Toxicity Screens in Human Retinal Organoids for Pharmaceutical Discovery
Summary
Here we present a step-by-step protocol to generate mature human retinal organoids and utilize them in a photoreceptor toxicity assay to identify pharmaceutical candidates for the age-related retinal degenerative disease macular telangiectasia type 2 (MacTel).
Abstract
Organoids provide a promising platform to study disease mechanism and treatments, directly in the context of human tissue with the versatility and throughput of cell culture. Mature human retinal organoids are utilized to screen potential pharmaceutical treatments for the age-related retinal degenerative disease macular telangiectasia type 2 (MacTel).
We have recently shown that MacTel can be caused by elevated levels of an atypical lipid species, deoxysphingolipids (deoxySLs). These lipids are toxic to the retina and may drive the photoreceptor loss that occurs in MacTel patients. To screen drugs for their ability to prevent deoxySL photoreceptor toxicity, we generated human retinal organoids from a non-MacTel induced pluripotent stem cell (iPSC) line and matured them to a post-mitotic age where they develop all of the neuronal lineage-derived cells of the retina, including functionally mature photoreceptors. The retinal organoids were treated with a deoxySL metabolite and apoptosis was measured within the photoreceptor layer using immunohistochemistry. Using this toxicity model, pharmacological compounds that prevent deoxySL-induced photoreceptor death were screened. Using a targeted candidate approach, we determined that fenofibrate, a drug commonly prescribed for the treatment of high cholesterol and triglycerides, can also prevent deoxySL toxicity in the cells of the retina.
The toxicity screen successfully identified an FDA-approved drug that can prevent photoreceptor death. This is a directly actionable finding owing to the highly disease-relevant model tested. This platform can be easily modified to test any number of metabolic stressors and potential pharmacological interventions for future treatment discovery in retinal diseases.
Introduction
Modeling human disease in cell culture and animal models has provided invaluable tools for the discovery, modification, and validation of pharmacologic therapeutics, allowing them to advance from candidate drug to approved therapy. Although a combination of in vitro and non-human in vivo models has long been a critical component of the drug development pipeline they frequently fail to predict the clinical performance of novel drug candidates1. There is a clear need for the development of technologies that bridge the gap between simplistic human cellular monocultures and clinical trials. Recent technological advances in self-organized three-dimensional tissue cultures, organoids, have improved their fidelity to the tissues they model making them promising tools in the preclinical drug development pipeline2.
A major advantage of human cell culture over non-human in vivo models is the ability to replicate the specific intricacies of human metabolism which can vary considerably even between higher order vertebrates such as humans and mice3. However, this specificity can be overshadowed by a loss in tissue complexity; such is the case for retinal tissue where multiple cell types are intricately interwoven and have a unique symbiotic metabolic interplay between cellular subtypes that cannot be replicated in a monoculture4. Human organoids, which provide a facsimile of complex human tissues with the accessibility and scalability of cell culture, have the potential to overcome the deficiencies of these disease modeling platforms.
Retinal organoids derived from stem cells have proven to be particularly faithful in modeling the complex tissue of the human neural retina5. This has made the retinal organoid model a promising technology for the study and treatment of retinal disease6,7. To date much of the disease modeling in retinal organoids has focused on monogenic retinal diseases where retinal organoids are derived from iPSC lines with disease-causing genetic variants7. These are generally highly penetrant mutations that manifest as developmental phenotypes. Less work has been effectively done on aging diseases where genetic mutations and environmental stressors impact tissue that has developed normally. Neurodegenerative diseases of aging can have complex genetic inheritance and contributions from environmental stressors that are inherently difficult to model using short-term cell cultures. However, in many cases these complex diseases can coalesce on common cellular or metabolic stressors that, when tested on a fully developed human tissue, can provide powerful insights into neurodegenerative diseases of aging8.
The late-onset macular degenerative disease, macular telangiectasia type II (MacTel), is a great example of a genetically complex neurodegenerative disease that coalesces on a common metabolic defect. MacTel is an uncommon retinal degenerative disease of aging that results in photoreceptor and Müller glia loss in the macula, leading to a progressive loss in central vision9,10,11,12,13. In MacTel, an undetermined, possibly multifactorial, genetic inheritance drives a common reduction in circulating serine in patients, resulting in an increase in a neurotoxic lipid species called deoxysphingolipids (deoxySL)14,15. To prove that accumulation of deoxySL is toxic to the retina and to validate potential pharmaceutical therapeutics, we developed this protocol to assay photoreceptor toxicity in human retinal organoids14.
Here we outline a specific protocol for differentiating human retinal organoids, establishing a toxicity and rescue assay using organoids, and quantifying outcomes. We provide a successful example where we determine the tissue-specific toxicity of a suspected disease-causing agent, deoxySL, and validate the use of a safe generic drug, fenofibrate, for the potential treatment of deoxySL-induced retinal toxicity. Previous work has shown that fenofibrate can increase the degradation of deoxySL and lower circulating deoxySL in patients, however, its efficacy in reducing deoxySL-induced retinal toxicity has not been tested16,17. Although we present a specific example, this protocol can be utilized to evaluate the effect of any number of metabolic/environmental stressors and potential therapeutic drugs on retinal tissue.
Protocol
1. Thawing, passaging, and expanding iPSCs/ESCs
NOTE: For all cell culturing steps, use best practices to maintain a sterile cell culture.
- Coat a 6-well cell culture plate with basement membrane matrix medium.
- To prepare 1x of this medium, follow product specifications or dilute 75 µL cold matrix medium with 9 mL of DMEM/F12. Add 1.5 mL of freshly prepared 1x medium per well in a 6-well plate. Incubate at 37 °C for 30 min.
- Aspirate off the basement membrane matrix medium and rinse each well with 3 mL of DMEM/F12. Add 2 mL of DMEM/F12 and incubate at 37 °C until use. Use the plate the day it is prepared.
- Prepare a 10 mM stock solution of rock inhibitor (Y-27632 dihydrochloride) in PBS, and store at -20 °C until use.
