Design of a Biocompatible Drug-Eluting Tracheal Stent in Mice with Laryngotracheal Stenosis
Summary
Laryngotracheal stenosis results from pathologic scar deposition that critically narrows the tracheal airway and lacks effective medical therapies. Using a PLLA-PCL (70% poly-L-lactide and 30% polycaprolactone) stent as a local drug delivery system, potential therapies aimed at decreasing scar proliferation in the trachea can be studied.
Abstract
Laryngotracheal stenosis (LTS) is a pathologic narrowing of the subglottis and trachea leading to extrathoracic obstruction and significant shortness of breath. LTS results from mucosal injury from a foreign body in the trachea, leading to tissue damage and a local inflammatory response that goes awry, leading to the deposition of pathologic scar tissue. Treatment for LTS is surgical due to the lack of effective medical therapies. The purpose of this method is to construct a biocompatible stent that can be miniaturized to place into mice with LTS. We demonstrated that a PLLA-PCL (70% poly-L-lactide and 30% polycaprolactone) construct had optimal biomechanical strength, was biocompatible, practicable for an in vivo placement stent, and capable of eluting drug. This method provides a drug delivery system for testing various immunomodulatory agents to locally inhibit inflammation and reduce airway fibrosis. Manufacturing the stents takes 28−30 h and can be reproduced easily, allowing for experiments with large cohorts. Here we incorporated the drug rapamycin within the stent to test its effectiveness in reducing fibrosis and collagen deposition. Results revealed that PLLA-PCL tents showed reliable rapamycin release, were mechanically stable in physiological conditions, and were biocompatible, inducing little inflammatory response in the trachea. Further, the rapamycin-eluting PLLA-PCL stents reduced scar formation in the trachea in vivo.
Introduction
Laryngotracheal stenosis (LTS) is a pathologic narrowing of the trachea most often due to iatrogenic post-intubation injury. The combination of bacterial colonization, foreign body response to a tracheostomy or endotracheal tube, and patient-specific factors lead to an aberrant inflammatory response. This maladaptive immune response leads to the deposition of collagen in the trachea, resulting in luminal narrowing of the trachea and subsequent stenosis1,2. As current treatment for this disease is primarily surgical, developing an alternative medically-based treatment paradigm targeting the aberrant inflammatory and profibrotic pathways that lead to excessive collagen deposition has been studied. Rapamycin, which inhibits the mTOR signaling complex, has been shown to have immunosuppressive effects as well as a robust antifibroblast effect. However, when rapamycin is systemically administered, common side effects (e.g., hyperlipidemia, anemia, thrombocytopenia) can be pronounced3. The purpose of our methodology is to develop a vehicle for local drug delivery practicable for use in the airway that would lessen these systemic effects. Our assessments focus on investigating the local immune response to the drug delivery construct as well as its capacity to inhibit fibroblast function and alter the local immune microenvironment. Disease-specific outcomes include in vivo testing that evaluate markers of fibrosis.
Biodegradable drug-eluting stents have been used in animal models of disease in multiple organ systems, including the airway4. For the management of airway stenosis or collapse, previous investigations have used drug-coated silicone and nickel-based stents5. A PLLA-PCL construct was chosen for this particular method because of its drug elution profile and mechanical strength in physiological conditions over a period of 3 weeks, which has been demonstrated in previous published studies6. PLLA-PCL is also a biocompatible and biodegradable material already approved by the FDA4. Biocompatible stents eluting cisplatin and MMC have been studied in large animal models such as rabbits and dogs. However, in these animal models, stents were not placed in an animal model of disease and were implanted transcervically. This study provides a unique method for assessing a biocompatible drug-eluting stent placed transorally in a mouse model of airway injury and laryngotracheal stenosis. A biocompatible stent that elutes an immunomodulatory drug locally and can be miniaturized for study in a murine model is valuable for translational preclinical research. Previous attempts at stent utilization with other material constructs generated robust foreign body responses worsening the underlying inflammation that distinguishes LTS7. This methodology, to our knowledge, is the first of its kind to study the immunomodulatory and antifibrotic effects of a stent-based drug delivery system in a murine model of LTS. The murine model itself offers several advantages for studying the effects of an immunomodulatory drug on the trachea. Genetically modified mice and experimental cohorts of healthy and diseased mice can be studied, which can lead to experimental reproducibility and improve cost-effectiveness. Moreover, the delivery of the stent transorally into the mouse trachea mimics clinical delivery of such a stent in humans, which further highlights the translational advantage of this method. Finally, the relative ease with which the PLLA-PCL stent with the drug can be produced allows for modifications to deliver alternate drug therapies aimed at reducing scar formation in the trachea.
Protocol
NOTE: All methods described here were approved by the Johns Hopkins University Animal Care and Use Committee (MO12M354).
