Generation of Hook Ischemia-Reperfusion Model using a Three-Day Developing Chick Embryo
Summary
This paper describes ischemia-reperfusion (I/R) modeling in a 3-day chick embryo using a spinal needle customized hook to better understand I/R development and treatment. This model is simple, quick, and inexpensive.
Abstract
Ischemia and reperfusion (I/R) disorders, such as myocardial infarction, stroke, and peripheral vascular disease, are a few of the leading causes of illness and death. Many in vitro and in vivo models are currently available for studying the I/R mechanism in disease or damaged tissues. However, to date, no in ovo I/R model has been reported, which would allow for a better understanding of I/R mechanisms and faster drug screening. This paper describes I/R modeling using a spinal needle customized hook in a 3-day chick embryo to understand I/R development and treatment mechanisms. Our model can be used to investigate anomalies at the DNA, RNA, and protein levels. This method is simple, quick, and inexpensive. The current model can be used independently or in conjunction with existing in vitro and in vivo I/R models.
Introduction
Ischemia-reperfusion tissue injury has been linked to a number of pathologies, including heart attacks, ischemic stroke, trauma, and peripheral vascular disease1,2,3,4,5. This is primarily due to a lack of a comprehensive understanding of the disease progression and the lack of an effective research model. Ischemic injury occurs when the blood supply to a specific area of the tissue is cut off. As a result, ischemic tissue eventually necrotizes, though the rate varies depending on the tissue. Hence, restoring the blood supply may help to mitigate the damage. However, it has been observed, in some cases, that reperfusion causes more tissue damage than ischemia alone does6,7,8. Therefore, understanding the molecular and cellular mechanisms of ischemia-reperfusion is required to develop an effective therapeutic intervention. Currently, no effective treatment for I/R injuries is known. This disparity has prompted the creation of new experimental models, ranging from in vitro a in vivo models, to address the existing problem9,10,11,12,13.
Chick embryos (Gallus gallus domesticus) are widely used in research because of their ease of access, ethical acceptability, relatively large size (compared to other embryos), low cost, and rapid growth14. We used a chick embryo at 72 h of development to create an in ovo I/R by occluding and releasing the right vitelline artery with the assistance of a spinal needle. We named it the Hook-I/R ischemia-reperfusion model (Figure 1). The model utilized in this study is capable of accurately simulating all downstream processes, including oxidative and inflammatory pathways, which are frequently associated with I/R damage15,16,17.
Protocol
The Institutional Animal Ethical Committee at Era's Lucknow Medical College and Hospital issued a written waiver stating that no formal approval was required to conduct these experiments in accordance with the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA). However, Standard Operating Procedures were followed to minimize any potential for embryonic distress.
1. Buffer preparation (Table 1)
- Prepare Ringer's solution
- To prepare Ringer's solution, dissolve 0.72 g of NaCl (123 mM), 0.017 g of CaCl2 (1.53 mM), 0.037 g of KCl (4.96 mM) in 70 mL of sterile distilled H2O, with a final volume of 100 mL. Adjust the pH to 7.4. Let it dissolve completely and autoclave. Then, filter through a 0.22 µm filter, aliquot into single-use amounts (about 10 mL) and store at room temperature.
- Prepare normal saline (0.9% Sodium Chloride, NaCl).
- In 70 mL of sterile distilled H2O, dissolve 0.9 g of NaCl (154 mM). Make up the volume to 100 mL. Autoclave for 15 min at 121 °C. Adjust the pH to 7.4 with 0.1 N HCl or 0.1 N NaOH if necessary. Make 10 mL aliquots in a 15 mL sterile centrifuge tube and store at room temperature.
- Prepare 70% ethanol (v/v).
- Mix 70 mL of pure ethanol (Mol. wt. 46.07 g/L) to 30 mL of sterile H2O. Prepare as needed or store at room temperature. There is no need for sterilizing.
- Prepare 1x Phosphate Buffer Saline (1x PBS).
- Prepare 100 mL of 1x PBS by adding 0.144 g of Na2HPO4·7H2O (5.37 mM), 0.8 g of NaCl (136.8 mM), 0.2 g of KCl (26.8 mM), 0.2 g of KH2PO4 (14. 6 mM) to 70 mL of distilled water. Dissolve and make up the volume to 100 mL and autoclave for 15 min at 121 °C. Bring the pH to 7.4, adding a couple of drops of 0.1 N HCl or 0.1 N NaOH, if needed. Make aliquots of 10 mL in a 15 mL sterile centrifuge tube and store at room temperature.
2. Day 1
- Arrange all the tools necessary for egg sterilization (70% ethanol, cleaning wipes, an egg rack, and an OHP marker).
- Clean the 0-day eggs with 70% ethanol using tissue paper wipes. Use only a 0-day egg, as older eggs may not give rise to an embryo.
- Write the current date on eggs with an OHP marker.
- Place the eggs in an egg incubator set to a temperature of 36-37 °C and a humidity level of 60%-65%. Incubate the eggs for the next 24 h.
3. Day 2
- Arrange the necessary equipment for the withdrawal of 5-6 mL of albumin (sharp edge scissors, 5 mL syringe, 18 G needle, syringe discarder, and adhesive tape).
- Wipe the surgical scissors with 70% ethanol or sterilize using an autoclave after wiping them with 70% ethanol.
- Now take the egg from the 37 °C egg incubator for layering.
- Place the egg on a clean egg rack.
- Attach a small piece of adhesive tape (size: about 1-inch length x width) to the egg's edge.
- Make a small hole in the edge of the eggshell using sharp-pointed edge scissors. Insert a 5 mL syringe at an approximate angle of 75°.
NOTE: The 5 mL syringe comes with a 24 G x 1 needle (sterile), but it is good to replace the 24 G x 1 needle with an 18 G x 1.5 needle (sterile). The 18 G x 1.5 needle is 1.25 x 38 mm in width. Therefore, it will facilitate the removal of albumin. - After inserting the needle into the yolk sac, slowly withdraw 5-6 mL of albumin.
NOTE: This provides the embryo with a bed on which it can grow. Withdrawing albumin prevents overspill of albumin while establishing a window. Finally, the risk of the embryo being damaged during windowing is mitigated by eliminating 5-6 mL of albumin. - After removing the albumin, reseal the opening with adhesive tape and leave the eggs to incubate at 37 °C for 48 h.
4. Day 4
- Prepare the Ringer's solution, 0.9% normal saline, and 1x PBS as described in section 1 of the protocol. Then, autoclave the three solutions. Following autoclaving, place the respective solution at room temperature.
