Optimizing the Use of a Liquid Handling Robot to Conduct a High Throughput Forward Chemical Genetics Screen of Arabidopsis thaliana
Summary
A high throughput screen of synthetic small molecules was conducted on the model plant species, Arabidopsis thaliana. This protocol, developed for a liquid handling robot, increases the speed of forward chemical genetics screens, accelerating the discovery of novel small molecules affecting plant physiology.
Abstract
Chemical genetics is increasingly being employed to decode traits in plants that may be recalcitrant to traditional genetics due to gene redundancy or lethality. However, the probability of a synthetic small molecule being bioactive is low; therefore, thousands of molecules must be tested in order to find those of interest. Liquid handling robotics systems are designed to handle large numbers of samples, increasing the speed with which a chemical library can be screened in addition to minimizing/standardizing error. To achieve a high-throughput forward chemical genetics screen of a library of 50,000 small molecules on Arabidopsis thaliana (Arabidopsis), protocols using a bench-top multichannel liquid handling robot were developed that require minimal technician involvement. With these protocols, 3,271 small molecules were discovered that caused visible phenotypic alterations. 1,563 compounds induced short roots, 1,148 compounds altered coloration, 383 compounds caused root hair and other, non-categorized, alterations, and 177 compounds inhibited germination.
Introduction
In the past 20 years researchers in the field of plant biology have made great strides using chemical genetics approaches, both forward and reverse, improving our understanding of cell wall biosynthesis, the cytoskeleton, hormone biosynthesis and signaling, gravitropism, pathogenesis, purine biosynthesis, and endomembrane trafficking1,2,3,4,5. Employing forward chemical genetics techniques enables the identification of phenotypes of interest and allows researchers to understand the genotypic underpinnings of particular processes. Conversely, reverse chemical genetics seeks out chemicals that interact with a pre-determined protein target6. Arabidopsis has been at the forefront of these discoveries in plant biology because its genome is small, mapped, and annotated. It has a short generation time, and there are multiple mutant/reporter lines available to facilitate the identification of aberrant subcellular machinery7.
There are two major bottlenecks that slow the progress of forward chemical genetic screens, the initial screening process and determining the target of the compound of interest8. A major aid in increasing the speed of small molecule selection is the use of automation and automated equipment9. Liquid handling robots are an excellent tool for handling large libraries of small molecules and have been instrumental in driving progress in the biological sciences10. The protocol presented here is designed to alleviate the bottleneck associated with the screening process, enabling the identification of bioactive small molecules at a rapid rate. This technique decreases the burden of labor and time on behalf of the operator while also decreasing the economic cost to the principle investigator.
Thus far, most chemical libraries analyzed have held between 10,000 and 20,000 compounds, some with as many as 150,000 and some with as few as 709,11,12,13,14,15,16. The protocol introduced herein was implemented on a small molecule library of 50,000 compounds (see Table of Materials), one of the larger forward chemical genetics screens conducted on Arabidopsis to date. This protocol fits with the current trend towards increased efficiency and speed regarding forward chemical genetics, especially as it pertains to herbicide discovery, insecticide discovery, fungicide discover, drug discovery, and cancer biology17,18,19,20,21. Though implemented here with Arabidopsis, this protocol, could easily be adapted to cell cultures, spores, and potentially even insects in liquid medium within 96-, 384-, or 1536-well plates. Due to its small size, Arabidopsis is amenable to screening in 96 well plates. However, distributing seeds evenly among wells is a challenge. Hand seeding is accurate but labor intensive, and though there are devices designed to dispense seeds into 96-well plates, they are expensive to purchase. Here, we show how this step can be circumvented with just a small loss in accuracy.
The overall goal of this method was to make screening a large chemical library against Arabidopsis more manageable, without compromising accuracy, via the use of a liquid handling robot. The use of this method improves the efficiency of the researcher by decreasing the time taken to complete initial dilution series management and subsequent phenotypic screens, allowing quick visualization of samples under a dissecting microscope, and rapid identification of novel bioactive small molecules. Figure 1 depicts this protocol's key outcomes in 4 steps.

Figure 1: Overall workflow of the forward chemical genetics screen. An overview of the protocol to be described with some detail for each of the 4 key steps. 1: Receiving the Chemical Library, 2: Making the Dilution Library, 3: Making the Screening Plates, and 4: Incubating and visualizing the Screening Plates. Please click here to view a larger version of this figure.
Protocol
1. Creating a Dilution Library
- Label 625 Dilution Library plates by hand, ensuring that they match to their corresponding plate from the chemical library. Additionally, connect in flow and out flow hoses to the Multichannel Tip Wash Automated Labware Positioner (ALP) by passing them through the Console Drive to the 5 Gallon Reservoir (see Table of Materials).