- Thaw a vial of iPSCs/ESCs into DMEM/F12 medium and centrifuge at 400 x g for 5 min. Aspirate the supernatant and resuspend the pellet in mTeSR medium. Plate 1 vial of cells on to a coated 6-well well in mTeSR media with 10 µM of rock inhibitor (Y-27632 dihydrochloride) and incubate overnight in the incubator. The next day, aspirate off media and add back just mTeSR.
NOTE: Change media every day until passaging. - Prepare 0.5 mM EDTA in PBS by diluting 500 µL of 0.5 M EDTA in 500 mL of PBS.
- Passage cells when they reach 80-90% confluency. Split cells 1:3 or 1:6 per well, depending on the growth rate of the cells.
- To remove adhered cells from the plate, aspirate off media and incubate with 0.5 mM EDTA in PBS for 5 min at room temperature. Following incubation, remove EDTA solution and lift off cells by forcefully ejecting 1 mL of mTeSR medium on to cells with a p1000 pipette.
- Continue to suck up and forcefully eject mTeSR medium and cells, up to 5x, to remove the adhered remaining cells. Plate cells onto a fresh basement membrane matrix-coated 6-well plate, with mTeSR medium. Continue replacing media daily until plates again reach 80-100% confluency.
- Once cells have expanded to a full 6-well plate and have reached 80-100% confluency, set aside one well to expand the cell line for subsequent differentiations or to freeze for cryopreservation. Use the remaining five wells to begin the differentiation process by making embryoid bodies.
2. Making embryoid bodies (EBs)
NOTE: EB formation and differentiation media recipes are derived from protocols in Cowan et al.5, Ohlemacher et al.18, and Zhong et al.19.
- Prepare 1x Dispase solution and Neural Induction Media (NIM) as described below.
- Prepare 10 mg/mL of 5x stock solution of Dispase by dissolving the powder in DMEM/F12. Sterile filter using a 0.22 µm filter. Store 1 mL aliquots at -20 °C until use.
- Using the 5x Dispase solution, prepare 1 mL/well of 2 mg/mL Dispase in DMEM/F12. Warm the solution at 37 °C for 30 min.
- Prepare NIM by supplementing DMEM/F12 with 1% N2 supplement, 1% MEM NEAA, and 2 mg/mL heparin at 180 U/mg. NIM can be stored at 4 °C for up to one month.
- Prepare and warm 16 mL of 3:1 mix of mTeSR:NIM (12mL:4mL) solution to room temperature.
- Remove differentiated cells and add warm Dispase solution.
- Remove differentiating cells from the iPSCs/ESCs cell culture. Under a dissection scope, scrape off opaque white colonies with a p10 pipette tip. Aspirate off mTeSR medium from the culture wells. This will remove the differentiated clumps that were just scraped off.
- Immediately add the pre-warmed Dispase solution and incubate at 37 °C until most of the edges of the cell colonies begin to curl up under a microscope. This will take 4-8 min.
NOTE: The incubation time for cells in Dispase will have to be determined for each lab. The duration can differ between cell lines and a longer incubation is required for cells that have been plated for 3 days or more. Slow-growing iPSCs/ESCs that take greater than 3 days to reach confluency will begin to adhere with greater strength to the plate, making it difficult to lift the cells off. For slow-growing cells, try passaging cells at 1:2 for expansion into a full 6-well plate to reduce the time from plating to confluency.
- Aspirate the solution and gently add back at least 2 mL of DMEM/F12 medium per well. Add DMEM/F12 medium slowly to the side of the well taking care not to dislodge the cells.
- Aspirate DMEM/F12 medium then forcefully add back fresh 1 mL of DMEM/F12 directly on to the cells to lift cell colonies from the plate. With a p1000 pipette repeatedly suck up and forcefully eject DMEM/F12 in the well, up to 5x, to dislodge as many cell colonies as possible.
NOTE: Differentiating/differentiated iPSCs/ESCs stick to the plate more than undifferentiated iPSCs/ESCs. Cells that do not come off with repeated pipetting are best left behind. Excess pipetting kills cells and reduces efficiency of EB production. Cell clumps will have between 100-400 cells per clump.
- Aspirate DMEM/F12 medium then forcefully add back fresh 1 mL of DMEM/F12 directly on to the cells to lift cell colonies from the plate. With a p1000 pipette repeatedly suck up and forcefully eject DMEM/F12 in the well, up to 5x, to dislodge as many cell colonies as possible.
- Transfer floating colonies to a 15 mL conical tube and rinse wells with an additional 1 mL DMEM/F12 medium per well to collect any remaining floating colonies.
- Allow the floating colonies to settle by gravity for no more than 5 min. Once settled, remove all but 1-2 mL supernatant. Take care not to agitate the soft pellet at the bottom.
- Resuspend the pellet in pre-warmed 3:1 mTeSR:NIM medium and transfer to an ultra-low-attachment T75 flask. Incubate overnight to allow EBs to form. This will be considered Day 0 of differentiation.
- Gradually change media to NIM to begin the differentiation of EBs into neural progenitors.
- The following day, remove media and replace with 10 mL of 1:1 mTeSR:NIM medium. To remove media from free-floating EB culture, tip the flask up so that the EBs collect into the bottom corner of the flask. Aspirate off the supernatant, making sure not to aspirate any of the EBs in the pellet at the bottom.
- The following day, replace the media with 10 mL of 1:3 mTeSR:NIM medium.
- The next day, replace the media with full NIM medium. Continue changing media every 2 days with NIM medium until 7 days following Dispase treatment.
3. Plating EBs and initiating neural retinal differentiation
- One week following Dispase treatment, plate free-floating EBs onto a 6-well plate coated with growth factor-reduced basement membrane matrix medium. To coat plate, refer to step 1.1.
- Aspirate spent media from EBs and replace with 12 mL of NIM.
- Ensure an even distribution of EBs into each well by agitating the EB-containing media to resuspend the EBs, then quickly remove 1/6th of the media (2 mL) and dispense into one well. Repeat for the remaining wells.
