Microsurgical Skills of Establishing Permanent Jugular Vein Cannulation in Rats for Serial Blood Sampling of Orally Administered Drug
Summary
Detailed microsurgical techniques are demonstrated to establish a longer-term jugular vein cannulation rat model for sequential blood collection in the same animal. Physiological and hematological parameters have been monitored during the rat’s recovery phase. This model has been applied to study pharmacokinetics of orally administered polyphenol without inducing animal stress.
Abstract
Blood sampling in small laboratory animals is necessary for pharmaceutical lead optimization but can cause great harm and stress to experimental animals, which could potentially affect results. The jugular vein cannulation (JVC) in rats is a widely used model for repeated blood collection but requires adequate training of surgery skills and animal care. This article details the microsurgical procedures for establishing and maintaining a permanent JVC rat model with specific focus on the placement and sealing of the jugular cannula. The importance of monitoring physiological (e.g., body weight, food, and water intake) and hematological parameters, was highlighted with results presented for 6 days post-surgery during the rat's recovery. The drug-plasma concentration-time profile of orally administered natural phenol ellagic acid was determined in the JVC rat model.
Introduction
Repeated acquisition of blood samples from small laboratory animals, such as rodents, guinea pigs, and rabbits, is an important aspect for pharmaceutical lead optimization and also for reducing the number of animals used in research1,2. The pipeline for developing new diagnostic tools and drug formulation (e.g., vaccine) requires access to different volumes of blood in order to evaluate their robustness and performance in vivo, such as pharmacokinetics (PK), toxicity, and sensitivity3,4,5.
The laboratory approach to blood sample collection is broadly classified into two types, surgical and nonsurgical6. The nonsurgical approach is relatively easy to grasp for the researcher, which includes common techniques, such as cardiac puncture, orbital sinus puncture, and bleeding of the saphenous and tail vein. Multiple blood sampling is possible by some non-surgical methods, but the sample volume is small and can cause physical wound and psychological stress to the animals1. On the other hand, the surgical approach is a favorite alternative to repeated venipuncture, and it involves placement of a temporary or permanent cannula in the blood vessels of animals7,8,9. The large blood volume could be repeatedly withdrawn through the cannula in conscious rats while avoiding the stress and pain due to the handling technique, restrain, and anesthesia7,8,10,11. However, the cannula implantation requires an experienced researcher with adequate training in order to successfully collect the blood.
Blood collection through jugular vein cannulation (JVC) in rats is the most widely used method to study the drug PK6,10,12,13. Yet, establishment of the JVC rat model needs careful practice of microsurgical skills and knowledge of postsurgical care and maintenance. Especially, after the surgery, the rat requires administration of analgesics and sufficient recovery time to reach stable physiological condition for further experiments13,14,15. Although the body weight gain (i.e., >10 g) is a valid and commonly applied indicator for the rat's recovery, it is not uncommon that the rats have unexpected death postoperatively due to dehydration, infection, and inflammation, which could be subtle to notice at the early onset14,15. In addition, catheter obstruction in the JVC model remains to be an issue during the blood collection.
The present protocol has demonstrated in detail the microsurgical procedures for JVC in an anesthetized rat with specific focus on the identification, isolation, and cannulation of the jugular vein. The importance of physiological and hematological monitoring of the rats during the recovery phase is highlighted. Finally, serial blood samples were collected through the venous catheter to study the PK of the orally administered natural phenol ellagic acid with poor bioavailability (i.e., low systemic concentration) to verify the JVC rat model.
Protocol
The procedures described below were performed as part of a protocol approved by the Institutional Animal Care and Use Committee of Northwestern Polytechnical University (No. 202101117).
1. Preoperative preparation (the day before the surgery)
NOTE: Required solutions: normal saline (0.9% w/v sodium chloride), heparinized saline (1% w/v heparin sodium), catheter lock solution, non-steroidal anti-inflammatory drug (NSAID), such as meloxicam solution (2 mg/mL).
- Solution preparation
- Aliquot 200 µL of pre-packaged catheter lock solution in a 1.5 mL sterile microcentrifuge tube.
NOTE: Catheter lock solution is composed of heparinized saline (0.4% v/v heparin sodium) mixed with glycerol (v/v,1:1). - Mix 1 g of heparin sodium in 100 mL of the normal saline to prepare 1% heparinized saline.
- Dissolve meloxicam in normal saline to prepare a 2 mg/mL concentration solution for postoperative pain relief.