- Access the computer and turn on the wash pump through the connection of the Device Controller to the Multichannel Tip Wash ALP in order to circulate water. This will turn off automatically at the end of the protocol.
- Load, by hand, the Stacker 10's attached to the Stacker Carousel, in the following order in Hotels A – D (Figure 4, Stacker); one box of AP96 P20 Pipette Tips in Room 1, four 96-Well V-Bottom Plates in Rooms 2 – 5 with the two upper plates containing stock concentrations from the ordered library and the two lower plates empty (Figure 5, Stacker). Additionally, load one box of AP96 P20 Pipette Tips in Room 6, and four 96-Well V-Bottom Plates in Rooms 7 – 9 with the two upper plates containing stock concentrations of the ordered library and the two lower plates empty (Figure 5, Stacker).
- Set up, by hand, the deck with a 300 mL water reservoir on P3, a 300 mL 70% EtOH bath on P7, Tip Loader ALP (TL1), and Multichannel Tip Wash ALP (TW1) (Figure 4, Deck and Figure 5, Deck).
- Using the operating software, present AP96 P20 Pipette Tips from the Stacker 10 and move them to the Tip Loader ALP.
NOTE: 1.5 through 1.12 all are done with the liquid handling robot's operating software; see Table of Materials. - Present Room 2 from Hotel A and separate all four 96-Well V-Bottom Plates on the deck, placing the bottom two on P4 and P8 and the top two on P5 and P9 (Figure 4).
- Load AP96 P20 Pipette Tips with the Tip Loader ALP onto the 96-Channel 200 µL Head. Aspirate 90 µL from the 300 mL water reservoir and dispense into the 96-Well V-Bottom Dilution Plate on P4. Repeat this step for the plate on P8.
- Mix the chemical library plate on P5 by repetitively aspirating and dispensing 15 µL three times. Additionally, aspirate 10 µL from the chemical library plate on P5 and dispense 10 µL into the dilution plate on P4.
- Mix the solutions of the plate on P4 by repetitively aspirating and dispensing 50 µL a total of three times. Once mixed, clean the AP96 P20 Pipette Tips by aspirating and dispensing 70 µL of 70% EtOH from P7, then washing them in the Multichannel Tip Wash ALP by aspirating and dispensing a 110% volume of water four times.
- Repeat steps 1.8 – 1.9 for the second pair of plates on P8 and P9. Upon creating the second 96-Well V-Bottom Dilution Plate, stack the plates in the following order from bottom to top: P9, P5, P8, and P4. Then, place the stack on one empty Static ALP; either P1, P2, P6, P10, P11, P12, or P13.
- Repeat steps 1.6 – 1.10 until Room 5 in Hotel A is empty. Repeat step 1.5 upon reaching Room 6, moving new AP96 P20 Pipette Tips to the Tip Loader ALP and placing the used AP96 P20 Pipette Tips on an empty Static ALP.
- Repeat steps 1.6 – 1.10 until Room 9 of Hotel A is empty. However, in order to proceed to Hotel B, the plates and tips on the deck must be reloaded into Hotel A.
- Re-fill, by hand, the 300 mL water reservoir. This step is crucial, and the computer program can incorporate a pause detailing this message, requiring the user to hit 'continue', before carrying out the next step.
- Repeat steps 1.5 – 1.13 for the remaining hotels, ensuring a full 300 mL water reservoir each time prior to proceeding to the next hotel.
2. Adding Media-seed Mixture to Screening Plates
- Make ½ Murashige and Skoog (MS) Media with 0.1% Agar by the addition of 4.3 g MS Salts, 0.50 g MES, 1.0 g Agar to 1 L DI H2O. Adjust the pH to 5.7 though the addition of 5 M potassium hydroxide while monitoring with a pH probe.
- Sterilize seeds by shaking them in 1% bleach and SDS between 15 and 30 min, and then rinse 4 times with an equal volume of water by centrifugation. Once seeds are sterile, place them at 4 °C from 24 hours to 7 days for vernilization. The Arabidopsis Biological Resource Center describes additional methods of sterilization, vernilization, and growth22.
- Add seeds to media by hand at a density of 0.1 g/100 mL. This density results in an average of 3 – 10 seeds per well of a 96-well plate.
- Place, by hand, four 96-Well Flat-Bottom Plates in Rooms 1 and 2 of Hotel A (Figure 6, Hotel A). Place a box of AP96 P250 Pipette Tips on the Tip Loader ALP, a 300 mL reservoir filled with the media-seed mixture created in steps 2.1 – 2.3 on P3, and a 300 mL reservoir filled with 70% EtOH on P7 (Figure 4, Deck and Figure 6, Deck).