- Before incubating, gently shake the plate, back and forth then side to side, to evenly distribute the EBs. If the EBs clump in the middle they will not differentiate properly.
NOTE: Plating density is critical to differentiation. Make sure to plate greater than 30 EBs per well. If EBs production was not efficient, distribute EBs into fewer wells.
- Change the media daily with NIM medium. Keep the level of media in the wells at ~3 mL. When changing media, remove all but 1 mL of media to not dry out the plated EBs, and add 2 mL of media gently to the side of the well to not lift the cells off the plate.
- Nine days following the plating of EBs, begin changing the media from NIM to Retinal Differentiation Media (RDM) over the course of two days.
- Prepare RDM by mixing the following: 48% DMEM/F12 (1:1) and 48% DMEM supplemented with 2% B27 supplement without vitamin A, 1% MEM NEAA and 1% Pen-Strep. Filter sterilize using 0.20 µm filter. RDM can be stored at 4 °C for up to one month.
- The first day, switch media to a 1:1 mix of NIM:RDM. On the second day, change the media to RDM. All subsequent days, feed cells RDM.
NOTE: Over the next few weeks, plated EBs will form neural retinal progenitors that begin to generate visibly pigmented RPE.
4. Making free-floating organoids and maintaining free-floating organoid cultures
- Fragment plated EBs at 28 days following initial Dispase treatment (21 days after EB plating) using a sterile scalpel to cut a 1-2 mm2 grid into plated EBs. This will separate the retinal progenitor colonies into small chunks so that later in the development they will not get too big and necrose from insufficient nutrient exchange in their center.
- Detach plated EBs.
- To do so, first add an additional 2 mL of RDM to each well. Then aspirate ~1000 µL of media from the wells using a p1000 pipette. Now forcefully eject the media directly onto plated EBs with the tip fully submerged.
- Repeat this until all the cells have been lifted from the plate. Keep the tip submerged during ejection of media and do not push the pipette plunger past the first stop to avoid bubbles.
- Resuspend nascently detached EB chunks into a sterile plastic 125 mL Erlenmeyer flask and fill the flask with ~40 mL of RDM media. Place the flask on an orbital shaker placed in a standard incubator. Set the shaker speed at 130-140 RPM (speed setting 3).
NOTE: Culturing organoids on a shaker prevents them from sticking together while culturing at a higher concentration of organoids. Bioreactors also achieve the same ends, but a shaker is less expensive. - Feed organoids with a partial media change, replacing approximately 12 mL of RDM 2-3 times per week, depending on how yellow the media gets. Early organoids will not require much feeding but as they grow, they will require more frequent media changes with more media.
- Prune organoids every two weeks to remove non-retinal, overgrown, and dying organoids. This prevents wasting media and improves the health of the remaining retinal organoids.
- Transfer cells from their flask onto a 6-well plate or 10 cm dish using a 5 mL pipette. Keep organoids that display stratified neuroepithelium (Figure 1A, asterisk). These organoids are transparent and white.
- Sort out and remove ill-formed or overgrown retinal organoids, and non-retinal organoids with a 5 mL pipette. These will generally be opaque and yellowish (Figure 1A). Also remove anything that does not look like a stratified neuroepithelium. The first few prunings will be labor intensive but will become much easier every subsequent time as organoids become larger and more homogenous (Figure 1B).
5. Maintaining mature organoids and differentiating them to a post-mitotic retinal tissue
- At day 56 (week 8) following the initial Dispase treatment for EB formation (28 days after culturing dislodged EBs in a flask) change organoid media to RDM+. This will provide extra nutrients to fully mature organoids as retinal tissue.
- Prepare 100 mM taurine in PBS. Store aliquots in -20 °C until use.
- Prepare RDM+ by supplementing RDM with 10% FBS, 100 µM Taurine, and 2 mM commercial glutamine supplement. Filter sterilize using 0.20 µm filter.
- Continue to change RDM+ media 2-3 times a week.
- At week 17-18 (post-Dispase treatment) transfer organoids from the Erlenmeyer flask to an ultra-low attachment 6-well plate using a 5 mL pipette. For the first week continue to prune and separate organoids from each other daily until organoids no longer stick together. Optimal density is 10-15 organoids per well. Change media 2-3 times a week.
NOTE: Reduce the number of organoids per well if they organoids frequently adhere to each other or if the media is turning very yellow between media changes. - Expect organoids to become fully post-mitotic by week 185 when neural retinal cells are present throughout. By week 24, check for the formation of rudimentary outer segments on the outside of the organoid. By week 26-28 ensure that outer segments are thick on the outside of the organoids (Figure 1C). Perform toxicity assays after week 26 when organoids have defined outer segments signifying a mature differentiation state.
NOTE: Use only well-differentiated retinal organoids for toxicity assay. Performing toxicity assay on organoids with well-defined outer segments will allow for their identification.
6. Deoxysphinganine (deoxySA) and drug treatment
NOTE: Presented here is a single treatment of fenofibrate to rescue deoxySA toxicity over the period of 4 days (Figure 2). Concentration of deoxySA added to organoids, duration of deoxySA treatment on organoids and the type of drug used to rescue toxicity14 can, however, be modified as per the experimental needs to assay toxicity and toxicity rescue.
- Prepare 1 mM stock solution of deoxySA in ethanol. Store at -20 °C for 1 month. Alternatively, a stock solution of deoxySA can be prepared in DMSO.
- Perform a serial dilution of deoxySA to determine an appropriate toxic concentration of deoxySA for drug rescue.
NOTE: First determine a concentration of deoxySA that will result in sufficient cell death to measure a rescue effect. If the concentration is too high, the toxic response will result in disintegration of the organoid and no rescue will be observed. If the toxic response is too low, it will be difficult to observe a significant rescue effect. The deoxySA toxic response can vary from lab-to-lab and batch-to-batch of deoxySA.- Dilute 1 mM deoxySA stock solution to concentrations of 0.5 µM, 1 µM, and 2 µM in 3 mL of RDM+, each. Add 3 µL of ethanol to RDM+ to prepare a control treatment.