- Take out the egg from the 37 °C egg incubator and cut the shell into a circular shape. Before cutting the eggshell, cover the area to be cut with adhesive tape.
NOTE: Covering the window area with adhesive tape prevents the breaking of the eggshell into undesired places. However, if you break into an unwanted place, seal the area with the adhesive tape. Covering the places to be cut with adhesive tape prevents shell pieces from falling onto the yolk sac. - Create a small hole in the eggshell with a sharply pointed edge scissor at the place where the windowing is desired and begin cutting a circular opening. This process is known as windowing.
NOTE: Ensure that the circular cut is large enough to allow easy access to the embryo from any direction. If needed, alter the egg position to accommodate the embryo's position. - Next, using a stereo zoom surgical microscope, locate the right vitelline artery (RVA).
NOTE: Chicken embryos normally undergo thoracic torsion (along with cervical flexure, etc.) as they develop, such that the left side of the head is against the yolk at the 72 h stage. More caudally, where the vitelline arteries exit the body, the embryo is not much twisted, and this part of the body lies ventral side down toward the yolk. So, viewing directly, the right of the embryo is to the right of the researcher. - Once the RVA is located, create two small holes on the left and right sides of the RVA using a 26 G needle (Figure 2).
- Place the Doppler blood flow imaging probe above the RVA. Ensure that the Doppler blood flow imaging probe is placed 5 ± 1 mm from the site of ischemia and toward the distal end of the RVA. Take a flux reading for 2 min and 30 s (or longer if desired). This will be the normoxic phase reading.
- In the meantime, using a nose plier and toothed forceps, manually mold the spinal needle's edge into the shape of a hook (Figure 3). Do this by bending the edge of the spinal needle for approximately 1 mm. A larger size will make it more difficult to insert and remove the spinal needle during the I/R procedure.
- Insert the spinal needle directly beneath the right vitelline artery using a micromanipulator.
NOTE: Insert the spinal needle with extreme caution to avoid damaging the RVA or any adjacent arteries. The optimal technique is to adjust the spinal needle's custom-designed hook exactly above the right side of the RVA hole, followed by gradually inserting the spinal needle's custom-designed edge into the yolk sac with the assistance of a micromanipulator under the guidance of a stereo zoom surgical microscope through the right hole. Once the spinal needle's hook is in the yolk sac, gradually adjust the hook beneath the RVA so that its edge is exactly placed beneath the left hole. Now is the time to lift the spinal needle. - Now, with the assistance of the micromanipulator, gradually lift the artery until the Doppler blood flow flux indicates a minimum decrease of 80% in arterial flow.
- Once a dropdown of 80% or more in Doppler flux is achieved, leave the spinal needle lifted (pulling the artery upward) for 5 min. This will be the period of ischemia in the RVA.
NOTE: It is critical to monitor the Doppler flux during the duration of ischemia. If a significant amount of fluctuation is found, terminate the tests. - After the 5 min ischemia period, gradually release the artery to restore normal blood flow levels. Ensure a Doppler blood flowmeter reading displays values comparable to those obtained during normoxia. This will be the period of reperfusion in the RVA (Figure 4).
- After the I/R procedure, apply a few drops (2-3) of 1x PBS to the embryo and watch it for 2-3 min.
NOTE: The use of 1x PBS helps prevent the embryo from drying out. - Finally, reseal the window with adhesive tape and place the egg back in the egg incubator for 5 h and 55 min.
- After 5 h and 55 min, take the egg from the egg incubator, place it on the egg rack, reopen the window, and follow the downstream treatment protocol.
5. Treatments
- For the treatment of the arteries with drugs, activators, or inhibitors, excise the RVA after 1 h of the I/R process.
- For the downstream studies, first remove the embryo from the eggshell by placing it on a sterile 90 mm Petri dish.
- Once the embryo is released into the Petri dish, excise the RVA using the ocular iris under the guidance of a stereo zoom surgical microscope.
- Ensure that the excision dimension of RVA is up to 15 ± 1 mm (distal from the trunk), 5 ± 1 mm each on the left and right side of the artery, and 2 ± 1 mm toward the trunk.
NOTE: A ruler can be utilized to measure the area to be excised (optional). - After excising the RVA, wash it with 1x PBS in a sterile Petri dish containing 1x PBS.
- For the desired treatments, place the artery in a 1.5 mL centrifuge tube (sterilized) filled with 500 µL of Ringer's solution. Place the RVA in the centrifuge tube and place it in a 37 °C incubator for 5 h and 55 min.
NOTE: Depending on the treatments, either use the Ringer's solution without any treatment, or treatment with the desired volume and concentration of drug, activator, or inhibitor. - After 5 h and 55 min of incubation, take out the RVA from the 37 °C laboratory incubator, and proceed with the desired treatments.
Representative Results
The Doppler Blood Flow Imaging technique was used to evaluate the effectiveness of our model. In short, we compared the data from the control group with the data from the RVA group to determine the success of our creation. Figure 4A depicts a typical flux associated with the control animal, while Figure 4B depicts the results obtained from an RVA. The numeric 1-8 represents the various events associated with I/R phases. In brief, numeric 1-3 correspond to the phase of normoxia, while a sharp decline in flux at points 3 and 4 represents the events associated with the decrease in blood flow in the RVA. Once an 80% or greater drop-down had been achieved, the RVA was left raised for the next 5 min. This was the phase of ischemia (numeric 4-5). Following a 5 min lifting of the RVA, the RVA was released, which is represented by numeric 6 and 7. The flux from point 7 onward represents the reperfusion phase, which occurs after the RVA has achieved normal blood flow levels, which was the phase of reperfusion. This particular experiment demonstrated the effectiveness of I/R modeling in the 3-day developed chick embryo.
To verify the utility of our model, we have studied the expression patterns of proteins, RNA, and DNA through ELISA, western blotting, qRT-PCR, and gel electrophoresis analysis. In brief, we divided the 3-day developed eggs into three experimental groups: control, I/R, and Treatments + I/R. A significant difference in expression of proteins, genes, and DNA integrity was observed between their respective I/R and control groups. As discussed below, drug treatment to the I/R group effectively improved the outcome observed in this group compared to the I/R treated group alone; this is consistent with our prior publication on which this protocol is based1. Standard laboratory procedures were followed for ELISA18, western blotting19, qRT-PCR20, and gel electrophoresis21, which are not covered in this paper (Figure 5, Figure 6, Figure 7, Figure 8).