NOTE: Prepared heparinized saline and meloxicam solution are filtered through a 0.22 µm filter. All the solutions are sterilized and stored at 4°C for future use.
- Aliquot 200 µL of pre-packaged catheter lock solution in a 1.5 mL sterile microcentrifuge tube.
- Surgical instruments and materials
- Pack all clean surgical tools in a pouch and tape it with a piece of autoclave sterilization tape. Refer to Figure 1A for the specific surgical instruments used.
- Autoclave the surgical pouch at 121 °C for 30 min for the next day use.
- Animal preparation
- Prior to the surgery, house all male Sprague-Dawley (SD) rats in the standard Animal Room with controlled temperature at 22 ± 1 °C. Feed them with the standard laboratory food and water ad libitum for at least 7 days.
NOTE: Both male and female rats can be used for the JVC model, and their typical ages and body weights vary from 9-14 weeks and 294 ± 57 g, respectively. - Anesthetize the rat with 3%-3.5% isoflurane mixed with oxygen in a pre-anesthesia chamber. Determine whether the rat becomes unconscious by the lack of response to foot pinch.
- Gently take the rat out, place the rat's nose into an anesthetic nosepiece supplying 2%-2.5% isoflurane.
- In the ventral and dorsal position, remove the fur thoroughly around its right shoulder and posterior areas of neck with depilatory cream and a pet razor. Return the rat to the cage for surgery to be performed on the next day.
- Prior to the surgery, house all male Sprague-Dawley (SD) rats in the standard Animal Room with controlled temperature at 22 ± 1 °C. Feed them with the standard laboratory food and water ad libitum for at least 7 days.
2. Before the surgery on the day
- Prepare the aseptic workstation
- Spray 75% medical alcohol to disinfect the operation area, and then place the heating pad covered with a clean cushion. Set the LED lamp with a cold light source beside the workstation.
- Pre-warm the required solutions (step 1.1) to room temperature.
- Fill 0.6 mL of heparinized saline and 0.15 mL of catheter lock solution in two sterile 1.0 mL blunt tipped syringes, respectively. Withdraw 2.5 mL of the normal saline using a sterile 5.0 mL syringe.
- Soak the cotton balls in 75% medical alcohol. Squeeze out excess ethanol before use.
- Weigh and record the rat's body weight.
3. During the surgery
- Surgical preparation
- Wear the surgical coat, sterile gloves, and facial mask. Then open the sterilized surgical pouch, leave all surgical tools in 75% medical alcohol, and dry them before use.
- Jugular vein isolation
NOTE: The estimated operation time for this part is 10 min.- Anesthetize the surgery-ready and shaved rat with 3%-3.5% isoflurane mixed with oxygen in an induction chamber and determine whether the rat becomes unconscious by the lack of response to foot pinch.
- Place the rat's nose into the nosepiece supplied with 2%-2.5% isoflurane to maintain the anesthesia.
- Subcutaneously inject (s.q.) meloxicam solution at a dose of 2 mg/kg.
NOTE: Make sure to select analgesics that do not interact with the drug compound of interest in the pharmacokinetics study. - Using adhesive tape, restrain the rat's forearms in their ventral position to each side of the surgical platform.
- Gently scrub the surgical area by alternating between cotton balls soaked in 75% medical alcohol and iodine-based scrub for a total of three times.
- Carefully lift the skin near the clavicle on the right side of the midline of the neck with forceps and make an incision towards the chest about 1.5-2.0 cm in length with a pair of surgical scissors.
- Blunt dissect the thin tissue cover with iris scissors to expose the underneath jugular vein. The proximal cephalic end of the external jugular vein consists of two branches, which can be visually identified.
NOTE: Depending on the age and sex of the rat, the soft tissue (e.g., salivary glands, lymphatic nodes, and fatty tissues) covering the jugular vein varies. Compared to the young rats, the old rats are fatter (e.g., BW > 300 g), and thus need more tissue separation before the jugular vein is visible. - Lift the jugular vein along with its connective membranous tissues to visualize the lymph gland attached to the jugular vein. Carefully separate the vein along the vascular direction from surrounding muscle, fat, and other tissues.
- Nudge the forceps under the jugular vein without damaging the collateral blood vessels and pass two pieces of 6-0 suture under the vein to mark the two ends of the blood vessel individually.
- Pull one piece of the suture as far as possible toward the rat head and ligate the vein cranially with 2-3 knots using forceps.
- Place the second ligature on the caudal end of the vein with 1 loose knot.