NOTE: 2.5 through 2.8 are done with the operating software. - Present Rooms 1 and 2 in Hotel A, and separate the stacks of four plates. Place one plate on each of the empty static ALPs (P4, P5, P6, P8, P9, P10, P11, and P12). Load AP96 P250 Pipette Tips on the 96-Channel 200 µL Head.
- Aspirate 90 µL from the 300 mL media-seed reservoir on P3 and dispense into the first 96-Well Flat-Bottom Plate. Repeat this process until all eight plates contain the media-seed mixture.
- Clean the AP96 P250 Pipette Tips by aspirating and dispensing 70 µL from the 300 mL reservoir filled with 70% EtOH on P7. Wash the tips in the Multichannel Tip Wash ALP by aspirating and dispensing a 110% volume of water four times, unload the tips at TL1, and gather the plates by hand.
3. Adding Small Molecules to Screening Plates
- Load, by hand, a box of AP96 P250 Pipette Tips into Room 1 of Hotel A, two 96-Well V-Bottom Dilution Library Plates into Rooms 2, 4, 6, and 8, and two 96 Well Flat-Bottom Screening Plates into Rooms 3, 5, 7, and 9 (Figure 4, Stacker and Figure 7, Hotel A). Additionally, connect hoses to and from the Multichannel Tip Wash ALP to the 5 Gallon Reservoir.
NOTE: 3.2 through 3.10 are done with the operating software. - Configure the deck to contain a 300 mL 70% EtOH wash reservoir at P7; the media-seed reservoir can be left on the deck at P3 (Figure 4, Deck and Figure 7, Deck). Additionally, turn on the Console Drive through connection of the Device Controller to circulate water through the Multichannel Tip Wash ALP. This will turn off automatically at the end of the protocol.
- Present the AP96 P250 Pipette Tip Box from Hotel A and move it to the Tip Loader ALP.
- Present the 96-Well V-Bottom Dilution Library Plates from Room 2 of Hotel A to the deck and place one on Static ALP P4 and one on P8. Present the 96-Well Flat-Bottom Screening Plates from room 3 of Hotel A to the deck and place one on Static ALP P5 and one on P9.
- Load AP96 P250 Pipette Tips with the Tip Loader ALP onto the 96-Channel 200 µL Head.
- Mix the 96-Well V-Bottom Dilution Plate on P4 by aspirating and dispensing 50 µL three times. Following that, aspirate 10 µL from this plate and dispense into the 96-Well Flat-Bottom Screening Plate on P5.
- Mix the solutions in the plate at P5 by aspirating and dispensing 50 µL three times. Clean the AP96 P250 Pipette Tips with ethanol by aspirating and dispensing 70 µL of 70% EtOH from the reservoir at P7 and then wash the tips in the Multichannel Tip Wash ALP by aspirating and dispensing a 110% volume of water four times.
- Repeat steps 3.5 and 3.6 for the second 96-Well V-Bottom Dilution Library Plate (P8) and 96-Well Flat-Bottom Screening Plate (P9).
- Stack the two 96-Well V-Bottom Dilution Library Plates together and the two 96-Well Flat Bottom Screening Plates together. Move the plates to Static ALPs P1, P2, P6, P10, P11, P12, or P13.
- Repeat steps 3.4 – 3.9 three times, adding diluted chemicals to screening plates a total of eight times. Finally, check the number of seeds in each well of the screening plates through visual conformation and supplement those wells with fewer than three seeds by additional sterilized and vernalized seed.
4. Incubation and Visualization of Screening Plates
- Incubate the 96-well Flat-Bottom Screening Plates for four days in an environmental chamber at 22 °C on a 16/8 light/dark cycle in a desiccation proof container. Visualize the 96-well Flat-Bottom Screening Plates under a dissecting microscope. Record all aberrant phenotypes for further investigation.
Representative Results
The ability to accurately and efficiently characterize phenotypes based on the addition of small molecules at screening concentrations under a dissecting microscope is the end goal of this method of forward chemical genetics on Arabidopsis. The phenotypes observed when all 50,000 compounds had been screened was diverse and could be broken into several distinct classes (Figure 2). Figure 3A-F depicts examples of phenotypes that were observed at low magnification under a dissecting microscope. Some phenotypes provided inconclusive results (Figure 3G, H). These had to be retested at lower concentrations to ensure that the chemical didn't provide a different phenotype at a lower dose.
Poor results can arise for many reasons. One is poor germination rates of the seeds. This can cause a screening plate to predominantly exhibit no germination or incomplete germination phenotypes (Figure 3G, H), which can be misleading. To overcome this, pre-test germination rates for all seeds used. Once germination rates have been established, and are greater than 95% for Arabidopsis, vernalization of seed prior to chemical addition is a key step ensuring simultaneous germination. Lack of simultaneous germination can lead to false positives in phenotyping. In addition to this, poor results can arise if media is allowed to evaporate during incubation. This lack of hydration prevents seeds from germinating and can be avoided through the use of desiccation-proof containers. Additionally, DMSO and media solutions are in the two external columns of every plate, ensuring proper micro climates and germination rates are obtained.
