Imaging-Guided Bioreactor for Generating Bioengineered Airway Tissue
Summary
The protocol describes an imaging-enabled bioreactor that allows the selective removal of the endogenous epithelium from the rat trachea and homogenous distribution of exogenous cells on the lumen surface, followed by long-term in vitro culture of the cell-tissue construct.
Abstract
Repeated injury to airway tissue can impair lung function and cause chronic lung disease, such as chronic obstructive pulmonary disease. Advances in regenerative medicine and bioreactor technologies offer opportunities to produce lab-grown functional tissue and organ constructs that can be used to screen drugs, model disease, and engineer tissue replacements. Here, a miniaturized bioreactor coupled with an imaging modality that allows in situ visualization of the inner lumen of explanted rat trachea during in vitro tissue manipulation and culture is described. Using this bioreactor, the protocol demonstrates imaging-guided selective removal of endogenous cellular components while preserving the intrinsic biochemical features and ultrastructure of the airway tissue matrix. Furthermore, the delivery, uniform distribution, and subsequent prolonged culture of exogenous cells on the decellularized airway lumen with optical monitoring in situ are shown. The results highlight that the imaging-guided bioreactor can potentially be used to facilitate the generation of functional in vitro airway tissues.
Introduction
The luminal surface of the respiratory tract is lined by a layer of epithelium that mainly consists of multi-ciliated, club, goblet, and basal stem cells1,2. The epithelial layer serves as a primary defense mechanism of the lung, acting as a biophysical barrier that protects the underlying airway tissue against inhaled pathogens, particulates, or chemical gases. It protects the airway tissue via multiple mechanisms, including intercellular tight junction formation, mucociliary clearance, and antimicrobial and antioxidant secretion3,4. The defective airway epithelium is associated with devastating respiratory diseases, such as chronic obstructive pulmonary disease (COPD)5, primary ciliary dyskinesia (PCD)6, and cystic fibrosis (CF)7.
Advances in lung-on-chip (LOC) technology represent an opportunity to study human lung development, model various lung diseases, and develop new therapeutic materials in tightly regulated in vitro environments. For example, airway epithelium and endothelium can be cultured on opposite sides of a thin, porous membrane to mimic the gas exchanging lung tissue, allowing faithful disease modeling and drug testing8. Similarly, in vitro disease models have been created to model airway diseases in vitro, such as COPD9 and cystic fibrosis10. However, a major challenge of LOC devices is recapitulating the complex three-dimensional (3D) architecture of the lung tissue and dynamic cell-tissue matrix interactions in vitro11.
Recently, innovative tissue engineering methodologies have been developed that allow manipulation of ex vivo lung tissues12. Using these methodologies, denuded allogenic or xenogeneic tissue grafts can be prepared by removing the endogenous cells from the lung tissue via chemical, physical, and mechanical treatments13. In addition, the preserved native tissue extracellular matrix (ECM) in the decellularized lung scaffolds provide the physio-mimetic structural, biochemical, and biomechanical cues for implanted cells to attach, proliferate, and differentiate14,15.
Here, an imaging-guided bioreactor system created by combining LOC and tissue engineering technologies to allow in vitro tissue manipulation and culture of explanted rat tracheal tissues is reported. Using this airway tissue bioreactor, the protocol demonstrates selective removal of the endogenous epithelial cells without disrupting the underlying subepithelial cellular and biochemical components of the airway tissue. We next show the homogenous distribution and instantaneous deposition of the newly seeded exogenous cells, such as mesenchymal stem cells (MSCs), on the denuded airway lumen by instilling the cell-loaded collagen I pre-gel solution. In addition, by using the micro-optical imaging device integrated into the bioreactor, the visualization of the trachea lumen during epithelium removal and endogenous cell delivery is also done. Further, it is shown that the trachea and newly implanted cells can be cultured in the bioreactor without noticeable cell death and tissue degradation for 4 days. We envision that the imaging-enabled bioreactor platform, the thin film-based de-epithelialization technique, and the cell delivery method used in this study can be useful for generating airway tissues for in vitro disease modeling and drug screening.