- Jugular vein cannulation
NOTE: The estimated operation time for this part is 15 min.- Open the package containing 11 cm polyurethane (PU) catheter (I.D. 0.6 mm x O.D. 0.9 mm, Figure 1B) and attach the catheter to the prepared blunt tipped syringe filled with the heparinized saline.
- Slowly push the heparinized saline into the catheter to avoid air bubbles.
- Nudge the non-tip flat side of the forceps under the jugular vein to exit on the other side. Make a small v-shaped cut on the vein near the cranial tie with a pair of castroviejo micro scissors and gently open the incision with the tip of the elbow vessel dilator forceps.
NOTE: Rinse the incision with pre-warmed normal saline (37 °C) if a small amount of blood gushes out. - Cut out the oblique opening of the front end of the jugular vein catheter. Clamp the oblique end of the tube with forceps and slide it into the jugular vein.
NOTE: This step may need another person to facilitate the catheter sliding. - While advancing the catheter, slowly withdraw the elbow microsurgical forceps and clamp the outer surface of the vessel with forceps.
NOTE: If the right blood vessel is selected and the tip of the catheter is successfully slid into the blood vessel, the entire catheter insertion process should not feel any flow resistance. - Stop inserting the catheter upon hitting the first blue mark of the PU tube (Figure 1B), which is approximately 3.0 cm in length.
- Secure the inserted catheter to the vein with both caudal and rostral ligatures using forceps.
- Thread a 6-0 suture through the exposed tissue on the right side of the incision using a suture needle (1/2 curved cutting, 12 mm) and tie the ligature with a hemostat.
- Bend the catheter at the second blue mark (Figure 1B) to bind with the same ligature (in step 3.3.8) and to avoid occluding the PU-tubing.
- Snip all the extra suture thread and close the catheter by replacing the blunt tipped syringe with a 22 G stainless-steel plug.
- Catheter exteriorization
NOTE: The estimated operation time for this part is 10 min.- Place the rat in the dorsal position and gently clean the area between the scapulae with the cotton ball soaked in 75% medical alcohol.
- Make a very small incision at the center of the dorsal neck with surgical scissors. Through the dorsal incision, guide and gently push the trochar underneath the skin toward the ventral incision on the right side of the neck.
- Put the venous catheter into the trochar and then pull out and guide the venous catheter toward the dorsal incision.
- Secure the exteriorized catheter into the muscle layer in the same way with the suture (see the procedure in steps 3.3.8 and 3.3.9).
- Close the skin layer of ventral and dorsal incisions with the 6-0 nylon suture and suture needle (3/8 curved cutting, 17 mm). Swab all surgical incisions with iodophor.
NOTE: The wound-clips is an alternative method to close the skin incision. - Remove the catheter plug by clasping the catheter with fingertips. Place a new blunt tipped syringe and slowly draw back the syringe to test the blood flow.
NOTE: Since the rat is in the supine position, one may not be able to obtain blood samples. Blood samples could be obtained by changing to a side body position. - Hold the catheter again with fingertips and inject 0.2 mL of heparinized saline and 0.1 mL of lock solution into the catheter using the blunt tipped syringe.
- Hold the catheter with fingertips and replace the syringe with a stainless-steel plug. Unclamp the catheter and push the plug slightly in to ensure the tightness of the catheter.
4. Immediate post-surgical care
- Recover the rat in the dorsal decubitus position by caging it individually with fresh corncob bedding. Often, provide a temperature-regulated heating pad to maintain the core body temperature.
NOTE: For animal welfare, leaving food and water on the bedding is an effective way to alleviate the pain caused by neck movements when eating and drinking. - Record the end time of the surgery and monitor the rat at 2 h intervals for at least 4 h. Provide additional analgesia for the recovery if the rat shows signs of pain or distress.
5. Physiological and hematological monitoring during recovery phase
- Monitor the body weight and the food and water intake daily and record the data.
- To collect a small volume of fresh blood for hematological test, place the rat in a restrainer. Open the plug and insert the syringe into the venous PU catheter to ensure the catheter is not obstructed.
NOTE: The blood collection was performed at the same time daily for 6 consecutive days. - Discard the initial withdrawn blood, which contains a mixture of blood, heparinized saline, and catheter lock solution.
- Use a new syringe to collect 150 µL of fresh blood sample and transfer the blood sample to the 0.5 mL tube containing K2EDTA (1.8 mg/mL blood) spray dried on the tube wall.