Satisfactory experimental results are achieved when germination rates are >95%, seeds are vernalized prior to addition into 96-well flat-bottom plates, ensuring simultaneous germination, and media does not evaporate during incubation. Ideally, chemicals would be tested at a concentration that allows all seeds to germinate and phenotypes to be assessed accurately (Figure 3A-F). The majority of the treatments produced seedlings with phenotypes that were visually indistinguishable from mock controls with no morphological phenotypes (Figure 3A), with the vast majority of aberrant phenotypes consisting of bleached and severely stunted roots (Figure 2).
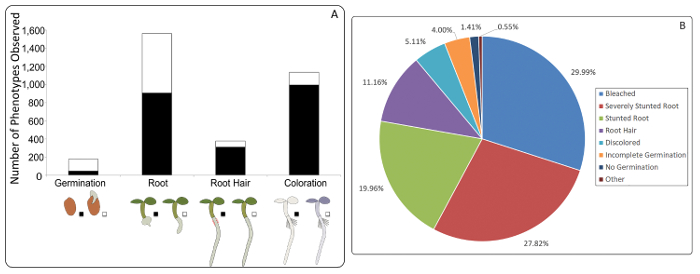
Figure 2: The most common phenotypes observed and the proportion of each phenotype observed. A) A total of 3,271 small molecules were found to be bioactive at 100 µM after four days of incubation. The color indicates the severity of the phenotype (black = more severe, white = less severe). The most commonly observed phenotype pertained to root morphology, with more than 1,500 compounds inducing stunted roots of varying severity. Coloration was also commonly affected by the compounds in this library, with 1,148 seedlings recorded as entirely bleached or partially discolored. Just under 400 compounds produced distinctive root hair phenotypes – either stunted or both stunted and brightly colored. Finally, germination was affected by just under 200 compounds. In these cases, seeds either did not complete germination or did not even begin to germinate. B) Seedlings exhibiting abnormalities in root morphology, either being stunted or severely stunted, constituted almost half of all phenotypically aberrant seedlings. The next largest group were those that resulted in bleached or discolored seedlings. Phenotypes that pertained to root hair abnormalities also made up a sizeable portion while the inhibition of germination, either no germination or incomplete germination, only occurred in a small percentage of all bioactive compounds. Finally, there were a number of phenotypes that occurred at such a low frequency, they were grouped into the category 'other', an example of which was the production of green mucilage, seen in Figure 3. Please click here to view a larger version of this figure.

Figure 3: Images of representative phenotypes observed during the forward chemical genetics screen. No visible morphological abnormalities (A), brown root hairs (B), stunted root (C), severely stunted root (D), bleached (E), green mucilage (F), incomplete germination (G), and no germination (H). Please click here to view a larger version of this figure.
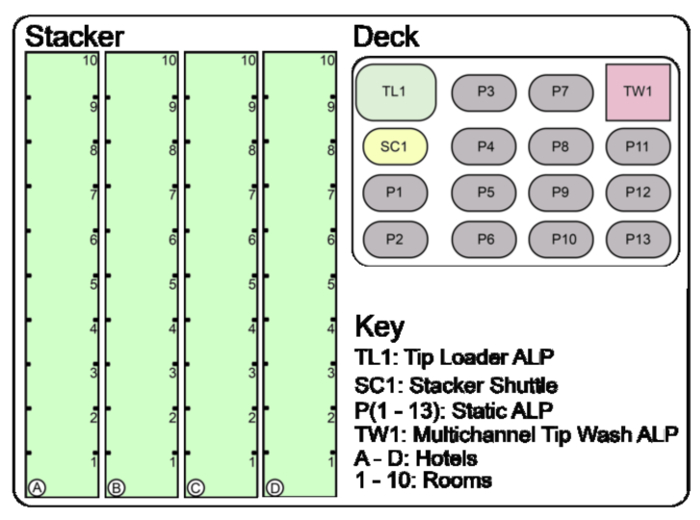
Figure 4: Overview of the Stacker 10 and deck set up before initiation of protocol. The Stacker Carousel is comprised of four Stacker 10's (Hotels A – D) which each accommodate ten Rooms. The Deck holds a variety of ALPs: the Tip Loader, the Stacker Shuttle, the Multichannel Tip Wash, and 13 static ALPs. Please click here to view a larger version of this figure.
