A Simple and Effective Method to Consistently Isolate Mouse Cardiomyocytes
Summary
The gold standard in cardiology for cellular and molecular functional experiments are cardiomyocytes. This article describes adaptations to the non-Langendorff technique to isolate mouse cardiomyocytes.
Abstract
The need for reproducible yet technically simple methods yielding high-quality cardiomyocytes is essential for research in cardiac biology. Cellular and molecular functional experiments (e.g., contraction, electrophysiology, calcium cycling, etc.) on cardiomyocytes are the gold standard for establishing mechanism(s) of disease. The mouse is the species of choice for functional experiments and the described technique is specifically for the isolation of mouse cardiomyocytes. Previous methods requiring a Langendorff apparatus require high levels of training and precision for aortic cannulation, often resulting in ischemia. The field is shifting toward Langendorff-free isolation methods that are simple, are reproducible, and yield viable myocytes for physiological data acquisition and culture. These methods greatly diminish ischemia time compared to aortic cannulation and result in reliably obtained cardiomyocytes. Our adaptation to the Langendorff-free method includes an initial perfusion with ice-cold clearing solution, use of a stabilizing platform that ensures a steady needle during perfusion, and additional digestion steps to ensure reliably obtained cardiomyocytes for use in functional measurements and culture. This method is simple and quick to perform and requires little technical skill.
Introduction
For decades, an essential idea in cardiac biology literature is the molecular mechanism of action. The mechanism of action must be established in order to publish reliable studies. A well-established strategy to determine molecular mechanism is isolated cardiomyocyte studies, which require high-quality cardiomyocytes for attaining trustable data. Cellular and molecular experiments performed on cardiomyocytes to determine mechanism of action are the gold standard for investigating contraction1, electrophysiology2, calcium (Ca2+) cycling3, myofilament Ca2+ sensitivity4, cytoskeleton5, metabolism6, effects of hormones7, signaling molecules8, drug studies9, etc. The mouse has become the species of choice for most cardiac biology experiments due to the ease of genetic manipulation, its small size, its relatively short lifespan, low cost, etc10. However, the reliable isolation of high-quality mouse cardiomyocytes is not trivial with current techniques.
Labs have been isolating cardiomyocytes for almost 70 years11. Virtually all techniques to isolate cardiomyocytes rely on digestion of the heart via various enzymes (collagenase, protease, trypsin, etc.). In the early periods (1950s-1960s), the chunk method was employed, which involved removing the heart, cutting into much smaller pieces and incubating in solution with collagenase/protease/trypsin12. In the 1970s labs implemented the ameliorated “Langendorff” method13, which isolated cardiomyocytes using a coronary artery perfusion-based isolation technique (retrograde perfusion with enzyme via the Langendorff apparatus); this technique remains the dominant method of myocyte isolation in the field today, ~50 years later14,15,16. Recent work has shifted to cannulating the heart in vivo to limit hypoxia time and ischemic damage resulting in superior cardiomyocyte isolations (better yields and higher quality)17. Recently, this has evolved into performing in vivo, Langendorff-free heart perfusions18,19,20,21,22. We have evolved the Langendorff-free cardiomyocyte isolation technique based on the Ackers-Johnson et al.18 technique and adapted various components from the many previous isolation techniques. These key adaptations include the injection of an ice-cold clearing buffer and the incorporation of a supporting platform to stabilize the needle, allowing for decreased manipulation of the heart. Also detailed in this technique is temperature control of injected buffers (37 °C), which decreased the time between in vivo injection and digestion due to less EDTA perfusion as previously published18. By decreasing manipulation of the heart and therefore minimizing puncture site size, thorough and constant perfusion of the coronary arteries is obtained. We also refined the technique with a secondary chunk method digestion, the amount of EDTA in the injected clearing buffer, and changed the pH. Our described technique is more reliable, more efficient, and does not require the extensive training/practice compared to the using the Langendorff apparatus (Table 1).
Protocol
All procedures performed in this study were approved by the Institutional Animal Care and Use Committee at the Ohio State University in accordance with NIH guidelines.
1. Solution preparation
NOTE: Please see Table 2 for buffer concentrations.
