| 22 x 22 mm coverslips | Corning | 2850-22 | |
| 96 well plate | Corning | 3628 | |
| 6 well plate | Corning | 3506 | |
| Calcium Green-5N | Invitrogen | C3737 | |
| MitoTracker green FM | Invitrogen | M7514 | |
| Rhod-2, AM | Invitrogen | R1244 | |
| DMSO | Invitrogen | D12345 | |
| Pluronic F-127 | Invitrogen | P3000MP | |
| D-Mannitol | Sigma | M9546 | |
| Sucrose | EMD Millipore | 8510 | |
| HEPES | Sigma | H3375 | |
| EGTA | Sigma | E8145 | |
| Potassium chloride | Fisher | BP366-500 | |
| Potassium phosphate monobasic | Sigma | P0662 | |
| Magnesium chloride | Sigma | M2670 | |
| Sodium pyruvate | Sigma | P2256 | |
| L-malic acid | Sigma | M1125 | |
| Calcium chloride | Sigma | C4901 | |
| Potassium acetate | Fisher | BP364-500 | |
| Adenosine 5′-triphosphate magnesium salt | Sigma | A9187 | |
| Phosphocreatine disodium salt | Sigma | P7936 | |
| Saponin | Sigma | S7900 | |
| Ru360 | Calbiochem | 557440 |
Riferimenti
- Deluca, H. F., Engstrom, G. W. Calcium uptake by rat kidney mitochondria. P Natl Acad Sci USA. 47, 1744-1750 (1961).
- Lehninger, A. L., Rossi, C. S., Greenawalt, J. W. Respiration-dependent accumulation of inorganic phosphate and Ca ions by rat liver mitochondria. Biochem Biophys Res Co. 10, 444-448 (1963).
- Kwong, J. Q. The mitochondrial calcium uniporter in the heart: energetics and beyond. J Physiol. 595 (12), 3743-3751 (2017).
- Denton, R. M. Regulation of mitochondrial dehydrogenases by calcium ions. Biochim Biophys Acta. 1787 (11), 1309-1316 (2009).
- Jouaville, L. S., Pinton, P., Bastianutto, C., Rutter, G. A., Rizzuto, R. Regulation of mitochondrial ATP synthesis by calcium: evidence for a long-term metabolic priming. P Natl Acad Sci USA. 96 (24), 13807-13812 (1999).
- Haworth, R. A., Hunter, D. R. The Ca2+-induced membrane transition in mitochondria. II. Nature of the Ca2+ trigger site. Arch Biochem Biophys. 195 (2), 460-467 (1979).
- Hunter, D. R., Haworth, R. A. The Ca2+-induced membrane transition in mitochondria. I. The protective mechanisms. Arch Biochem Biophys. 195 (2), 453-459 (1979).
- Kwong, J. Q., Molkentin, J. D. Physiological and pathological roles of the mitochondrial permeability transition pore in the heart. Cell Metab. 21 (2), 206-214 (2015).
- Luongo, T. S., et al. The mitochondrial Na+/Ca2+ exchanger is essential for Ca2+ homeostasis and viability. Nature. 545 (7652), 93-97 (2017).
- De Stefani, D., Patron, M., Rizzuto, R. Structure and function of the mitochondrial calcium uniporter complex. Biochim Biophys Acta. 1853 (9), 2006-2011 (2015).
- Baughman, J. M., et al. Integrative genomics identifies MCU as an essential component of the mitochondrial calcium uniporter. Nature. 476 (7360), 341-345 (2011).
- De Stefani, D., Raffaello, A., Teardo, E., Szabo, I., Rizzuto, R. A forty-kilodalton protein of the inner membrane is the mitochondrial calcium uniporter. Nature. 476 (7360), 336-340 (2011).
- Pan, X., et al. The physiological role of mitochondrial calcium revealed by mice lacking the mitochondrial calcium uniporter. Nat Cell Biol. 15 (12), 1464-1472 (2013).
- Kwong, J. Q., et al. The Mitochondrial Calcium Uniporter Selectively Matches Metabolic Output to Acute Contractile Stress in the Heart. Cell Rep. 12 (1), 15-22 (2015).
- Luongo, T. S., et al. The Mitochondrial Calcium Uniporter Matches Energetic Supply with Cardiac Workload during Stress and Modulates Permeability Transition. Cell Rep. 12 (1), 23-34 (2015).
- Brown, D. A., et al. Expert consensus document: Mitochondrial function as a therapeutic target in heart failure. Nat Rev Cardiol. 14 (4), 238-250 (2017).
- Logan, C. V., et al. Loss-of-function mutations in MICU1 cause a brain and muscle disorder linked to primary alterations in mitochondrial calcium signaling. Nat Genet. 46 (2), 188-193 (2014).
- Lewis-Smith, D., et al. Homozygous deletion in MICU1 presenting with fatigue and lethargy in childhood. Neurol Genet. 2 (2), e59 (2016).
- Tosatto, A., et al. The mitochondrial calcium uniporter regulates breast cancer progression via HIF-1alpha. EMBO Mol Med. 8 (5), 569-585 (2016).
- Cardenas, C., et al. Selective Vulnerability of Cancer Cells by Inhibition of Ca(2+) Transfer from Endoplasmic Reticulum to Mitochondria. Cell Rep. 15 (1), 219-220 (2016).
- Dedkova, E. N., Blatter, L. A. Calcium signaling in cardiac mitochondria. J Mol Cell Cardiol. 58, 125-133 (2013).
- Florea, S. M., Blatter, L. A. The role of mitochondria for the regulation of cardiac alternans. Front Physiol. 1, 141 (2010).
- Palmer, A. E., Tsien, R. Y. Measuring calcium signaling using genetically targetable fluorescent indicators. Nat Protoc. 1 (3), 1057-1065 (2006).
- Bonora, M., et al. Subcellular calcium measurements in mammalian cells using jellyfish photoprotein aequorin-based probes. Nat Protoc. 8 (11), 2105-2118 (2013).
- Zima, A. V., Kockskamper, J., Mejia-Alvarez, R., Blatter, L. A. Pyruvate modulates cardiac sarcoplasmic reticulum Ca2+ release in rats via mitochondria-dependent and -independent mechanisms. J Physiol. 550 (Pt 3), 765-783 (2003).
- Kruger, N. J. The Bradford method for protein quantitation. Methods Mol Biol. 32, 9-15 (1994).
- Zazueta, C., Sosa-Torres, M. E., Correa, F., Garza-Ortiz, A. Inhibitory properties of ruthenium amine complexes on mitochondrial calcium uptake. J Bioenerg Biomembr. 31 (6), 551-557 (1999).
- Davidson, S. M., Duchen, M. R. Imaging mitochondrial calcium signalling with fluorescent probes and single or two photon confocal microscopy. Methods Mol Biol. 810, 219-234 (2012).
- Matlib, M. A., et al. Oxygen-bridged dinuclear ruthenium amine complex specifically inhibits Ca2+ uptake into mitochondria in vitro and in situ in single cardiac myocytes. J Biol Chem. 273 (17), 10223-10231 (1998).
- Chamberlain, B. K., Volpe, P., Fleischer, S. Inhibition of calcium-induced calcium release from purified cardiac sarcoplasmic reticulum vesicles. J Biol Chem. 259 (12), 7547-7553 (1984).