Ischemia-reperfusion stimulates several processes, including the formation of reactive oxygen species (ROS) that are detrimental to tissue. The detrimental effects caused by the intrinsic antioxidant body defense systems' response to ROS is an important feature of I/R injury. We examined the activities of oxygen regulatory proteins 150 (ORP150), cytoplasmic superoxide dismutase 1 (SOD1), and catalase in ischemic vessels. When compared to the control group, I/R elevated the activity of ORP150, cytoplasmic SOD1, and catalase (Figure 5A–C). However, supplementation with N-Acetyl-L-Cysteine (NAC), a ROS quencher, mitigated the I/R group's oxidative stress.
To assess our model's inflammatory potential, we used ELISA to examine IL-1β and TNF-α expression (Figure 6). Both interleukins were overexpressed in the I/R group as compared to the control group, indicating that our model has the potential to be used in inflammatory investigations. Next, we tested the expression of the NOD-like receptor pyrin domain-containing protein 3 (NLRP3) inflammasome pathway22,23, and pro-inflammatory molecule, NF-kβ24,25, which are involved in amplifying inflammation, to confirm the usefulness of this model for inflammatory studies. In response to I/R generated in the RVA, this investigation found evidence of activation of the NLRP3 inflammasome (Figure 7A), as well as NF-kβ (Figure 7B). However, treatment with Naringenin, a well-known anti-inflammatory drug, ameliorated the inflammatory effects, as observed in the drug-treated groups.
Ischemia-reperfusion activates programed cell death pathways26,27,28. We studied the effects of I/R on apoptosis and autophagy pathways in the chick embryo. Figure 8A depicts the effect of I/R on caspase-3 and zVAD-fmk. Figure 8B–D demonstrate that the I/R group had higher expression of LC3II as well as LC3II/I ratio (autophagosomes marker), Beclin1 (a significant regulator of autophagy in mammalian cells), and Atg7 (needed for basal autophagy) than the control group, respectively, at protein levels. In contrast, Figure 8E shows the effect of the I/R on mRNA levels. The addition of 3-MA, an autophagy inhibitor, however, reversed the results. These findings suggest that the model might be used to investigate different cell death processes (e.g., necroptosis). Figure 9 shows how efficiently the changes at the DNA level can be studied using our Hook-I/R model.
Finally, we compare the results from the Hook-I/R model and the middle cerebral artery occlusion (MCAO) model to see how effective our model is in comparison to other models. To summarize, I/R-treated RVAs had higher levels of the apoptotic protein cleaved caspase-3, the autophagy proteins Beclin1 and Atg7, and the inflammatory interleukins TNF-α and IFN-Ƴ than control RVAs. Interestingly, whether analyzing inflammatory stress or cellular death pathways, the Hook-I/R model produced results that were extremely similar to those obtained by the MCAO model (Figure 10).
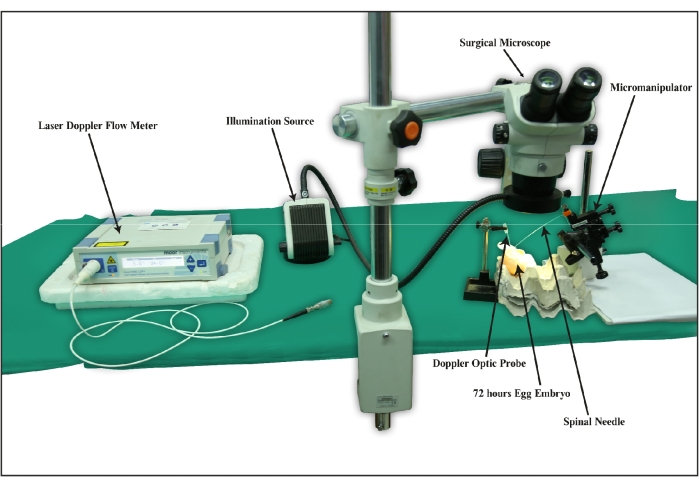
Figure 1: A schematic diagram of a typical set-up of the Hook-I/R chick embryo ischemia-reperfusion model. The picture shows the Hook-I/R chick embryo ischemia-reperfusion experimentation requirements, such as the stereo zoom surgical microscope, laser Doppler blood flowmeter, micromanipulator, spinal needle, 72 h developed chick embryos, and an illumination source.”Please click here to view a larger version of this figure.
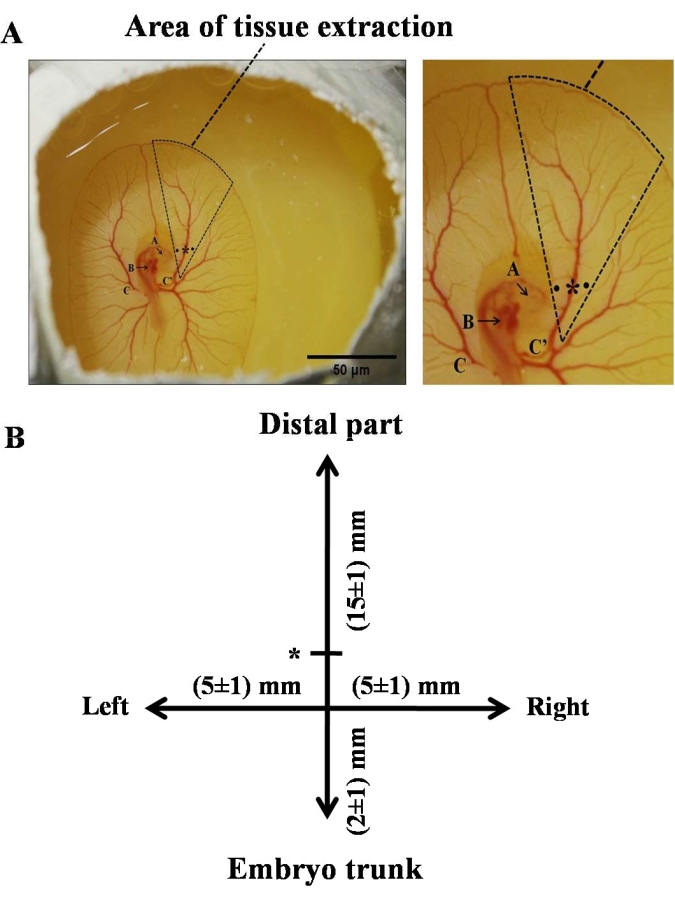
Figure 2: Representative image of a 3-day chick embryo-exhibiting site of occlusion and area of tissue excision. (A) The triangle shows the location of excision of the tissue for western blotting, RNA, and DNA isolation. The star and dots represent the site of the occlusion and the holes created on the left and right side of the RVA to insert the needle beneath the artery to lift it, respectively. A magnified image of the area of tissue extraction is also provided along with it. The notation A represents the eye, B represents the heart, C represents the left of the vitelline artery, and C' represents the right of the vitelline artery. (B) A schematic representation of the right side of the chick embryo demonstrates the area of tissue excision in the vicinity of RVA. The straight line represents the RVA emerging from the embryo trunk. The star on the vertical line symbolizes the position of the Laser Doppler blood flow probe. The intersection denotes the site of occlusion. Excision of the arteries was done up to 15 ± 1 mm (distal from the trunk), 5 ± 1 mm each on the left and right side of the artery, and 2 ± 1 mm toward the trunk. All the measurements show distances from the site of occlusion. Please click here to view a larger version of this figure.
