Probe Type II Band Alignment in One-Dimensional Van Der Waals Heterostructures Using First-Principles Calculations
Summary
Calculations performed by the Vienna Ab initio Simulation Package can be used to identify the intrinsic electronic properties of nanoscale materials and predict the potential water-splitting photocatalysts.
Abstract
Computational tools based on density-functional theory (DFT) enable the exploration of the qualitatively new, experimentally attainable nanoscale compounds for a targeted application. Theoretical simulations provide a profound understanding of the intrinsic electronic properties of functional materials. The goal of this protocol is to search for photocatalyst candidates by computational dissection. Photocatalytic applications require suitable band gaps, appropriate band edge positions relative to the redox potentials. Hybrid functionals can provide accurate values of these properties but are computationally expensive, whereas the results at the Perdew-Burke-Ernzerhof (PBE) functional level could be effective for suggesting strategies for band structure engineering via electric field and tensile strain aiming to enhance the photocatalytic performance. To illustrate this, in the present manuscript, the DFT based simulation tool VASP is used to investigate the band alignment of nanocomposites in combinations of nanotubes and nanoribbons in the ground state. To address the lifetime of photogenerated holes and electrons in the excited state, nonadiabatic dynamics calculations are needed.
Introduction
The worldwide demand for clean and sustainable energy has spurred research for promising materials to reduce dependence on finite petroleum resources. Simulations are more efficient and economical than experiments in accelerating the search for new functional materials1. Material design from a theoretical perspective2,3,4 is now more and more popular due to rapid advances in computational resources and theory developments, making computational simulations more reliable5. The density functional theory (DFT) calculations implemented in many codes are becoming more robust and yield reproducible results6.
The Vienna Ab initio Simulation Package (VASP)7 presents one of the most promising DFT codes for predicting molecular and crystalline properties and more than 40,000 studies making use of this code have been published. Most work is performed at the Perdew-Burke-Ernzerhof (PBE) functional level8, which underestimates the band gap sizes, but captures the essential trends in band alignment and band offsets3. This protocol aims to outline the details of investigating the band edge profiles and bandgaps of nanoscale materials for clean and renewable energy using this computational tool. More examples using VASP are available at https://www.vasp.at.
This report presents the computational screening of one-dimensional (1D) vdW heterostructures with type II band alignments9 for a promising application in photocatalytic water splitting4. Specifically, nanoribbons (NRs) encapsulated inside nanotubes (NTs) are examined as an example10. To address noncovalent interactions, vdW corrections using the DFT-D3 method are included11. The DFT calculations in steps 1.2, 2.2, 3.2, 3.5.2, and section 4 by VASP are performed using a Portable Batch System (PBS) script by the high-performance research computers in the CenTOS system. An example of a PBS script is shown in the Supplementary Materials. The data postprocessing by the P4VASP software in step 3.3 and the figure plot by the xmgrace software in step 3.4 are carried on a local computer (laptop or desktop) in the Ubuntu system.
Protocol
1. Optimize the atomic structure.
- Prepare four input files for structure relaxation calculation by VASP: INCAR, POSCAR, POTCAR, and KPOINTS.
NOTE: There are specified parameters in the INCAR file that define the calculation. The line "EDIFFG = 0.02" in the INCAR file indicates that all atoms are relaxed until the force on each atom is <0.02 eV/Å. The POSCAR file contains the atomic geometry information. The initial lattice parameters in the POSCAR file can be chosen from theoretical3 or experimental references12,13. The KPOINTS file defines the k point mesh and POTCAR is the pseudopotential file. The order of atom types in POSCAR should be the same as that in POTCAR. Examples of input files for structure relaxation are shown in the Supplementary Materials (except the pseudopotential file, which needs a license from VASP).- Generate the initial structure of boron nitride (BN) nanoribbons (NR) for "POSCAR".
- Download the POSCAR file for the BN bulk unit from https://materialsproject.org.
- Use v2xsf to convert the POSCAR file to a file in xsf format that can be read by xcrysden. Type v2xsf POSCAR on the terminal in the Ubuntu system to get "POSCAR.xsf.gz". Type gunzip POSCAR.xsf.gz and output the POSCAR.xsf file.