1. Preparation of rapamycin in PLLA-PCL
- Prepare two glass vials (with caps) of 70:30 PLLA-PCL polymer solutions (inherent viscosity 1.3−1.8 DL/G; Table of Materials) solutions, with one vial containing 1.0% rapamycin and the other without rapamycin.
- Make a 1.0% rapamycin containing polymer solution by adding 6 mg of rapamycin to 600 mg of 70:30 PLLA-PCL in a glass vial.
- For polymer solution without rapamycin (control), only add 600 mg of 70:30 PLLA-PCL to the glass vial.
- Under a fume hood, add 6 mL of dichloromethane to each glass vial.
CAUTION: Dichloromethane is a corrosive material and only glass pipettes should be used. Appropriate safety precautions include personal eye protection and gloves. - Add 120 µL of glycerol to each glass vial (for 2% glycerol solution).
NOTE: The addition of glycerol allows for increased flexibility and decreased stiffness in the stent construct. - Recap the glass vials and allow 70:30 PLLA-PCL with and without rapamycin to dissolve and homogenize for 6−12 h.
NOTE: Glass vials can also be placed on a rotating shaking platform for faster dissolution.
2. Rapamycin elution testing
- Pipette 1 mL of 70:30 PLLA-PCL solution containing 1% rapamycin into a glass Petri dish.
NOTE: This volume of 70:30 PLLA-PCL solution containing 1% rapamycin will contain 120 µg of rapamycin and represents the total amount of rapamycin in each construct. Alternative volumes and concentrations can be used. - Allow 70:30 PLLA-PCL with 1% rapamycin to harden into a flat disk in the glass Petri dish.
- Once hardened, place the disk in 2 mL of phosphate-buffered saline (PBS, pH 7.4) and incubate in a 37 °C chamber.
- Collect and replace PBS (2 mL) every 24 h and use collected PBS for high-performance liquid chromatography (HPLC) analysis of rapamycin content.
- Use an autosampler and a C18 4.6 cm x 250 mm HPLC column to run samples through the autosampler injector along with serial dilutions of rapamycin to create a calibrated standard curve. Run each sample in triplicate.
NOTE: Serial dilutions of rapamycin to be tested include 10%, 1%, 0.1%, and 0.01%. - Use a mobile phase of HPLC grade water and acetonitrile with 10/90 volume/volume with a flow rate of 2.0 mL/min. Set absorbance during HPLC for rapamycin to 280 nm.
NOTE: Figure 1 shows the elution of rapamycin over a 14 day period.
3. Creation of rapamycin-eluting PLLA-PCL murine airway stents
NOTE: Perform steps 3.2−3.9 using sterile materials and sterile technique to avoid contamination that would influence in vivo and in vitro applications.
- Prepare PLLA-PCL solutions containing rapamycin as described in section 1.
- Using a glass Pasteur pipette with a rubber balloon, apply 1 mL of PLLA-PCL solution containing 1% rapamycin onto the venous cannula of a 22 G fluorinated ethylene propylene based angiocatheter (Table of Materials). Hold the angiocatheter in one hand and use the glass pipette to drop the PLLA-PCL solution onto the angiocatheter. Slowly rotate the angiocatheter to ensure a homogenous coverage of the angiocatheter with the PLLA-PCL solution.
NOTE: At first, the PLLA-PCL solution will be thin, but as the dichloromethane evaporates during the application onto the angiocatheter, the solution will become more viscous to mold onto the angiocatheter. Continuously turning the angiocatheter during the application is beneficial. This step must be done slowly and meticulously to ensure the consistent thickness of the stent. - Prop the molded angiocatheter with the tip of the angiocatheter facing downward and the hilt on the edge of a glass Petri dish for drying.
- Allow stents to dry for 24 h in a vacuum hood at room temperature (Figure 2A).
- Remove the stent construct from the underlying angiocatheter by gently twisting the underlying catheter free and sliding it out of the molded stent (Figure 2B).
- Check the stent circumferentially for any defects that may have resulted during the casting process. If a defect in the casted stent is present, discard it.
- Trim each end of the casted stent with fine straight scissors so that the edges are axial.
- Using fine straight scissors, cut 3 mm axial segments of the cast stent for use in a mouse model of LTS (Figure 2C).
NOTE: Approximately 8 stents can be made from one casted angiocatheter, with each 3 mm stent containing 120 µg of rapamycin. - Load the 3 mm stent onto a new 22 venous catheter for use in a mouse model of LTS (Figure 1).
4. Laryngotracheal stenosis induction in mice
- Prior to conducting in vivo mouse studies obtain approval from the Animal Care and Use Committee.