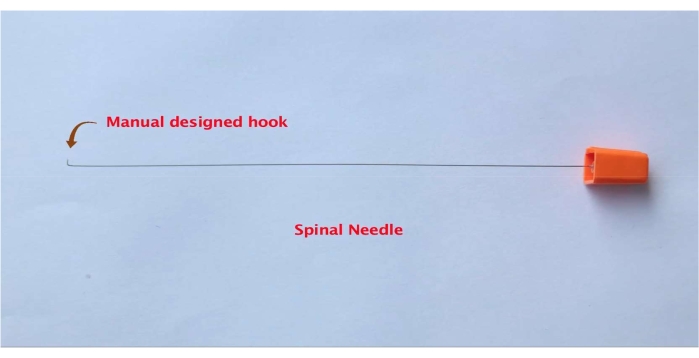
Figure 3: Representative picture of a spinal needle with a custom-made hook. Please click here to view a larger version of this figure.
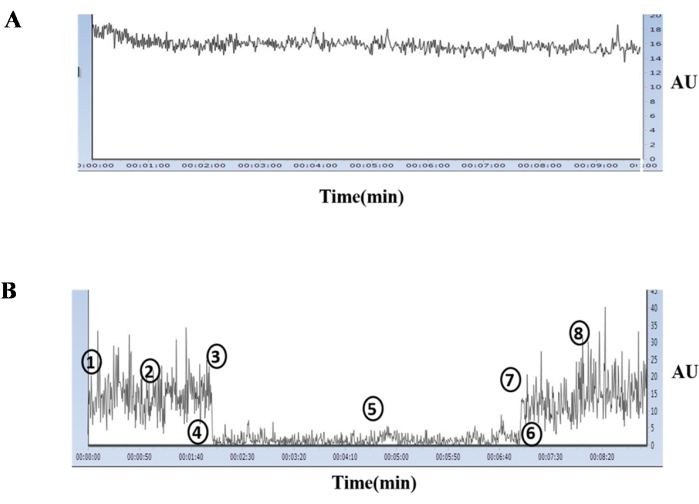
Figure 4: Representation of a typical Laser Doppler blood flowmeter signal. A typical Laser Doppler blood flowmeter signal was measured on a 3-day chick embryo artery from a control group egg (A). Events at baseline (1), during normoxia (2), immediate pre-lifting of RVA (3), immediate post-lifting of RVA (4), during ischemia (5), immediate pre-releasing of RVA (6), immediate post-reperfusion (7), during reperfusion (8) in the ischemia treated group as recorded by the Laser Doppler blood flowmeter (B). This figure was adopted from Fauzia et al., 2018 (Frontiers in Pharmacology; under a CC-BY license)1. Please click here to view a larger version of this figure.
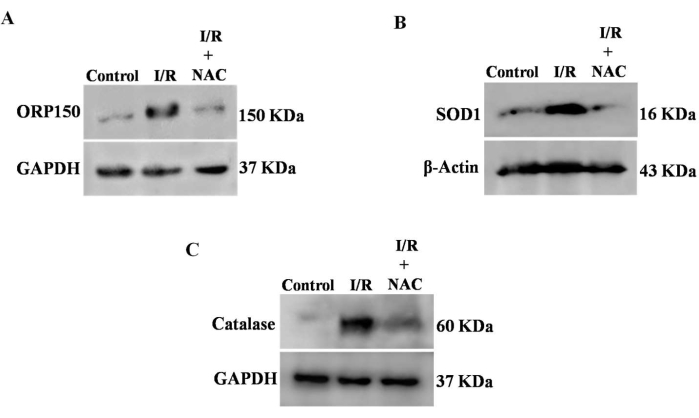
Figure 5: Effect of ischemia-reperfusion on the expression of oxidative stress marker proteins. Western blotting was used to determine the ORP150, SOD1, and catalase expression levels. (A) ORP150 expression was increased after I/R damage. NAC, a pan-ROS inhibitor, lowered ORP150 to a level comparable to that of control. (B) Following I/R, SOD1 expression was elevated. SOD1 expression was likewise decreased following NAC treatment. (C) The I/R event induced the expression of catalase. RVA treated with I/R and exposed to NAC showed a decrease in catalase levels. GAPDH (A,C) and β-Actin (B) were used as internal controls. Please click here to view a larger version of this figure.
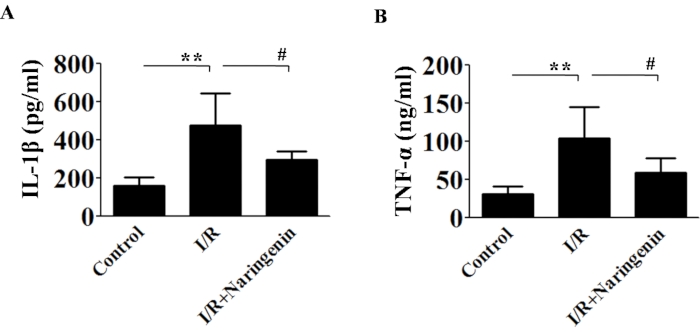
Figure 6: Estimation of IL-1β and TNF-α using ELISA. ELISA was used to quantify IL-1β and TNF-α, and the results revealed that there was an increase in IL-1β and TNF-α expression, which was mitigated by the addition of Naringenin. For this study, the ANOVA followed by the Newman-Keuls test was applied. Error bars represent mean ± SD, n = 3. Please click here to view a larger version of this figure.

Figure 7: Effect of ischemia-reperfusion on the expression of inflammation marker proteins. (A) I/R treatment resulted in increased expression of NLRP3. Incubation of I/R treated RVA with Naringenin decreased the expression of NLRP3. (B) Similar to NLRP3, the expression of NF-kβ was also increased after I/R treatment. The treatment of Naringenin reduced the expression of NF-kβ. GAPDH was used as an internal control. Please click here to view a larger version of this figure.
