Exploring Biomolecular Interaction Between the Molecular Chaperone Hsp90 and Its Client Protein Kinase Cdc37 using Field-Effect Biosensing Technology
Summary
Field-effect biosensing (FEB) is a label-free technique for detecting biomolecular interactions. It measures the electric current through the graphene biosensor to which the binding targets are immobilized. The FEB technology was used to evaluate biomolecular interactions between Hsp90 and Cdc37 and a strong interaction between the two proteins was detected.
Abstract
Biomolecular interactions play versatile roles in numerous cellular processes by regulating and coordinating functionally relevant biological events. Biomolecules such as proteins, carbohydrates, vitamins, fatty acids, nucleic acids, and enzymes are fundamental building blocks of living beings; they assemble into complex networks in biosystems to synchronize a myriad of life events. Proteins typically utilize complex interactome networks to carry out their functions; hence it is mandatory to evaluate such interactions to unravel their importance in cells at both cellular and organism levels. Toward this goal, we introduce a rapidly emerging technology, field-effect biosensing (FEB), to determine specific biomolecular interactions. FEB is a benchtop, label-free, and reliable biomolecular detection technique to determine specific interactions and uses high-quality electronic-based biosensors. The FEB technology can monitor interactions in the nanomolar range due to the biocompatible nanomaterials used on its biosensor surface. As a proof of concept, the protein-protein interaction (PPI) between heat shock protein 90 (Hsp90) and cell division cycle 37 (Cdc37) was elucidated. Hsp90 is an ATP-dependent molecular chaperone that plays an essential role in the folding, stability, maturation, and quality control of many proteins, thereby regulating multiple vital cellular functions. Cdc37 is regarded as a protein kinase-specific molecular chaperone, as it specifically recognizes and recruits protein kinases to Hsp90 to regulate their downstream signal transduction pathways. As such, Cdc37 is considered a co-chaperone of Hsp90. The chaperone-kinase pathway (Hsp90/Cdc37 complex) is hyper-activated in multiple malignancies promoting cellular growth; therefore, it is a potential target for cancer therapy. The present study demonstrates the efficiency of FEB technology using the Hsp90/Cdc37 model system. FEB detected a strong PPI between the two proteins (KD values of 0.014 µM, 0.053 µM, and 0.072 µM in three independent experiments). In summary, FEB is a label-free and cost-effective PPI detection platform, which offers fast and accurate measurements.
Introduction
Biomolecular interactions:
Proteins are essential parts of organisms and participate in numerous molecular pathways such as cell metabolism, cell structure, cell signaling, immune responses, cell adhesion, and more. While some proteins perform their function(s) independently, most proteins interact with other proteins using a binding interface to coordinate proper biological activity1.
Biomolecular interactions can mainly be classified based on the distinct structural and functional characteristics of proteins involved2, for example, based on the protein surfaces, the complex stability, or the persistence of interactions3. Identifying essential proteins and their roles in biomolecular interactions is vital for understanding biochemical mechanisms at the molecular level4. Currently, there are various approaches to detect these interactions5: in vitro6, in silico7, in live cells8, ex vivo9, and in vivo10 with each having its own strengths and weaknesses.
The in vivo assays are performed using the whole animal as an experimental tool11, and the ex vivo assays are performed on tissue extracts or whole organs (e.g., heart, brain, liver) in a controlled external environment by providing minimal alterations in natural conditions. The most common application of in vivo and ex vivo studies is to evaluate the pharmacokinetics, pharmacodynamics, and toxicity effects of potential pharmacological agents before human trials by ensuring their overall safety and efficacy12.
Biomolecular interactions can also be detected within living cells. Imaging live cells allow us to observe dynamic interactions as they execute the reactions of a particular biochemical pathway13. Moreover, detection techniques, such as bioluminescence or fluorescence resonance energy transfer, can provide information about where and when these interactions occur within the cell14. Although detection in live cells offers crucial details, these detection methodologies rely on optics and labels, which may not reflect the native biology; they are also less controlled than in vitro methods and require specialized expertise to perform15.
The in silico computational methods are primarily used for large-scale screening of target molecules before the in vitro experiments. Computational prediction methods, computer-based databases, molecular docking, quantitative structure-activity relationships, and other molecular dynamics simulation approaches are among the well-established in silico tools16. Compared to laborious experimental techniques, the in silico tools can easily make predictions with high sensitivity, but with reduced accuracy in predictive performance17.
In vitro assays are performed with microorganisms or biological molecules outside of their standard biological context. Portraying biomolecular interactions through in vitro methods is critical to understanding protein functions and the biology behind the complex network of cell functioning. The preferred assay methodology is chosen according to the protein's intrinsic properties, kinetic values, and the mode and intensity of interactions18,19.
The Hsp90/Cdc37 interaction:
The chaperone-kinase pathway, connecting Hsp90 and Cdc37, is a promising therapeutic target in tumor biology20. Hsp90 plays a central role in cell cycle control, protein assembly, cell survival, and signaling pathways. Proteins that rely on Hsp90 for their functions are delivered to Hsp90 for complexation through a co-chaperone, such as Cdc37. The Hsp90/Cdc37 complex controls the folding of most protein kinases and serves as a hub for a multitude of intracellular signaling networks21. It is a promising anti-tumor target due to its elevated expression in various malignancies, including acute myeloblastic leukemia, multiple myeloma, and hepatocellular carcinoma22,23.
Commonly used in vitro biomolecular interaction detection techniques
Co-immunoprecipitation (co-IP) is a technique relying on antigen-antibody specificity to identify biologically relevant interactions24. The primary disadvantage of this method is its inability to detect low-affinity interactions and kinetic values24. Biophysical methods such as isothermal titration calorimetry (ITC), surface plasmon resonance (SPR), biolayer interferometry (BLI), and FEB technology are preferred to determine the kinetic values.
ITC is a biophysical detection method based on the determination of binding energy along with a complete thermodynamics analysis to characterize biomolecular interactions25. The primary advantage of ITC is that it does not require any labeling or fixation of the target protein. The main difficulties encountered by ITC are the high concentration of target protein required for one experiment and the difficulty in analyzing non-covalent complexes due to small binding enthalpies26. Both SPR and BLI are label-free biophysical techniques that rely on the immobilization of the target molecule on the sensor surface, followed by subsequent injections of the analyte over the immobilized target27,28. In SPR, alterations in the refractive index during biomolecular interactions are measured27; in BLI, the interference in reflected light is recorded in real-time as a change in wavelength as a function of time28. Both SPR and BLI share common advantages of offering high specificity, sensitivity, and detection capabilities29. In both methods, the target protein is immobilized on biosensor surfaces, and hence, there may be some loss of the native conformation of the target, which makes it difficult to discriminate between specific vs. non-specific interactions30. BLI uses expensive disposable fiber-optic biosensors to immobilize the target, and is, therefore, a costly technique31. Compared to these well-established biomolecular detection tools, FEB technology offers a reliable and label-free platform by using low nanomolar concentrations for biomolecular detection in real-time with kinetic characterization. The FEB technology also overcomes the bubbling challenges faced in ITC and is more cost-effective compared to SPR or BLI.
The field-effect transistor (FET) based biosensors is an emerging field for detecting biomolecular interactions by offering varied biomedical applications. In the FET system, targets are immobilized to the biosensor chips and interactions are detected by changes in conductance32. The unique feature to be considered in the development of an efficient electronic biosensor is the physicochemical properties such as the semi-conductive nature and chemical stability of the coating material used to fabricate the sensor surface33. Conventional materials like silicon used for FET have limited the sensitivity of sensors because it requires oxide layers sandwiched between the transistor channel and a specific environment for proper functioning34. Moreover, silicon transistors are sensitive to high salt environments, thus making it hard to measure biological interactions in their natural environment. The graphene-based biosensor is presented as an alternative as it offers excellent chemical stability and electric field. Since graphene is a single atomic layer of carbon, it is both extremely sensitive as a semi-conductor and chemically compatible with biological solutions; both of these qualities are desirable to generate compatible electronic biosensors35. The remarkable ultrahigh loading potential of biomolecules offered by graphene-coated biosensors lead to the development of graphene-based biosensors FEB technology.
Principle of FEB technology: FEB is a label-free biomolecular detection technique that measures the electric current through the graphene biosensor to which the binding targets are immobilized. Interactions between the immobilized protein and the analyte result in alterations in current that are monitored in real-time, enabling accurate kinetic measurements36.