- Use xcrysden to build the BN supercell.
- Tipo xcrysden –xsf POSCAR.xsf on the terminal in the Ubuntu system. Select the menu Modify/Number of Units Drawn and extend the cell in the X and Y directions.
- Select the menu File/Save XSF Structure to export the supercell structure, named "supercell".
NOTE: The name of the structure is an arbitrary definition.
- Use xmakemol to open the supercell. Type xmakemol -f supercell on the terminal in the Ubuntu system. Select the menu Edit/Visible. Click Toggle to delete the atoms inside the region and cut the NR to the desired width and chirality.
- Generate the initial structure of the BN nanotube (NT) for POSCAR. Download "NanotubeModeler" from http://www.jcrystal.com/products. Open NanotubeModeler.exe in the Windows system. Select the menu Select type/B-N and specify the chirality. Select the menu File/Save XYZ table to export the structure.
- Generate the initial structure of the nanocomposite by encapsulating the NR (from step 1.1.1) inside the NT (from step 1.1.2).
NOTE: The encapsulation can be finished by adjusting the Cartesian coordinates of the NR and the NT10,14,15. - Use the vmd software to check the atomic structure before submitting the calculation job.
- Tipo vmd on the terminal in the Ubuntu system. In the opened vmd main window, select the menu File/New Molecule and find the POSCAR file through the Esplora window. Load POSCAR by typing VASP_POSCAR.
- Display the structure in different styles in the Graphical Representations/Drawing Method window.
NOTE: For example, once the CPK is chosen, each atom (bond) is represented by a sphere (stick). The installation guide and full tutorial of vmd are available at http://www.ks.uiuc.edu/Research/vmd.
- Generate the initial structure of boron nitride (BN) nanoribbons (NR) for "POSCAR".
- Tipo qsub job.pbs on the terminal in the Linux system to submit the job to the computer cluster.
NOTE: "job.pbs" represents the name of the PBS script. The name of the PBS script is an arbitrary definition. The four input files together with the PBS script should be at the working directory. The command qsub job.pbs will be used in steps 2.2, 3.2, 3.5.2, and section 4. An example of a PBS script can be found in the supplementary coding file. After the submitted job is finished, if "reached required accuracy – stopping structural energy minimization" appears at the end of the output log, the converged result is obtained. The resulting CONTCAR file will be used as the input file POSCAR in steps 2.1, 3.1, 3.5.1, 3.5.3.1, 4.1.1, 4.1.4, and section 4.2.
2. Calculate the encapsulation energy.
- Tipo mkdir nanocomposite isolated-nanoribbon isolated-nanotube to create three folders for the nanocomposite, the NR, and the NT on a terminal in a Linux system. Prepare one PBS script "job.pbs" and four input files INCAR, POSCAR, POTCAR, and KPOINTS for the energy calculation in each folder.
NOTE: The input file POSCAR is the file named CONTCAR with the relaxed structure from step 1. Examples of input files are given in Supplementary Materials (except POTCAR). - Go to each folder and type qsub job.pbs on the terminal in the Linux system.
NOTE: The three submitted jobs will perform the static self-consistent energy calculations for the nanocomposite, isolated NR, and isolated NT, respectively. - Extract the total energy from the file OUTCAR for each system after finishing the static self-consistent calculations. Type grep “free energy TOTEN” ./nanocomposite/OUTCAR | tail -n 1, grep “free energy TOTEN” ./isolated-nanoribbon/OUTCAR | tail -n 1, and grep “free energy TOTEN” ./isolated-nanotube/OUTCAR | tail -n 1. Define the three displayed values as ENT+NR, ENR, and ENT, respectively. Calculate the encapsulation energy per angstrom: EL = (ENT+NR – ENT -ENR)/L14,15.
NOTE: The periodical direction in each system is along the Z axis and L is the lattice constant of the unit cell along the Z axis. Test calculations of the energy dependence on the plane wave cutoff energy and the k point mesh are needed. The encapsulation energy can be used as an estimate for the energetic stability of the nanocomposite.
3. Extract the electronic properties from the band structure.
- Prepare one PBS script "job.pbs" and six input files: INCAR, POSCAR, POTCAR, KPOINTS, CHGCAR, and CHG for band calculation. Set ICHARG = 11 in INCAR.