- Pre-isolation (up to 2 weeks in advance of myocyte isolation)
- Perfusion buffer base: Prepare 1 L of perfusion buffer base (NaCl, KCl, NaH2PO4, HEPES, Glucose, and BDM) in 1 L of ultrapure 18.2 MΩ·cm H2O. Adjust to pH 7.4 prior to 0.22 µm sterile filtration. Store at 4 °C until the day of isolation.
- Clearing buffer: Prepare 1 L of clearing buffer (NaCl, KCl, NaH2PO4, HEPES, Glucose, EDTA, and BDM) in 1 L of ultrapure 18.2 MΩ·cm H2O. Adjust to pH 7.4 prior to 0.22 µm sterile filtration. Store at 4 °C until the day of isolation.
- 100 mM CaCl2 stock solution: Weigh out 1.11 g of CaCl2 and dissolve into 100 mL of ultrapure 18.2 MΩ·cm H2O. Store at room temperature until the day of isolation.
- Liberase aliquots: Dissolve 5 mg of digestive enzymes in 5 mL of sterile, RNase-free water. Prepare 0.8 mL aliquots and store at -20 °C until the day of isolation.
- Laminate the dishes: Apply 100 µL of 40 µg/mL mouse laminin to sterile, glass-bottom 30 mL Petri dishes. Allow to sit for 30 min and remove excess laminin. Set the laminin on a Petri dish with UV incubation at room temperature for 30 min. Store at 4 °C until the day of isolation (laminated Petri dishes can be used for up to 2 weeks).
- DMEM media: In a biosafety cabinet, add 2.5 mL of FBS, 0.5 mL of Penicillin-Streptomycin (50 U/mL-50 µg), and 1 mL of 500 mM BDM to 46 mL of DMEM. Store at 4 °C until the day of isolation (media can be used for up to 2 weeks).
- M199 media: Prepare 1 L of M199 media (NaHCO3, HEPES, L-glutathione, M199, BDM, and BSA) in 1 L of ultrapure 18.2 MΩ·cm H2O. Adjust to pH 7.4 prior to sterile filtration (0.22 µm filter). Store at 4 °C until the day of isolation.
- 500 mM BDM stock: Add 0.5 g of BDM to 10 mL of ultrapure 18.2 MΩ·cm H2O. Store at 4 °C until the day of isolation.
- Stabilizing platform creation: Mold playdough around the base of the needle screwed into the Luer stopcock. Mold to the Petri dish that will contain the heart and ensure that the needle is at proper height for ventricle injection (~5 mm above bottom of dish).
- The day of isolation
- Perfusion buffer: On the day of the isolation, add 9.5 mg of MgCl2 to 100 mL of perfusion buffer base. Allow to mix completely before removing 30 mL of completed perfusion buffer and place in a clean, wide-mouth, 100 mL glass bottle with a screw cap lid (digestion buffer).
NOTE: These additions can be made on the day of isolation without additional pH adjustments as their addition negligibly alters the pH.- Remove 12 mL of perfusion buffer and set to the side (stop buffer). Pour the remaining 58 mL of perfusion buffer into another clean, wide-mouth, 100 mL glass bottle with a screw cap lid. Store the remaining perfusion buffer at 37 °C throughout isolation.
- Digestion buffer: Add 7.5 µL of 100 mM CaCl2 to 30 mL of perfusion buffer for a final concentration of 25 µM. Immediately prior to the isolation, add 770 µL of Liberase to the digestion buffer for a final concentration of 26 µg/mL.
- Stop buffer: Dissolve 240 mg of BSA in 12 mL of perfusion buffer. Store at 37 °C throughout isolation.
- Clearing buffer: Prepare a 3 mL syringe of clearing buffer and clear the needle of bubbles. Store on ice until isolation.
- Perfusion buffer: On the day of the isolation, add 9.5 mg of MgCl2 to 100 mL of perfusion buffer base. Allow to mix completely before removing 30 mL of completed perfusion buffer and place in a clean, wide-mouth, 100 mL glass bottle with a screw cap lid (digestion buffer).