Instrumentation: The FEB system comprises a graphene field-effect transistor (gFET) sensor chip and an electronic reader that applies a constant voltage throughout the experiment (Figure 1). The analyte is applied in solution to the target protein immobilized on the biosensor surface. When an interaction occurs, an alteration in the current is measured and recorded in real-time. As the analyte concentration increases, the fraction of bound analyte will also increase, causing higher alternations in the current. Using the automated analysis software provided with the instrument (Table of Materials), I-Response is measured and recorded in terms of biosensing units (BU)37. I-Response is defined as the alteration in the current (I) through the biosensor chip measured in real-time upon the interaction of the immobilized target with the analyte. The FEB automated analysis software can analyze both the I-Response and C-Response to dynamic interaction events, where the C-Response records the alterations in the capacitance (C). The variations in both the I-Response and C-Response correspond directly to the fraction of bound analyte and can be further analyzed to generate KD values. The automated analysis software's default preference is I-Response.
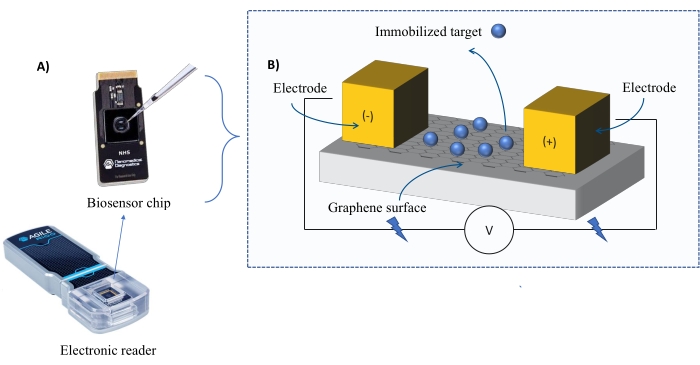
Figure 1: Overview of the experimental setup. (A) Graphene-based chip and an electronic reader. (B) An overview of the chip components. The chip is attached to two electrodes that supply current to the system. The surface of the chip is covered with graphene, which when activated can bind the target. Please click here to view a larger version of this figure.
Methodology:
Initially, the activated biosensor chip is inserted into the FEB device (Figure 1) followed by the execution of the steps outlined below: (1) Calibration: The experiment starts with system calibration using 1x phosphate-buffered saline (PBS; pH = 7.4) to create the baseline equilibration response. (2) Association: The analyte is introduced in the chip, and the I-Response is monitored until binding saturation is reached. (3) Dissociation: The analyte is dissociated using 1x PBS. (4) Regeneration: Remnants of the analyte are removed using 1x PBS. (5) Washing: A total of five washes are performed using 1x PBS for the thorough removal of the bound and unbound analytes from the chip.
Analysis:
Data analysis is performed using the fully automated software provided with the instrument. The automated analysis software generates a Hill fit plot with a KD value. The Hill fit plot describes the association of an analyte to the target protein as a function of analyte concentrations. The concentration at which a half-maximal response is achieved is proportional to the KD value. A low KD value represents high binding affinity and vice versa.
To validate the data obtained from the FEB experiment, I-Responses are extracted from each readout point for each analyte concentration using the data review/export software and can be exported to other statistical analysis software (see Table of Materials) as explained below.
Protocol
NOTE: The recombinant proteins used in this study, Hsp90 and Cdc37, were commercially obtained (see Table of Materials).
1. Chip activation
NOTE: All materials to be used in the experiment are listed in the Table of Materials. Filter all prepared solutions through a sterile 0.2 μm filter.
- Prepare 1-Ethyl-3-(3-dimethylamino propyl) carbodiimide (EDC) solution by adding 2 mg of EDC to 2.5 mL of 1 M 2-(N-morpholino) ethane sulfonic acid (MES) buffer (pH = 6.0) in a 15 mL tube. Prepare N-Hydroxysulfosuccinimide (sulfo-NHS) solutions by adding 6 mg of sulfo-NHS to 2.5 mL of 1 M MES buffer (pH = 6.0) in a separate 15 mL tube. Aliquot 50 µL of each solution in independent tubes and store at -20 °C for future use.
- Mix equal volumes of EDC sand sulfo-NHS solution (50 µL of EDC + 50 µL of sulfo-NHS) by pipetting up and down (do not vortex).
NOTE: The mixed solution of EDC/sulfo-NHS must be used within 30 min to maintain effective cross-linking for proper chip functionalization. - Place the biosensor chip (5.7 cm x 2.4 cm; see Table of Materials) supplied by the company in a glass Petri dish with a fitted lid. All the functionalization steps involved in chip activation are suggested to be done within the Petri dish. Apply 50 µL of 1 M MES buffer (pH = 6.0) to the biosensor chip, incubate for 1 min at room temperature, and then aspirate the buffer.
- Apply 50 µL of EDC/sulfo-NHS solution immediately to the sensor chip. Cover the Petri dish and incubate for 15 min at room temperature. Aspirate EDC/sulfo-NHS solution from the chip.
- Rinse the chip once with 50 µL of 1 M MES buffer (pH = 6.0); aspirate the MES buffer.
2. Target protein immobilization
- Rinse the chip 2x with 50 µL of 1x PBS (pH = 7.4). Aspirate the PBS from the chip and add the target molecule, Hsp90 (50 µL; 500 nM).
NOTE: Buffer mismatch may ruin the entire experiment; hence, before the experiment, it is important to make sure that the target molecule is in the same buffer as that used for calibration (e.g., 1x PBS (pH = 7.4)). If needed, perform a buffer exchange by overnight dialysis before the experiment. In this experiment, overnight dialysis was performed for both recombinant proteins, Hsp90 and Cdc37, against 1x PBS (pH = 7.4; see Table of Materials) with proper buffer exchange at 4 °C. The concentration of the target material (Hsp90 in this case) may vary according to different experimental protocols and the nature of target materials (protein/peptide/ligands). - Cover the glass Petri dish and incubate for 30 min at room temperature. Aspirate the solution containing the target molecule and rinse 3x with 50 µL of 1x PBS (pH = 7.4). Aspirate the 1x PBS (pH = 7.4) solution from the chip.
- Add 50 µL of Quench 1 (3.9 mM amino-PEG5-alcohol in 1x PBS (pH = 7.4)) solution to the chip. Cover the glass Petri dish and incubate for 15 min at room temperature. Aspirate the Quench 1 solution from the chip.
- Add 50 µL of Quench 2 (1 M ethanolamine (pH = 8.5)) solution to the chip. Cover the glass Petri dish and incubate for 15 min at room temperature. Aspirate Quench 2 solution from the chip and rinse the chip 5x using 50 µL of 1x PBS, leaving the last PBS droplet on the sensor.
3. Preparing analyte samples
- Prepare analyte dilution series for Cdc37 in the desired concentration range. For the first experiment, the following concentrations were used: 25 nM, 50 nM, 100 nM, 200 nM, 400 nM, 800 nM, 1,000 nM, 2,000 nM, 3,000 nM, and 5,000 nM. For the second experiment, a different set of concentrations ranging from 0.4 nM to 200 nM was used.
- Design the experiment to include at least eight different analyte concentrations to obtain a reliable KD value. Prepare the different dilutions of the analyte protein in the same buffer as that used for calibration and target protein; here it is 1x PBS (pH = 7.4).
4. Loading of the activated biosensor chip into the FEB device
NOTE: The FEB device consists of a reader featured with LED light indications and a cartridge to insert the biosensor chip.
- After target protein immobilization, insert the activated chip into the cartridge of the device, which is connected via USB to a computer. After the chip insertion, a green LED light will be displayed on the reader indicating that the FEB device is ready for the experiment. Install the automated software (see Table of Materials) supplied by the company on the computer, to which the FEB device is connected, to monitor the experiment step by step as described below.
5. Run the experiment
- Press the Run Experiment module on the automated software and choose 10 Points with Regeneration or any other desired protocol. Fill in the following details: operator name, experiment name, date (e.g., Yana, Hsp90 + Cdc37, 14.03.2021); regeneration buffer (e.g., PBS buffer); immobilized target (e.g., Hsp90); analyte in solution (e.g., Cdc37). See Supplementary Figure S1 for details.
- Press the Begin the Experiment button displayed on the software and follow the instructions shown by the automated software as described below.