- Under a dissection microscope, use a sterile probe to select 4 groups of organoids with a minimum of 5 organoids per group. Split the groups into separate wells of an ultra-low attachment 6-well plate. Select organoids that have approximately the same size and shape.
- Aspirate off as much RDM+ from the organoids as possible. To each well of organoids, add one concentration of deoxySA in RDM+. Add vehicle control to the fourth well of organoids.
- After two days of culture in deoxySA or vehicle, replace the media with freshly prepared corresponding deoxySA dilutions in RDM+.
- On the fourth day of treatment, proceed to step 7 to process and assay organoids for cell death. Once a deoxySA concentration has been selected, continue to step 6.3 to treat with fenofibrate.
NOTE: Target cell death in the selected deoxySA treatment group is 5 – 20 TUNEL positive cells per 10,000 µm2. The organoids should not disintegrate by the touch of a probe over the course of the treatment.
- Treat organoids with the selected toxic dose of deoxySA and fenofibrate.
- Prepare a 40 mM stock solution of fenofibrate in DMSO. Store at -20 °C for up to 1 year.
- Prepare the drug treatment media: In 3 mL RDM+, dilute 1 mM deoxySA stock solution to 1 µM deoxySA and dilute 40 mM fenofibrate stock solution to 20 µM.
- Prepare deoxySA treatment media: In 3 mL RDM+, dilute 1 mM deoxySA stock solution to 1 µM deoxySA and add 1.5 µL of DMSO.
- Prepare the control media: In 3 mL RDM+, add 3 µL ethanol and 1.5 µL of DMSO.
- Under a dissection microscope using a sterile probe select three groups of organoids with a minimum of five organoids per group. Split the groups into separate wells of an ultra-low attachment 6-well plate.
- Add the prepared treatments (deoxySA, deoxySA + fenofibrate, or control) to the organoids. After two days, change the media in the wells with freshly prepared RDM+ supplemented with corresponding deoxySA / fenofibrate / control solutions.
- On the fourth day of treatment proceed to step 7 to process and assay organoids for cell death (Figure 2).
7. Embedding and cryosectioning of organoids
- Prepare fresh 4% PFA by adding 1 mL of 16% PFA ampoule to 3 mL of PBS.
- Remove RDM+ media from organoids and rinse once with PBS. Remove as much PBS as possible from organoids. Add 2 mL of freshly prepared 4% PFA (in PBS) to the organoids and incubate for 15 min at room temperature.
- Following 15 min incubation, rinse organoids twice with 2 mL of PBS, then wash in PBS for 10 min.
NOTE: Perform fixation in a chemical fume hood and discard PFA solution and two subsequent PBS rinses into an appropriately labeled PFA waste container stored in the fume hood. - Aspirate all the PBS, then add 20% sucrose in PBS to the fixed organoids. Ensure the organoids are mixed into the sucrose solution and do not remain in a PBS bubble on top. Keep the organoids in the sucrose solution until they sink to the bottom of the sucrose solution, i.e., ~1 h at room temperature. Fixed organoids can be stored in sucrose solution overnight at 4 °C.
- Embed the organoids in OCT solution in a cryosection mold. Multiple organoids per condition can be embedded into one mold. Orient the organoids ensuring that their best retinal regions will be crosssectioned on the cryostat, and the middle of the organoids are all roughly in the same plane. Freeze the embedded organoids at -20 °C. Embedded organoids can be stored at -80 °C for years.
NOTE: Organoids are difficult to see in frozen OCT; orient RPE clusters towards the side of the mold that will be facing the blade to facilitate organoid identification. - Cryosection organoids into 10-14 µm thick sections and adhere sections to poly-L lysine treated slides. While cryosectioning, repeatedly check the slides under a microscope to identify sections that contain the middle of the organoids. Only collect the middle slices of the organoids where all the layers are evenly represented (Figure 2A-C, Figure 3). Once sectioned, slides can be stored in a slide box at -80 °C for years.
NOTE: Slices taken on the ends of the organoid will over-sample outermost photoreceptors and are not appropriate for quantification. Average-sized organoids will provide 10-15 slices of the middle of the organoid which represents a cross section of the stratified retinal layers.
8. TUNEL staining for apoptotic cells
- Thaw frozen slides for 30 min at 37 °C. Immediately placing slides at 37 °C prevents condensation. Tissue samples soaked in pure water can distort the tissue. A 37 °C incubation also improves adhesion of tissue to the glass slide.
- Create a continuous border on the glass slide around the tissue sections using a hydrophobic pen. This will provide a border and ensure adequate fixation and antibody incubations with small volume of solutions. Add approximately 100-200 µL (the volume will fill the hydrophobic-bordered region without spilling over) of freshly prepared 4% PFA for 20 min. Perform fixation in a chemical fume hood and discard PFA solution into appropriately labeled PFA waste container stored in the fume hood.
- Rinse slides once in PBS then wash in PBS for 25 min at room temperature.
- Prepare the TUNEL mixture. Thaw both violet and blue vials from the in situ cell death detection kit. Remove 100 µL of violet vial solution (can use for a negative control) and combine the remaining 450 µL of solution from the violet vial with 50 µL of solution from the blue vial. Mix well and keep in the dark. Each set of vials (500 µL total) can stain 5-10 slides.
- Permeabilize tissue slices.
- Prepare Permeabilization Solution: PBS with 0.1% TritonX-100 and 0.1% Sodium Citrate
- Remove PBS from sample slides then add Permeabilization Solution to samples. Incubate for 2 min on ice block or at 4 °C. Following that, rinse once in PBS then wash in PBS for 10 min.
- Remove PBS and dry away as much solution as possible using the corner of a folded tissue. Add 50-100 µL of prepared TUNEL mixture to the samples, depending on the size of the area drawn by the hydrophobic pen. Cover with glass cover slip and incubate at 37 °C for 1 h in the dark.