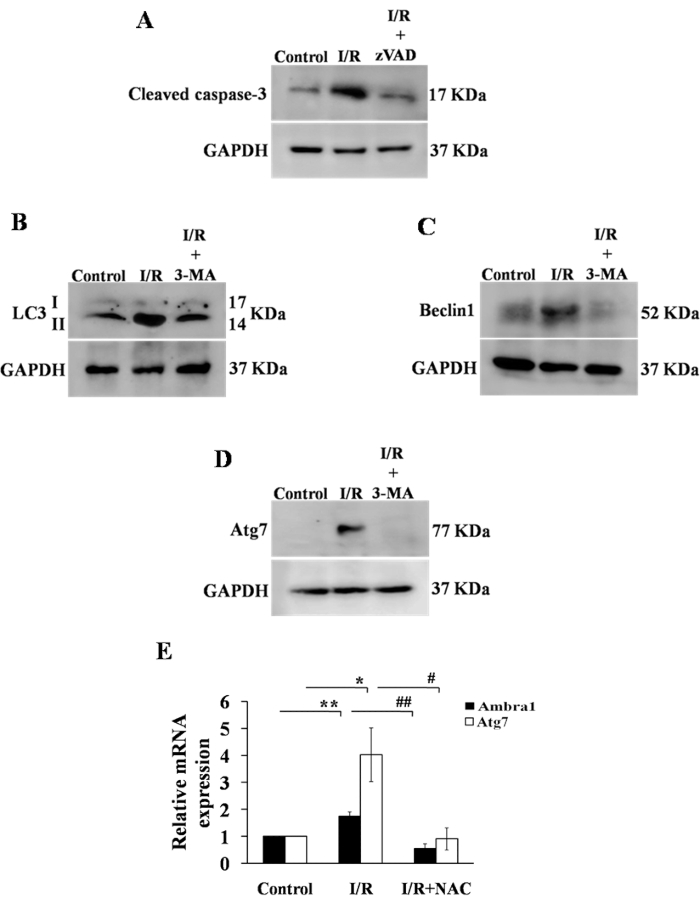
Figure 8: Effect of ischemia-reperfusion on the apoptotic and autophagic status of RVA cells. (A) To explore the induction of apoptosis by the I/R process, the expression of cleaved caspase 3 was evaluated using western blotting. Following I/R damage, the level of cleaved caspase 3 was increased. However, treatment with zVAD-fmk, an apoptosis inhibitor, decreased cleaved caspase 3 expression. (B–D) To evaluate the induction of autophagy following I/R, the expression of LC3II/I, Beclin1, and Atg7 was determined. Following I/R, the expression of all autophagic markers increased, while the exposure of I/R-treated RVA to the autophagy inhibitor 3-MA decreased the expression of all proteins to control levels. As an internal control, GAPDH was used. (E) qRT-PCR was used to confirm the induction of autophagy and determine the Ambra1 and Atg7 mRNA expression levels. Both genes showed a considerable increase in expression in the I/R group compared to control, which was restored to near-normal levels following NAC therapy. GAPDH was used as an internal control for qRT-PCR experiments. For experiment (E), the ANOVA, followed by the Newman-Keuls test, was applied. Error bars represent mean ± SD, n = 3. Please click here to view a larger version of this figure.
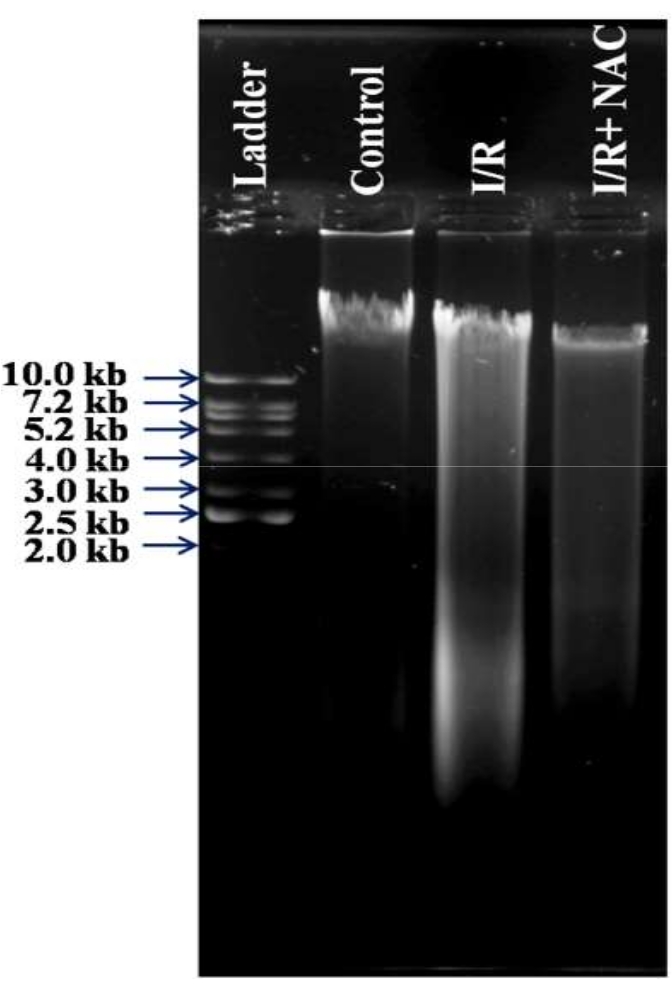
Figure 9: Effect of ischemia-reperfusion on DNA damage. To determine if I/R induces DNA nicks, we examined DNA damage using gel electrophoresis. I/R resulted in genomic DNA degradation, as shown by smearing in the I/R sample. NAC therapy of I/R-damaged RVA reduced DNA damage. A 1 kb DNA ladder was employed. Please click here to view a larger version of this figure.