NOTE: If the catheter is blocked, 0.2 mL of heparinized saline can be injected into the catheter to flush the catheter a few minutes before the next blood collection time. - Inject sterile saline in the same volume to compensate for the withdrawn blood. Inject 150 µL of pre-warmed normal saline (37 °C) and infuse 0.2 mL sterile heparinized normal saline through the catheter.
- Inject 100 µL of the lock solution into the catheter to ensure the sealing and sterility of the catheter before the next sample collection.
- Analyze the blood samples within 2 h of collection using an automated blood cell counter.
6. Repeated blood sampling for pharmacokinetic studies of oral administered drug
NOTE: Rats with weight gain >10 g and stable hematological level are suggested to be enrolled for future study. Following the current protocol, the JVC rats required 4 to 6 days to recover.
- After 4-6 days of surgery, fast the rat for 12 h with free access to water.
NOTE: Depending on the experimental goal, fasting the animal is optional. - Orally gavage the fasted rat with natural phenol bioactive ellagic acid at a dose of 6 mg/kg with a straight gavage needle16.
- Collect 200 µL of blood samples in the heparinized tubes via the jugular vein cannula at pre-determined time points over 24 h post-oral administration. The blood collection process follows the procedure in step 5.5.
NOTE: The catheter does not need to be closed with the lock solution until the blood collection is completed. - Immediately centrifuge the blood sample at 3000 x g at 4 °C for 10 min.
- Analyze the extracted plasma sample by liquid chromatography-mass spectroscopy17,18.
Representative Results
This protocol has thoroughly demonstrated how to establish a long-term JVC model using microsurgical skills for serial blood collection. Figure 1A shows the essential surgical instruments and materials used to conduct the surgery. The specification of PU catheter with three blue marks is also illustrated, which is helpful for guiding the researcher to place the vein cannula in step 3.3., how to use the marks on the PU catheter to guide the cannulation (Figure 1B). It is also important to be aware of the timeline required to establish the JVC rat model (Figure 1C). Although the operating time for the JVC is approximately 35 min, if the researcher is skillful, it takes 10-14 days (the adaptation and recovery phase) for the JVC rat model to be ready for use, compared to the non-surgical approach, such as the tail snipping or orbital sinus puncture, which can be used immediately with proper training.
The physiological and hematological conditions over 6 days postoperatively was also investigated (Figure 2). The rat's body weight gain, food and water intake, and complete blood cell count were variable during the recovery phase (Figure 2A,B). It was found that the majority of rats under the present study condition recover within 4-6 days post-surgery as evidenced by restored levels of some key features, such as body weight gain >10 g, regular diet intake, and selected blood components relating to infection, dehydration, and inflammation, including white blood cell count, red blood cell count, hemoglobin and platelet count (Figure 2C–F). It is worth noting that the amount of water intake in rats was relatively large on the first day post-operation, indicating dehydration.
Pharmacokinetics of the natural polyphenol, ellagic acid was studied in the established JVC rat model (Figure 3). The ellagic acid is characterized with poor drug bioavailability. When administered in a low dose (e.g., 6 mg/kg), a large volume of blood sample is required to detect its concentration in the plasma. Figure 3 shows low plasma-concentration ellagic acid concentration in ng/mL over 24 h and its varied gastro-intestinal tract (GIT) absorption owing to its poor solubility and permeability.
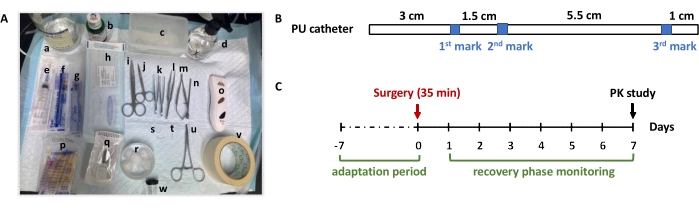
Figure 1: Overview of the main surgical instruments and supplies used for JVC rat model establishment. (A) Top: a-d is normal saline, iodophor, plastic ware, spray bottle with 75% medical alcohol, respectively; Middle: e-o is 5.0 mL syringe, 1.0 mL syringe, blunt tipped syringe, sterile cannula, surgical scissors, iris scissors, half-curved forceps, vessel dilator balanced forceps, castroviejo micro scissors, stainless steel trochar, pet razor, respectively; bottom: p-w is cotton swabs, 6-0 sterile non-absorbable nylon suture thread, cotton balls, two types of suture needle, stainless steel plug, curved hemostat, adhesive tape, anesthetic nosepiece, respectively. (B) Specification of the PU catheter used for cannulation of jugular vein in rats. The catheter is 11 mm in total length with O.D 0.6 mm x I.D 0.9 mm. The catheter has three blue marks to serve as an anchor point during the cannulation; (C) Suggested timeline of establishing JVC rat model. In this study, the rat's body weight, as well as the food and water intake, were recorded daily during the recovery phase, and blood samples were collected once daily for routine hematological monitoring. Please click here to view a larger version of this figure.