NOTE: All incubations and washes for the following steps are to be done in the dark to prevent bleaching of fluorophores. Use either a dark box or cover the slides with aluminum foil to block out light. - Label the photoreceptors. Rinse once with PBS then wash for 20 min in PBS. Remove PBS then add primary Recoverin antibody (rabbit anti-Recoverin) diluted 1:500 in PBS with 0.1% Tween and 5% donkey serum. Incubate at 4 °C overnight.
- After removing primary antibody, rinse once in PBS then wash in PBS for 20 min. Add secondary antibody, donkey anti-rabbit with an orange or red fluorophore, diluted 1:1,000 in PBS with 0.1% Tween and 5% donkey serum and incubate at room temperature for 2 h. Rinse once with PBS then wash for 20 min in PBS. Add DAPI at 1:1000 in PBS, incubate at room temp for 10 min.
- Rinse once with PBS then wash for 20 min in PBS. Remove PBS and add the mounting medium. Add cover slip, seal with nail polish, and leave to dry in the dark. Slides can be stored at 4 °C in a dark container for up to 1 week.
NOTE: For the best image quality take images the next day.
9. Imaging organoid slices and quantifying death.
NOTE: Imaging requires a confocal microscope with capabilities to distinguish between three fluorophore channels. This experiment uses green (Alexa Fluor 488), orange (Alexa Fluor 555), and UV (DAPI) channels. Any combination of fluorophores can be used ensuring that emissions do not bleed into the other channels.
- Locate retinal sections for imaging and quantification. Under a low magnification of the microscope (5x or 10x) use Recoverin staining, which labels the cytoplasm of photoreceptors, and DAPI staining, which labels the nuclei of all cells, to locate a region of the sliced organoid that is intact, well-formed, and in a plane that samples a representative cross-section of the organoid (Figure 2 and Figure 3). Find a section that has a distinct outer nuclear layer of photoreceptors that is at least a few cells thick, and a separate and distinct layer of nuclei that do not have Recoverin staining (Figure 2 and Figure 3).
- Frame the image. Increase the magnification of the microscope to a 20x objective and frame the slide to fill the image with as much of the photoreceptor layer as possible (Figure 3).
- Set the Z-plane. If using a microscope that images Z-stacks set the upper and lower limits of the Z-range to image the entire depth of the slice. Use the same Z-stack thickness for all images in an experimental set so that the same amount of tissue is assayed in all samples. If not using a microscope that images Z-stacks, focus the image to the middle of the slice.
- Image all three channels of fluorophores in a Z-stack acquisition. All three channels are required to positively identify a dying cell. Image one area per organoid.
- Save the image set in a file format that preserves the ratio of area per pixel.
10. Quantifying dying cells
- Open image stacks in FIJI (Image J) software. In the Bio-Formats Import Options window, ensure that no boxes under the "split into separate windows" section are selected. Scrolling between aligned image channels is critical to appropriately identifying cells.
- Flatten the Z-stack image set by clicking on Image > Stacks then select 'Z project' from the dropdown menu. This will create a new image for quantification. If the organoids were not imaged in Z-series, skip this step.
- Select the image channel that shows Recoverin staining (red, in this example). Click on the polygon selection tool in the toolbar and outline a continuous region of photoreceptors in the image (Figure 3A). Click on Analyze > Set Measurements. In the Set Measurements pop-up window select the box next to Area, click on Analyze and select Measure to measure the area (or press "m" as a short-cut) of photoreceptors to count dying cells. The area measurement will appear in a pop-up measurements window.
- Count the TUNEL-positive nuclei in the photoreceptor region previously selected (Figure 3B,C). Right click on the point tool in the tool bar and select multi-point tool. In the TUNEL channel only, click on TUNEL positive staining that overlaps with a DAPI positive nuclei and Recoverin staining. Toggle between the channels to validate positive cells.
- Divide the number of TUNEL positive cells by the area to get a normalized value for the cell death in photoreceptors per organoid (Figure 2D,E).
Representative Results
Retinal organoids were generated from a non-MacTel control iPSC line. After organoids reached 26 weeks in culture they were selected and split into experimental groups. Organoids were treated with varying concentrations of deoxySA to determine if deoxySA is toxic to photoreceptors. Four concentrations of deoxySA were tested, from 0 to 1 µM (Figure 2) and organoids were treated for 8 days, with media changes every other day. Cell death in response to deoxySA is concentration-dependent and detectible in as little as 50 nM deoxySA (Figure 2D). The highest concentration tested, 1 µM deoxySA, gave a robust effect while maintaining the integrity of the retinal organoid (Figure 2B,D). A higher concentration of 5 µM deoxySA caused disintegration of organoids within a few days (data not shown). The results of the deoxySA dose response determined the optimal concentration of deoxySA for future toxicity experiments. The 1 µM deoxySA dose resulted in a substantial cell death without oversaturating toxicity, as was observed at 5 µM.
To test the ability of fenofibrate to rescue deoxySA-induced cell death, retinal organoids were treated with either 1 µM deoxySA, 1 µM deoxySA plus 20 µM fenofibrate, or a no treatment vehicle control (Figure 2A-C,E). The 20 µM fenofibrate treatment prevented deoxySA-induced toxicity in the photoreceptors of retinal organoids, significantly reducing cell death by approximately 80% after 4 days of treatment (Figure 2A-C,E)16. Additional lipid-altering drugs were tested using the same protocol, including fumonosin-B1, which showed a complete rescue of deoxySA toxicity. Related lipid species were also tested to identify the specific downstream sphingolipid metabolite that leads to photoreceptor cell death14.
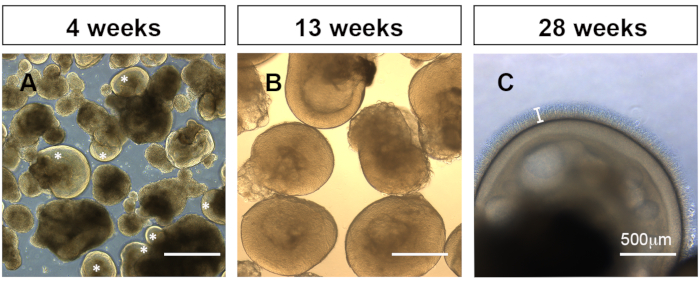
Figure 1: Representative bright field images of organoids under an inverted light microscope. (A) 4-week-old organoids at 2 days following detachment show appropriate retinal layering (white *) or non-retinal organoid development. (B) Developing organoids at week 13. (C) Retinal organoids at week 28 with clear retinal layering and well-formed outer segments projecting from the outer photoreceptor layer (white bar). Please click here to view a larger version of this figure.