The bioreactor includes a rectangular chamber connected to a programmable syringe pump, perfusion pump, and ventilator for culturing isolated rat trachea. The bioreactor features inlets and outlets connected to the trachea or the tissue culture chamber to separately supply reagents (e.g., culture media) to the internal and external spaces of the trachea (Figure 1). A custom-built imaging system can be used to visualize the interior of the in vitro-cultured rat trachea at the cellular level (Figure 2). The endogenous epithelium of the trachea is removed via the instillation of a detergent-based decellularization solution followed by vibration-assisted airway washing (Figure 3). Hydrogel solution, such as type I collagen, is used as a delivery vehicle for seeding exogenous cells uniformly and instantaneously across the denuded trachea lumen (Figure 4). All the materials used to construct the bioreactor and conduct the experiments are provided in the Table of Materials.
Protocol
The animal tissue protocol below has been approved by the animal welfare guideline and regulations of the Institute for Animal Care and Use Committee (IACUC) at Stevens Institute of Technology, and it complies with the National Institutes of Health (NIH) guidelines for the use of experimental animals.
1. Design and construction of imaging-guided rat trachea bioreactor
- Designing and fabrication of rat trachea bioreactor
- Create a computer-aided design (CAD) model of the bioreactor chamber with relevant design, such as inlets, outlets, and tissue culture chamber, using CAD generator software. For this study, use the geometry and dimensions presented in Figure 1A-C. The tutorial of CAD generator software can be found in16,17.
- Export the generated CAD model to a computer numerical control (CNC) controller software and cut the polytetrafluoroethylene (PTFE) plastic using a CNC machine to create the bioreactor chamber. The tutorial of the CNC controller software can be found in18.
NOTE: In addition to PTFE, other plastic materials, such as ultra-high molecular weight polyethylene (UHMWPE) and polyetherimide can be used to fabricate the culture chamber. - Sterilize all bioreactor components, such as Luer adapters and screws, to avoid contamination of the tissues and cells cultured in the device. Assemble all bioreactor components to the main tissue culture chamber in a clean environment (Figure 1A).
- Construction of gradient index (GRIN) lens-based imaging device
- To create the in situ imaging device, insert a tube lens into a stackable lens tube and secure it using a retaining ring. Mount the lens-tube assembly onto a scientific CMOS camera via a C-mount adapter.
- Use software, such as Micro-Manager, to operate the camera and acquire photos and videos. Aim an object located at a long distance (e.g., 10 m from the camera) and adjust the distance between the tube lens and imaging sensor of the camera until a focused image of the object is formed on the computer screen by the imaging software being used.
- Mount a filter lens on a dual-edge super-resolution dichroic mirror and a laser to the device using optical cage system components, including assembly rod, threaded cage plate, and cage cube.
- Connect the objective lens (20x) to the device. Mount a GRIN lens (diameter = 500 µm) at the distal end of the lens tube via the XY translator. Adjust the distance between the GRIN lens and the objective lens to form focused microscopic images (Figure 2A,B).
2. Isolation of the rat trachea
- Sanitize the surgical area and sterilize the surgical instruments using an autoclave at 121 °C for 30 min prior to the surgery.
- Place a rat in the induction chamber and deliver 2.5% of isoflurane for 15 min using a small vaporizer to anesthetize the animal. Assess the anesthesia depth by pedal reflex. To do this, firmly pinch the toe and confirm that the animal does not respond to the toe pinch.
- After anesthesia, remove the isoflurane from the chamber and administer 1 mL of 1,000 unit/mL heparin through the lateral tail vein to prevent blood clotting in the pulmonary vasculature.