- Anesthetize a 9-week old male C57BL/6 by giving an intraperitoneal injection of ketamine (80−100 mg/kg) and xylazine (5−10 mg/kg).
NOTE: Other strains of mice can be substituted, but it is recommended that the mice weight be between 20−27 g. - Randomize mice into an experimental group (chemomechanical injury with a placement of a 1% rapamycin-containing PLLA-PCL stent) and two control groups: 1) chemomechanical injury with the placement of a PLLA-PCL stent, 2) chemomechanical injury without the placement of a stent.
- Place a mouse on a surgical platform in a supine position. Use a small loop of thread affixed to the top of the platform to extend the cervical spine by looping it around the central incisors.
- Affix the hands and legs of the mouse to the table using 2 inch pieces of tape. Pinch mouse paw to ensure it has had adequate anesthesia.
- The surgical site is then prepped. The overlying fur should be removed and the skin should be cleaned (alternating iodine or chlorhexidine scrub with alcohol or diluted skin disinfectant) three times. The area should be draped and sterile gloves be worn. The sterilized instruments should be laid on a sterile surface when not in use.
- Make a 1.5 cm midline vertical incision in the neck of the mouse using fine curved iris scissors. Divide the overlying thymus and lateralize the two resulting lobes to visualize the trachea. Divide the overlying sternohyoid and sternothyroid (strap) muscles at the superior attachment bilaterally.
NOTE: The entire laryngotracheal complex should be completely exposed after this. - Pass a 22 G angiocatheter transorally through the larynx into the trachea. Use small forceps to apply pressure to the anterior larynx of the mouse to assist in the correct placement of the angiocatheter. Visualize the white angiocatheter through the trachea to ensure correct placement (non-esophageal).
NOTE: The mouse trachea is extremely thin and the white angiocatheter will be visualized through the translucent trachea. - Pass a bleomycin-coated wire brush through the inserted angiocatheter.
- Slowly withdraw the angiocatheter so that only the wire brush remains in the trachea.
- Apply counter pressure using fine forceps on the trachea to mechanically disrupt the tracheal lumen with the wire brush.
- Reinsert the angiocatheter into the trachea over the wire brush.
- Remove the wire brush and reapply bleomycin.
- Repeat steps 4.8−4.12 a total of 5x. Apply bleomycin to the brush between each application.
NOTE: Validation and description of this model can be found in Hillel et al.8. - Remove the angiocatheter from the mouse trachea.
NOTE: If no stent is to be placed, the incision can be closed with tissue glue.
5. Transoral PLLA-PCL stent placement in mice
- Load one 3 mm casted stent onto an empty 22 G angiocatheter (Figure 3A).
NOTE: The stent should rest 5 mm from the tip of the angiocatheter. - Draw a thin black vertical line on the 3 mm casted stent.
NOTE: This line allows for improved visualization of the stent in the trachea. - Transorally intubate the mouse with the angiocatheter that has been preloaded with the stent.
- Visualize the stent on the angiocatheter in the trachea through the transcervical incision (Figure 3B).
NOTE: The trachea is very thin, allowing the black dye on the stent to be visualized through the trachea. Use this to confirm correct tracheal placement. - Hold the stent in place by gripping the trachea through the transcervical incision with fine forceps.
- Remove the angiocatheter transorally while maintaining a grip on the stent with the fine forceps.
NOTE: It is extremely important not to apply too much force to the stent to not crush the lumen when the angiocatheter is removed. However, enough force is required to ensure the stent is not removed with the angiocatheter. A 0.8 mm sialendoscope can be used to visualize the location and placement of the stent in the trachea. - Close the transcervical incision with tissue glue.
- Allow each mouse to recover to its original activity level before placing it back in its cage.
6. Histologic preparation of samples
- Anesthetize mice with inhaled anesthetic and sacrifice mice with cervical dislocation per animal protocol after 7, 14, or 21 days.
NOTE: Based on the chronicity of the study, different time intervals can be used (e.g., 28, 30 days, etc.). - Position a mouse on the surgical platform as described in steps 4.4 and 4.5.
- Reopen the neck incision on the mouse using fine curved iris scissors.
- Expose the trachea per step 4.6.
- Divide the distal trachea below the level of the stent using fine curved iris scissors.
- Divide the proximal trachea below the larynx and above the stent using fine curved iris scissors.
NOTE: It is important to divide the distal trachea first to prevent the trachea from retracting into the thorax. - Separate the divided trachea from the esophagus posteriorly and remove trachea from the mouse.
NOTE: Stents will still be retained within the mouse trachea. - Remove stent from the trachea and fix in 10% formalin for 24 h.
- Embed formalin-fixed mouse trachea specimens in paraffin with the distal end oriented superiorly so that axial cuts of the trachea can be made in the next step.
- Cut 5 μm sections of the paraffin-embedded mouse trachea using a microtome.