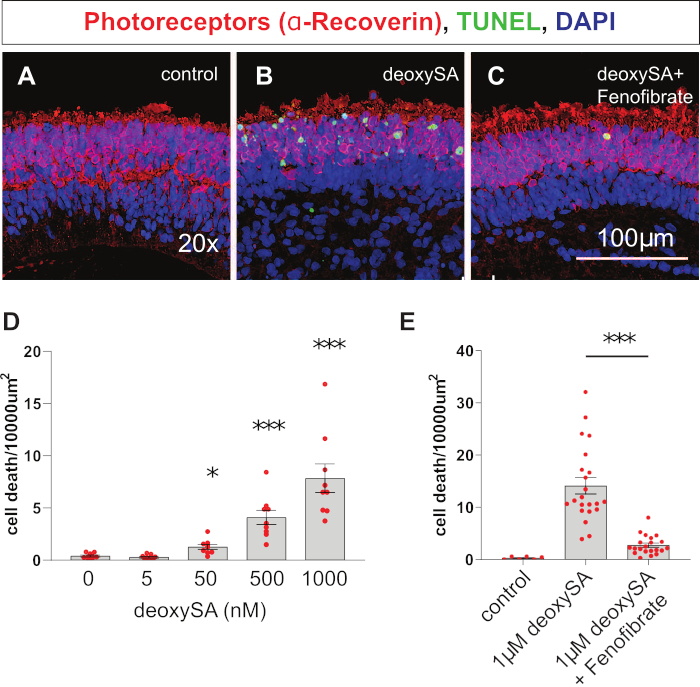
Figure 2: Representative confocal images of cell death. Representative confocal images of cell death (TUNEL, green) within the photoreceptor layer (Recoverin, red) of the retinal organoid following 4 days of treatment with either control media (A), 1 µM deoxySA (B), or 1 µM deoxySA with 20 µM fenofibrate (C). (D) Quantification of cell death in human photoreceptors following treatment of retinal organoid tissue with varying concentrations of deoxySA. (E) Quantification of cell death in human photoreceptors following treatment of retinal organoid tissue with control media (n=6), 1 µM deoxySA (n=22), 1 µM deoxySA + 20 µM fenofibrate (n=21). *p<0.05, ***p<0.001 with one way ANOVA and post hoc Tukey test. Data derived from New England Journal of Medicine, Gantner, M., Eade, K., and Wallace. M., et al. Serine and lipid metabolism in macular disease and peripheral neuropathy, 381(15), 1422-1433, Copyright © (2019) Massachusetts Medical Society. Reprinted with permission.14 Please click here to view a larger version of this figure.
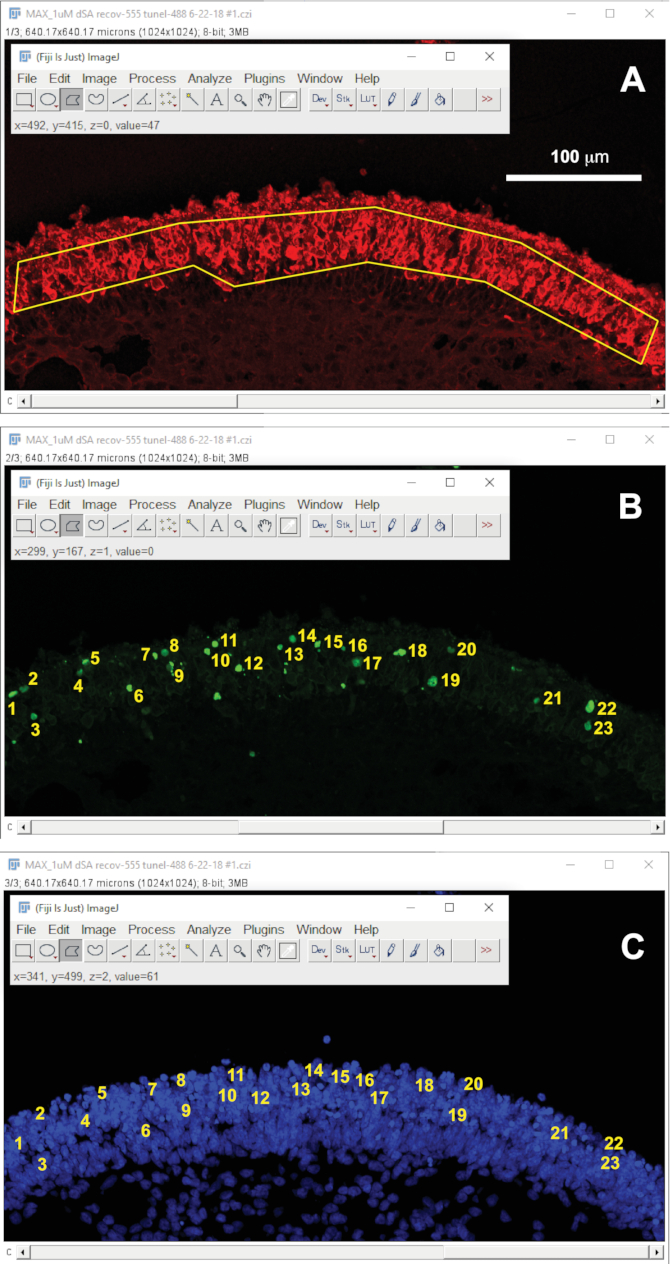
Figure 3: Screen shots showing a confocal image of a sectioned and stained organoid treated with 1 µM deoxySA viewed using FIJI software. Fluorescent channels have been split in to α-Recoverin (A, red), TUNEL (B, green), and DAPI (C, blue). (A) The photoreceptor area has been outlined using the polygon tool selected in the toolbar (top left). (B,C) Cell counts using the multi-point tool, selected in the tool bar, of TUNEL positive (B, green) and DAPI positive (C, blue) cells within the photoreceptor layer. Please click here to view a larger version of this figure.