- To euthanize the animal, expose the animal to 5% isoflurane for an additional 15-20 min. Remove the animal from the induction chamber and place it on the surgical board in a face-up supine position.
- Fix the rat on the surgical board by immobilizing the legs and tail using adhesive tape. Next, sterilize the rat's neck and chest regions using 70% isopropyl alcohol (IPA). Open the abdominal cavity by making a 3-4 cm incision using scissors in the skin.
NOTE: Be careful not to cut the skin deeply by pointing the tips of the scissors upward. - Transect the inferior vena cava (IVC) using the scissors and confirm the euthanasia by exsanguination.
- Perform the tracheotomy by making an incision using scissors in the midline of the neck and exposing the trachea. Perform midline thoracotomy by making an incision in the chest wall and cutting between the ribs to reach the trachea end connected to the lung. Next, use a bicep and scissors to cut both ends of the trachea and isolate the trachea.
- After isolation, rinse the trachea using 20 mL of 1x phosphate-buffered saline (1x PBS). Transfer the trachea to the bioreactor using sterilized biceps. Secure the two ends of the trachea to the Luer connector using a 4-0 suture thread.
- Deliver 5 mL of culture medium to the bioreactor chamber using the programmable syringe pump through the tubing connected to the bioreactor chamber at a flow rate of 5 mL/min.
NOTE: The culture medium is composed of Dulbecco's modified Eagle's medium (DMEM), recombinant human FGF-basic (0.1 ng/mL), fetal bovine serum (FBS, 10%), and antibiotic-antimycotic (1%). - Tightly close the bioreactor lid with the acrylic plastic lid and screws. Close the bioreactor chamber tubing connections with the male/female Luer plugs to prevent the flow of the culture medium in the tubing.
3. Imaging-guided removal of the trachea epithelium
- To visualize the trachea lumen either in bright-field or fluorescent, place the bioreactor on the imaging stage (Figure 3A).
- Prepare carboxyfluorescein succinimidyl ester (CFSE) solution (concentration: 100 μM) in the CFSE cell labeling kit by diluting the CFSE solution with 1x PBS.
NOTE: The concentration of the original CFSE solution is 10 mM (in dimethyl sulfoxide (DMSO)). - Infuse 500 µL of the CFSE solution through the trachea via the syringe pump through the tubing connected to the trachea cannula at a flow rate of 5 mL/min. Stop the pump when the CFSE solution fills the trachea. Wait for 10 min, and then wash the trachea lumen by infusion of 10 mL of 1x PBS using the syringe pump to remove the residual unincorporated CFSE reagent.
- Insert the distal imaging end of the GRIN lens into the trachea via the Luer connector attached to one end of the trachea. Then, gently move the GRIN lens inside the trachea until the trachea surface is focused and take photos and videos in bright-field or fluorescence at 20x magnification.
- For capturing bright-field images in the Micro-Manager software follow the steps below.
- Introduce white light through the cage cube to illuminate the trachea lumen. Click on the Live icon to show the luminal surface of the trachea in real-time. Use Imaging Setting > Exposure (ms) to change exposure time to the desired value. In this study, the exposure time used was in the range of 10 ms and 50 ms.
- To adjust the contrast and brightness of images, use the Histogram and Intensity Scaling window to move black and white arrows at the endpoint of the interactive histogram display. Alternatively, use the Auto Stretch option that allows the software to adjust the brightness and contrast automatically to optimal levels.
- Click on the Snap icon to freeze the image. Then, use Setting > Export Images as Displaced to save the images in the desired format. Alternatively, use Setting > ImageJ to directly export the image to the ImageJ software and save it.
- To obtain photos and videos in fluorescent mode, illuminate the trachea lumen with CFSE-specific laser light (CFSE: excitation wavelength: 488 nm, emission wavelength: 515 nm) through the cage cube. Follow the steps 3.5.2-3.5.3 to acquire the images. After taking photos and videos, gently remove the GRIN lens from the trachea.