NOTE: The preconverged CHGCAR and CHG files are from the static self-consistent calculations in step 2.2. The band analysis is at the PBE level. The k point sampling in the KPOINTS file is in line-mode. Examples of input files for this step can be found in the Supplementary Materials (except POTCAR). - Tipo qsub job.pbs on the terminal in the Linux system to submit the job.
- Use P4VASP to generate the projected band.
- Load "vasprun.xml" by typing p4v vasprun.xml on terminal in the Ubuntu system.
NOTE: "p4v" is used to start P4VASP. The file "vasprun.xml" should be at the working directory. - Select the menu Electronic/Local DOS+bands control and then Select/Bands.
- Specify the atomic numbers of NT in the section Atom selection. Get the atomic number by pointing to the corresponding atoms using vmd as mentioned in step 1.1.4. Specify the color, type, and size of the symbol for the projected band structure through the menu Symbol and Symbol size. Press the menu Add new line.
NOTE: The graph will show the band structure with contributions from the NT. - Repeat the same procedure following step 3.3.2.1 to get the projected band with contributions from the NR.
- Specify the atomic numbers of NT in the section Atom selection. Get the atomic number by pointing to the corresponding atoms using vmd as mentioned in step 1.1.4. Specify the color, type, and size of the symbol for the projected band structure through the menu Symbol and Symbol size. Press the menu Add new line.
- Select the menu Graph/Export. Export the graph into a file with an agr format (for example, as "11-4.agr").
NOTE: The output data of the projected bands by P4VASP are in three columns where the third one represents the weighting.
- Load "vasprun.xml" by typing p4v vasprun.xml on terminal in the Ubuntu system.
- Use xmgrace to edit the projected band.
- Tipo xmgrace 11-4.agr on the terminal to start xmgrace in the Ubuntu system. Select the menu Plot/Axis properties to edit the label and range of the axis.
- Select the menu Plot/Set appearance to read the energy value at the specified band number and k point.
NOTE: The valence band maximum (VBM) and conduction band minimum (CBM) of NR/NT can be read from the projected band with contributions on NR/NT, respectively. According to the band alignments, heterostructures can be classified into three types: type I (VBMNT <VBMNR <CBMNR <CBMNT or VBMNR <VBMNT <CBMNT <CBMNR), type II (VBMNT <VBMNR <CBMNT <CBMNR or VBMNR <VBMNT <CBMNR <CBMNT), or type III (VBMNT <VBMNT <CBMNR <CBMNR or VBMNR <VBMNR <CBMNT <CBMNT)9. - Calculate the valence band offset (VBO), conduction band offset (CBO), and the band gap following Kang et al.16.
- Select the menu File/Print to export the graph with eps format.
- Calculate the band decomposed charge density for the VBM and CBM.
- Prepare one PBS script "job.pbs" and seven input files: INCAR, POSCAR, POTCAR, KPOINTS, WAVECAR, CHGCAR, and CHG. Specify the band numbers for the CBM and VBM by the tag IBAND in INCAR. Use the single corresponding k point for each band edge.
NOTE: The preconverged CHGCAR, CHG, and WAVECAR files are from the static self-consistent calculations in step 2.2. Examples of input files for this step are given in the Supplementary Materials (except POTCAR). - Tipo qsub job.pbs on the terminal in the Linux system to submit the job.
- Use vmd to plot the VBM and CBM in real space after the job is finished.
- Start a vmd session and load the POSCAR file as in step 1.1.4.
- Select the menu File/New Molecule in the vmd main window. Find the PARCHG file through the Esplora window. Load PARCHG by typing VASP_PARCHG.
- Select the menus Draw/Solid Surface and Show/Isosurface in the Graphical Representations window. Change the isovalue to an appropriate value (for example, 0.02). Change the color of the isosurface through the menu Coloring Method.
NOTE: This is an intuitive analysis for band types with respect to that in step 3.4. Generally, the atomic structure is arranged away from the boundary, otherwise the visualized charge density is not shown in a continuous manner. Please see Supplemental Figure 1 for details.