Discussion
Differentiation protocol variations
Since the invention of self-forming optic cups by Yoshiki Sasai's group20, many labs have developed protocols to generate retinal organoids that can vary at almost every step5,18,19,21. An exhaustive list of protocols can be found in Capowski et al.22. The differentiation protocol we provide here is a simple, low-intervention protocol that provides a good starting point for any lab attempting to differentiate mature retinal organoids. Users are encouraged to explore and adopt variations on this protocol. Common steps for protocol variation are the production and early differentiation of EBs, and the use of differentiation factors such as BMP4, DKK-1, or retinoic acid to improve efficiency5,19,23.
Culturing organoids on a shaker for much of the late-stage development has been an effective step to increase differentiation efficiency and reduce the time spent handling organoids. Bioreactors achieve the same ends24, however, using a shaker with disposable Erlenmyer flasks is more cost-effective and can be done with common lab equipment. The main advantage of keeping the organoids in mobilized suspension is that it removes the need to separate organoids that stick together on the plate while they are growing. The suspended cultures also facilitate greater nutrient exchange, so organoids have a tendency to grow larger than those on a standard still plate. For this reason, it is good to start out by fragmenting plated EBs as small as possible in step 4.1.
Since obtaining mature retinal organoids requires differentiating a single culture for nearly 6 months, performing toxicity assays on retinal organoids can seem time-consuming compared to using a 2-D monoculture. However, toxicity assays are performed on a single control line, and differentiations can be set up routinely. Following an initial 6-month investment, a regular supply of organoids to perform additional experiments and protocol modifications will be at hand without delay. Initiating 2-3 rounds of retinal organoid differentiation every 1-2 months provides ample tissue for complex and thorough sets of experiments.
Organoids provide a versatile model for targeted drug testing.
This protocol describes a photoreceptor toxicity assay to test the efficacy of lipid-altering drugs to rescue deoxySL-induced cell death. Although this is a specific application showing a pharmacological rescue for a disease-causing agent in MacTel, this protocol can be utilized to test any number of insults (e.g., nutrient deficiency, hypoxic conditions, light toxicity) and pharmacological rescues. Therefore, it can be adapted to model other retinal diseases. In our own work we have shown that by using this same protocol and substituting various related sphingolipid species and drugs that directly block sphingolipid metabolism we were also able to provide insights to the MacTel disease mechanism by determining the specific toxic downstream sphingolipid metabolite that leads to photoreceptor cell death14. Unlike modeling diseases at the level of genetic mutations, which require establishing variant iPSC lines, using organoids to model toxic metabolic conditions and environmental stressors allows for the use of a common control iPSC line as a dynamic model with abundant and readily available tissue source.
The human organoid models are particularly advantageous when studying metabolic diseases in the retina, where different cell types have a unique symbiotic metabolic interplay that cannot be replicated in a monoculture of photoreceptor-like cells4. This complex metabolic model has allowed us to discover the effectiveness of the drug fenofibrate in preventing the toxic effects of deoxySLs directly in humans. In mouse models of elevated deoxySLs14 (unpublished data) and mouse embryonic fibroblast cell culture models of deoxySL metabolism fenofibrate proved to be ineffective16. Testing drugs in the context of complex human tissue models will allow us to identify effective treatments and discount ineffective treatments that would have otherwise been missed had we only relied on mice or simplistic monocultures.
Future development for drug screening
In this protocol we quantify apoptosis in photoreceptors, the most abundant cell type. However, by utilizing any of the proven antibodies that specifically label the other retinal cell types this protocol is modifiable to assay cell death in any retinal cell type, or cell subtype (e.g., cone vs rod). The disadvantage of quantifying apoptosis using TUNEL staining in tissue slices is that it is a low throughput technique to quantify cell death, limiting the application of this assay to screening small lists of candidate drugs. A larger screen of untargeted drug libraries would not be feasible using an IHC approach. Future developments in this protocol to facilitate larger drug screens would require the integration of a more readily quantifiable cell death marker. While these are available, the irregularity of retinal organoids in size, presence or absence of non-retinal tissue appendages, and variability in photoreceptor layer quality makes it difficult to normalize results across organoids. We expect that future developments to increase the throughput of human organoid disease models to screen large drug libraries will vastly improve our ability to discover, modify, and validate drugs that will advance from preclinical trials to approved therapeutics.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
Supported by the Lowy Medical Research Institute. We would like to thank the Lowy family for their support of the MacTel project. We would like to thank Mari Gantner, Mike Dorrell, and Lea Scheppke for their intellectual input and assistance preparing the manuscript.