- Perform de-epithelialization as described below.
- Prepare 2% sodium dodecyl sulfate (SDS) decellularization solution in distilled (DI) water. Instill 50 µL of SDS through the trachea at a flow rate of 6.3 mL/min, by using the syringe pump through the trachea cannula to generate a thin film of the detergent solution on the trachea lumen.
- Close the bioreactor's tubing connections using male/female Luer plugs and transfer the bioreactor to an incubator. Allow the SDS to dwell within the trachea for 10 min at 37 °C. Instill another 50 µL of SDS solution through the trachea and incubate for 10 min.
- Remove lysed epithelium and SDS by irrigating the trachea lumen with 500 µL of 1x PBS via syringe pump at a flow rate of 10 mL/min for 3x. Place the bioreactor on a shaker and mechanically vibrate the bioreactor at 20 Hz frequency and displacement amplitude of approximately 0.3 mm to physically promote detachment of SDS-treated epithelial cells from the trachea lumen.
NOTE: In this study, the shaker was custom-built by assembling a subwoofer speaker, a subwoofer plate amplifier, and an accelerometer. A sinusoidal waveform was generated by a computer and fed into the shaker via the amplifier, while the response of the shaker was monitored via the accelerometer (Figure 3B). In addition, conventional shakers, such as electromagnetic and inertia shakers, can be used to promote the detachment of the cells. To do this, set the frequency and acceleration of the shaker to 20 Hz and 0.5 g, respectively (equivalent to 0.3 mm displacement amplitude). - Instill 500 µL of 1x PBS twice through the trachea lumen to remove residual SDS and cell debris while the trachea is mechanically vibrated. Following the epithelium removal procedure, evaluate the clearance of the epithelial layer by measuring the intensity of CFSE using the GRIN lens imaging device as in step 3.6 (Figure 3C).
4. Trachea tissue preparation for further analyses
- To confirm the epithelium removal, perform further tissue analyses, such as hematoxylin and eosin (H&E) staining, trichrome, pentachrome, and immunostaining. To do this, remove the trachea from the bioreactor, and fix it in 30 mL of 4% neutral buffered paraformaldehyde solution in 1x PBS (pH = 7.4) at 4 °C overnight.
- Dehydrate and embed the fixed trachea tissue by following the steps below.
- After fixation, wash the trachea tissue with 10 mL of 1x PBS, transfer the trachea to 30 mL of 70% alcohol, and keep it at 4 °C overnight. Cut the trachea into small cylindrical sections (~5 mm) using a sharp blade and insert the tissue sections in the tissue embedding cassettes (length x width x height: 4 cm x 2.5 cm x 0.5 cm). Keep two trachea sections in each cassette.
- Dehydrate the sections in a series of isopropyl alcohol (IPA) solutions – 85%, 90%, 95%, 100% – for 1 h in 30 mL of each solution. Remove the previous solution before adding the next solution.
- Upon completion, submerge the cassettes in 30 mL of clearing agent (e.g., xylene) for 2 h to displace the IPA solution from the tissue sections completely. Perform this step in a fume hood with proper ventilation.
- Submerge the cassettes in paraffin for 2 h, and then embed them in paraffin at 4 °C overnight. Next, cut the paraffin-embedded tissues into thin sections (5-8 μm) using a microtome device for the H&E, trichrome, pentachrome, and immunostaining.
- To prepare the tissues for scanning electron microscopy (SEM) analysis, fix the trachea in 30 mL of 2.5% glutaraldehyde solution in 1x PBS (pH = 7.4) at 4 °C overnight. Then, dehydrate the fixed trachea tissue for SEM by following the steps below.
- After fixation, rinse the trachea tissue with 10 mL of 1x PBS. Cut the trachea tissue longitudinally into small semi-cylinder sections (length: ~5 mm) using scissors and insert the tissue sections in the cassettes.