NOTE: The software is fully automated, user-friendly, and guides the user throughout the experiment step by step. A pop-up window will appear on the screen with instructions to proceed further at each step of the experiment. The software will provide instructions for each repetitive step consecutively from calibration, analyte association, dissociation, regeneration, and wash (5x) for each analyte concentration throughout the experiment. - Perform instrument calibration. To do so, aspirate the remaining PBS solution from the chip and apply 50 µL of calibration buffer (1x PBS; pH = 7.4). Press the Continua button and wait for 5 min until the calibration step is finished. The software displays the endpoint determined for the calibration step (5 min) with a warning alarm to follow-up.
- Next, perform an analyte association. To do so, aspirate the calibration buffer from the chip and apply 50 µL of the lowest analyte concentration (25 nM of Cdc37). Press the Continua button and wait for 5 min until the association step is finished. The software displays the endpoint for the association step (5 min) with a warning alarm to proceed.
- Perform an analyte dissociation. To do so, aspirate the analyte solution from the chip and apply 50 µL of the dissociation buffer (1x PBS; pH = 7.4). Press the Continua button and wait for 5 min until the dissociation step duration (5 min) is finished. The software displays the endpoint for the dissociation step (5 min) with a warning alarm to follow-up.
- Next, perform chip regeneration. Aspirate the dissociation solution from the chip and apply 50 µL of regeneration buffer (1x PBS; pH = 7.4). Press the Continua button and wait for 30 s until the regeneration step duration (30 s) is finished. The software displays the endpoint for the regeneration step (30 s) with a warning alarm to follow-up.
- Finally, wash the chip. Aspirate the regeneration solution from the chip and apply 50 µL of wash buffer (1x PBS; pH = 7.4) to the chip. Aspirate the solution from the chip and repeat this 5x. Leave the last drop of wash buffer on the chip and press the Continua button and wait for 30 s until the wash step duration is finished in the software display.
NOTE: The software displays the endpoint for the wash step (30 s) with a warning alarm to proceed with the next cycle of the experiment. - Repeat the steps for each analyte concentration used; the five steps of calibration, analyte association, dissociation, regeneration, and wash (5x) constitute one cycle. For the experiment shown here, we performed 10 cycles for 10 analyte concentrations (ranging from 25 nM to 5,000 nM or 0.4 nM to 200 nM; Figure 2).
6. Analysis
- Press the Analysis button seen at the top of the automated analysis software at the end of the experiment. A display window containing all the experimental points will appear. In the window, ensure that the analyte concentrations used for the prescribed protocol are correct.
- Press the Run Analysis button to generate the KD value automatically. The software generates a Hill fit plot by plotting the analyte concentrations against the corresponding I-Responses from which the dissociation constant at equilibrium, KD value, is calculated.
- Export the raw data for analysis in other statistical analysis software by using the data review/export software as described below.
- Copy the R1R file created automatically at the end of the experiment (e.g., Hsp90 + Cdc37 14.03.2021) to a new folder on the desktop. Open the Data Review/Export software supplied by the company (see Supplementary Figure S2A).
- Click on Data Processing > Process R1R files > OK on the home screen of the data review/export software.
- Select the folder containing R1R files created on the desktop in step 6.3.1. and press the OK button. This creates a copy of the original R1R data, to review and edit the data without overriding the original file.
- Press the data processing icon seen at the home screen of the data review/export software. Press Load Processed R1R Files > OK. Select the same folder created in step 6.3.1 containing processed R1R files. Press OK. At this step, the folder containing the experiment files is ready to be reviewed.
- At the home screen of data review/export software, press Data Analysis. Select Calibration > Calibrate edited R1R files > OK (see Supplementary Figure S2B). This step calibrates all the data points according to the first calibration step, to create a baseline.
- At the home screen of data review/export software, press Data Analysis. Select Review and Edit R1R Files > OK. Review the data points, delete points, or add steps by using the Delete/Add Step button. Make sure all the steps are in the correct place, e.g., the calibration step is on the baseline, the association step is at the peak (see Supplementary Figure S3), and then press the Save button to save all the changes made.
- At the home screen of data review/export software, press Data Analysis. Select Analyze/Plot Data, choose the steps to export (see Supplementary Figure S4). Add each association step (e.g., step 2, step 7, and so on) after subtracting the calibration step (e.g.,step 1, step 6, and so on) to the export list before exporting the data.
- Press Export this Data. The software generates a spreadsheet file containing the I-Response for each analyte concentration data point from each transistor (if all transistors are working, we will have three different I-response values for each concentration point). Use this spreadsheet file to further analyze the data in the statistical software.
- Open the statistical analysis software. Create an XY table with three Y values; press Create. The data for this table (X, Y values) is copied from the spreadsheet file created in step 6.3.8. The x-axis corresponds to analyte concentration (depending on the concentration used), and the three Y values correspond to the I-Responses, obtained from the Data Review/Export software supplied by the company.
- Press Analyze this Data. Choose XY Analyses > Nonlinear Regression (Curve Fit) > Binding Saturation > One Site > Total. The software will automatically analyze the data, generate the KD value, and create a graph of the data points.
Representative Results
Results from experiment 1:
The target protein Hsp90 (500 nM) was immobilized to the chip following the target immobilization protocol as described above. For the first experiment, 10 concentrations of the analyte protein, Cdc37, ranging from 25 nM to 5,000 nM, were prepared based on the data available in the literature (see Table 1).
The steps of the experiment can be monitored in real-time by following the alterations occurring in the I-Response (Figure 3A). One can visualize how each step included in the protocol affects the I-Response throughout the experiment. For example, the I- Response is close to zero during calibration, and in the association step, the I-Response increases gradually with increasing analyte (Cdc37) concentrations. Looking at the data from this experiment (Figure 3A), a sudden increase in the I-Response was observed in low analyte concentrations (50 BU at 25 nM). A low I-Response was expected at low analyte concentration that will increase gradually with increasing concentrations. This sudden increase suggested that an experiment including lower analyte concentration points should be designed.
Following the protocol steps discussed above, the data was analyzed in the provided automated analysis software, which automatically generated a Hill fit plot with a calculated KD value (Figure 3B). The KD value calculated from the automated analysis software for the first experiment was 350,000 ± 76,000 µM. Most of the common biological interactions showed a KD value in the low µM or even in the nM range; thus, the KD value generated for the first experiment is very high.
There can be several explanations for the observed high KD value: there may be low-affinity binding between the two specific proteins studied (Hsp90/Cdc37), which means that a high ligand concentration is required for the molecular interactions; the experiment might have been designed with non-ideal, out-of-range analyte concentrations; the high value can be due to the built-in parameters used in the fitting module of the analysis software. The automated analysis software uses a dose-response analysis module to fit the data points, but some interactions may not fit well in this model, thus giving false data, resulting in high KD values.
To obtain an accurate KD value with a saturated Hill plot, standardization of the experiment by trial and error is required which includes both lower and higher analyte concentrations points, to acquire the lowest KD value with a low error rate.
Considering all these factors, the data from analysis software was exported to additional statistical analysis software. The I-Response for each association point was extracted using the automated data review/export software and is summarized in Table 2 by following the steps discussed above in the analysis step (step 6).
The statistical analysis software results (Figure 3C) showed a relatively low KD value with a high standard deviation (0.011 ± 0.494 µM) and a low R2 value of 0.78. The significant variation observed in the KD value and low R2 value suggested that the concentration points chosen for this experiment are not optimal. In addition, it was also noticed that some graph points were not fitting well with the plot trendline (Figure 3C); this strengthened the need to design a second experiment by including different analyte concentration points.
The built-in parameters used by the two analysis software are entirely different, and it is reflected in the KD values generation process. The differences in the KD values obtained from the two analysis software (350,000 ± 76,000 µM vs. 0.011 ± 0.494 µM) can be attributed to the fact that different analysis modules were used for calculation. The analysis performed in the statistical analysis software may be a better fit for this interaction, thus giving a lower KD value.
Nevertheless, if the results from both analyses are combined (350,000 ± 76,000 µM and 0.011 ± 0.494 µM), it can be concluded that the first experiment was not optimal and that additional concentration points would be required.
To validate this point, the uncorrelated graph point, 3,000 nM was excluded (see Supplementary Figure S5). The results of the analysis showed a KD value of 0.006 ± 0.081 µM with an R2 value of 0.98. These results indicated that the next experiment should be designed focusing on more relevant concentrations. Therefore, a second experiment was performed to evaluate Hsp90/Cdc37 PPI using different test points with concentrations below and above the predicted KD.