Materials
| 0.5M EDTA | Invitrogen | 15575020 | |
| 125mL Erlenmeyer Flasks | VWR | 89095-258 | |
| 1-deoxysphinganine | Avanti | 860493 | |
| B27 Supplement, minus vitamin A | Gibco | 12587010 | |
| Beaver 6900 Mini-Blade | Beaver-Visitec | BEAVER6900 | |
| D-(+)-Sucrose | VWR | 97061-432 | |
| DAPI | Thermo-fisher | D1306 | |
| Dispase II, powder | Gibco | 17105041 | |
| DMEM, high glucose, pyruvate | Gibco | 11995073 | |
| DMEM/F12 | Gibco | 11330 | |
| Donkey anti-rabbit Ig-G, Alexa Fluor plus 555 | Thermo-fisher | A32794 | |
| donkey serum | Sigma | D9663-10ML | |
| FBS, Heat Inactivated | Corning | 45001-108 | |
| Fenofibrate | Sigma | F6020 | |
| Glutamax | Gibco | 35050061 | |
| Heparin | Stemcell Technologies | 7980 | |
| In Situ Cell Death Detection Kit, Fluorescin | Sigma | 11684795910 | |
| Matrigel, growth factor reduced | Corning | 356230 | |
| MEM Non-Essential Amino Acids Solution | Gibco | 11140050 | |
| mTeSR 1 | Stemcell Technologies | 85850 | |
| N2 Supplement | Gibco | 17502048 | |
| Penicillin-Streptomycin | Gibco | 15140122 | |
| Pierce 16% Formaldehyde | Thermo-fisher | 28906 | |
| Rabbit anti-Recoverin antibody | Millipore | AB5585 | |
| Sodium Citrate | Sigma | W302600 | |
| Steriflip Sterile Disposable Vacuum Filter Units | MilliporeSigma | SE1M179M6 | |
| Taurine | Sigma | T0625 | |
| Tissue Plus- O.C.T. compound | Fisher Scientific | 23-730-571 | |
| Tissue-Tek Cryomold | EMS | 62534-10 | |
| Triton X-100 | Sigma | X100 | |
| Tween-20 | Sigma | P1379 | |
| Ultra-Low Attachment 6 well Plates | Corning | 29443-030 | |
| Ultra-Low Attachment 75cm2 U-Flask | Corning | 3814 | |
| Vacuum Filtration System | VWR | 10040-436 | |
| Vectashield-mounting medium | vector Labs | H-1000 | |
| wax pen-ImmEdge | vector Labs | H-4000 | |
| Y-27632 Dihydrochloride (Rock inhibitor) | Sigma | Y0503 |
Riferimenti
- Waring, M. J., et al. An analysis of the attrition of drug candidates from four major pharmaceutical companies. Nature Review Drug Discovery. 14 (7), 475-486 (2015).
- Khalil, A. S., Jaenisch, R., Mooney, D. J. Engineered tissues and strategies to overcome challenges in drug development. Advances in Drug Delivery Reviews. 158, 116-139 (2020).
- Demetrius, L. Of mice and men. When it comes to studying ageing and the means to slow it down, mice are not just small humans. EMBO Reports. , 39-44 (2005).
- Lindsay, K. J., et al. Pyruvate kinase and aspartate-glutamate carrier distributions reveal key metabolic links between neurons and glia in retina. Proceedings of the National Academy of Sciences U. S. A. 111 (43), 15579-15584 (2014).
- Cowan, C. S., et al. Cell types of the human retina and its organoids at single-cell resolution. Cell. 182 (6), 1623-1640 (2020).
- Kruczek, K., Swaroop, A. Pluripotent stem cell-derived retinal organoids for disease modeling and development of therapies. Stem Cells. 38 (10), 1206-1215 (2020).
- Sinha, D., Phillips, J., Phillips, M. J., Gamm, D. M. Mimicking retinal development and disease with human pluripotent stem cells. Investigative Ophthalmology and Visual Science. 57 (5), 1-9 (2016).
- Gan, L., Cookson, M. R., Petrucelli, L., La Spada, A. R. Converging pathways in neurodegeneration, from genetics to mechanisms. Nature Neuroscience. 21 (10), 1300-1309 (2018).
- Aung, K. Z., Wickremasinghe, S. S., Makeyeva, G., Robman, L., Guymer, R. H. The prevalence estimates of macular telangiectasia type 2: the Melbourne collaborative cohort study. Retina. 30 (3), 473-478 (2010).
- Chew, E. Y., et al. Effect of ciliary neurotrophic factor on retinal neurodegeneration in patients with macular telangiectasia type 2: A randomized clinical trial. Ophthalmology. 126 (4), 540-549 (2019).
- Gass, J. D., Blodi, B. A. Idiopathic juxtafoveolar retinal telangiectasis. Update of classification and follow-up study. Ophthalmology. 100 (10), 1536-1546 (1993).
- Klein, R., et al. The prevalence of macular telangiectasia type 2 in the Beaver Dam eye study. American Journal of Ophthalmology. 150 (1), 55-62 (2010).
- Powner, M. B., et al. Loss of Muller’s cells and photoreceptors in macular telangiectasia type 2. Ophthalmology. 120 (11), 2344-2352 (2013).
- Gantner, M. L., et al. Serine and lipid metabolism in macular disease and peripheral neuropathy. New England Journal of Medicine. 381 (15), 1422-1433 (2019).
- Scerri, T. S., et al. Genome-wide analyses identify common variants associated with macular telangiectasia type 2. Nature Genetics. 49 (4), 559-567 (2017).
- Alecu, I., et al. Cytotoxic 1-deoxysphingolipids are metabolized by a cytochrome P450-dependent pathway. Journal of Lipid Research. 58 (1), 60-71 (2017).
- Othman, A., et al. Fenofibrate lowers atypical sphingolipids in plasma of dyslipidemic patients: A novel approach for treating diabetic neuropathy. Journal of Clinical Lipidology. 9 (4), 568-575 (2015).
- Ohlemacher, S. K., Iglesias, C. L., Sridhar, A., Gamm, D. M., Meyer, J. S. Generation of highly enriched populations of optic vesicle-like retinal cells from human pluripotent stem cells. Curr Protoc Stem Cell Biol. 32, 1-20 (2015).
- Zhong, X., et al. Generation of three-dimensional retinal tissue with functional photoreceptors from human iPSCs. Nature Communication. 5, 4047 (2014).
- Eiraku, M., et al. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature. 472 (7341), 51-56 (2011).
- Wahlin, K. J., et al. Photoreceptor outer segment-like structures in long-term 3D retinas from human pluripotent stem cells. Science Reports. 7 (1), 766 (2017).
- Capowski, E. E., et al. Reproducibility and staging of 3D human retinal organoids across multiple pluripotent stem cell lines. Development. 146 (1), (2019).
- Luo, Z., et al. An optimized system for effective derivation of three-dimensional retinal tissue via Wnt signaling regulation. Stem Cells. 36 (11), 1709-1722 (2018).
- Ovando-Roche, P., et al. Use of bioreactors for culturing human retinal organoids improves photoreceptor yields. Stem Cell Research Therapy. 9 (1), 156 (2018).