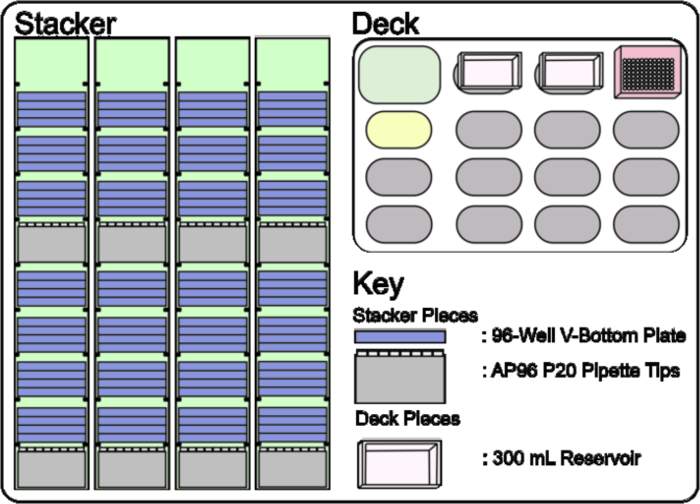
Figure 5: Stacker 10 and Deck set up required for Creating a Dilution Library. The four Stacker 10's are loaded with boxes of AP96 P20 Pipette Tips in Rooms 1 and 6 of Hotels A – D and stacks of four 96-Well V-Bottom Plates in Rooms 2 – 5 and rooms 7 – 9 of Hotels A – D. The deck layout consists of two 300 mL Reservoirs on static ALPs P3 and P7. Please click here to view a larger version of this figure.
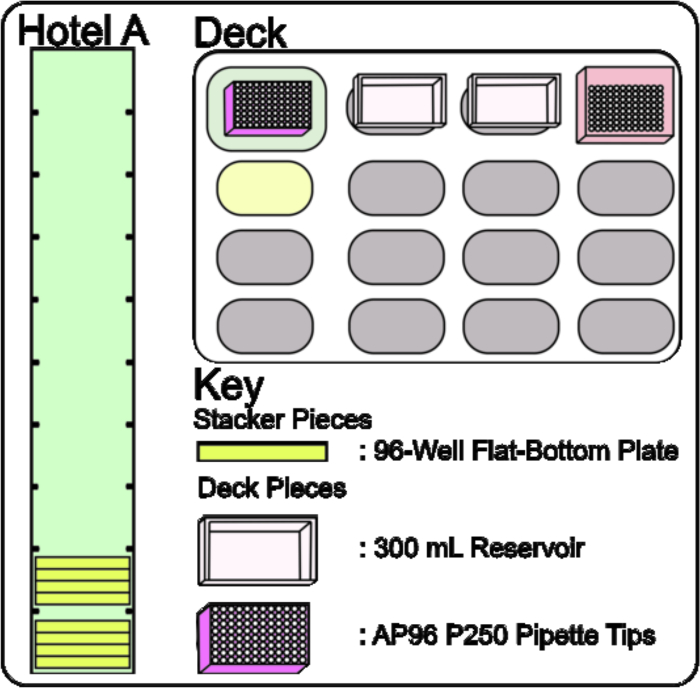
Figure 6: Stacker 10 and Deck set up for Adding Media-Seed Mixture to Screening Plates. The Stacker 10 is loaded with four 96-Well Flat-Bottom Plates in Rooms 1 and 2 of Hotel A. The deck layout consists of two 300 mL Reservoirs on Static ALPs P3 and P7 and a box of AP96 P250 Pipette Tips on TL1. Please click here to view a larger version of this figure.
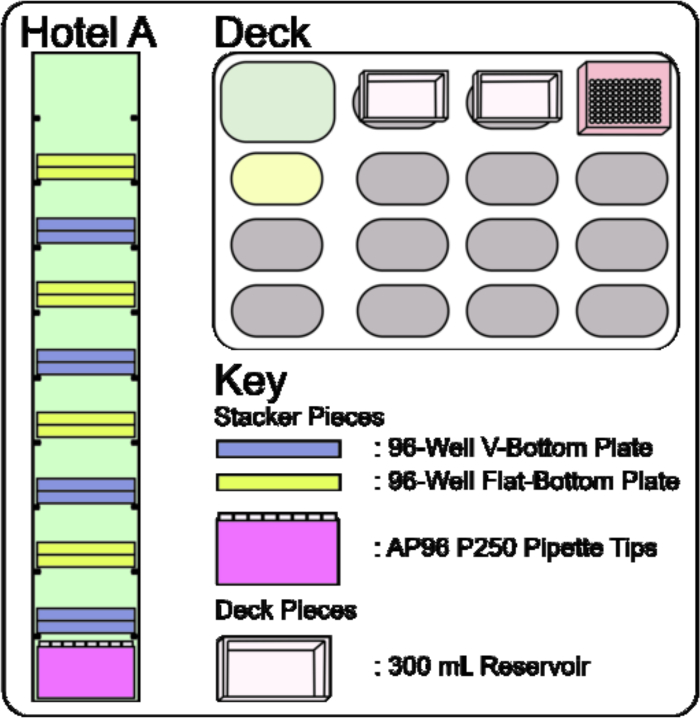
Figure 7: Stacker 10 and Deck set up for Adding Small Molecules to Screening Plates. The Stacker 10 is loaded with a box of AP96 P250 Pipette Tips in Room 1, a stack of two 96-Well V-Bottom Dilution Plates in Rooms 2, 4, 6, and 8, and a stack of two 96-Well Flat-Bottom Plates filled with the media-seed mixture in Rooms 3, 5, 7, and 9. The deck layout consists of two 300 mL reservoirs on Static ALPs P3 and P7. Please click here to view a larger version of this figure.