Results from experiment 2:
The target protein, Hsp90 (500 nM) was immobilized to the chip using the protocol as described above. A total of 10 concentrations of the analyte (Cdc37) were prepared as follows: 0.4 nM, 0.8 nM, 1.6 nM, 3.2 nM, 6.4 nM, 12.5 nM, 25 nM, 50 nM, 100 nM, and 200 nM. The second experiment was designed by including different analyte concentration points, by considering all the insights gained from the previous experiment as mentioned above.
During the experiment, each step was monitored in real-time (Figure 4A). Here, the real-time I-Response graph (derived from the low concentrations of the analyte-0.4 nM, 0.6 nM, 1.6 nM) starts initially from zero and increases gradually upon the increase of analyte concentrations at each cycle of the experiment as compared to the sudden increase of I-Response observed in the first experiment.
Following the protocol mentioned above, the data were analyzed using the automated analysis software (Figure 4B). The KD value calculated from the automated analysis software for this experiment was 0.0673 ± 0.0002 µM, which suggested a strong interaction between target protein Hsp90 and analyte Cdc37. The standard deviation of 0.0002 µM was much smaller than that obtained in the first experiment, which gives confidence that the new data is much more reliable.
To validate this result, we exported the data points (Table 3) to another statistical analysis software as explained previously; the results from the analysis are summarized in Figure 4C.
Using the statistical analysis software, it was found that the KD value for Hsp90/Cdc37 PPI is 0.014 ± 0.005 µM (Figure 4C). The low values of KD and standard deviation indicate a strong interaction between the two proteins, Hsp90 and Cdc37. Moreover, the R2 value of 0.99 indicates that the analysis perfectly fits the experimental graph points. Both analysis software showed consistency in the results, with low KD values of about 0.0673 ± 0.0002 µM and 0.014 ± 0.005 µM, respectively.
To confirm the experiment's reproducibility and accuracy, the same experiment was performed in two replicates using Hsp90 (500 nM) as target protein and Cdc37 as analyte with the same concentrations ranging from 0.4 nm to 200 nm as used in the second experiment. During the experiment, each step was monitored in real-time (Supplementary Figure S6A and Supplementary Figure S7A). The KD value calculated from the automated analysis software is depicted in Supplementary Figure S6B and Supplementary Figure S7B (0.053 ± 0.002 µM and 0.0719 ± 0.0007 µM, respectively). As mentioned previously, the data points were exported (Supplementary Table S1 and Supplementary Table S2) to another statistical analysis software, and the results are shown in Supplementary Figure S6C and Supplementary Figure S7C (Supplementary Figure S6C: KD is 0.003 ± 0.009 µM and R2 is 0.99; Supplementary Figure S7C: KD is 0.004 ± 0.009 µM and R2 is 0.99). Results of the two replicates from both the analysis software corroborated well and confirmed the accuracy, repeatability, and reproducibility of the Hsp90/Cdc37 FEB experimental data.
A control study was also performed in which the biomolecular interaction between Hsp90 and a control protein, Bovine serum albumin (BSA) was elucidated. The control study using BSA as an analyte was done using an identical experimental setup and analyte concentrations used in the second experiment. A total of 10 concentrations of the control protein, BSA, ranging from 0.4 nM to 200 nM were prepared in 1x PBS (pH = 7.4) and each step of the experiment was monitored in real-time.
Similar to the experiment with Hsp90 and Cdc37, the real-time I-Response graph initially starts from 0; however, in the control study, the I-Response did not increase gradually upon the increase of analyte concentrations (Supplementary Figure S8A). The data were also analyzed using the automated analysis software (Supplementary Figure S8B), and the software was unable to calculate the KD value from the data points of this experiment. It is evidenced, from Hill Plot generated from the software, that there is no interaction between Hsp90 and the control BSA analyte. To validate this result, we exported the data points (Supplementary Table S3) to another statistical analysis software as explained previously. Using the statistical analysis software, it was found that the KD value for Hsp90/BSA PPI is negative (Supplementary Figure S8C; KD is -0.009 ± -0.0003 µM), again indicating that there is no interaction between the two proteins, Hsp90 and BSA. Moreover, the R2 value (0.82) indicates that the analysis did not fit the experimental graph points. Both analysis software showed consistency in the results that Hsp90 and BSA do not interact.
When comparing Hsp90/Cdc37 FEB experimental data with other experimental data available in the literature, it was found that the KD values obtained in this study (KD = 0.014 ± 0.005 µM, KD = 0.053 ± 0.002 µM, and KD = 0.072 ± 0.001 µM) were in two orders of magnitude lower than previously published data (KD = 1.46 µM38 or 6 µM39). These differences can be attributed to the use of full-length human Hsp90 and Cdc37 constructs in the current experiments. Previous studies were performed on yeast Hsp90 constructs that contained only partial protein38 or C. elegans homolog proteins39 (Table 1). To validate the results obtained in the FEB system, ITC experiments were performed.
Evaluating Hsp90/Cdc37 PPI using the ITC system:
ITC is considered the gold standard to investigate biomolecular interactions. We further performed ITC to determine the thermodynamic kinetics of the Hsp90/Cdc37 PPI, and as validation for the FEB experiment. The Hsp90 and Cdc37 are subjected to dialysis against PBS (pH = 7.0) before ITC. About 0.25 µM solution of the target protein (Hsp90) and 2.5 µM solution of analyte protein (Cdc37) were prepared in PBS for the calorimetric titrations. A total volume of 80 µL of Cdc37 solution at consecutive intervals was titrated from the rotating syringe to the cell containing 150 µL of Hsp90 solution at 298.15 K. Each injection occurred over a 4 s duration, and the time interval between consecutive injections was 150 s. A total of 19 injections of 2 µL each were made with a stirring speed of 500 rpm and a reference power of 41.9 µW, respectively. The heat variation resulting from Hsp90/Cdc37 interaction was monitored, and the final data obtained at the end of the Cdc37 analyte injections were fitted using ITC analysis software supplied by the company. The heat variations along with the thermodynamic parameters such as molar enthalpy change (ΔH), entropy change (ΔS), the number of binding sites (n), and the equilibrium dissociation constant (KD) between Hsp90/Cdc37 PPI were estimated. The Gibbs free energy change (ΔG) was also calculated from the equation ΔG = ΔH – TΔS after obtaining thermal parameters from the ITC thermogram.
The ITC plot reveals the amount of heat used up or liberated during the biological interactions to elucidate the nature of interactions and intermolecular forces governing the PPI40. Cdc37 (2.5 µM) was titrated into Hsp90 (0.25 µM), and the corresponding curves are depicted in Figure 5A,B. The ITC plot obtained was found to be exothermic and was fitted into a single binding site model exhibiting a binding stoichiometry (n) of one, binding energy, ΔG = -45.9 kJ/mol, and KD value = 0.009 µM. The slight inconsistency observed in the form of an endothermic shift at the beginning of the ITC thermogram (Figure 5A) suggests the formation of bubbles while titrating the analyte protein to the measurement cell41,42. Among the Hsp90/Cdc37 PPI, ITC experiments performed at lower and higher concentrations, the 10-fold excess of Cdc37 (Hsp90 concentration = 0.25 µM and Cdc37 concentration = 2.5 µM) exhibited acceptable binding parameters by attaining a saturation curve. The KD value obtained from ITC (KD = 0.009 µM) correlated well with the KD value calculated using FEB technology (KD = 0.014 µM, 0.053 µM, and 0.072 µM), strongly supporting FEB's sensitivity and accuracy in detecting Hsp90/Cdc37 PPI.
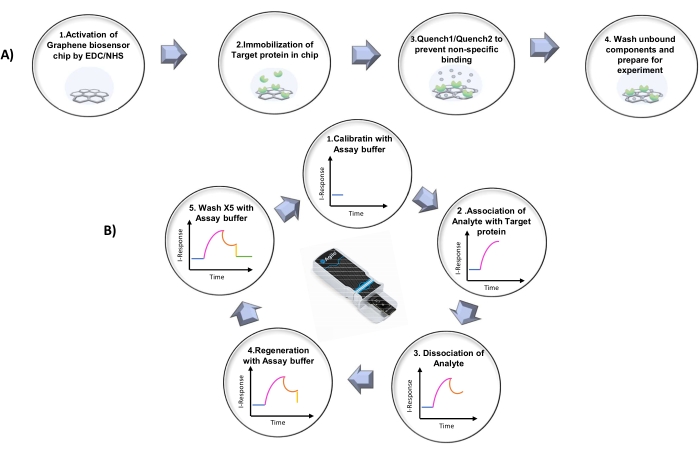
Figure 2: Protocol summary. (A) Summary of the steps for the chip activation process. (B) Graphical representation of the five repeating steps in the protocol. Please click here to view a larger version of this figure.