- Prepare one PBS script "job.pbs" and seven input files: INCAR, POSCAR, POTCAR, KPOINTS, WAVECAR, CHGCAR, and CHG. Specify the band numbers for the CBM and VBM by the tag IBAND in INCAR. Use the single corresponding k point for each band edge.
4. Modulate the electronic properties of the nanocomposite (NT encapsulated inside NR) by external fields.
- Add a transverse electric field to the nanocomposite17.
- Prepare one PBS script "job.pbs" and four input files: INCAR, POSCAR, POTCAR, and KPOINTS.
- Define the strength of the electric field by the tag "EFIELD" in units of eV/Å.
- Set LDIPOL = T. Specify IDIPOL with an exact value (1, 2, or 3).
NOTE: These two tags are added to include dipole corrections. The electric field will be applied along the X, Y, or Z axis by setting the value of IDIPOL to 1, 2, or 3. - Perform the static self-consistent calculations and band structure calculations following sections 2 and 3 without structural optimization.
NOTE: Previous studies indicate that electric fields over 5 V/Å can be used to modify the band gap of BN-NT and BN-NR without deforming the structure18,19.
- Add a longitudinal tensile strain to the nanocomposite.
- Change the lattice parameters along the periodical direction to reflect the strain effect.
NOTE: For example, the optimized lattice parameter of the nanocomposite along the Z axis is 2.5045 Å. If 1% uniaxial tensile strain is applied along the Z direction, change the lattice parameter in POSCAR to 2.5045 x 1.01 = 2.529545 Å. - Relax the modified structure following section 1.
- Perform static self-consistent calculations and band structure calculations following sections 2 and 3.
- Change the lattice parameters along the periodical direction to reflect the strain effect.
Representative Results
Zigzag BN-NRs encapsulated inside armchair BN-NTs (11,11) were chosen as representative examples for a 1D vdW heterostructure. The lattice parameters were taken from Sahin et al.20. For convenience, zigzag NRs are abbreviated Zn, where n represents the III–V dimers along the width14. The encapsulation energy EL from step 2.3 was used as a rough estimate for the energetic stability of the nanocomposite. The EL values of Z2, Z3, and Z4 encapsulated inside BN-NT (11,11) were -0.033 eV/Å, -0.068 eV/Å, and -0.131 eV/Å, respectively10, as shown in Figure 1. Although EL varied by an order of magnitude with BN-NR size, all three nanocomposites presented type II band structures (from step 3.4) superior to the all-carbon cases14, where type II only emerged for NR with only one appropriate size inserted in NT14.
The band structure of the nanocomposite from step 3.2, BN-NT (11,11) + Z4, is shown in Figure 2. VBM/CBM locates at NT/NR (from step 3.5), respectively. The staggered band alignment was beneficial for light harvesting. The main mechanism of charge transfer is as follows: the photo generates electrons and a hole in Z4 at the X point, shown in Figure 3, and then the hole dissociates from Z4 (kX) to NT (11,11) (kVBM, the k point of VBM for this nanocomposite), shown in Figure 4. The calculated VBO (from step 3.4.3) is 317 meV, larger than the thermal energy at 300 K (KT ~30 meV), and effectively decreases the recombination rate of the photogenerated carriers10.
To enhance light harvesting through a wide spectrum, both transverse electric fields and longitudinal tensile strains are applied to BN-NT (11,11) + Z4. The evolution of band edges relative to the vacuum level from step 4 is shown in Figure 5. A substantial gap reduction up to near 0.95 eV is observed in this nanocomposite by external fields. More importantly, the staggered band alignment is preserved10. Based on these results, such a 1D system is expected to integrate photocatalytic hydrogen generation and safe capsule storage21. The photogenerated electrons could be collected by NR. Driven by electrostatic attraction, protons penetrate through the NT to generate a hydrogen molecule. The produced hydrogen is completely isolated within the nanotube to avoid an unwanted reverse reaction or explosion.