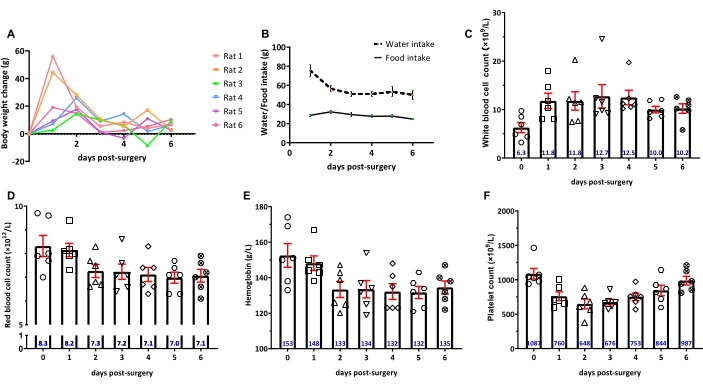
Figure 2: Physiological and hematological monitoring of rats over 6 days post-operatively. (A) Body weight change; (B) The change in water and food intake; (C–F) White blood cell count, red blood cell count, hemoglobin, and platelet count, respectively. The data represent the mean ± SEM with n = 6. The numeric values in blue represent the mean value. Please click here to view a larger version of this figure.
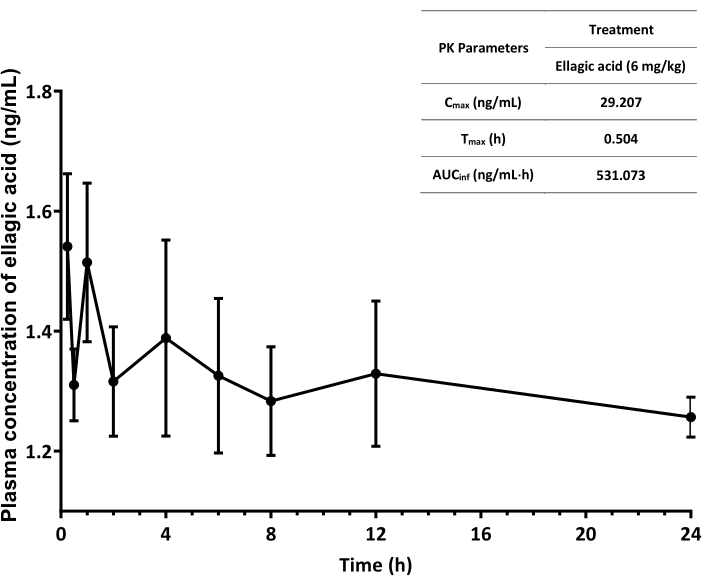
Figure 3: Plasma ellagic acid concentration-time profiles of rats over 24 h after oral gavage. The data represent the mean ± SEM with n = 3. The values of PK parameters are obtained using add-in program PKSolver in a spreadsheet software (e.g., Microsoft Excel)19. Cmax: peak concentration, Tmax: time to reach Cmax; AUCinf: area under the plasma concentration-time curve from time zero to infinity. Please click here to view a larger version of this figure.
Discussion
Mastering the technique of vessel cannulation requires significant practice and learning the lesson from each operation. Christakis et al. using cumulative sum (CUSUM) analysis, found that a researcher needs to practice 200 rats over a period of one year before being ready for the PK evaluation of drug candidates20. Yet, the operating time required for the vein cannulation can be significantly reduced by the number of rats performed13,20. Using our protocol, the success rate of effectively cannulating the jugular vein and collecting the blood sample increased from approximately 50% to above 80% (total rats performed were 15), and the initial operating time was reduced to 35 min from 2 h.