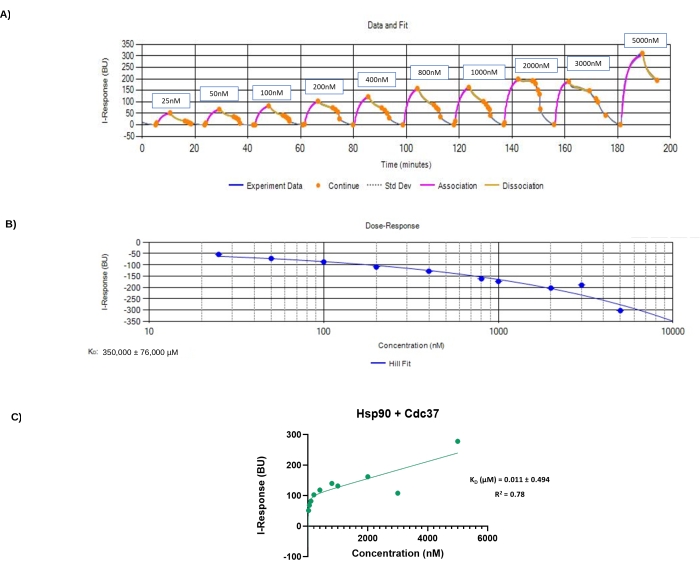
Figure 3: Data files generated from experiment 1. (A) Experiment data monitored in real-time. The y-axis corresponds to the I-Response in biosensor units (BU), and the x-axis corresponds to the different time points and concentrations of the analyte in the experiment. This figure corresponds to the data from the first experiment. (B) Hill Fit plot generated from the automated analysis software. The y-axis corresponds to I-Response, the x-axis corresponds to concentrations of analyte (Cdc37) in nM. For this experiment, the KD value is 350,000 ± 76,000 µM. (C) Association plot generated using the statistical analysis software. The y-axis corresponds to I-Response at the end of the association phase, the x-axis corresponds to the different analyte, Cdc37, concentrations. This plot was generated using the data points provided in Table 2. Summary of the analysis results are provided in the graph; for this experiment, the KD value is 0.011 ± 0.494 µM and the R2 value is 0.78. Please click here to view a larger version of this figure.
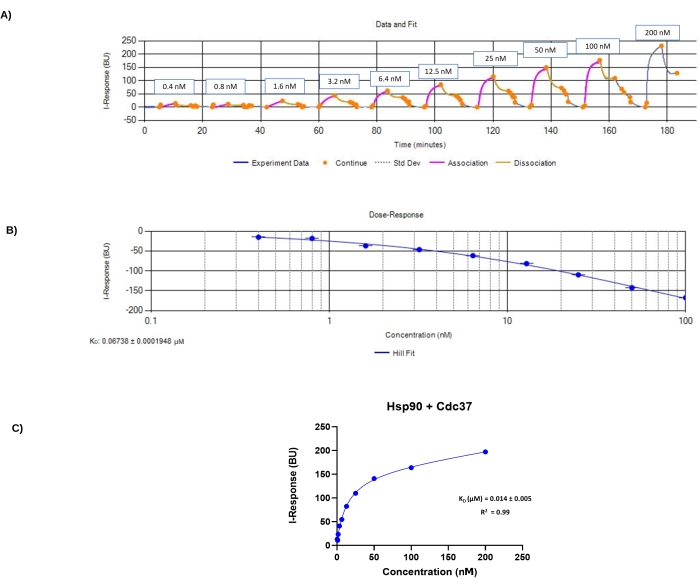
Figure 4: Data files generated from experiment 2. (A) Experiment data monitored in real-time. The y-axis corresponds to the I-Response in biosensor units (BU), and the x-axis corresponds to the different time points and concentrations of the analyte in the experiment. This figure corresponds to the data from the second experiment. (B) Hill Fit plot generated from the automated analysis software. The y-axis corresponds to I-Response, the x-axis corresponds to concentration of analyte (Cdc37) in nM. For this experiment, the KD value is 0.0673 ± 0.0002 µM (C) Association plot generated using the statistical analysis software. The y-axis corresponds to I-Response at the end of the association phase, the x-axis corresponds to the different analyte Cdc37 concentrations. This plot was generated using the data points provided in Table 3. Summary of the analysis results are provided in the graph; for this experiment, the KD value is 0.014 ± 0.005 µM and the R2 value is 0.99. Please click here to view a larger version of this figure.
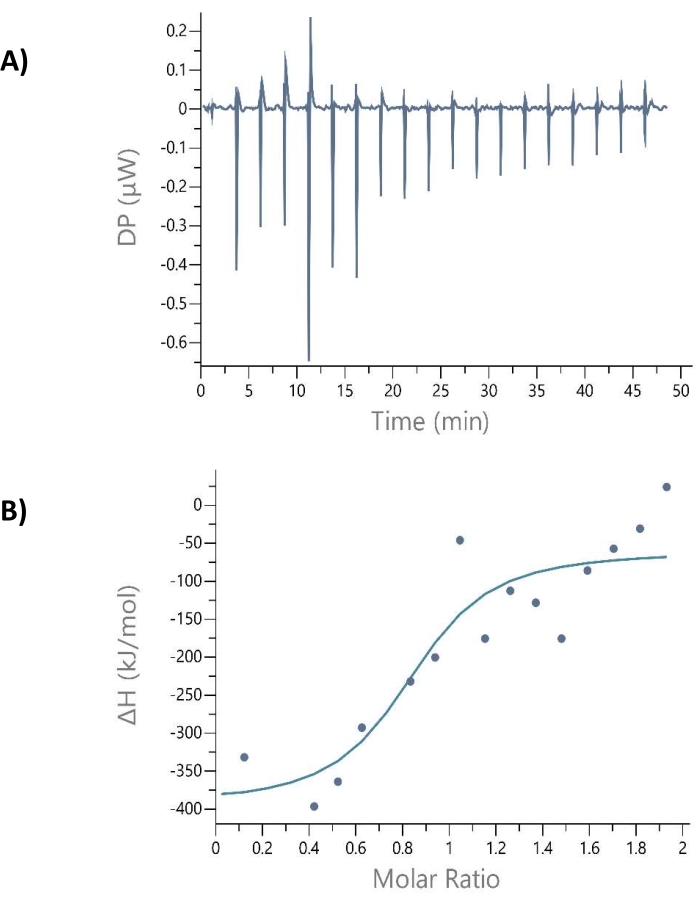
Figure 5: ITC thermogram of Hsp90 and Cdc37 interaction. (A) The top panel represents the corresponding heat evolving curves obtained due to consecutive injections of Cdc37 (2.5 µM) into Hsp90 (0.25 µM) at 298.15 K. The differential power (DP) represents differential power as a function of time. (B) The integrated data points in the bottom panel designate the corresponding normalized heat versus the molar ratio of Hsp90/Cdc37. Please click here to view a larger version of this figure.
| Assays performed | FEB analysis KD | Prism analysis KD | Construct Origin | Amino Acids | Riferimenti |
| ITC | 1.46 μM, and 1.32 μM | NA | Yeast Hsp90 | N-terminal domain of Hsp90 | 36 |
| Human Cdc37 | |||||
| SPR | 6 µM | NA | C. elegans | Full length | 37 |
| FEB – First repetion | 0.014 μM | 0.067 μM | Human Hsp90 | Full length | Performed in this study |
| Human Cdc37 | |||||
| FEB – Second repetition | 0.053 μM | 0.003 μM | Human Hsp90 | Full length | Performed in this study |
| Human Cdc37 | |||||
| FEB – Third repetition | 0.072 μM | 0.004 μM | Human Hsp90 | Full length | Performed in this study |
| Human Cdc37 | |||||
| ITC | 0.009 µM | NA | Human Hsp90 | Full length | Performed in this study |
| Human Cdc37 | |||||
| FEB- Control study | No data (There was insufficient data to calculate the KD) | -0.009 μM (KD value with negative values indicates that there is no interaction) | Human Hsp90 | Full length | Performed in this study |
| BSA |
Table 1: Hsp90/Cdc37 PPI evaluation using different methods reported in this study and the literature.