NOTE: Specimens should be cut axially starting from the distal trachea. - Obtain a 5 μm representative section every 250 μm along the length of the specimen.
- Complete H&E stain on each representative section.
- Deparaffinize the slides by placing in xylene 2x per slide for 3 min.
- Place the slides in 100% ethanol for 2 min, 95% ethanol for 2 min, and 70% ethanol for 2 min to rehydrate.
- Wash the slides with running tap water for 2 min.
- Stain the slides in hematoxylin for 3−5 min.
- Wash the slides with running tap water for 5 min.
- Place the slides in 1% acid alcohol for 1 min.
- Wash the slides with running tap water for 1 min.
- Rinse in 0.2% ammonia water for 30 s.
- Rinse the slides in running tap water for 5 min. Dip in 95% ethanol 10x.
- Eosin stain by placing slide in eosin phloxine for 30 s.
- Dehydrate the slides by placing in 95% ethanol for 5 min, followed by absolute ethanol 2x for 5 min.
- Place in xylene 2x for 5 min.
- Mount the slides with xylene-based mounting medium.
- Measure the thickness of the lamina propria as described in Hillel et al.8.
7. Stent biocompatibility in vivo
- Prepare tissue sections as in steps 6.1−6.4. Do not remove stent from these tracheal sections in order to visualize the changes in inflammation relative to the location of the stent within the lumen of the trachea.
- To determine the acute foreign body response to the stent, observe macrophage and T cell activity using CD3 and F4/80 markers.
NOTE: Other markers can also be used to determine acute inflammatory reaction to the stent. - Obtain 5 μm cut sections of the paraffin-embedded trachea on commercially available hydrophilic plus slides (Table of Materials). Place two sections on each slide.
NOTE: These sections should be from an area of the trachea where the PLLA-PCL stent was placed.
- To determine the acute foreign body response to the stent, observe macrophage and T cell activity using CD3 and F4/80 markers.
- Place the slides in xylene 2x for 5 min each.
- Place the slides in 100% ethanol 2x for 3 min each.
- Place the slides in 95% ethanol for 1 min.
- Place the slides in a histology staining rack so that they are immersed in antigen retrieval buffer (Table of Materials) and then place in a vegetable steamer with boiling water for 20 min.
- Remove the staining racks from the steamer and discard the antigen retrieval buffer.
- Replace the buffer with 1 mL of PBS.
- Place the slides in a wet staining box for 1 min.
- Discard the PBS and incubate the slides with Dulbecco's modified eagle medium (DMEM) with 10% FBS for 30 min.
- Discard the DMEM and add a mixture of two primary antibodies raised in different species. Cover the slides with paraffin film and incubate overnight at 4 °C in a dark room.
NOTE: In this protocol rabbit anti-CD3 and rat anti-F4/80 (Table of Materials) were used. - Wash the slides in PBS 3x for 5 min.
- Incubate the slides with secondary antibodies specific to the primary antibody species (Table of Materials) diluted in DMEM with 10% FBS for 0.5−1 h at room temperature in the dark.
- Wash the slides in PBS 3x for 5 min in the dark.
- Mount the slides on coverslips with a drop of mounting medium with 4'6-diamidino-2-phenylindole (DAPI). Store the slides in the dark at either 20 °C or 4 °C.
- Observe the stained slides on a laser scan confocal microscope and photograph.
- Quantify the total number of cells staining with DAPI and antibodies of interest for comparison to uninjured trachea.
8. Mouse trachea quantitative gene expression analysis
- Collect mouse trachea as described in steps 6.1−6.7 and remove stent.
NOTE: Harvested mouse tracheas can be stored in a -80 °C freezer for up to 2 years. - Desiccate the tracheal tissue harvested in step 8.1 using fine scissors and further homogenize using a bead mill homogenizer with 1.4 mm ceramic beads (Table of Materials) as part of a commercially available column-based RNA extraction kit (Table of Materials).
NOTE: Bead mill homogenizer settings = one 40 s cycle at 6 m/s. - Extract RNA from tracheal lysate using a column-based extraction and purification kit (Table of Materials) following manufacturer's instructions.
- Quantify RNA from each sample using a spectrophotometer (Table of Materials).
NOTE: RNA purity is assessed using the 260/230 nm ratio with a pure RNA sample reading 2.0. - Create complementary DNA from extracted RNA using reverse transcriptase (Table of Materials) and a thermocycler with the following settings: 5 min at 25 °C (priming), 46 °C for 30 min (reverse transcription), and then 95 °C for 1 min (inactivation of reverse transcriptase).
- Dilute complementary DNA samples with RNase free water to a concentration of 10−25 ng/mL for use in quantitative real-time PCR.