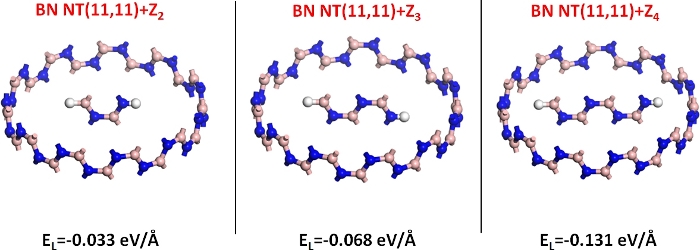
Figure 1: Zigzag BN nanoribbons Z2, Z3, and Z4 encapsulated inside a BN nanotube (11,11). The encapsulation energy (EL) is listed under each structure. Please click here to view a larger version of this figure.
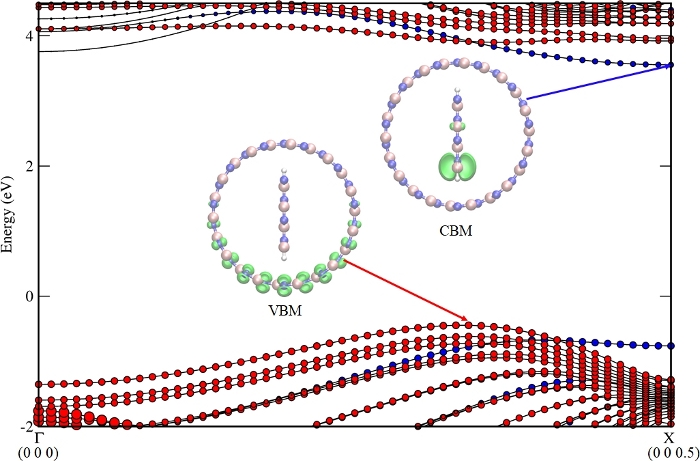
Figure 2: Band structure of BN nanotube (11,11) + BN nanoribbon Z4. The contributions from the nanotube and nanoribbon to the energy bands are represented in red and blue spheres, respectively. The left insets show the charge density distributions of the CBM and the VBM states (isovalue 0.02 e/Å3). This figure was adapted from Gong et al.10 with permission from The Royal Society of Chemistry. Please click here to view a larger version of this figure.
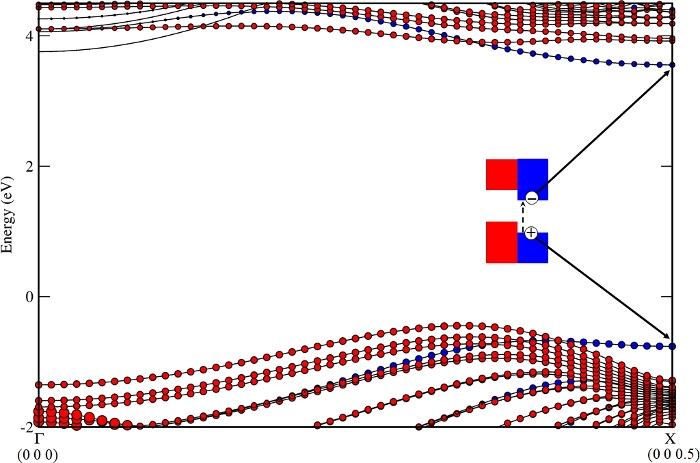
Figure 3: The photo generates electrons and a hole in the BN nanoribbon Z4 at the X point. Please click here to view a larger version of this figure.
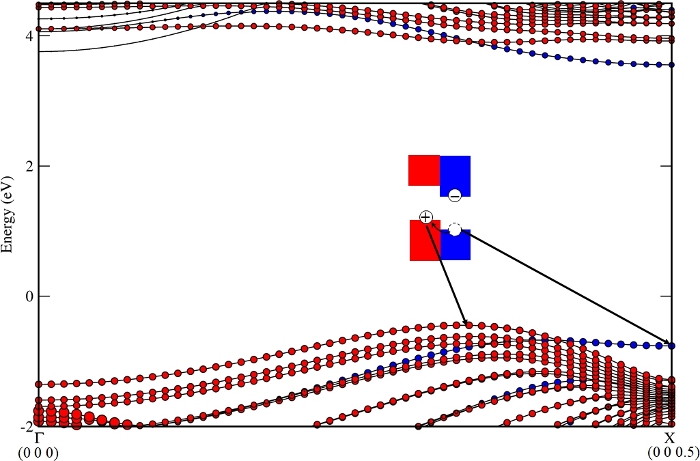
Figure 4: The hole dissociates from the BN nanoribbon Z4 (kX) to the BN nanotube (11,11) (kVBM, the k point of VBM for this nanocomposite). Please click here to view a larger version of this figure.