2. Manifold preparation
- Clear the temperature-controlled manifold with freshly prepared perfusion buffer, being careful to ensure no bubbles remain. Using a Luer lock connector, screw in a 27 G needle, and clear of bubbles. A cleared manifold is essential for a successful isolation.
3. Animal preparation
- Inject the mouse intraperitoneally with 100 mg/kg ketamine and 20 mg/kg xylazine by body weight immediately prior to the isolation. This procedure used a male, 4-month-old C57Bl/6 mouse.
- If needed, place the mouse on a heated surgical pad to encourage sedation. Tent the arms of the mouse, and tape the limbs and base of the tail to a blue lab diaper. Ensure that the animal is fully anesthetized by the withdrawal of toe-pinch reflex. Apply a sterile drape around the surgical area.
4. Cardiomyocyte isolation procedure
- Expose the sternum of the mouse and, lateral to the midline, cut proximally through the ribs and to the axilla. Gently cut fully through the diaphragm, ensuring shallow cuts to avoid the heart. Clamp the sternum with a hemostat and fold the ribs backwards to expose the thoracic cavity.
- Gently remove the pericardium from the heart and cut fully through the inferior vena cava, immediately distal to the heart. Use a 3 mL syringe with a 27 G needle to quickly inject 3 mL of ice-cold clearing buffer into the right ventricle of the heart over 1 min.
- Gently hold the heart using tweezers and pull away from the body, exposing as much of the aorta as possible. Using a hemostat, clamp the ascending aorta, being careful not to clamp the atria, and excise the heart from the chest.
- Quickly transfer the clamped heart to the lid of a polypropylene Petri dish with approximately 10 mL of warm perfusion buffer. Place the clamped heart into the supporting platform and use a stabilized 27 G needle attached to the manifold to inject 10 mL of temperature-controlled 37 °C perfusion buffer into the left ventricle over 5 min.
- Transfer the clamped heart and supporting platform to a Petri dish with approximately 5 mL of digestion buffer. Exchange input syringes for a 50 mL syringe with 25 mL of digestion buffer.
- Clear the manifold of any bubbles and remaining perfusion buffer before injection. Carefully replace the needle into the same needle position in the left ventricular apex.
- Use a perfusion pump to inject temperature-controlled 37 °C digestion buffer into the left ventricle for 15 min using a 50 mL syringe. Control the temperature of the solution with a water jacket previously calibrated to eject 37 °C solution at the end of the needle.
- Remove the ventricles from the atria and transfer to a 10 mL beaker. Add 3 mL of digestion buffer to the beaker and, using sharp scissors, cut the ventricles into large chunks. Cover the beaker with aluminum foil and place in a shaking water bath preheated to 37 °C for 5 min.
- Discard the supernatant, being careful to avoid removing any chunks of tissue. Resuspend the tissue chunks in 3 mL of digestion buffer and triturate for approximately 4 min or until a homogenous mixture is achieved.
- Filter the cells through a 70 µm nylon cell strainer into a 50 mL polypropylene tube.
- Return the filtrate to a 14 mL round-bottom polypropylene tube and centrifuge for 1 min at 100 rpm. Discard the supernatant and resuspend the pellet in 3 mL of stop buffer.
- Add 54 µL of 100 mM CaCl2 stock to the resuspended cells for a final CaCl2 concentration of 1.8 mM. This step can also be performed stepwise to increase cell yield.
- Allow live cells to settle by gravity for 10 min before removing the supernatant containing dead cells. Resuspend the pellet in storage solution (perfusion solution with 200 µM CaCl2). These cells can now be used for functional experiments (i.e., calcium imaging/contraction, patch clamp, etc.), culture, etc.
NOTE: The cells may also be resuspended in lab- or experiment-dependent solutions.
5. Cell culture
- Count the cells using a hemocytometer or by a field view count using a gridded cover slip. As myocytes are very large, counting using a hemocytometer is not always accurate. This is used to get an idea of approximately how many cells were obtained from the isolation.
- Dilute the cells to ~25,000 cells/mL with DMEM media. Add 1 mL of cell solution to each well.
- Incubate at 37 °C, 95% O2, 5% CO2 for 2 h.