- Mix 1 µL of the complementary DNA (10−25 ng/mL) with 10 µL of a PCR mastermix (Table of Materials), 8 µL of ddH2O and 0.5 μL of 10 μM forward and reverse primers for the gene of interest in one well of a 96-well PCR plate (Table of Materials).
NOTE: The total volume in each well should be 20 μL. Each well on the 96 well plate should contain only one sample and one gene of interest. - Repeat steps 8.1–8.7 for all genes of interest and run each sample for each gene in triplicate.
NOTE: The gene-specific forward and reverse primers (Table of Materials) for collagen 1, collagen 3, and the reference gene β-actin are commonly used to investigate markers of fibrosis. - Place the 96 well plate on the real-time quantitative PCR machine and initiate the following protocol: denature at 95 °C for 15 s and anneal and extend at 60 °C for 60 s for 40 cycles.
- Normalize the cycle threshold value (CT) for gene product detection of each gene of interest (GOI) to the CT value for the reference gene β-actin for all samples to obtain a ΔCT for each GOI. Then, compare the ΔCT values for each GOI for treated and untreated samples to generate a ΔΔCT.
NOTE: The cycle threshold is the point on the amplification curve when a predetermined amount of gene product is present (ΔRn). The ΔRn value for each reaction should be determined for each gene of interest and should fall on the linear portion of the amplification curve.
NOTE: ΔCT = CT (GOI) – CT (reference gene); ΔΔCT = ΔCT (treated) – ΔCT (untreated). - Calculate the relative change in gene expression between treated and untreated samples as an expression of fold change by calculating 2ΔΔCT.
Representative Results
The biodegradable PLLA-PCL stent construct loaded with rapamycin used in this study was capable of eluting rapamycin in a consistent and predictable fashion in physiological conditions (Figure 1). Figure 2 shows the PLLA-PCL stent casted around a 22 G angiocatheter for use in a murine model of LTS. To determine if the effects of rapamycin elution in the trachea is efficacious in attenuating fibrosis, measured changes in fibrosis-related gene expression and markers of acute inflammation can be assessed through gene expression analysis, flow cytometry, immunofluorescence, and ELISA. Successful placement of a miniaturized stent into the trachea of the mouse using the method described above has been demonstrated. A schematic of the method is shown in Figure 3. Figure 4 shows the miniaturized biocompatible stent in situ in the trachea as indicated by the black marker on the stent, which is visualized through the translucent mouse trachea. In initial experiments, using a 0.8 mm sialendoscope to confirm placement of the stent in the trachea was helpful. After 21 days, to confirm that the transoral placement of the stent was efficacious and the stent did not migrate from its originally placed position, the neck incision was reopened to determine the placement of the stent. As shown in Figure 4B, the black dye marker of the stent showed the stent maintained its position in the trachea. Resection of the trachea after 21 days of treatment with the stent is shown in Figure 4C.
Representative images of biocompatibility testing using immunofluorescent staining for markers of acute and chronic inflammation are shown in Figure 5. This demonstrated that the PLLA-PCL stent construct (without rapamycin) was not immunoreactive as determined by the minimal number of immune cells present after placement. It is important to note that in this method, normal uninjured tracheas were used and a PLLA-PCL stent without rapamycin was placed to determine the inflammatory response to the construct itself.
Next, to determine whether the rapamycin-eluting PLLA-PCL stent was effective in mitigating scars, we previously demonstrated gene expression changes in markers of acute inflammation and fibrosis6. Specifically, there is a 90.3 fold reduction (SEM ± 26.0; n = 4; p < 0.01) reduction in col1a1 at day 4, as well as the acute inflammatory markers INF-γ, CD11b, Arg-1, and IL-1B6. Though the differences in fold change for some genes were not significant, it is possible that with a greater cohort of mice, significance could be achieved. To determine whether there were changes to the trachea due to drug elution histologically, or whether there were changes to the trachea due to radial force exertion by the stent, we demonstrated that there was a decrease in the width of the lamina propria in those tracheas treated with rapamycin-eluting stents compared to those without rapamycin-eluting stents6.