- Dehydrate the sections in a series of IPA solutions – 35%, 50%, 70%, 85%, 95%, and 100% – for 10 min in 30 mL of each solution. Remove the previous solution before adding the next solution.
- Perform hexamethyldisilazane (HMDS)-based drying method by submerging the tissues in the following solutions: 100% IPA: HMDS (2:1; v/v) for 10 min, followed by 100% IPA: HMDS (1:2; v/v) for 10 min, and finally 100% HMDS for overnight.
NOTE: HMDS is toxic. Work under a fume hood during all drying steps. - Remove the tissues from the HMDS solution and allow them to dry under the fume hood for 1 h. Mount the section on aluminum pin stubs using a carbon double-sided conductive tape or silver conductive paste for SEM imaging.
5. Homogeneous distribution of exogenous cells onto the denuded tracheal lumen
- Prepare a de-epithelialized rat trachea using the protocol in step 3. Thaw frozen mesenchymal stem cells (MSCs) for 30 s in a 37 °C water bath.
NOTE: In this study, we used mesenchymal stem cells (MSCs) as a model cell to show the distribution of exogenous cells onto the de-epithelialized trachea. Ideally, primary airway epithelial cells, basal cells, or induced-human pluripotent cells (iPSCs) can be used for epithelium regeneration purposes. - Count the cells with a hemocytometer and prepare a cell solution with a concentration of 5 x 106 cells/mL. Label the cells fluorescently by incubating the cells with 2 mL of CFSE solution (concentration: 100 µM) at room temperature for 15 min. Rinse the cells with 5 mL of 1x PBS for 3x and resuspend the cells in fresh culture medium at a final concentration of 3 x 107 cells/mL.
- Prepare hydrogel pre-gel solution as a vehicle for cell delivery. For this study, use collagen I as a delivery vehicle for MSCs cells and follow the manufacturer's instructions to prepare the pre-gel. For example, to obtain 3.6 mg/mL collagen pre-gel, mix one part of the chilled neutralization solution with nine parts of the rat tail collagen in a 1.5 mL sterile tube. Then, gently pipet the mixture up and down for adequate mixing.
NOTE: Other biocompatible hydrogels may be used instead of collagen I according to the study and user needs. - Once the hydrogel solution is prepared, add the cells quickly to the solution with desired concentration (e.g., 5 x 106 cells/mL). To obtain a uniform cell-hydrogel mixture, mix the cells and gel solution by gently pipetting with a micropipette.
- Attach one end of the trachea within the bioreactor to a programmable syringe pump through a Luer connector. Deliver 5 mL of fresh culture medium at 37 °C into the bioreactor chamber to cover the exterior surface of the trachea using the syringe pump at a flow rate of 5 mL/min.
- Administer a 10 µL bolus of the cell-hydrogel mixture into the de-epithelialized trachea within the bioreactor using the syringe pump at a flow rate of 5 mL/min to generate a cell-hydrogel layer on the trachea lumen (Figure 4).
- After cell injection, place the bioreactor in a sterile cell culture incubator at 37 °C and 5% CO2 for gelation. For collagen I, the gelation occurs in 30 min.
- To visualize the distribution of implanted cells, sterilize the GRIN lens by wiping with 70% IPA or ethanol and place the bioreactor on the imaging stage. Take photos and videos in both bright-field and fluorescent modes as needed.
- After 30 min of cell seeding, infuse 1 mL of culture medium into the trachea lumen using a syringe pump at a flow rate of 1 mL/min.
- Culture the cell-seeded trachea within the bioreactor in an incubator at 37 °C for the desired time. For long-term culture, refresh the culture medium in the trachea lumen and the bioreactor chamber every 24 h. During the cell culture, keep the media inside the lumen static and change it every 24 h, while the media outside the trachea is continuously perfused via a unidirectional flow at 5 mL/min.