- Gently aspirate DMEM media from each well and add 2 mL of M199 media.
- Incubate at 37 °C, 95% O2, 5% CO2 for up to 24 h.
Representative Results
There are a few elements to examine when determining the success of an isolation. First, the cardiomyocytes must be rod-shaped with no membrane blebs, such as the cells isolated in Figure 1. A typical isolation will yield ~80% of the myocytes being rod-shaped. If the isolation yields anything less than 50% rod-shaped cells, then it is considered an unsuccessful isolation and cardiomyocytes are not used. Lastly, the cardiomyocytes should be quiescent. Spontaneously contraction myocytes demonstrate Ca2+ intolerance and will produce unreliable data. Of all the isolations performed using the above technique, nearly all isolations are considered successful. Cultured cardiomyocytes are considered successful if they are rod-shaped with no membrane blebs or rounded edges with high survival rates (Figure 2).
Table 1. A table of noted differences between our method, Langendorff methods, and previously published Langendorff-free methods of isolating mouse cardiomyocytes. Please click here to download this Table.
Table 2: Buffer media concentrations. Please click here to download this Table.

Figure 1. (A) 4-month-old C57Bl/6 mouse cardiomyocytes imaged using bright field microscopy. Yield: ~80%; 20x magnification. (B) Bright field image of a 4-month-old C57Bl/6 mouse cardiomyocyte. Cardiomyocytes are quiescent, exhibiting rod-shape with no membrane blebs. Scale bar is 100 µm. Please click here to view a larger version of this figure.
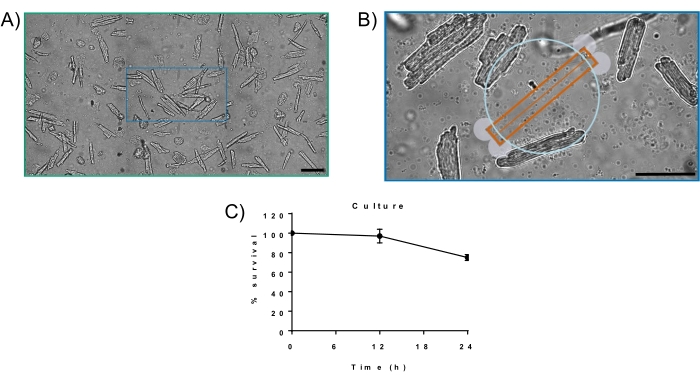
Figure 2. (A) The 4-month-old C57Bl/6 mouse cardiomyocytes cultured for 24 h in M199 media. Viability: ~80%; 20x magnification. (B) Myocytes cultured for 24 h. At 24 h, cardiomyocytes exhibit rod-shape with no membrane blebs. (C) % survival curve for cardiomyocytes cultured for 24 h using the described method. Scale bar is 100 µm. Please click here to view a larger version of this figure.
Discussion
The principal advantage of our Langendorff-free cardiomyocyte isolation technique is that it limits hypoxia and ischemic time by not requiring cannulation to a Langendorff apparatus. Alternatively to classical Langendorff techniques that take several minutes to remove, clean, and hang the heart, often resulting in ischemic damage to the myocyte, our method includes an in vivo clearing of blood via an ice-cold clearing solution. The ice-cold clearing buffer contains ethylenediaminetetraacetic acid (EDTA), irreversibly chelates divalent cations to efficiently remove calcium, making it an excellent anticoagulant, and therefore stops contraction23. The use of ice cold solutions inhibits contraction by slowing ion exchange and cellular metabolic rate, preventing damage caused by ischemia24. By removing the need for aortic cannulation, Langendorff-free techniques produce robust myocytes resulting in a more reliable preparation to yield trustworthy data, which is not always the case for Langendorff methods of isolation.