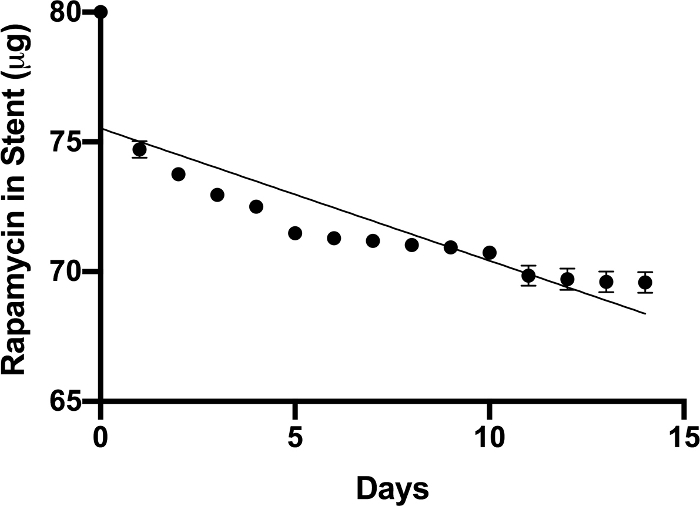
Figure 1: Rapamycin PLLA-PCL elution. The PLLA-PCL construct containing 1% rapamycin demonstrated a consistent and predictable release of rapamycin over a 14 day period. Data points represent the mean ± SEM of sampled elution (n = 3). Please click here to view a larger version of this figure.
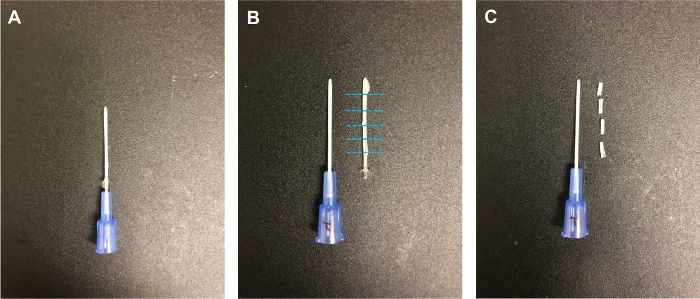
Figure 2: Stent casting. (A) The PLLA-PCL solution was allowed to dry around a 22 G angiocatheter. (B) The cast was then removed from the angiocatheter. (C) Stents were cut to 3 mm lengths for use in the mouse model6. Please click here to view a larger version of this figure.
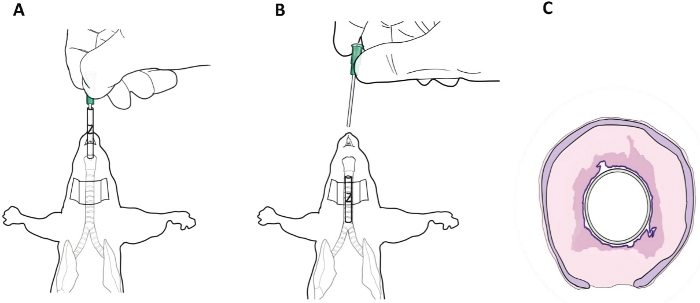
Figure 3: Transoral stent placement in mice. (A) The stent was loaded onto an empty angiocatheter and placed transorally into the trachea. (B) The black dye marking on the stent may be seen through a transcervical incision to confirm its position in the mouse trachea. (C) Representative drawing of the stent in situ in the diseased mouse trachea6. Please click here to view a larger version of this figure.
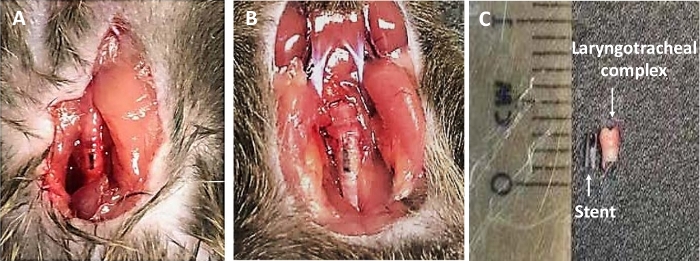
Figure 4: In situ images of stent. (A–B) The stent with black dye markings may be seen in situ in the murine trachea. (C) An image of the stent and the murine laryngotracheal complex after harvest at 21 days6. Please click here to view a larger version of this figure.
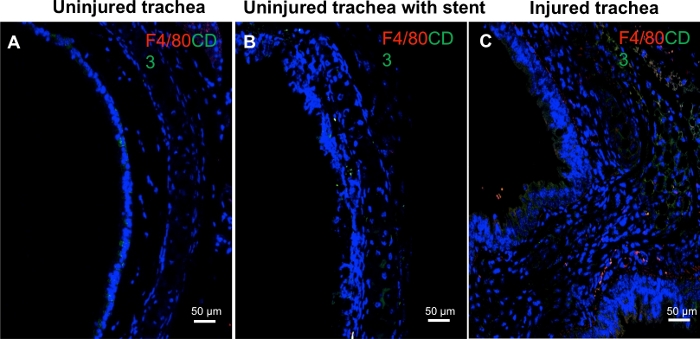
Figure 5: Stent biocompatibility. Immunofluorescent staining for F4/80 (macrophages, red chromophore) and CD3 (green chromophore, T-lymphocytes) at day 4 revealed minimal inflammatory cells in the (A) uninjured trachea and (B) trachea with PLLA-PCL stent. This contrasts with (C) an injured trachea, with a thickened lamina propria and the presence of numerous cells with positive F4/80 and CD3 staining6. Please click here to view a larger version of this figure.