| Analyte concentrations (nM) | I-Response transistor 1 | I-Response transistor 2 | I-Response transistor 3 | Average |
| I-Response | ||||
| 25.00 | 53.540 | 49.406 | 50.590 | 51.178 |
| 50.00 | 71.427 | 65.943 | 67.568 | 68.313 |
| 100.00 | 85.305 | 79.226 | 81.074 | 81.868 |
| 200.00 | 106.652 | 99.320 | 101.502 | 102.491 |
| 400.00 | 122.572 | 114.837 | 117.128 | 118.179 |
| 800.00 | 144.293 | 136.374 | 138.872 | 139.846 |
| 1000.00 | 135.165 | 128.657 | 130.425 | 131.416 |
| 2000.00 | 164.487 | 159.401 | 161.697 | 161.862 |
| 3000.00 | 109.997 | 106.705 | 107.205 | 107.969 |
| 5000.00 | 282.637 | 274.207 | 276.247 | 278.422 |
Table 2: Summary of the I-Response values generated from three different transistors of each chip at each concentration point of the first experiment.
| Analyte concentrations (nM) | I-Response transistor 1 | I-Response transistor 2 | I-Response transistor 3 | Average I-Response |
| 0.40 | 13.097 | 14.721 | 11.714 | 13.177 |
| 0.80 | 10.183 | 8.861 | 13.240 | 10.762 |
| 1.60 | 24.826 | 20.377 | 25.240 | 23.481 |
| 3.20 | 40.746 | 41.047 | 40.783 | 40.858 |
| 6.40 | 54.938 | 54.224 | 54.913 | 54.691 |
| 12.80 | 83.057 | 81.452 | 82.596 | 82.369 |
| 25.60 | 110.440 | 109.152 | 110.343 | 109.978 |
| 51.20 | 141.161 | 140.363 | 141.125 | 140.883 |
| 102.40 | 164.215 | 162.679 | 164.572 | 163.822 |
| 200.00 | 196.373 | 197.007 | 198.495 | 197.292 |
Table 3: Summary of the I-Response values generated from three different transistors of each chip at each concentration point of the second experiment.
Supplementary Figure S1: Screenshot of the main screen of automated software before the beginning of the experiment. Please click here to download this File.
Supplementary Figure S2: Screenshot of the main screen of automated software for data processing and analysis. (A) Main screen display of data review/export software showing data processing options. (B) The main screen display of data review/export software showing data analysis options. Please click here to download this File.
Supplementary Figure S3: The review data points obtained from review/export software. In this step, review the experiment and add or delete data points. Make sure the association point (2, 7, 12, 17, 22, and so on) is always at the pick. For example, the first pick from the left is data point number 2, and then data point number 10 (but it should be number 7). To solve this just delete points 3, 4, and 5. Please click here to download this File.
Supplementary Figure S4: Export data list for example. In this step, chose what experiment points to be exported. Chose an association point (2, 7, 12, 17, 22, and so on) and subtract the previous calibration steps (1, 6, 11, 16, 21, and so on). Add all the points to the export list and export this data to get a spreadsheet with the I-Response for each data point. Please click here to download this File.
Supplementary Figure S5: Association plot generated using the statistical analysis software. The y-axis corresponds to I-Response at the end of the association phase, the x-axis corresponds to the different analyte Cdc37 concentrations. This plot was generated using the data points from the first experiment and excluded the 3,000 nM concentration data point. Summary of the analysis results are provided in the graph; for this experiment, the KD value is 0.006 ± 0.081 µM and the R2 value is 0.98. Please click here to download this File.
Supplementary Figure S6: Data files generated from experiment 2 replicate 1. (A) Experiment data monitored in real-time. The y-axis corresponds to the I-Response in biosensor units (BU), and the x-axis corresponds to the different time points and concentrations of the analyte in the experiment. This figure corresponds to the data from the second repetition performed in the second experiment where Hsp90 was immobilized on the chip (500 nM) and 10 concentrations of Cdc37 analyte were prepared (0.4-200 nM). The real-time I-Response graph starts initially from 0 and increases gradually upon the increase of analyte concentrations at each cycle of the experiment, suggesting the experiment is successful. (B) Hill Fit plot generated from the automated analysis software. The y-axis corresponds to I-Response, the x-axis corresponds to concentrations of analyte (Cdc37) in nM. The results from the automated analysis showed a relatively low KD value and low standard deviation of 0.0531 ± 0.0002 µM, suggesting a strong interaction between Hsp90 and Cdc37. (C) The association plot generated using the statistical analysis software. The y-axis corresponds to I-Response at the end of the association phase, the x-axis corresponds to the different analyte Cdc37 concentrations. This plot was generated using the data points provided in Table S1. Summary of the analysis results are provided in the graph; for this experiment, the KD value is 0.003 ± 0.009 µM and the R2 value is 0.99. Please click here to download this File.
Supplementary Figure S7: Data files generated from experiment 2 replicate 1. (A) Experiment data monitored in real-time. The y-axis corresponds to the I-Response in biosensor units (BU), and the x-axis corresponds to the different time points and concentrations of the analyte in the experiment. This figure corresponds to the data from the third repetition in the second experiment where Hsp90 was immobilized on the chip (500 nM) and 10 concentrations of Cdc37 analyte were prepared (0.4-200 nM). (B) Hill Fit plot generated from the automated analysis software. The y-axis corresponds to I-Response, the x-axis corresponds to concentrations of analyte (Cdc37) in nM. The results from the automated analysis showed a relatively low KD value and a low standard deviation of 0.0719 ± 0.0007 µM, suggesting a strong interaction between Hsp90 and Cdc37. (C) Association plot generated using the statistical analysis software. The y-axis corresponds to I-Response at the end of the association phase. The x-axis corresponds to the different analyte Cdc37 concentrations. This plot was generated using the data points provided in Table S2. Summary of the analysis results are provided in the graph; for this experiment, the KD value is 0.004 ± 0.009 µM and the R2 value is 0.99. Please click here to download this File.
Supplementary Figure S8: Data files generated from the control experiment. (A) Experiment data monitored in real-time. The y-axis corresponds to the I-Response in biosensor units (BU), and the x-axis corresponds to the different time points and concentrations of the analyte in the experiment. This figure corresponds to the data from the control experiment where Hsp90 was immobilized on the chip (500 nM) and 10 concentrations of BSA (control protein) analyte were prepared (0.4-200 nM). The real-time I-Response graph starts from low I-Response around 10-15 BU and does not increase upon an increase in the analyte concentrations at each cycle of the experiment, suggesting there is no interaction between the Hsp90 and BSA. (B) Hill Fit plot generated from the automated analysis software. The y-axis corresponds to I-Response; the x-axis corresponds to concentrations of analyte (BSA) in nM. The results from the automated analysis showed that there is insufficient data to calculate the KD value suggesting there is no interaction between Hsp90 and BSA. (C) Association plot generated using the statistical analysis software.The y-axis corresponds to I-Response at the end of the association phase, the x-axis corresponds to the different analyte BSA concentrations. This plot was generated using the data points provided in Table S4. Summary of the analysis results are provided in the graph; for this experiment, the KD value is -0.009 ± -0.0003 µM and the R2 value is 0.82. A negative KD value close to zero indicates that there is no interaction between Hsp90 and BSA. Please click here to download this File.
Supplementary Table S1: Summary of the I-Response values generated at each concentration point and read from each chip's transistors (three different transistors for each chip). This table corresponds to the data extracted from the second repetition performed for the second experiment where Hsp90 was immobilized on the chip (500 nM) and 10 concentrations of Cdc37 analyte were prepared (0.4-200 nM). The data points from this table were used for analysis in the statistical analysis software. Please click here to download this Table.
Supplementary Table S2: Summary of the I-Response values generated at each concentration point and read from each chip's transistors (three different transistors for each chip). This table corresponds to the data extracted from the third repetition performed for the second experiment where Hsp90 was immobilized on the chip (500 nM) and 10 concentrations of Cdc37 analyte were prepared (0.4-200 nM). The data points from this table were used for analysis in the statistical analysis software. Please click here to download this Table.
Supplementary Table S3: Summary of the I-Response values generated at each concentration point and read from each chip's transistor (three different transistors for each chip). This table corresponds to the data from the control experiment where Hsp90 was immobilized on the chip (500 nM) and 10 concentrations of BSA analyte were prepared (0.4-200 nM). The data points from this table were used for analysis in the statistical analysis software. Please click here to download this Table.