The demonstration of establishing a JVC rat model involves several critical steps. Firstly, the incision area around the neck is important for initially locating the jugular vein. If the right JVC is performed, the incision area is generally selected on the upper side of the clavicle along the right side of the neck midline (see section 3.2 jugular vein isolation). Secondly, JVC depends on preparation of a clean segment of the vein. Upon blunt dissection of soft tissue, the jugular vein is visible and identified by these two features: 1) two branches at the proximal end, and 2) a lymph node attached to it. Thirdly, while sliding the catheter into the jugular vein (see section 3.3 jugular vein cannulation), trimming the front end of the catheter, and supporting the blood vessel with steady external force could greatly improve the success rate of cannulation. Moreover, proper analgesia and heat must be provided for comforting the rat, as stress and pain can cause alterations in animal's behavior that may influence their post-operative recovery. Lastly, the duration of anesthesia, heat loss, and the complication can cause unexpected rat death; thus, it is important to closely monitor the rats during and after the surgery for at least 3 days. Evaluation of multiple health indicators, such as the body weight gain, diet, and drinking status, and hematological components of rats during the recovery period, could provide information that can be compared with reference values of interest of healthy SD rats in the database21,22,23,24. If rats experience dehydration, sterile isotonic fluids at 3%-5% of the body weight can be injected subcutaneously at the end of the surgery to compensate for the fluid loss. Most rats gain their body weight (e.g., >10 g) by day 3 post-surgery and thus, should be ready for use. Yet, for studies involving blood biomarkers evaluation (e.g., leukocyte, cytokines), it is recommended to enroll the rats by day 4-6 post-surgery, to ensure the normal hematological indexes for rats.
Despite its usefulness in PK study, depending on the catheter materials, not all drug candidates are suitable for the single cannulation. Gaud et al. found high log P compounds were bound to the PE catheter material, resulting in altered PK25. In addition, the analgesics (e.g., meloxicam) is often applied to reduce the pain in rat post-surgery. Considering the elimination half-life of meloxicam is around 19-23 h26,27, the single dose of meloxicam (2 mg/kg) injected s.q. is almost cleared out of the body after 24 h. Yet, potential drug-drug interactions can occur in use of meloxicam. For example, meloxicam can compete with other drugs for Cytochrome P450 metabolism28,29. Thus, the dose and type of analgesics selected should be screened depending on the drug chosen for the pharmacokinetic study. If the drug of interest interacts with meloxicam, other painkillers (e.g., buprenorphine) may be used.
In conclusion, this protocol has thoroughly demonstrated how to establish a long-term JVC rat model for blood collection at the laboratory setting and to investigate the physiological status of rats during the postsurgical recovery phase. The highlighted vital surgical steps and experiences could be helpful for the researcher to efficiently achieve the application of the cannulation model.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work is supported by National Natural Science Foundation of China (No. 82003692) to R.X. Zhang; Top Academic Scholarship at Northwestern Polytechnical University to R. Miao.
Materials
| 0.5 mL test tube containing EDTA anticoagulant | Xinkang | N/A | collecting blood samples for hematology test |
| 0.5*20 mm 1.0-mL syringe | KLMEDICAL | N/A | washing or replacing the fluid with saline |
| 0.6*28.5 mm 5.0-mL syringe | HD | N/A | Subcutaneous injection |
| 1.0-mL Blunt tipped syringe (22G) | skillsmodel | S4-PKT22G | Inject the saline and collect blood samples through catheter |
| 1.5 mL sterile microcentrifuge tube | Axygen | MCT-150-C-S | Store sterile catheter lock solution heparinized saline and meloxicam solution |
| 1.5 mL microcentrifuge tubes | Biosharp | BS-15-M | blood collection |
| 1/2 circle cutting 5*12 mm suture needle | skillsmodel | S4-FHZ | Thread the muscle layer to fix the catheter |
| 3/8 circle cutting 7*17 mm suture needle | skillsmodel | S5-FHZ | Suture the incision of rat cortex |