Discussion
The most critical steps for successfully constructing and using a drug-eluting stent in vivo are 1) determining the optimal PLLA-PCL ratio for the desirable drug elution rate, 2) determining the appropriate concentration of drug to be eluted, 3) molding the stents around the angiocatheter for in vivo use, and 4) transorally delivering the stent into the mice after LTS induction without causing fatal airway obstruction.
While there are several methods for drug delivery using stents in animal models of airway disease, development of a biocompatible stent capable of drug delivery in a diseased murine model is a first. In developing this method for drug delivery, several modifications were made to the method. It was important to determine the appropriate PLLA-PCL composition for making stents that were mechanically rigid enough to be placed into the trachea and to remain in the trachea despite physiological secretions. A 70:30 composition of PLLA-PCL was decided upon for stent construction with an angiocatheter molding because it could successfully elute rapamycin in a predicable nature, be placed in a safe and reliable fashion in our mouse model, and not degrade in physiological conditions. A difficult and potentially manufacturer-dependent portion of this method is molding the stent around the angiocatheters. Initially, in constructing the in vivo stents, 22 G angiocatheters were placed inside the tip of a glass pipette and the polymer solution was poured into the space between the angiocatheter and the glass pipette, forming a cast of the space between. However, the wall of the stents resulting from this method often were too thin and were not able to be reliably placed into the trachea. A limitation of the current method for molding stents on the angiocatheter is the dependence on the manufacturer for consistency, and the need for meticulous attention to detail to ensure homogeneity in stent production. The potential for variance in thickness between casted stents needs to be addressed in future studies. With further studies, we hope to design a mold for a 22 G angiocatheter surrounded by another glass or noncorrosive material such that the space between the angiocatheter and the glass encasing is 50 µm, which we determined to be an adequate wall thickness of the stent for easy placement into the trachea.
There are several benefits to using a biocompatible stent to study drug delivery to the affected trachea. Overall, stents composed of metal or silicone that have been coated with a polymer containing the drug have been shown to produce granulation tissue and further inflammatory response to an already scarred portion of the trachea. This method, which interrogates the use of a stent made entirely of biomaterial that is biocompatible and also releases an immunomodulatory drug in a reliable manner is advantageous. The PLLA-PCL stent is also shown to be biocompatible in the murine model and does not elicit an acute inflammatory response. In studying drugs that can combat fibrosis, using a stent entirely composed of biocompatible material such as PLLA-PCL as described in this method is beneficial.
The advantage and ease of using this method to construct usable stents is that the composition of the PLLA-PCL can be varied, allowing for differences in release profiles for the drug mixed in the composition. Previous studies of the PLLA-PCL material show that variation in PLLA-PCL blends can lead to greater degradation of the material, allowing for faster drug release9. Moreover, using this method to construct stents that can be placed in mice is advantageous, as most stent studies for airway disease were done in larger animals10,11,12. The use of a mouse model that can be modified for different disease paradigms and allow for testing of the drug-eluting stent in a diseased state is ideal. Testing the drug-eluting stent in a diseased state and being able to compare its efficacy to those in animals without disease allows for greater experimental rigor. This method also shows how a drug-eluting stent for the airway can be placed transorally, as opposed to previous studies where stents were implanted surgically.
This study demonstrates a very usable platform for stent development and testing in a small animal model. However, other factors for use in human subjects must be considered. Given the firm and rigid nature of the stent it likely cannot be placed through a channeled flexible bronchoscope but will need to be placed transorally with direct laryngoscopy and rigid bronchoscopy. Ideally, the stent would be placed after balloon dilation of the airway to help mitigate restenosis. From the prospective of patient safety, the potential for the stent to migrate is a large concern as it could potentially lead to life threatening airway compromise. Secondarily, the potential for stent obstruction secondary to the accumulation of mucus or blood must also be considered. Further testing and development are needed to minimize these risks.
Future studies can utilize this method to test different drugs in the PLLA-PCL blend to understand further how different immunosuppressive treatments could mitigate scar formation in the trachea. Because such stents can be placed in mice, using a larger cohort of mice or mice with genetic modifications to scar-forming genes can also be tested. Future experiments can also include understanding the changes to the trachea that can occur after chronic implantation of the stent (3–6 months) and the changes to the lumen of the trachea as well as the gene expression profiles of inflammatory markers and fibrosis markers.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
National Institute on Deafness and Other Communication Disorders of the National Institutes of Health under award numbers 1K23DC014082 and 1R21DC017225 (Alexander Hillel). This study was also financially supported by the Triological Society and American College of Surgeons (Alexander Hillel), the American Medical Association Foundation, Chicago, IL (Madhavi Duvvuri) and a T32 NIDCD training grant (Kevin Motz).