Discussion
In this study, the feasibility of using the FEB technology (a real-time kinetic characterization approach) was evaluated to determine the biomolecular interaction between Hsp90 and Cdc37. The initial exploratory experiment (first experiment) suggested that choosing the proper analyte concentrations is a critical part of the experiment and that the experiment should be designed by including concentration points above and below the KD value, which were predicted based on the data available in the literature.
Nevertheless, if there is no preliminary information about the interaction, we suggest designing a preliminary experiment by including a broad range of concentration points in both lower (0.08 nM, 0.16 nM, 0.4 nM, 2 nM) and higher (1,000 nM, 2,500 nM, 5,000 nM) ranges. The preliminary experiment provides a rough estimate of the KD value and enables to plan the subsequent experiments using the right concentration ranges to obtain an accurate KD value. The FEB system offers an edit protocol function in which one is free to choose the number of concentration points for the experiment.
From our experience, the most critical steps involved in the FEB protocol are as follows. The target protein and analyte buffer should be the same as the calibration buffer. Even a small buffer mismatch can cause fluctuations in the I-Response and give unreliable results. Thus, we suggest performing dialysis overnight by proper buffer exchange. Careful pipetting on the biosensor chip while aspirating or applying a solution is critical throughout the experiment. Careless pipetting may lead to broken transistors or fluctuations in the I-Response; while pipetting, make sure to not touch the sensitive surface of the chip. The EDC/sulfo-NHS solution used in the chip activation process should be mixed fresh before each use. Using an old mixture may result in a non-functionalized chip. Make sure to use the same buffer for all the five repetitive steps (calibration, association, dissociation, regeneration, and wash) involved in all cycles of the experiment.
When comparing FEB with well-established biomolecular detection systems in the field, such as BLI or SPR, we found that all three methods share similar advantages43,44,45 which are: they are label-free systems, which do not require any expensive fluorescent dyes to detect the interactions; they can detect interactions even in small nM ranges; the chips are reusable in both SPR and FEB and they can be regenerated and reused efficiently for up to 10 experiments, and all three methods provide kinetic data in real-time, and the experiment time is short.
The main advantage of SPR is that it provides more extensive data from the experiment so that one can calculate KD, Kon, Koff, stoichiometry, and thermodynamic parameters44. The major advantage of BLI is its ability to test multiple concentrations in one experiment. The kinetic measurements can be done simultaneously for up to eight concentrations by using different sensors. However, the BLI sensors are not reusable, thus making the cost of the experiment extremely high43.
The main advantage of FEB is its automated, user-friendly software that guides the user throughout the experiment, and its open-hand pipetting platform that allows the user to add the analyte to the immobilized target at each step. This open-pipetting approach combined along with a simple software makes the FEB easy to learn and requires little or even no training. However, it does require that the user has a basic mastery of hand pipetting, and the lack of automated liquid handling means that the reproducibility of step timing depends entirely on the user. This same lack of integrated liquid handling means that the chips and instruments are cost-effective compared to SPR or BLI. The FEB also exhibits high reproducibility and reliability in detecting biomolecular interactions due to the defined fabrication process involved in biosensor chip preparation35.
Compared to the well-established and standard ITC detection method, FEB utilizes only 50 µL of analyte volume at nanomolar concentrations for the whole experiment, decreasing the chance of nucleation or aggregation of proteins. One of the biggest problems with ITC is degassing the proteins before loading to avoid air bubbles. During the ITC experiment, there is a chance of developing bubbles inside the measurement cell due to continuous stirring and intense purging, which may adversely splatter the sample solution or cause sample loss42. The FEB technology overcomes these challenges of ITC by offering robust target immobilization chemistry, reducing aggregation and bubbling, increasing stability, and improving concentration-dependent problems faced by ITC.
A potential drawback of FEB devices is their susceptibility to buffer composition and concentration changes, which may cause a drift in the baseline that should be corrected during the fitting and analysis of the results. The biosensor chip is also sensitive to pH and salt, so buffers used during calibration and measurement of the analyte must be the same throughout the experiment. Chemicals used as preservatives (e.g., sodium azide) and hydrophobic molecules can damage the sensor and may even cause an increase in non-specific binding. These drawbacks can be mitigated with attention to buffer consistency throughout the measurement steps, dialysis of interfering compounds from the analyte stock, and appropriate passivation of the surface (e.g., use of detergents, protein blockers, or different PEG molecules).
In summary, we successfully demonstrated strong PPI between Hsp90/Cdc37 by using the FEB technology and validated their interaction using ITC, providing encouraging data to support FEB as an innovative alternative method for PPI detection. The FEB technology offers a wide range of applications in diverse fields of drug discovery, pharmaceuticals, biomolecular analysis of small molecules, peptides, and proteins, antibody development, drug validation, and lead optimization at our fingertips by translating in vitro biological activity readily into in vivo efficacy. Considering the advantages of FEB technology and its immense applications in exploring PPI, protein, and DNA or RNA interactions, as well as protein and small molecule (e.g., drug) interactions, we propose that it is a unique, promising technology in the PPI detection toolbox. In our instance, FEB technology offers a valuable tool to readily assess the binding efficacy of Hsp90/Cdc37 PPI candidate antagonists, which then aids in selecting promising candidates for further in vitro and in vivo evaluations pertaining to various diseases in which this PPI is critical.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This research was supported by a grant from the Binational Science Foundation (BSF) to S.K.S. and N.Q.
Materials
| Automated analysis software | Agile plus software, Cardea (Nanomed) | NA CAS number: NA |
Referred to in the text as the automated analysis software supplied with the instrument. Generates automated analysis. |
| COOH-BPU (Biosensing Processing Unit) | Agile plus software, Cardea (Nanomed) | NA CAS number: NA |
biosensor chip |
| Data review software | Datalign 1.0, Cardea (Nanomed) | NA CAS number: NA |
Referred to as the supplied data review software in the text. Supplied with the instrument and allows to review and export the information data points. |
| Dialysis bag | CelluSep, Membrane filtration products | T2-10-15 CAS number: NA |
T2 tubings (6,000-8,000 MWCO), (10 mm fw, 6.4mm Ø, 0.32ml/cm, 15m) |
| EDC (1-Ethyl-3-(3-dimethylamino propyl) carbodiimide) | Cardea (Nanomed) | EDC160322-02 CAS number: 25952-53-8 |
White powder |
| ITC (Isothermal titration calorimetry) system | Microcal-PEAQ-ITC (Malvern, United Kingdom) | NA CAS number: NA |
|
| MES (2-(N-morpholino) ethane sulfonic acid) buffer | Merck | M3671-50G CAS number: 4432-31-9 |
White powder |
| NHS (N-Hydroxysulfosuccinimide) chips | Cardea (Nanomed) | NA CAS number: NA |
Graphene-based chip |
| PBS (Phosphate-buffered saline) X 10 | Bio-Lab | 001623237500 CAS number: 7758-11-4 |
Liquid transparent solution |
| Pipete | Thermo Scientific | 11855231 CAS number: NA |
Finnpipette F3 5-50 µL, yellow |
| Quench 1 (3.9 mM amino-PEG5-alcohol in 1 X PBS) | Cardea (Nanomed) | 0105-001-002-001 CAS number: NA |
Liquid, transparent solution |
| Quench 2 (1 M ethanolamine (pH=8.5)) | Cardea (Nanomed) | 0105-001-003-001 CAS number: NA |
Liquid, transparent solution |
| Recombinant protein Cdc37 | Abcam | ab256157 CAS number: NA |
|
| Recombinant protein Hsp90 beta | Abcam | ab80033 CAS number: NA |
|
| Spreadsheet | Excel, Microsoft office | NA CAS number: NA |
|
| Statistical software | GraphPad, Prism | NA CAS number: NA |
Referred to as the other statistical software. Sigma plot, phyton or other statistical programes may also be used |
| Sulfo-NHS | Cardea (Nanomed) | NHS160321-07 CAS number: 106627-54-7 |
White powder |
| Tips | Alex red | LC 1093-800-000 CAS number: NA |
Tip 1-200 µl, in bulk, 1,000 pcs |
Riferimenti
- Tuncbag, N., Gursoy, A., Guney, E., Nussinov, R., Keskin, O. Architectures and functional coverage of protein-protein interfaces. Journal of Molecular Biology. 381 (3), 785-802 (2008).
- Berggård, T., Linse, S., James, P. Methods for the detection and analysis of protein–protein interactions. Proteomics. 7 (16), 2833-2842 (2007).