- After culturing the cells for a certain time period (e.g., 1 and 4 days in this study), remove the trachea from the bioreactor. Use scissors to cut the trachea longitudinally into two semi-cylinder sections (i.e., upper and lower sections) on days 1 and 4 of in vitro culture to expose the inner surfaces for monitoring cell growth and calculating cell density. Use a conventional fluorescent microscope to visualize the cells on the inner surfaces.
- To acquire the images using Micro-Manager software, follow steps 3.5 and 3.6. Use a fluorescein isothiocyanate (FITC) filter to visualize the CFSE-labeled cells in fluorescence mode. Analyze the images taken by the fluorescent microscope and calculate cell densities on upper and lower sections using the ImageJ software. To calculate the number of cells and the cell density, follow the steps below.
- Open an image in ImageJ software and convert the image to a binary image (16-bit) using Image > Type > 16-bit. Set the image scale with the scale bar using Analyze > Set Scale.
- Adjust the threshold of the image to highlight the structures of the cells to count. Use Image > Adjust > Threshold. Use Analyze > Analyze Particles to count the number of the cells. Calculate the density of the cells by dividing the number of cells by the surface area of the image.
- To assess the viability after cell seeding, use a cell viability kit. For this study, incubate the trachea tissue and the cells in the bioreactor at 37 °C for 6 h and stain the cells with 4 mM calcein-AM and 2 mM ethidium homodimer-1 in 1 mL of 1x PBS at room temperature for 15 min.
- After rinsing with 1x PBS, visualize the cells using a fluorescent microscope. Count the live and dead cells using the steps described in step 5.12. Calculate cell viability as the percentage of live cells (stained with calcein-AM) per total cells (both live and dead cells).
Representative Results
The GRIN lens-based in situ imaging modality can allow visualization of the tracheal inner lumen in situ (Figure 5A). Using this imaging method, both bright-field and fluorescent images of the native and de-epithelialized tracheas can be obtained (Figure 5B,C). No fluorescent signal was observed from the native trachea prior to CFSE labeling (Figure 5Bii). However, when the tracheal epithelium was labeled with CFSE dye, a uniform fluorescent signal (green) was observed throughout the epithelium (Figure 5Ci). Following de-epithelialization via SDS and vibration-assisted airway irrigation, the intensity of the fluorescent signal was decreased significantly (Figure 5Cii), indicating ablation of the epithelium.
The H&E images of de-epithelialized trachea showed the removal of the pseudostratified epithelium from the trachea lumen and preservation of the cells and ECM microstructure in underlying tissue layers (Figure 6A). Moreover, pentachrome (Figure 6B) and trichrome staining (Figure 6C) confirmed the maintenance of the trachea tissue architecture and the ECM components, such as collagen and proteoglycans. Furthermore, immunofluorescence of epithelial cells (epithelial cell adhesion molecule, EpCAM; Figure 6D) and collagen I (Figure 6E) revealed complete removal of the epithelium and preservation of collagen I within the subepithelial tissue. SEM imaging of native trachea showed that the luminal surface of native rat trachea mainly was populated with multi-ciliated cells and goblet cells. On the other hand, SEM images of de-epithelialized trachea lumen showed that the basement membrane was exposed as indicated by a mesh network of ECM fibers and the absence of epithelial cells (Figure 6F).
Using de-epithelialized rat tracheas and fluorescently labeled cells, we investigated whether incorporating a hydrogel as a cell delivery vehicle could achieve homogeneous cell distribution onto the de-epithelialized tracheal lumen (Figure 7A,B). In this study, the mesenchymal stem cells (MSCs) were used as the model cells for the cell delivery and culture study. As described in the protocol (step 5), we instilled MSC-loaded collagen I pre-gel into a de-epithelialized rat trachea and monitored the distribution of the cells on the tracheal lumen using the GRIN lens imaging system. As a control experiment, we suspended MSCs in the culture medium, infused the cell-loaded culture medium into the trachea, and visually inspected the distribution of the cells. In situ imaging results showed that the fluorescently labeled cells delivered via hydrogel remained adhered more uniformly across the lumen than those seeded via culture medium (Figure 7B). We then tested the cell viability before and after cell delivery to evaluate potential cell damage and death due to shearing stress during cell infusion. The result showed that cell viability was not significantly affected by the cell delivery procedure as over 90% of cells remained viable (Figure 7C).