This is a variation of a previously published Langendorff-free isolation technique18 with adaptations as highlighted in Table 1. Most importantly, this technique introduces a stabilizing platform that limits the number of times the needle must be removed and replaced from the heart. Any unnecessary movement introduced to the needle while inserted into the ventricle widens the puncture site. A wider puncture site results in backflow out of the ventricle from the puncture site, decreased pressure in the heart, and decreased flow to the coronaries. This issue is fixed by using the stabilizing platform. By limiting the needle movement and the number of times the needle is removed and replaced (one compared to three times as previously described18), we maintain consistent flow to the coronaries and consistently achieve digestion. Another key adaptation was the addition of a temperature-controlled jacket to maintain enzyme solutions at 37 °C upon entering the heart (temperature bath was calibrated to eject at 37 °C from the needle). The digestion enzyme used has optimum reaction activity at 37 °C, and the addition of temperature control increases enzyme activity, therefore decreasing digestion time. Liberase also has minimal lot-to-lot variability and has higher reproducibility compared to other enzymes. Another adaptation of previous techniques is a decreased amount of injected clearing solution. Decreasing the amount of clearing buffer to enter the heart and allowing for more blood to be cleared by perfusion buffer decreases inactivation of the digestion enzyme by EDTA. Another reason that our method consistently achieves successful myocyte isolations is by the usage of BDM. BDM is a myosin inhibitor that prevents the power stroke mechanism of the sarcomere, thereby inhibiting contraction and limiting reoxygenation injury during digestion. By limiting contraction, we prevent calcium cycling and inherent reactive oxygen species production in contracting myocytes in culture. Since the culture media contains calcium, it is possible that the myocytes could contract and decrease subsequent yield after culture. We elect to culture the myocytes in BDM to prevent contraction and increase yield25,26. BDM can always be washed out of myocytes for functional measurements after culture with great success. Alternatively, all steps of this procedure can be performed without the addition of BDM, but isolations may not be considered as "successful" on BDM-free isolated myocytes.
Besides ischemic time, there are many other factors that will determine if an isolation will produce robust myocytes. Since there are different conditions in the individual labs, the persons performing the isolation may need to modify the amount of enzyme and/or calcium, the perfusion time and/or pump speed, and the rocking water bath speed and/or time. These parameters will be dependent upon the mouse model used, such as healthy or diseased, aging, etc.
While this technique does not require the surgical skill needed for the Langendorff based techniques, there are still critical steps to make the technique successful. The most common issue that yields poor myocytes with this technique is due to bubbles entering the perfusion system and blocking the myocardial tissue from access to the perfusion buffer. The solution to this problem is careful practice to avoid bubbles (via proper syringe clearance technique, proper needle clearance technique, burping the manifold of bubbles when changing syringes, etc.), as well as multiple exit points from the perfusion system in case a bubble is lodged in the manifold. Without bubbles, in our experience, we get near 100% successful myocyte isolations (defined as above 50% rod shaped myocytes; the average is ~80%).
While the method has been simplified by the removal of the surgical skill required for the Langendorff method, this adapted method has only been tried in mice. Another advantage of the Langendorff-free method is that it can be used for many murine models (i.e., from neonate to senescence), as previously described19. Unfortunately, larger mammals (e.g., dogs, pigs, etc.) will not be suitable for this technique due to the size of their hearts. It would be impossible to perfuse the heart with enough solution to acquire reliable myocytes.
Our adaption of the Langendorff-free method is a dependable method for the successful isolation of mouse cardiomyocytes. Compared to the Langendorff method, this alternative approach requires little technical skill to consistently obtain cardiomyocytes.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work was supported by National Institutes of Health Grants R01 HL114940 (Biesiadecki), R01 AG060542 (Ziolo), and T32 HL134616 (Sturgill and Salyer).