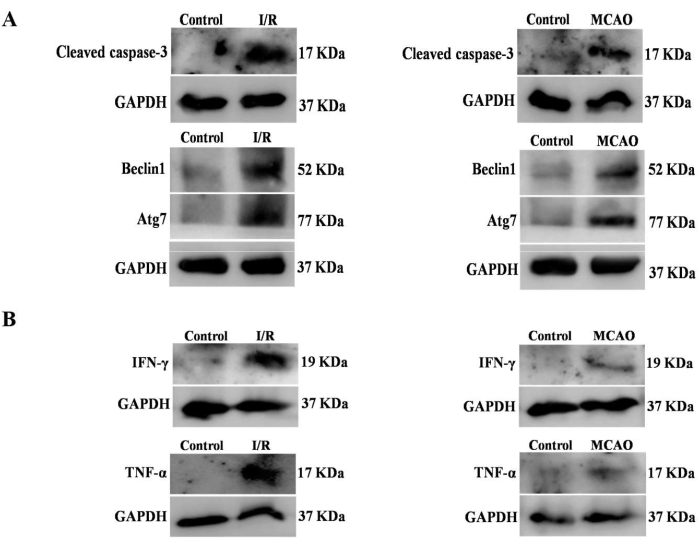
Figure 10: Comparison of the Hook-I/R model vs. the MCAO model. A comparison was made between the data generated by the Hook-I/R model and the data generated by the MCAO model. (A) The expression of the apoptotic protein caspase-3 and the autophagy proteins Beclin1 and Atg7 was higher in I/R treated RVAs when compared to the expression of the same proteins in control RVAs, which was in agreement with the findings obtained in the MCAO experiments. (B) TNF-α and IFN-Ƴ levels were found to be elevated in both the RVA and MCAO groups when compared to their respective controls. Please click here to view a larger version of this figure.
| Name of Material/Equipment | Concentration | Autoclave | Storage | ||
| Reagents for Ringer’s solution | |||||
| Sodium chloride | 123 mM | ||||
| Potassium chloride | 4.96 mM | ||||
| Calcium chloride | 1.53 mM | ||||
| Sterile distilled water | 100 mL | ||||
| pH | 7.4 | 15 min at 121 °C | R.T. | ||
| Reagents for 0.9% normal saline | |||||
| Sodium Chloride | 154mM | ||||
| Sterile distilled water | 100 mL | ||||
| pH | 7.4 | 15 min at 121 °C | R.T. | ||
| Reagents for 70% ethanol | |||||
| 70% ethanol | 70 mL | ||||
| Sterile distilled water | 30 mL | Not required | R.T. | ||
| Reagents for 1x Phosphate Buffer Saline | |||||
| Sodium chloride | 136.8 mM | ||||
| Potassium chloride | 26.8 mM | ||||
| Potassium phosphate monobasic anhydrous | 14.6 mM | ||||
| di-Sodium hydrogen phosphate heptahydrate | 5.37 mM | ||||
| Sterile distilled water | 100 mL | 15 min at 121 °C | R.T. | ||
Table 1: Recipe of solutions used in this study.
Discussion
The goal of ischemia-reperfusion research is to create therapeutic strategies that prevent cell death and promote recovery29,30. To overcome current constraints in I/R research, we designed a Hook I/R chick embryo model to produce a reliable and reproducible I/R model. To our knowledge, ours is the first I/R model ever created in a 3-day chick embryo for routine I/R experiments, besides studying stress signals (e.g., oxidative and inflammatory stress). Given the benefits of size and accessibility at day 3 of development31, the chick embryo was used at a developmental stage of 72 h, because of the model's high output, simplicity to employ, and adaptability for routine analysis32. In brief, at developmental day 3, the in ovo chick is a highly controlled, yet accessible and reasonably transparent model within the egg that can be used to visualize normal physiology, disease pathology, and the effects of experimental treatment. Its huge size makes it particularly useful for studying the formation and behavior of the embryo during normal physiology as well as stress33. Although older chick embryos (those incubated for 4 days or longer) can be employed, and we did try a later time-point, the use of older embryos as model systems is severely limited by the fact that the yolk sac grows so thick beyond 3 days of development that surgical procedures are difficult. It is noteworthy to mention that while the RVA is not fully formed at an early time window (say, on day 1 or 2), windowing could lead to teratogenicity34. Besides the limitations associated with an early or late time point of development, a 72 h chick embryo serves as an ideal model to research the I/R process since 3-day chick embryos have a well-defined circulatory system30.
Understanding the pathophysiology of I/R injury is a major goal of designing an I/R-model. Currently, there is not any clinically useful therapeutic for reducing ischemia-reperfusion damage1,35. As a result, a wide range of in vitro and in vivo models have been proposed. In this context, I/R modeling in a 3-day developing chick embryo in ovo was proposed for the first time. Previous research has shown that occlusion-reperfusion of arterial blood flow causes pathological alterations in experimental models that are similar to those seen in humans36,37,38, so we were hopeful that our model would do the same (such oxidative and inflammatory stress observed in in vivo models or in patients). With the findings of our study, the above hypothesis was shown to be correct.
The circulation of blood from embryos to the yolk sac is controlled by vitelline vessels in chick embryos39. Embryos get nutrients from the yolk and diffuse oxygen from the air via vitelline circulation; thus, restricting any of the vitelline vessels that can disrupt nutrition and oxygen transfer. Based on these facts, ischemia was induced by occluding the blood supply to the RVA for 5 min, followed by reperfusion for additional 5 h and 55 min. The proposed model can be utilized to examine a range of various disease processes associated with I/R and test a variety of drugs and their targets. The current model can be used to examine changes in DNA, RNA, and proteins. It has the potential to give high output. Besides its simplicity and adaptability for regular analysis, the model can also investigate short-term stem cell homing, which will be investigated in future studies.
In comparison to in vitro models, our model is easy to use, cost-effective, and grows quickly, as well as being ethically innocent32. The presence of microvasculature, the immune system, and physiological assessments are all benefits over in vitro models while low cost, time-effectiveness, and no ethical problems are advantages over in vivo models40. The above advantages indicate that our model has a comparable effect to the other I/R models currently in use (Figure 10). A potential shortcoming is the current model's inability to quantify the infarct area. Direct infarct assessment may be a valuable biomarker for I/R-mediated damage and a method for mapping the effects of various therapy medications. Thus, we sought to quantify the infarct area but could not do so because of the delicate structure of the 72 h-developed chicks. Therefore, additional research is required to assess the strategies and pathways for I/R processes comprehensively.
To summarize, the current model may be used to investigate various disease pathways linked to I/R and screen a variety of drugs and their targets. Due to its high reproducibility, cost-effectiveness, and simplicity, we anticipate that our model will be a valuable resource for basic science and translational research.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We want to express our gratitude to Hari Shankar for his critical inputs during videography and editing, Mr. Baqer Hussain for voice-over, Mr. Asghar Rizvi for video editing, Mr. Mohammad Haider for video shoots, Mr. Mohammad Danish Siddiqui for assistance during the experiments.