Discussion
This protocol is designed to aid researchers in accomplishing a forward chemical genetics screen on Arabidopsis. We provide representative results from a screen of 50,000 compounds (Figure 2 and Figure 3), one of the largest forward chemical genetics screens performed on Arabidopsis to date9,13,23. The use of a liquid handling robot enabled more efficient dilution library and screening library generation, improving the speed and efficiency of identification of novel compounds. Increasing the capacity to screen in a high throughput nature was accompanied by decreasing labor on behalf of the researcher. This technique was designed to be used with 96-well plates, which can accommodate small seeds or plants visible under a dissecting microscope. Utilizing plates with larger wells to fit larger seeds would require modifications to the throughput and design.
Additional limitations of this technique include the difficulty of using this piece of equipment with aseptic techniques; however, we did not encounter high percentages of contamination due to ½ MS media lacking sucrose. One could circumvent any contamination issue by placing the robot in a sterile room, allowing for sterile conditions and cell culture, or by using a liquid handling robot with a sterile chamber24,25. Another limitation is tip size and seed aspiration. A small pipette tip such as the AP96 P20 would clog with seeds; therefore, a larger pipette tip must be used for seed dispensing and solution mixing.
Critical steps within this protocol include the careful labeling of all plates in the dilution library and screening library, ensuring they are in the proper orientation along with correct order when feeding the robot. Clear labeling and systematic processing is straightforward and can overcome this issue. Another critical step is ensuring that the right equipment is in the correct place before starting the experiment, both within the Stacker 10's and on the deck. If the equipment is not properly placed on the deck, the 96-Channel 200 µL Head could crash, damaging the instrument and requiring maintenance. Another critical step is ensuring that the correct amount of liquid is placed within the 300 mL reservoirs and that this amount is entered correctly into the software. If the numbers don't match, the tips will not reach the liquid and aspiration will not occur.
It is also necessary to take steps to ensure that results obtained are accurate. One error that we noticed while developing the protocol was linked to tip life. After successive loading and unloading, the tips lose their ability to aspirate and dispense accurately. It is therefore imperative that each set of 96 tips is used a maximum of four times. It is also important to change the wash water regularly to avoid the potential for chemicals to be inadvertently added to screening plates. Finally, some chemicals have a tendency to precipitate out of solution26. To ensure each chemical is added at the correct concentration, mixing steps are incorporated into the dilution and screening protocol. Failure to mix could result in low quantities of chemicals being added from library to library, challenging interpretation of potential chemical induced phenotypes.
Using the correct plates for each part of the protocol is also very important. V-Bottom plates are designed to ensure that small volumes of liquid can be aspirated and are recommended for use in the creation of the Dilution Library. However, these plates are not suitable for the screening portion of the protocol, since their reflection of light leads to poor visualization of phenotypes. In order to observe the phenotypes of 3 – 4 day old seedlings, the screen must be carried out in flat-bottom plates.
Once the screening plates have been created, visualization is required. 96-well flat-bottom plates allow easy visualization under dissecting microscopes. It is imperative that the plates are stored in desiccation-proof containers to reduce the evaporation of the media. An alternative to microscopic visualization is using a high-resolution scanner. Images produced at high resolution reveal the majority of the phenotypes observed in this screen and provide an archive of the results that can be revisited in the future. Once visualization is complete, and the library of your choice screened, this method could then be performed on different organisms or with a different chemical library. Modifications to the equipment could allow for sterile culture, allowing ventures into the realms of stem cells, fungus, insects, and small plants2,18,25,27.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We thank Jozsef Stork, Mitchel Richmond, Jarrad Gollihue, and Andrea Sanchez for constructive and critical discussion. Dr. Sharyn Perry for the phenotypic photographs. This material is based upon work supported by the National Science Foundation under Cooperative Agreement No. 1355438.