Materials
| 10 cc Bd Luer-Lok Syringe | Fisher Sci | 14-827-52 | |
| 10 mL Pyrex Low-Form Beaker | Cole-Palmer | UX-34502-01 | |
| 100 mL polypropylene cap glass media storage bottle | DWK Life Sciences | UX-34523-00 | |
| 14 mL Round-Bottom Polypropylene Test Tubes With Cap | Fisher Sci | 14-959-11B | |
| 2,3-Butanedione Monoxime | Sigma | B0753 | >98% |
| 3 cc BD Luer-Lok Syringe | Fisher Sci | 14-823-435 | |
| 35 mm glass bottom dishes | MatTek Corporation | P35G-1.0-20-C | |
| 50 mL BD Syringe without Needle | Fisher Sci | 13-689-8 | |
| 50 mL Conical Centrifuge Tubes | Cole-Palmer | EW-22999-84 | |
| 95% O2 5% CO2 | |||
| AIMS Space Gel Heating Pad | Fisher Sci | 14-370-223 | |
| BD PrecisionGlide 27 G X 1/2" Hypodermic Needles | Becton Dickinson | 305109 | |
| Bovine Serum Albumin | Sigma | A3803 | Heat shock fraction, lyophilized powder, essentially fatty acid free, >98% |
| Calcium Chloride dihydrate | Sigma | C7902 | >99% |
| D-(+)-Glucose | Sigma | G7021 | Suitable for cell culture, >99.5% |
| DMEM | Fisher Sci | 11965092 | |
| EDTA | Fisher Sci | AAA1071336 | |
| Falcon 100 mm TC-treated Cell Culture Dish | Corning | 353003 | |
| FBS | R&D Systems (Bio-techne) | S11195 | |
| Fisherbrand Isotemp Heated Immersion Circulators | Fisher Sci | 13-874-432 | |
| Hartman Mosquito Hemostatic Forceps | World Precision Instruments | 15921 | |
| Hausser Scientific Hy-Lite Counting Chamber Set | Fisher Sci | 02-671-11 | |
| HEPES | Sigma | H4034 | >99.5% |
| Labeling Tape | Fisher Sci | 15-901-10R | |
| Legato 100 Syringe Pump | kdScientific | 788100 | |
| L-glutathione | Fisher Sci | ICN19467980 | |
| Liberase TH Research Grade | Sigma | 5401135001 | High thermolysin concentration |
| M199 | Fisher Sci | MT10060CV | |
| Magnesium Chloride | Invitrogen | AM9530G | |
| Mouse Laminin | Corning | 354232 | |
| Pen/Strep | Fisher Sci | ||
| Potassium Chloride | Sigma | P5405 | >99% |
| Precision Digital Reciprocating Water Bath | ThermoFisher Scientific | TSCIR19 | |
| Sodium Bicarbonate | Sigma | S5761 | Suitable for cell culture |
| Sodium Chloride | Sigma | S5886 | >99% |
| Sodium phosphate monobasic | Sigma | S5011 | >99% |
| Sterile Cell Strainer 70 µm | Fisher Sci | 22-363-548 | |
| Student Fine Scissors | Fine Science Tools | 91460-11 | |
| VWR Absorbent Underpads | Fisher Sci | NC9481815 |
Riferimenti
- Ziolo, M. T., Dollinger, S. J., Wahler, G. M. Myocytes isolated from rejecting transplanted rat hearts exhibit reduced basal shortening which is reversible by aminoguanidine. Journal of Molecular and Cellular Cardiology. 30 (5), 1009-1017 (1998).
- Ziolo, M. T., et al. Myocytes isolated from rejecting transplanted rat hearts exhibit a nitric oxide-mediated reduction in the calcium current. Journal of Molecular and Cellular Cardiology. 33 (9), 1691-1699 (2001).
- Traynham, C. J., et al. Diesterified nitrone rescues nitroso-redox levels and increases myocyte contraction via increased SR Ca(2+) handling. PLoS One. 7 (12), 52005 (2012).
- Nixon, B. R., et al. Combined troponin I Ser-150 and Ser-23/24 phosphorylation sustains thin filament Ca(2+) sensitivity and accelerates deactivation in an acidic environment. Journal of Molecular and Cellular Cardiology. 72, 177-185 (2014).
- Swager, S. A., et al. Claudin-5 levels are reduced from multiple cell types in human failing hearts and are associated with mislocalization of ephrin-B1. Cardiovascular Pathology. 24 (3), 160-167 (2015).
- Pinckard, K. M., et al. A novel endocrine role for the BAT-released lipokine 12,13-diHOME to mediate cardiac function. Circulation. 143 (2), 145-159 (2021).