| 6-0 sterile non-absorbable silk suture thread | JUNSHENG | N/A | ligature |
| 75% medical alcohol | HONGSONG | N/A | Disinfection |
| Adhensive tape | LIUTAI | N/A | positioning the rat |
| Autoclave sterilization tape | Biosharp | BS-QT-028 | Mark sterilized items |
| Automated blood cell counter | Sysmex | XN-550 | Hematology test |
| Castroviejo micro scissors | skillsmodel | WA1010 | Cut the opening in the blood vessel |
| Centrifuge | Thermo Fisher Scientific | 75002402 | Plasma preparation |
| Clean cushion | Qingjie | N/A | Prepare the operation area |
| Cotton balls | HC | N/A | Wound disinfection and sterilization |
| Cotton swabs | BEITAGOGO | N/A | Disinfection |
| Curved hemostat | skillsmodel | N/A | ligature |
| DN50 Stainless-steel rat restrainer | skillsmodel | S4-RGDQ1 | Restrict the movement of rats for easy operation |
| Ellagic acid | Aladdin | E102710-25g | natural phenol for oral administration |
| Half-curved forceps | skillsmodel | 53072 | Lift the muscle layer and tissue, isolate the jugular vein and tie the suture |
| Heating pad | Warm mate | N/A | preventing heat loss of animal |
| Heparin sodium | Solarbio | H8060 | anticoagulant |
| Iodophor | Xidebao | N/A | Clean the wound |
| Iris scissors | skillsmodel | 54002 | Bluent separation the muscle layer |
| Isoflurane | RWD | R510-22-16 | anaesthesia |
| LED lamp | EMPERORFEEL | N/A | sugery |
| Liquid chromatography-mass spectroscopy | Thermo Fisher Scientific | VQF01-20001/ TSQ02-10002 | detection of drug concentration in plasma |
| Meloxicam | Hongqiang | N/A | Analgesic |
| Normal saline | KL | N/A | Prepara the solution and protect blood vessels from drying out |
| Pet razor | Codos | 3180 | Shaving the fur |
| Phosphate-buffered saline | ZHHC | PW012 | Preparation of Ellagic acid solution |
| PU catheter | skillsmodel | RJVC-PU | Jugular vein cannulation |
| Small animal operation anesthesia console | RWD | 68620 | Operation workstation |
| Spray bottle | Altro | N/A | aseptic workstation |
| Stainless steel plug (22G) | skillsmodel | S4-PKD22G | Plug the catheter to ensure its sealing |
| Stainless steel trochar | skillsmodel | S$-PKDGZ | Guide the catheter exteriorization |
| Sterile lock solution | skillsmodel | SK-FB | lock the catheter to ensure its sterility |
| Straight feeding needle | skillsmodel | N/A | Oral gavage |
| Surgical pouch | BKMAM | N/A | container for sterilization of surgical instruments |
| Surgical scissors | skillsmodel | J21070 | Cut incision on rat skin |
| Vessel dilator balanced forceps | skillsmodel | WA3020 | Expand the blood vessel and guide the cannula to slide in |
| ZS-MV Small animal anesthesia machine | ZSLab | 1057003 | inducing and maintaining anaesthesia |
Riferimenti
- Parasuraman, S., Raveendran, R., Kesavan, R. Blood sample collection in small laboratory animals. Journal of Pharmacology and Pharmacotherapeutics. 1 (2), 87-93 (2010).
- Sadler, A. M., Bailey, S. J. Validation of a refined technique for taking repeated blood samples from juvenile and adult mice. Laboratory Animals. 47 (4), 316-319 (2013).
- Zhang, R. X., et al. Coordinating biointeraction and bioreaction of a nanocarrier material and an anticancer drug to overcome membrane rigidity and target mitochondria in multidrug-resistant cancer cells. Advanced Functional Materials. 27 (39), 12 (2017).
- Zhang, R. X., et al. Polymer-lipid hybrid nanoparticles synchronize pharmacokinetics of co-encapsulated doxorubicin-mitomycin C and enable their spatiotemporal co-delivery and local bioavailability in breast tumor. Nanomedicine-Nanotechnology Biology and Medicine. 12 (5), 1279-1290 (2016).
- Zhang, R. X., et al. Sample extraction and simultaneous chromatographic quantitation of doxorubicin and mitomycin C following drug combination delivery in nanoparticles to tumor-bearing mice. Journal of Visualized Experiments: JoVE. (128), e11 (2017).
- Bakar, S. K., Niazi, S. Simple reliable method for chronic cannulation of the jugular vein for pharmacokinetic studies in rats. Journal of Pharmaceutical Sciences. 72 (9), 1027-1029 (1983).
- Harms, P. G., Ojeda, S. R. A rapid and simple procedure for chronic cannulation of the rat jugular vein. Journal of Applied Physiology. 36 (3), 391-392 (1974).
- Thrivikraman, K. V., Huot, R. L., Plotsky, P. M. Jugular vein catheterization for repeated blood sampling in the unrestrained conscious rat. Brain Research Protocols. 10 (2), 84-94 (2002).