Materials
| (-80°C) freezer | Haier, China | – | |
| 1.5mL Centrifuge tube | TARSONS, India | 500010X | |
| 100mm Petri dish (sterile) | Tarsons, India | 460050 | |
| 18G Needle (18G×1.5 (1.25×38mm) | Ramsons, India | 13990 | |
| 1mL Syringe | DISPO VAN | – | |
| 26G Needle (26G×1/2 (10.45x13mm) | DISPO VAN, india | 30722D | |
| 37°C egg incubator with adjustable percentage humidity | Gentek, India | GL-100 | |
| 37°C laboratory incubator | SCIENCE TECH, India | CB 101-14 | |
| 3-Methyladenine (3-MA) | Sigma Aldrich, USA | M9281 | |
| 3mL Pasture Pipette | TARSONS, India | 940050 | |
| 50mL Beaker | TARSONS, India | – | |
| 5mL Syringe | DISPO VAN, India | IP53 | |
| 70% ethanol | Merck Millipore, United States | 64-17-5 | |
| Adhesive tape/Cello tape | Sunrise, India | – | |
| Ambra1 primers | Applied Biosystems, Foster city, USA | Hs00387943_m1 | |
| Anti-mouse IgG | Cell Signaling Technology, USA | 7076S | |
| Anti-Rabbit IgG | Jackson Immuno Research Laboratories, USA | 711-035-152 | |
| Atg7 | R&D Systems, USA | MAB6608 | |
| Atg7 primers | Applied Biosystems, Foster city, USA | Hs00893766_m1 | |
| Autoclave Bag | Tarsons, India | 550022 | |
| Autoclave Machine | Local made, Lucknow, India | – | |
| Beclin-1 | Proteintech, USA | 66665-1-Ig | |
| Beta Actin | ImmunoTag, USA | ITT07018 | |
| Bovine Serum Albumin | Himedia, Mumbai, India | TC194 | |
| Calcium Chloride | Himedia, Mumbai, India | GRM534 | |
| Catalase | ImmunoTag, USA | ITT5155 | |
| Cleaning wipes | Kimberly-Clark, India | 370080 | |
| Cleaved Caspase3 | ImmunoTag, USA | ITT07022 | |
| di-Sodium hydrogen phosphate heptahydrate | Himedia, Mumbai, India | GRM39611 | |
| Doppler blood flowmeter | Moors instrument, United Kingdom | moorVMS-LDF1 | |
| Egg rack | – | – | |
| Egg rack | – | – | |
| GAPDH | ImmunoTag, USA | M1000110 | |
| GAPDH primers | Applied Biosystems, Foster city, USA | Hs02758991_g1 | |
| Glycine | Himedia, Mumbai, India | MB013 | |
| Kidney tray | HOSPITO | – | |
| LC3A/B | Cell Signaling Technology, USA | 4108S | |
| Methanol | Rankem laboratories, Mumbai, India | M0252 | |
| Micromanipulator | Narishige, Japan | M-152 | |
| N-acetyl-L-cysteine (NAC) | Sigma Aldrich, USA | A7250 | |
| Naringenin | Sigma Aldrich, USA | 67604-48-2 | |
| NF-kβ | Thermo Fisher Scientific, USA | 51-0500 | |
| NLRP3 | ImmunoTag, USA | ITT07438 | |
| Nose plier | Local made, Lucknow, India | – | |
| Ocular forceps | Stoelting, Germany | 52106-40 | |
| Ocular iris | Tufft Surgical Instruments, Jaipur, India | Hard Age Vannas Micro Scissors Angled 8CM / 3 1/8" | |
| OHP marker pen | Camlin, India | – | |
| ORP-150 | ImmunoTag, USA | ITT08329 | |
| Pointed sharp edge scissor | Stoelting, Germany | 52132-11 | |
| Potassium Chloride | Himedia, Mumbai, India | MB043 | |
| Potassium phosphate monobasic anhydrous | Himedia, Mumbai, India | MB050 | |
| Protease Inhibitor | Abcam, United States | Ab65621 | |
| SOD-1 | ImmunoTag, USA | ITT4364 | |
| Sodium Chloride | Fisher Scientific, Mumbai, India | 27605 | |
| Sodium dodecyl sulphate | Himedia, Mumbai, India | GRM886 | |
| Spinal needle 25GA; 3.50 IN (90.51 X 90mm) | Ramson, India | GS-2029 | |
| Stereo Zoom surgical microscope | Olympus, Japan | SZ2-STU3 | |
| Syringe discarder | BIOHAZARD | 882210 | |
| Toothed forceps | Stoelting, Germany | 52102-30 | |
| Tris Base | G Biosciences, United States | RC1217 | |
| Tris Hydrochloric Acid | Himedia, Mumbai, India | MB030 | |
| Tween 20 | G Biosciences, United States | RC1227 | |
| White Leghorn Chicken 0-day eggs | – | – | |
| Z-Val-Ala-Asp(OMe)-FMK | MP Biomedicals, LLC, USA | FK009 |
Riferimenti
- Fauzia, E., et al. Chick Embryo: A Preclinical Model for Understanding Ischemia-Reperfusion Mechanism. Frontiers in Pharmacology. 21 (9), 1034 (2018).
- Eltzschig, H. K., Eckle, T. Ischemia and reperfusion–from mechanism to translation. Nature Medicine. 17 (11), 1391-1401 (2011).
- Raza, S. S., et al. Neuroprotective effect of naringenin is mediated through suppression of NF-κB signaling pathway in experimental stroke. Neuroscienze. 29 (230), 157-171 (2013).
- Raza, S. S., et al. Hesperidin ameliorates functional and histological outcome and reduces neuroinflammation in experimental stroke. Brain Research. 28 (1420), 93-105 (2011).
- Raza, S. S., et al. Silymarin protects neurons from oxidative stress associated damages in focal cerebral ischemia: a behavioral, biochemical and immunohistological study in Wistar rats. Journal of the Neurological Sciences. 15 (1-2), 45-54 (2011).
- Fan, L., Zhou, L. AG490 protects cerebral ischemia/reperfusion injury via inhibiting the JAK2/3 signaling pathway. Brain and Behavior. 11 (1), 01911 (2021).
- Wu, M. Y., et al. Current Mechanistic Concepts in Ischemia and Reperfusion Injury. Cellular Physiology and Biochemistry. 46 (4), 1650-1667 (2018).
- Collard, C. D., Gelman, S. Pathophysiology, clinical manifestations, and prevention of ischemia-reperfusion injury. Anesthesiology. 94 (6), 1133-1138 (2001).
- Allen, D. D., et al. Cell lines as in vitro models for drug screening and toxicity studies. Drug Development and Industrial Pharmacy. 31 (8), 757-768 (2005).