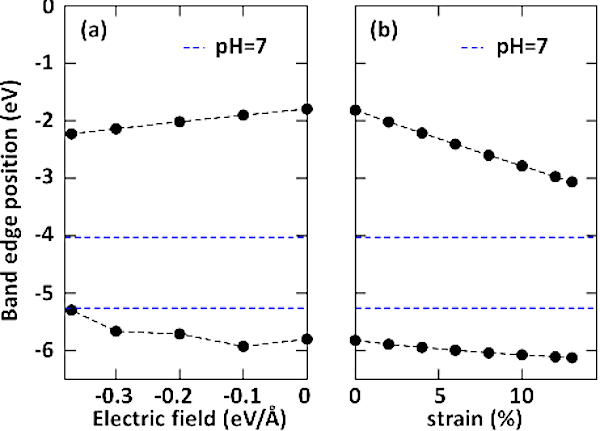
Figure 5: Band edge modulation of the BN nanotube (11,11) and the BN nanoribbon Z4 by external fields. Evolution of band edges relative to the vacuum level under (A) an electric field and (B) uniaxial tensile strain. The negative direction of electric field is denoted from the lower edge atom B to the upper edge atom N of Z4. The reduction potential of H+/H2 and the oxidation potential of O2/H2O are -4.44 eV and -5.67 eV at pH = 0, respectively. The pH = 7 shifts the water’s redox potentials (by pH x 0.059 eV) to -4.027 eV and -5.257 eV, respectively, shown as blue dashed lines. This figure was reproduced from Gong et al.10 with permission from The Royal Society of Chemistry. Please click here to view a larger version of this figure.
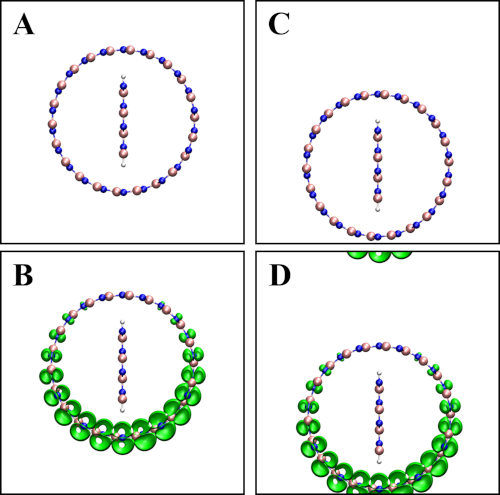
Supplemental Figure 1: (A) Atomic structure of a BN nanotube (11,11) + BN nanoribbon Z4 arranged away from the boundary and its corresponding conduction band minimum (B). (C) Atomic structure of a BN nanotube (11,11) and BN nanoribbon Z4 aligned with one boundary and its corresponding conduction band minimum (D). Please click here to view a larger version of this figure.
Supplementary Coding File: Please click here to view this file (Right click to download).
Discussion
The calculations for electronic properties in sections 2, 3, and 4 would be similar among various nanoscale materials. The initial atomic model in step 1 should be carefully designed to extract meaningful information. For example, the factor for selecting the model could be the size or chirality of the materials. Also, the initial atomic model in step 1.1 should be reasonably prepared for low-cost structure relaxation. Taking the nanocomposite in the protocol as an example, the NR should be encapsulated inside the NT in a symmetrical manner. Otherwise, it will be time consuming to search the optimized structure by VASP.
To consider the effect of an electric field, an artificial dipole sheet is added in the middle of the vacuum part in the periodic unit cell in VASP22. The vacuum region should not be too broad and the electric field should be weak enough to avoid artificial field emission23.
Whereas the effect of the strain can be simply realized by changing the lattice parameter in POSCAR, in the nanocomposite the situation would be more complex. The elastic responses of the NR and NT may be different from each other, undergoing the same strength. This will lead to a disproportionate structure. For example, when the uniaxial tensile strain is applied along the periodical direction, the optimized lattice parameter of the NT and NR along this direction changes from an initial 1.8 Å to 2.0 Å, and 2.2 Å, respectively. Large supercells are required for modeling: at least 11 unit cells of NT and 10 unit cells of NR in this case (11 x 2.0 Å = 10 x 2.2 Å = 22 Å).