- Weeks, J. R., Davis, J. D. Chronic intravenous cannulas for rats. Journal of Applied Physiology. 19 (3), 540-541 (1964).
- Goldkuhl, R., et al. Plasma concentrations of corticosterone and buprenorphine in rats subjected to jugular vein catheterization. Laboratory Animals. 44 (4), 337-343 (2010).
- Steffens, A. B. A method for frequent sampling of blood and continuous infusion of fluids in the rat without disturbing the animal. Physiology & Behavior. 4 (5), 833-836 (1969).
- Terao, N., Shen, D. D. Alterations in serum protein binding and pharmacokinetics of l-propranolol in the rat elicited by the presence of an indwelling venous catheter. Journal of Pharmacology and Experimental Therapeutics. 227 (2), 369-375 (1983).
- Feng, J., et al. Catheterization of the carotid artery and jugular vein to perform hemodynamic measures, infusions and blood sampling in a conscious rat model. Journal of Visualized Experiments: JoVE. (95), e51881 (2015).
- Karim, N., Ali, S. Jugular vein cannulation in rats – A mini review. Canadian Journal of Pure and Applied Sciences. 3, 929-935 (2009).
- Ling, S., Jamali, F. Effect of cannulation surgery and restraint stress on the plasma corticosterone concentration in the rat: application of an improved corticosterone HPLC assay. Journal of Pharmacy & Pharmaceutical Sciences. 6 (2), (2003).
- Lei, F., et al. Pharmacokinetic study of ellagic acid in rat after oral administration of pomegranate leaf extract. Journal of Chromatography B-Analytical Technologies in the Biomedical and Life Sciences. 796 (1), 189-194 (2003).
- Yan, L. L., et al. Method development and validation for pharmacokinetic and tissue distributions of ellagic acid using Ultrahigh Performance Liquid Chromatography-Tandem Mass Spectrometry (UPLC-MS/MS). Molecules. 19 (11), 18923-18935 (2014).
- Long, J. F., et al. Bioavailability and bioactivity of free ellagic acid compared to pomegranate juice. Food & Function. 10 (10), 6582-6588 (2019).
- Zhang, Y., et al. PKSolver: An add-in program for pharmacokinetic and pharmacodynamic data analysis in Microsoft Excel. Computer Methods and Programs in Biomedicine. 99 (3), 306-314 (2010).
- Christakis, I., et al. Learning curve of vessel cannulation in rats using cumulative sum analysis. Journal of Surgical Research. 193 (1), 69-76 (2015).
- Nm, S., Oduola, A. Haematological profile shows that Inbred Sprague Dawley rats have exceptional promise for use in biomedical and pharmacological studies. Asian Journal of Biomedical and Pharmaceutical Sciences. 4 (37), 33-37 (2014).
- Lillie, L. E., Temple, N. J., Florence, L. Z. Reference values for young normal Sprague-Dawley rats: weight gain, hematology and clinical chemistry. Human & Experimental Toxicology. 15 (8), 612-616 (1996).
- He, Q. L., et al. Sex-specific reference intervals of hematologic and biochemical analytes in Sprague-Dawley rats using the nonparametric rank percentile method. PLoS One. 12 (12), 18 (2017).
- EPA. Recommendations for and Documentation of Biological Values for Use in Risk Assessment. U.S. Environmental Protection Agency. , (1988).
- Gaud, N., et al. Single jugular vein cannulated rats may not be suitable for intravenous pharmacokinetic screening of high logP compounds. European Journal of Pharmaceutical Sciences. 99, 272-278 (2017).
- Turck, D., et al. Clinical pharmacokinetics of meloxicam. Arzneimittel-Forschung/Drug Research. 47 (3), 253-258 (1997).
- Aghazadeh-Habashi, A., Jamali, F. Pharmacokinetics of meloxicam administered as regular and fast dissolving formulations to the rat: Influence of gastrointestinal dysfunction on the relative bioavailability of two formulations. European Journal of Pharmaceutics and Biopharmaceutics. 70 (3), 889-894 (2008).
- Ludwig, E., et al. Activation of human cytochrome P-450 3A4-catalyzed meloxicam 5 ‘-methylhydroxylation by quinidine and hydroquinidine in vitro. Journal of Pharmacology and Experimental Therapeutics. 290 (1), 1-8 (1999).
- Zhang, R. X., et al. Nanoparticulate drug delivery strategies to address intestinal cytochrome P450 CYP3A4 metabolism towards personalized medicine. Pharmaceutics. 13 (8), (2021).