Furthermore, we cultured the cell-seeded tracheas for 4 days. The cells seeded via both collagen and culture medium (control) proliferated on the de-epithelialized trachea lumen surface as indicated by the increased number of cells expressing CFSE over time in both the upper and lower half of the tracheal lumen (Figure 7D). Notably, in the collagen hydrogel delivery group, the difference in the density of the cells between upper and lower lumens was smaller than that in the control group, indicating cell delivery via hydrogel promotes homogeneous cell distribution throughout the trachea lumen.
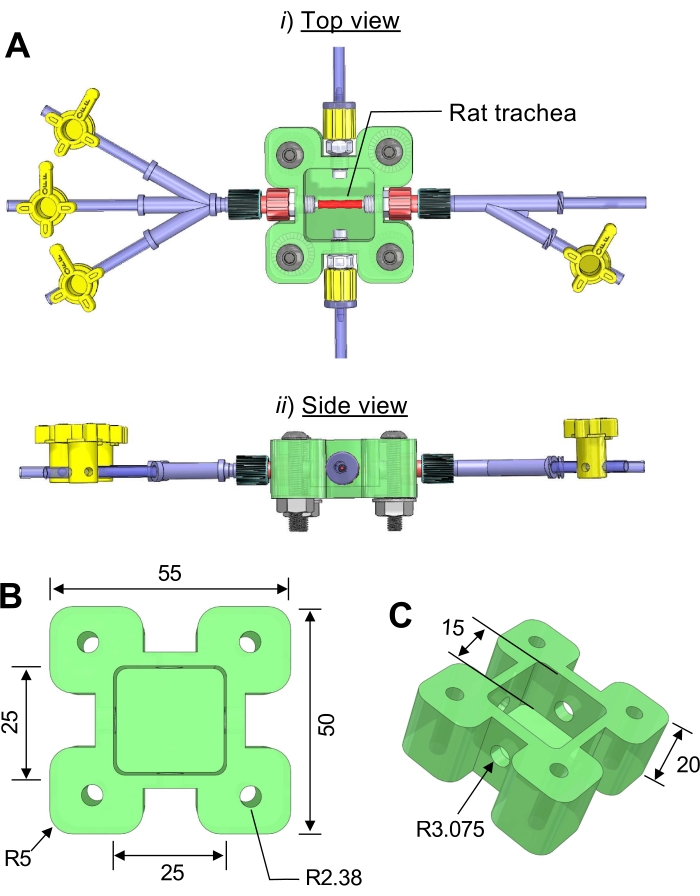
Figure 1: Three-dimensional (3D) computer drawing of the trachea bioreactor. (A) (i) side view, (ii) top view. (B) The dimensions of the bioreactor chamber. (C) A transparent acrylic plastic sheet is cut and attached to the top of the main chamber using screws. Unit = mm; R = radius. Please click here to view a larger version of this figure.