Materials
| 1. For stent | |||
| 22-gauge angiocatheter | Jelco | 4050 | |
| Dichloromethane | Sigma Aldrich | 270997-100ML | |
| Glycerol | Fisher Scientific | 56-81-5 | Available from other vendors as well. |
| PDLGA | Sigma Aldrich | 739955-5G | |
| PLLA-PCL (70 : 30) | Evonik Industries AG | 65053 | |
| Rapamycin | LC Laboratories | R-5000 | |
| 2. Animal surgery | |||
| Wire brush | Mill-Rose Company | 320101 | |
| 3. For immunohistochemistry staining | |||
| Antigen retrival buffer | Abcam | ab93678 | Available from other vendors as well; acidic pH needed |
| DAPI | Cell Signaling | 8961S | |
| DMEM | ThermoFisher Scientific | 11965-092 | Available from other vendors as well. |
| FBS (Fetal Bovine Serum) | MilliporeSigma | F4135-500ML | |
| Goat anti-rabbit-488 antibody | Lif technology | a11008 | |
| Goat anti-rat-633 antibody | Lif technology | a21094 | |
| Hydrophilic plus slide | BSB7028 | ||
| PBS | ThermoFisher Scientific | 100-10023 | Available from other vendors as well. |
| Rabbit anti-CD3 antibody | Abcam | ab5690 | |
| Rat antiF4/80 antibody | Biolengend | 123101 | |
| Zeiss LSM 510 Meta Confocal Microscope | Zeiss | ||
| 4. For quantative PCR | |||
| 0.5mm glass beads | OMNI International | 19-645 | |
| Bead Mill Homoginizer | OMNI International | ||
| Gene Specific Forward/Reverse Primers | Genomic Resources Core Facility | ||
| Nanodrop 2000 spectrophotometer | Thermo Scientific | ||
| Power SYBR Green Mastermix | Life Technologies | 4367659 | |
| RNeasy mini kit | Qiagen | 80404 | |
| StepOnePlus Real Time PCR system | Life Technologies |
Riferimenti
- Minnigerode, B., Richter, H. G. Pathophysiology of subglottic tracheal stenosis in childhood. Progress in Pediatric Surgery. 21, 1-7 (1987).
- Wynn, T. A. Fibrotic disease and the T(H)1/T(H)2 paradigm. Nature Reviews Immunology. 4 (8), 583-594 (2004).
- Kaplan, M. J., et al. Systemic Toxicity Following Administration of Sirolimus (formerly Rapamycin) for Psoriasis. Archives of Dermatology. 135 (5), 553-557 (1999).
- Kalra, A., et al. New-Generation Coronary Stents: Current Data and Future Directions. Currrent Atherosclerosis Reports. 19 (3), 14 (2017).
- Chao, Y. K., Liu, K. S., Wang, Y. C., Huang, Y. L., Liu, S. J. Biodegradable cisplatin-eluting tracheal stent for malignant airway obstruction: in vivo and in vitro studies. Chest. 144 (1), 193-199 (2013).
- Duvvuri, M., et al. Engineering an immunomodulatory drug-eluting stent to treat laryngotracheal stenosis. Biomaterials Science. 7 (5), 1863-1874 (2019).
- Mugru, S. D., Colt, H. G. Complications of silicone stent insertion in patients with expiratory central airway collapse. Annals of Thoracic Surgery. 84 (6), 1870-1877 (2007).
- Hillel, A. T., et al. An in situ, in vivo murine model for the study of laryngotracheal stenosis. JAMA Otolaryngolology Head Neck Surgery. 140 (10), 961-966 (2014).
- Can, E., Udenir, G., Kanneci, A. I., Kose, G., Bucak, S. Investigation of PLLA/PCL blends and paclitaxel release profiles. AAPS PharmSciTech. 12 (4), 1442-1453 (2011).
- Wang, T., et al. Paclitaxel Drug-eluting Tracheal Stent Could Reduce Granulation Tissue Formation in a Canine Model. Chinese Medical Journal (Engl). 129 (22), 2708-2713 (2016).
- Sigler, M., Klotzer, J., Quentin, T., Paul, T., Moller, O. Stent implantation into the tracheo-bronchial system in rabbits: histopathologic sequelae in bare metal vs. drug-eluting stents. Molecularand Cellular Pediatrics. 2 (1), 10 (2015).
- Robey, T. C., et al. Use of internal bioabsorbable PLGA “finger-type” stents in a rabbit tracheal reconstruction model. Archives of Otolaryngology Head Neck Surgery. 126 (8), 985-991 (2000).