Materials
| Keyboard | Local Provider | N/A | Used for protocol design and operating the Biomek FX |
| Mouse | Local Provider | N/A | Used for protocol design and operating the Biomek FX |
| Computer Screen | Local Provider | N/A | Used for protocol design and operating the Biomek FX |
| Computer | Local Provider | N/A | Used for protocol design and operating the Biomek FX |
| DIVERSet Diverse Screening Library | ChemBridge | N/A | Chemical library |
| Biomek Software | Beckman Coulter | N/A | Runs and designs the Biomek FX |
| Device Controller | Beckman Coulter | 719366 | Operates the water pump/tip washing station |
| Stacker Carousel Pendent | Beckman Coulter | 148240 | Manual operation of Biomek Stacker Carousel |
| Biomek Stacker Carousel | Beckman Coulter | 148520 | Rotary unit that houses all FX Stacker 10's |
| FX Stacker 10 | Beckman Coulter | 148522 | Elevator unit that houses components for screen |
| FX Stacker 10 | Beckman Coulter | 148522 | Elevator unit that houses components for screen |
| FX Stacker 10 | Beckman Coulter | 148522 | Elevator unit that houses components for screen |
| FX Stacker 10 | Beckman Coulter | 148522 | Elevator unit that houses components for screen |
| Biomek FX | Beckman Coulter | https://www.beckman.com/liquid-handlers | Robot that performs the desired operations |
| Accuframe | Artisan Technology Group | 76853-4 | Frames arm to place components corretly |
| Framing Fixture | Beckman Coulter | 719415 | Centers arm in the Accuframe |
| Multichannel Tip Wash ALP | Beckman Coulter | 719662 | Washes the tips after the ethanol bath |
| Tip Loader ALP | Beckman Coulter | 719356 | Pneumatically loads tips onto the arm |
| Air Compressor | Local Provider | N/A | Provides air for pneumatic tip loading |
| MasterFlex Console Drive | Cole-Parmer | 77200-65 | Pump used to circulate water through the Multichannel Tip Washer |
| Air Hose | Local Provider | N/A | Provides air from air compressor to Tip Loader |
| Water Hose | Local Provider | N/A | Provides water from 5 Gallon Reserviour to Tip Washer |
| Static ALP's | Beckman Coulter | Comes with Biomek FX | Supports equipment for the Screen |
| 5 Gallon Reserviour | Local Provider | N/A | Recirculates the dirty water from cleaning the tips |
| Grippers | Beckman Coulter | Comes with Biomek FX | Grabs and moves the equipment to the correct places |
| 96-Channel 200 µL Head | Beckman Coulter | Comes with Biomek FX | Holds the 96 tips used within the screen |
| AP96 P200 Pipette Tips | Beckman Coulter | 717251 | Used to make the screening library |
| 96 Well Flat Bottom Plate | Costar | 9018 | Aids in visulization of screen |
| 96 Well V-Bottom Plate | Costar | 3897 | Aids in storing of dilution library |
| AlumaSeal 96 Sealing Film | MedSci | F-96-100 | Seals for storage both the chemicle library and dilution library |
| Plastic ziplock sandwich bags | Local Provider | N/A | Used to ensure a humid environment for screen |
| AP96 P20 Pipette Tips | Beckman Coulter | 717254 | Used in the dilution library creation |
| Growth Chamber | Percival | AR36L3 | Germinates seeds for phenotypic visulization |
| Spatula | Local Provider | N/A | Holds seeds to add into wells where liquid seeding failed seed adequatly |
| Toothpick | Local Provider | N/A | Pushes seeds from spatula to wells |
| Murashige and Skoog Basal Salt Mixture | PhytoTechnology Laboratories | M524 | Add to MS media mixture |
| MES Free Acid Monohydrate | Fisher Scientific | ICN19483580 | Added to MS media to decrease pH |
| Agar Powder | Alfa Aesar | 9002-18-0 | Increases thickness of media to support seed suspension |
| 5M KOH | Sigma-Aldrich | 484016 | Increases pH to adequate levels |
| 1L Media Storage Bottle | Corning | 1395-1L | Holds enough media for a screen |
| Polypropylene Centrifuge Tubes | Corning | 431470 | Sterilizes seeds prior to vernilization |
| pH Probe | Davis Instruments | YX-58825-26 | Used for making media |
| ALPs (Automated Labware Positioners) Users Manual | Beckman Coulter | PN 987836 | Aids in setting up the accompaning equipment for the Biomek FX |
| Biomek 2000 Stacker Carousel Users Guide | Beckman Coulter | 609862-AA | Aids in setting up the Stacker Carousel |
| Biomek FX and FXP Laboratory Automation Workstations Users Manual | Beckman Coulter | PN 987834 | Used to frame the Multichannel Pod |
| Biomek FXP Laboratory Automation Workstation Customer Startup Guide | Beckman Coulter | PN B32335AB | Used to aid in setting up the Biomek FX |
| Biomek Software User's Manual | Beckman Coulter | PN 987835 | Used to set up and understand the Software |
Riferimenti
- Blackwell, H. E., Zhao, Y. Chemical genetic approaches to plant biology. Plant Physiol. 133 (2), 448-455 (2003).