While ground state electronic properties of materials can be determined by VASP quite well, to address the lifetime of photogenerated holes and electrons existing in an excited state, it is better to perform nonadiabatic dynamics calculation24. This is important to design photocatalysts with long lifetime carriers4.
The role of the computational approach performed by VASP plays into the discovery of novel materials and the screening for potential photocatalysts to assist experimental efforts. The band alignment at the PBE level in water splitting is not as convincing as quantitative experimental work. More accurate values of the band edges relative to the redox potentials, CBO, and VBO are needed. It would be best to use the Heyd-Scuseria-Ernzerhof (HSE) hybrid functional25, but it is more time consuming than PBE. Nevertheless, the results at the PBE level could be efficient for suggesting strategies for the enhancement of photocatalytic activity.
It should be mentioned that the computational design by VASP will also enable the prediction of solar cell materials, thermoelectric materials, lithium battery materials, gas capture materials, etc.2. High-throughput calculations have been combined with the machine learning procedures for better materials prediction and lower computational cost26,27.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work was supported from China Postdoctoral Science Foundation (Grant No. 2017M612348), Qingdao Postdoctoral Foundation (Grant No. 3002000-861805033070) and from the Young Talent Project at Ocean University of China (Grant No. 3002000-861701013151). The authors thank Miss Ya Chong Li for preparing the narration.
Materials
| Nanotube Modeler | Developed by Dr. Steffen Weber | NanotubeModeler1.8 | http://www.jcrystal.com/products/wincnt/NanotubeModeler.exe |
| P4VASP | Orest Dubay | p4vasp 0.3.30 | Open source, available at www.p4vasp.at |
| v2xsf | Developed by Dr. Jens Kunstmann | v2xsf | http://theory.chm.tu-dresden.de/~jk/software.html |
| VASP software | Computational Materials Physics, Dept. of Physics, University of Vienna | vasp.5.4.1 | https://www.vasp.at |
| VMD software | Theoretical and Computational Biophysics Group, University of Illinois at Urbana-Champaign | vmd1.9.3 | https://www.ks.uiuc.edu/Research/vmd |
| xcrysden | Dept. of Physical and Organic Chemistry, Jozef Stefan Institute | XCrySDen1.5.60 | http://www.xcrysden.org/ |
| Xmakemol | Developed by M. P. Hodges | xmakemol5.16 | https://www.nongnu.org/xmakemol/XmakemolDownloads.html |
| Xmgrace software | Grace Development Team under the coordination of Evgeny Stambulchik | xmgrace5.1.25 | http://plasma-gate.weizmann.ac.il/Grace/ |
Riferimenti
- Collins, C., et al. Accelerated discovery of two crystal structure types in a complex inorganic phase field. Nature. 546 (7657), 280-284 (2017).
- Jain, A., Shin, Y., Persson, K. A. Computational predictions of energy materials using density functional theory. Nature Reviews Materials. 1 (1), 15004 (2016).
- de Jong, M., et al. Charting the complete elastic properties of inorganic crystalline compounds. Scientific Data. 2, 150009 (2015).
- Fu, C. F., Wu, X. J., Yang, J. L. Material Design for Photocatalytic Water Splitting from a Theoretical Perspective. Advanced Materials. 30 (48), 1802106 (2018).
- Gu, T., Luo, W., Xiang, H. J. Prediction of two-dimensional materials by the global optimization approach. Wiley Interdisciplinary Reviews-Computational Molecular Science. 7 (2), e1295 (2017).
- Lejaeghere, K., et al. Reproducibility in density functional theory calculations of solids. Science. 351 (6280), aad3000 (2016).
- Kresse, G., Furthmüller, J. Efficient Iterative Schemes for ab Initio Total-Energy Calculations Using a Plane-Wave Basis Set. Physical Review B. 54 (16), 11169-11186 (1996).
- Perdew, J. P., Burke, K., Ernzerhof, M. Generalized Gradient Approximation Made Simple. Physical Review Letters. 77 (18), 3865-3868 (1996).