- Schmeer, C., Gamez, A., Tausch, S., Witte, O. W., Isenmann, S. Statins modulate heat shock protein expression and enhance retinal ganglion cell survival after transient retinal ischemia/reperfusion in vivo. Investigative Ophthalmology and Visual Science. 49 (11), 4971-4981 (2008).
- Huang, K. Y., et al. A systematic review and meta-analysis of acupuncture for improving learning and memory ability in animals. BMC Complementary and Alternative Medicine. 16 (1), 297 (2016).
- Sommer, C. J. Ischemic stroke: Experimental models and reality. Acta Neuropathologica. 133 (2), 245-261 (2017).
- Yang, W., Chen, J., Meng, Y., Chen, Z., Yang, J. Novel targets for treating ischemia-reperfusion injury in the liver. International Journal of Molecular Sciences. 19 (5), 1302 (2018).
- Seabra, R., Bhogal, N. In vivo research using early life stage models. In Vivo. 24 (4), 457-462 (2010).
- Liu, H., et al. Adiponectin peptide alleviates oxidative stress and NLRP3 inflammasome activation after cerebral ischemia-reperfusion injury by regulating AMPK/GSK-3beta. Experiments in Neurology. 329, 113302 (2020).
- Aboutaleb, N., Jamali, H., Abolhasani, M., Pazoki Toroudi, H. Lavender oil (Lavandula angustifolia) attenuates renal ischemia/reperfusion injury in rats through suppression of inflammation, oxidative stress and apoptosis. Biomedicine and Pharmacotherapy. 110, 9-19 (2019).
- Wallert, M., et al. alpha-Tocopherol preserves cardiac function by reducing oxidative stress and inflammation in ischemia/reperfusion injury. Redox Biology. 26, 101292 (2019).
- Ashafaq, M., et al. Catechin hydrate ameliorates redox imbalance and limits inflammatory response in focal cerebral ischemia. Neurochemical Research. 37 (8), 1747-1760 (2012).
- Gallagher, S., Chakavarti, D. Immunoblot analysis. Journal of Visualized Experiments. 20 (16), 759 (2008).
- Abt, M. A., Grek, C. L., Ghatnekar, G. S., Yeh, E. S. Evaluation of lung metastasis in mouse mammary tumor models by quantitative real-time PCR. Journal of Visualized Experiments. (107), e53329 (2016).
- Lee, P. Y., Costumbrado, J., Hsu, C. Y., Kim, Y. H. Agarose gel electrophoresis for the separation of DNA fragments. Journal of Visualized Experiments. (62), e3923 (2012).
- Wu, Y., et al. Cathelicidin aggravates myocardial ischemia/reperfusion injury via activating TLR4 signaling and P2X(7)R/NLRP3 inflammasome. Journal of Molecular and Cellular Cardiology. 139, 75 (2020).
- Franke, M., et al. The NLRP3 inflammasome drives inflammation in ischemia/reperfusion injury after transient middle cerebral artery occlusion in mice. Brain Behaviour and Immunity. 92, 223 (2021).
- Lawrence, T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harbor Perspectives in Biology. 1 (6), 001651 (2009).
- Liu, H., et al. Pterostilbene attenuates astrocytic inflammation and neuronal oxidative injury after ischemia-reperfusion by inhibiting NF-kappaB phosphorylation. Frontiers in Immunology. 10, 2408 (2009).
- Prakash, R., et al. Sivelestat-loaded nanostructured lipid carriers modulate oxidative and inflammatory stress in human dental pulp and mesenchymal stem cells subjected to oxygen-glucose deprivation. Materials Science and Engineering: C Materials for Biological Applications. 120, 111700 (2021).
- Prakash, R., et al. Oxidative stress enhances autophagy in stem cells through Erk1/2 signaling pathway – implications for neurotransplantations. Stem Cell Reviews and Reports. , (2021).
- Ahmad, A., et al. Gelatin-coated polycaprolactone nanoparticle-mediated naringenin delivery rescue human mesenchymal stem cells from oxygen glucose deprivation-induced inflammatory stress. ACS Biomaterials Science and Engineering. 5 (2), 683-695 (2019).
- Guan, X., et al. The neuroprotective effects of carvacrol on ischemia/reperfusion-induced hippocampal neuronal impairment by ferroptosis mitigation. Life Science. 235, 116795 (2019).
- Jin, Z., Guo, P., Li, X., Ke, J., Wang, Y., Wu, H. Neuroprotective effects of irisin against cerebral ischemia/ reperfusion injury via Notch signaling pathway. Biomedicine and Pharmacotherapy. 120, 109452 (2019).
- Wainrach, S., Sotelo, J. R. Electron microscope study of the developing chick embryo heart. Zeitschrift fur Zellforschung und mikroskopische Anatomie. 55, 622-634 (1961).
- Joshi, V. C., Wilson, A. C., Wakil, S. J. Assay for the terminal enzyme of the stearoyl coenzyme A desaturase system using microsomes. Journal of Lipid Research. 18 (1), 32-36 (1977).
- Kain, K. H., et al. The chick embryo as an expanding experimental model for cancer and cardiovascular research. Development Dynamics. 243 (2), 216-228 (2014).
- Mann, R. A., Moore, K. L., Persaud, T. V. N. Limitations in the u~e of the early chick embryo 88 a teratological model. Teratology. 7, 22-23 (1973).
- Chen, T., Vunjak-Novakovic, G. In vitro models of ischemia-reperfusion injury. Regenerative English and Translation Medicine. 4 (3), 142-153 (2018).
- Ma, R., et al. Animal models of cerebral ischemia: A review. Biomedicine and Pharmacotherapy. 131, 110686 (2020).
- Bromage, D. I., et al. Remote ischaemic conditioning reduces infarct size in animal in vivo models of ischaemia-reperfusion injury: a systematic review and meta-analysis. Cardiovascular Research. 113 (3), 288-297 (2017).
- Kalogeris, T., Baines, C. P., Krenz, M., Korthuis, R. J. Cell biology of ischemia/reperfusion injury. International Review of Cell and Molecular Biology. 298, 229-317 (2012).
- Hogers, B., DeRuiter, M. C., Baasten, A. M., Gittenberger-de Groot , A. C., Poelmann, R. E. Intracardiac blood flow patterns related to the yolk sac circulation of the chick embryo. Circ Res. 76 (5), 871-877 (1995).
- Rezzola, S., et al. angiogenesis-inflammation cross talk in diabetic retinopathy: novel insights from the chick embryo chorioallantoic membrane/human vitreous platform. Frontiers in Immunology. 11, 581288 (2020).