- Dejonghe, W., Russinova, E. Plant chemical genetics: From phenotype-based screens to synthetic biology. Plant Physiol. 174 (1), 5-20 (2017).
- McCourt, P., Desveaux, D. Plant chemical genetics. New Phytol. 185 (1), 15-26 (2010).
- Lumba, S., Cutler, S., McCourt, P. Plant nuclear hormone receptors: A role for small molecules in protein-protein interactions. Annu Rev Cell Dev Biol. 26, 445-469 (2010).
- Hicks, G. R., Raikhel, N. Opportunities and challenges in plant chemical biology. Nat Chem Biol. 5 (5), 268-272 (2009).
- De Rybel, B., et al. A role for the root cap in root branching revealed by the non-auxin probe naxillin. Nat Chem Biol. 8 (9), 798-805 (2012).
- Koornneef, M., Meinke, D. The development of Arabidopsis as a model plant. Plant J. 61 (6), 909-921 (2010).
- Serrano, M., Kombrink, E., Meesters, C. Considerations for designing chemical screening strategies in plant biology. Front Plant Sci. 6, 131 (2015).
- Yoshitani, N., et al. A structure-based strategy for discovery of small ligands binding to functionally unknown proteins: Combination of in silico screening and surface plasmon resonance measurements. Proteomics. 5 (6), 1472-1480 (2005).
- Macarron, R., et al. Impact of high-throughput screening in biomedical research. Nat Rev Drug Discov. 10 (3), 188-195 (2011).
- DeBolt, S., et al. Morlin, an inhibitor of cortical microtubule dynamics and cellulose synthase movement. Proc Natl Acad Sci U S A. 104 (14), 5854-5859 (2007).
- Christian, M., Hannah, W. B., Luthen, H., Jones, A. M. Identification of auxins by a chemical genomics approach. J Exp Bot. 59 (10), 2757-2767 (2008).
- Drakakaki, G., et al. Clusters of bioactive compounds target dynamic endomembrane networks in vivo. PNAS. 108 (43), 17850-17855 (2011).
- Armstrong, J. I., Yuan, S., Dale, J. M., Tanner, V. N., Theologis, A. Identification of inhibitors of auxin transcriptional activation by means of chemical genetics in Arabidopsis. Proc Natl Acad Sci U S A. 101 (41), 14978-14983 (2004).
- Brown, L. A., et al. A small molecule with differential effects on the PTS1 and PTS2 peroxisome matrix import pathways. Plant J. 65 (6), 980-990 (2011).
- De Rybel, B., et al. Chemical inhibition of a subset of Arabidopsis thaliana GSK3-like kinases activates brassinosteroid signaling. Chem Biol. 16 (6), 594-604 (2009).
- Arkin, M. R., Tang, Y., Wells, J. A. Small-molecule inhibitors of protein-protein interactions: progressing toward the reality. Chem Biol. 21 (9), 1102-1114 (2014).
- St Onge, R., Schlecht, U., Scharfe, C., Evangelista, M. Forward chemical genetics in yeast for discovery of chemical probes targeting metabolism. Molecules. 17 (11), 13098-13115 (2012).
- Vassilev, L. T., et al. In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science. 303 (5659), 844-848 (2004).
- Zhao, Y., et al. Chemical genetic interrogation of natural variation uncovers a molecule that is glycoactivated. Nat Chem Biol. 3 (11), 716-721 (2007).
- Walsh, T. A. The emerging field of chemical genetics: Potential applications for pesticide discovery. Pest Manag Sci. 63 (12), 1165-1171 (2007).
- . Seed Handling Available from: https://abrc.osu.edu/seed-handling (2013)
- Knoth, C., Salus, M. S., Girke, T., Eulgem, T. The synthetic elicitor 3,5-dichloroanthranilic acid induces NPR1-dependent and NPR1-independent mechanisms of disease resistance in Arabidopsis. Plant Physiol. 150 (1), 333-347 (2009).
- Conway, M. K., et al. Scalable 96-well Plate based iPSC culture and production using a robotic liquid handling system. J Vis Exp. , (2015).
- Daniszewski, M., et al. Automated cell culture systems and their applications to human pluripotent stem cell studies. SLAS Technol. , (2017).
- Popa-Burke, I., Russell, J. Compound precipitation in high-concentration DMSO solutions. J Biomol Screen. 19 (9), 1302-1308 (2014).
- Partridge, F. A., et al. An automated high-throughput system for phenotypic screening of chemical libraries on C. elegans and parasitic nematodes. Cold Spring Harb Protoc. , (2017).