- Roof, S. R., Shannon, T. R., Janssen, P. M., Ziolo, M. T. Effects of increased systolic Ca2+ and phospholamban phosphorylation during beta-adrenergic stimulation on Ca2+ transient kinetics in cardiac myocytes. American Journal of Physiology-Heart and Circulatory Physiology. 301 (4), 1570-1578 (2011).
- Harris, J. E., et al. Exercise-induced 3′-sialyllactose in breast milk is a critical mediator to improve metabolic health and cardiac function in mouse offspring. Nature Metabolism. 2 (8), 678-687 (2020).
- Roof, S. R., et al. CXL-1020, a novel nitroxyl (HNO) prodrug, is more effective than milrinone in models of diastolic dysfunction-a cardiovascular therapeutic: an efficacy and safety study in the rat. Frontiers in Physiology. 8, 894 (2017).
- Milani-Nejad, N. J. Small and large animal models in cardiac contraction research: advantages and disadvantages. Pharmacology and Therapeutics. 141 (3), 235-249 (2014).
- Harary, I., Farley, B. In vitro studies of single isolated beating heart cells. Science. 131 (3414), 1674-1675 (1960).
- Kono, T. Roles of collagenases and other proteolytic enzymes in the dispersal of animal tissues. Biochimica Biophysica Acta. 178 (2), 397-400 (1969).
- Baker, J. B. An improved apparatus for mammalian heart perfusion. The Journal of Physiology. 115 (1), 30-32 (1951).
- Powell, T., Twist, V. W. A rapid technique for the isolation and purification of adult cardiac muscle cells having respiratory control and a tolerance to calcium. Biochemical and Biophysical Research Communications. 72 (1), 327-333 (1976).
- Motayagheni, N. Modified Langendorff technique for mouse heart cannulation: Improved heart quality and decreased risk of ischemia. MethodsX. 4, 508-512 (2017).
- Zhang, Z., et al. An improved procedure for isolating adult mouse cardiomyocytes for epicardial activation mapping. Journal of Cellular and Molecular Medicine. 25 (24), 11257-11263 (2021).
- Jian, Z., et al. In vivo cannulation methods for cardiomyocytes isolation from heart disease models. PLoS One. 11 (8), 0160605 (2016).
- Ackers-Johnson, M., et al. A simplified, Langendorff-free method for concomitant isolation of viable cardiac myocytes and nonmyocytes from the adult mouse heart. Circulation Research. 119 (8), 909-920 (2016).
- Weldrick, J. J., Abdul-Ghani, M., Megeney, L. A., Burgon, P. G. A rapid and efficient method for the isolation of postnatal murine cardiac myocyte and fibroblast cells. Canadian Journal of Physiology and Pharmacology. 96 (5), 535-539 (2018).
- Myachina, T. A., Butova, X. A., Khohlova, A. D. A modified Langendorff-free method for isolation of cadiomyocytes from adult rat heart. AIP Conference Proceedings. 2174 (1), 020140 (2019).
- Omatsu-Kanbe, M., Yoshioka, K., Fukunaga, R., Sagawa, H., Matsuura, H. A simple antegrade perfusion method for isolating viable single cardiomyocytes from neonatal to aged mice. Physiological Reports. 6 (9), 13688 (2018).
- Omatsu-Kanbe, M., Fukunaga, R., Mi, X., Matsuura, H. An antegrade perfusion method for cardiomyocyte isolation from mice. Journal of Visualized Experiments. (171), e61866 (2021).
- Bers, D. M., Patton, C. W., Nuccitelli, R. A practical guide to the preparation of Ca2+ buffers. Methods in Cell Biology. 99, 126 (1994).
- Grosso, D. S., Frangakis, C. J., Carlson, E. C., Bressler, R. Isolation and characterization of myocytes from the adult rat heart. Preparative Biochemistry. 7 (5), 383-401 (1977).
- Thum, J. B. Butadione Monoxime increases the viability and yield of adult cardiomyocytes in primary cultures. Cardiovascular Toxicology. 1 (1), 61-72 (2001).
- Wolska, B. M., Solaro, R. J. Method for isolation of adult mouse cardiac myocytes for studies of contraction and microfluorimetry. American Journal of Physiology. 271 (3), 1250-1255 (1996).