- Ozcelik, V. O., Azadani, J. G., Yang, C., Koester, S. J., Low, T. Band alignment of two-dimensional semiconductors for designing heterostructures with momentum space matching. Physical Review B. 94 (3), 035125 (2016).
- Gong, M., et al. Robust staggered band alignment in one-dimensional van der Waals heterostructures: binary compound nanoribbons in nanotubes. Journal of Materials Chemistry C. 7 (13), 3829-3836 (2019).
- Grimme, S., Antony, J., Ehrlich, S., Krieg, H. A Consistent and Accurate ab Initio Parametrization of Density Functional Dispersion Correction (DFT-D) for the 94 Elements H-Pu. Journal of Chemical Physics. 132 (15), 154104 (2010).
- Zhang, L., Chen, Z. Q., Su, J., Li, J. F. Data mining new energy materials from structure databases. Renewable & Sustainable Energy Reviews. 107, 554-567 (2019).
- Zakutayev, A., et al. An open experimental database for exploring inorganic materials. Scientific Data. 5, 180053 (2018).
- Kou, L. Z., Tang, C., Frauenheim, T., Chen, C. F. Intrinsic Charge Separation and Tunable Electronic Band Gap of Armchair Graphene Nanoribbons Encapsulated in a Double-Walled Carbon Nanotube. Journal of Physical Chemistry Letters. 4 (8), 1328-1333 (2013).
- Kou, L. Z., Tang, C., Wehling, T., Frauenheim, T., Chen, C. F. Emergent properties and trends of a new class of carbon nanocomposites: graphene nanoribbons encapsulated in a carbon nanotube. Nanoscale. 5 (8), 3306-3314 (2013).
- Kang, J., Tongay, S., Zhou, J., Li, J. B., Wu, J. Q. Band offsets and heterostructures of two-dimensional semiconductors. Applied Physics Letters. 102 (1), 012111 (2013).
- Makov, G., Payne, M. C. Periodic boundary conditions in ab initio calculations. Physical Review B. 51 (7), 4014-4022 (1995).
- Chen, C., Lee, M., Clark, S. J. Band gap modification of singlewalled carbon nanotube and boron nitride nanotube under a transverse electric field. Nanotechnology. 15 (12), 1837 (2004).
- Zhang, Z. H., Guo, W. L. Energy-gap Modulation of BN Ribbons by Transverse Electric Fields: First-Principles Calculations. Physical Review B. 77 (7), 075403 (2008).
- Sahin, H., et al. Monolayer Honeycomb Structures of Group-IV Elements and III-V Binary Compounds: First-Principles Calculations. Physical Review B. 80 (15), 155453 (2009).
- Yang, L., et al. Combining Photocatalytic Hydrogen Generation and Capsule Storage in Graphene Based Sandwich Structures. Nature Communications. 8, 16049 (2017).
- Neugebauer, J., Scheffler, M. Adsorbate-substrate and adsorbate-adsorbate interactions of Na and K adlayers on Al(111). Physical Review B. 46 (24), 16067 (1992).
- He, W., Li, Z. Y., Yang, J. L., Hou, J. G. Electronic structures of organic molecule encapsulated BN nanotubes under transverse electric field. Journal of Chemical Physics. 124 (15), 154709 (2006).
- Zhang, R. Q., et al. Direct Z-Scheme Water Splitting Photocatalyst Based on Two-Dimensional Van Der Waals Heterostructures. Journal of Physical Chemistry Letters. 9 (18), 5419-5424 (2018).
- Paiera, J., Marsman, M., Hummer, K., Kresse, G. Screened hybrid density functionals applied to solids. Journal of Chemical Physics. 124 (15), 154709 (2006).
- Pyzer-Knapp, E. O., Suh, C., Gómez-Bombarelli, R., Aguilera-Iparraguirre, J., Aspuru-Guzik, A. What Is High-Throughput Virtual Screening? A Perspective from Organic Materials Discovery. Annual Review of Materials Research. 45, 195-216 (2015).
- Cerqueira, T. F. T., et al. Identification of Novel Cu, Ag, and Au Ternary Oxides from Global Structural Prediction. Chemistry of Materials. 27 (13), 4562-4573 (2015).