- Magliery, T. J., et al. Detecting protein-protein interactions with a green fluorescent protein fragment reassembly trap: Scope and mechanism. Journal of the American Chemical Society. 127 (1), 146-157 (2005).
- Xing, S., Wallmeroth, N., Berendzen, K. W., Grefen, C. Techniques for the analysis of protein-protein interactions in vivo. Plant Physiology. 171 (2), 727-758 (2016).
- Nguyen, T. N., Goodrich, J. A. Protein-protein interaction assays: Eliminating false positive interactions. Nature Methods. 3 (2), 135-139 (2006).
- Fernández-Suárez, M., Chen, T. S., Ting, A. Y. Protein-protein interaction detection in vitro and in cells by proximity biotinylation. Journal of the American Chemical Society. 130 (29), 9251-9253 (2008).
- Jiang, M., Niu, C., Cao, J., Ni, D. -. A., Chu, Z. In silico-prediction of protein–protein interactions network about MAPKs and PP2Cs reveals a novel docking site variants in Brachypodium distachyon. Scientific Reports. 8 (1), 15083 (2018).
- Yazawa, M., Sadaghiani, A. M., Hsueh, B., Dolmetsch, R. E. Induction of protein-protein interactions in live cells using light. Nature Biotechnology. 27 (10), 941-945 (2009).
- Wang, W., Goodman, M. T. Antioxidant property of dietary phenolic agents in a human LDL-oxidation ex vivo model: Interaction of protein binding activity. Nutrition Research. 19 (2), 191-202 (1999).
- Xing, S., Wallmeroth, N., Berendzen, K. W., Grefen, C. Techniques for the analysis of protein-protein interactions in vivo. Plant Physiology. 171 (2), 727-758 (2016).
- Qvit, N., Disatnik, M. -. H., Sho, E., Mochly-Rosen, D. Selective phosphorylation inhibitor of delta protein kinase C–Pyruvate dehydrogenase kinase protein–protein interactions: Application for myocardial injury in vivo. Journal of the American Chemical Society. 138 (24), 7626-7635 (2016).
- Alam, M. N., Bristi, N. J., Rafiquzzaman, M. Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharmaceutical Journal. 21 (2), 143-152 (2013).
- Paulmurugan, R., Gambhir, S. S. Novel fusion protein approach for efficient high-throughput screening of small molecule–mediating protein-protein interactions in cells and living animals. Ricerca sul cancro. 65 (16), 7413-7420 (2005).
- Boute, N., Jockers, R., Issad, T. The use of resonance energy transfer in high-throughput screening: BRET versus FRET. Trends in Pharmacological Sciences. 23 (8), 351-354 (2002).
- Deriziotis, P., Graham, S. A., Estruch, S. B., Fisher, S. E. Investigating protein-protein interactions in live cells using bioluminescence resonance energy transfer. Journal of Visualized Experiments: JoVE. (87), e51438 (2014).
- Ekins, S., Mestres, J., Testa, B. In silico pharmacology for drug discovery: Methods for virtual ligand screening and profiling. British Journal of Pharmacology. 152 (1), 9-20 (2007).
- Valerio, L. G. Application of advanced in silico methods for predictive modeling and information integration. Expert Opinion on Drug Metabolism & Toxicology. 8 (4), 395-398 (2012).
- Piehler, J. New methodologies for measuring protein interactions in vivo and in vitro. Current Opinion in Structural Biology. 15 (1), 4-14 (2005).
- Ideker, T., Sharan, R. Protein networks in disease. Genome Research. 18 (4), 644-652 (2008).
- Lu, H., et al. Recent advances in the development of protein–protein interactions modulators: mechanisms and clinical trials. Signal Transduction and Targeted Therapy. 5 (1), 213 (2020).
- Jarosz, D. Hsp90: A global regulator of the genotype-to-phenotype map in cancers. Advances in Cancer Research. 129, 225-247 (2016).
- Johnson, V. A., Singh, E. K., Nazarova, L. A., Alexander, L. D., McAlpine, S. R. Macrocyclic inhibitors of Hsp90. Current Topics in Medicinal Chemistry. 10 (14), 1380-1402 (2010).
- Mahalingam, D., et al. Targeting HSP90 for cancer therapy. British Journal of Cancer. 100 (10), 1523-1529 (2009).
- Stewart, A., Fisher, R. A. Co-Immunoprecipitation: Isolation of protein signaling complexes from native tissues. Methods in Cell Biology. 112, 33-54 (2012).
- Pierce, M. M., Raman, C. S., Nall, B. T. Isothermal titration calorimetry of protein-protein interactions. Methods. 19 (2), 213-221 (1999).
- Paketurytė, V., et al. Inhibitor binding to carbonic anhydrases by isothermal titration calorimetry. Carbonic Anhydrase as Drug Target. , 79-95 (2019).
- Grote, J., Dankbar, N., Gedig, E., Koenig, S. Surface plasmon resonance/mass spectrometry interface. Analytical Chemistry. 77 (4), 1157-1162 (2005).
- Kumaraswamy, S., Tobias, R. Label-free kinetic analysis of an antibody–antigen interaction using biolayer interferometry. Methods in Molecular Biology. , 165-182 (2015).
- Wallner, J., Lhota, G., Jeschek, D., Mader, A., Vorauer-Uhl, K. Application of bio-layer interferometry for the analysis of protein/liposome interactions. Journal of Pharmaceutical and Biomedical Analysis. 72, 150-154 (2013).
- Singh, A. N., Ramadan, K., Singh, S. Experimental methods to study the kinetics of protein–protein interactions. Advances in Protein Molecular and Structural Biology Methods. , 115-124 (2022).
- Frenzel, D., Willbold, D. Kinetic titration series with biolayer interferometry. PLoS One. 9 (9), 106882 (2014).
- Vu, C. -. A., Chen, W. -. Y. Field-effect transistor biosensors for biomedical applications: Recent advances and future prospects. Sensors. 19 (19), 4214 (2019).
- Bergveld, P. A critical evaluation of direct electrical protein detection methods. Biosensors & Bioelectronics. 6 (1), 55-72 (1991).
- Lowe, B. M., Sun, K., Zeimpekis, I., Skylaris, C. K., Green, N. G. Field-effect sensors – from pH sensing to biosensing: sensitivity enhancement using streptavidin–biotin as a model system. The Analyst. 142 (22), 4173-4200 (2017).
- Goldsmith, B. R., et al. Digital biosensing by foundry-fabricated graphene sensors. Scientific Reports. 9 (1), 434 (2019).
- Afsahi, S., et al. Novel graphene-based biosensor for early detection of Zika virus infection. Biosensors and Bioelectronics. 100, 85-88 (2018).
- Afsahi, S. J., et al. Towards novel graphene-enabled diagnostic assays with improved signal-to-noise ratio. MRS Advances. 2 (60), 3733-3739 (2017).
- Roe, S. M., et al. The mechanism of Hsp90 regulation by the protein kinase-specific cochaperone p50cdc37. Cell. 116 (1), 87-98 (2004).
- Gaiser, A. M., Kretzschmar, A., Richter, K. Cdc37-Hsp90 complexes are responsive to nucleotide-induced conformational changes and binding of further cofactors. The Journal of Biological Chemistry. 285 (52), 40921-40932 (2010).
- Popescu, A. I., Găzdaru, D. M., Chilom, C. G., Bacalum, M. Biophysical interactions: Their paramount importance for life. Romanian Reports in Physics. 65 (3), 1063-1077 (2013).
- Surya, S., Abhilash, J., Geethanandan, K., Sadasivan, C., Haridas, M. A profile of protein-protein interaction: Crystal structure of a lectin-lectin complex. International Journal of Biological Macromolecules. 87, 529-536 (2016).
- Velazquez-Campoy, A., Freire, E. ITC in the post-genomic era…? Priceless. Biophysical Chemistry. 115 (23), 115-124 (2005).
- Concepcion, J., et al. Label-free detection of biomolecular interactions using bioLayer interferometry for kinetic characterization. Combinatorial Chemistry & High Throughput Screening. 12 (8), 791-800 (2009).
- Helmerhorst, E., Chandler, D. J., Nussio, M., Mamotte, C. D. Real-time and label-free bio-sensing of molecular interactions by surface plasmon resonance: A laboratory medicine perspective. The Clinical Biochemist. Reviews. 33 (4), 161-173 (2012).
- Jacob, N. T., et al. Synthetic molecules for disruption of the MYC protein-protein interface. Bioorganic & Medicinal Chemistry. 26 (14), 4234-4239 (2018).