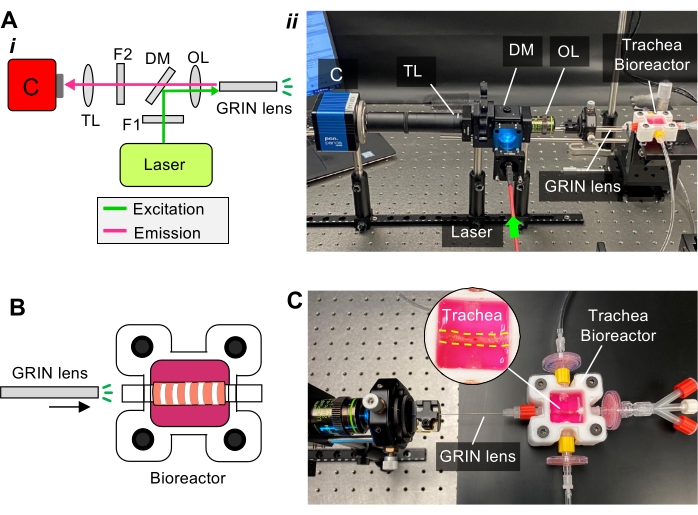
Figure 2: Custom-built micro-fiber imaging for in situ visualization of the rat trachea. (A) (i) Schematic of the imaging setup components of the light path. Abbreviations: C = camera, TL = tube lens, F = filter, DM = dichroic mirror, OL = objective lens; (ii) photograph of the imaging setup components showing the imaging probe (GRIN lens) is inserted in the Luer connector of the bioreactor. (B) Schematic showing that the GRIN lens is integrated with the bioreactor. (C) A photograph showing the imaging probe is used to visualize the trachea inside the bioreactor. Please click here to view a larger version of this figure.
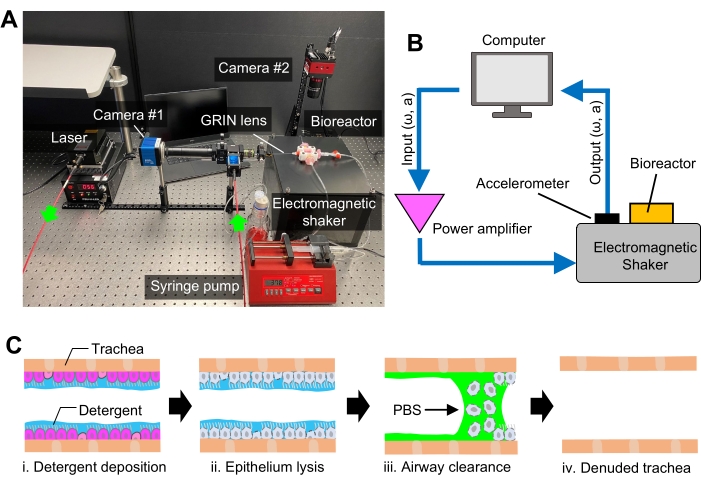
Figure 3: The platform for de-epithelialization of rat trachea. (A) Photograph showing the experimental setup for de-epithelialization and in situ optical fiber imaging. (B) Diagram showing the component and assembly of the custom-built electromagnetic shaker used to facilitate epithelium removal. ω: frequency, a: amplitude (C) Schematic showing the workflow of rat trachea de-epithelialization procedure. PBS = phosphate-buffered saline. Please click here to view a larger version of this figure.
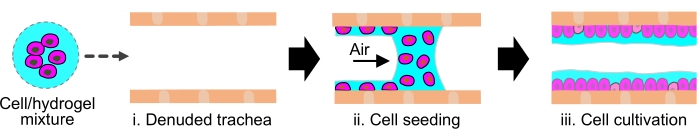
Figure 4: Delivery of exogenous cells into de-epithelialized rat trachea using hydrogel. Schematic showing hydrogel solution used as a delivery vehicle to topically deposit the cells onto the inner lumen of the de-epithelized rat trachea. Please click here to view a larger version of this figure.

Figure 5: In situ imaging of de-epithelialized trachea. (A) Photograph showing fluorescent imaging of the trachea lumen while the CFSE-labeled epithelium is excited with 488 nm blue laser through GRIN lens. (B) (i) Bright-field and (ii) fluorescence images of the trachea lumen before epithelium labeling. (C) Fluorescence images of the (i) native and (ii) de-epithelialized trachea (De-Epi trachea) while the epithelium is labeled with CFSE fluorescent dye. Please click here to view a larger version of this figure.
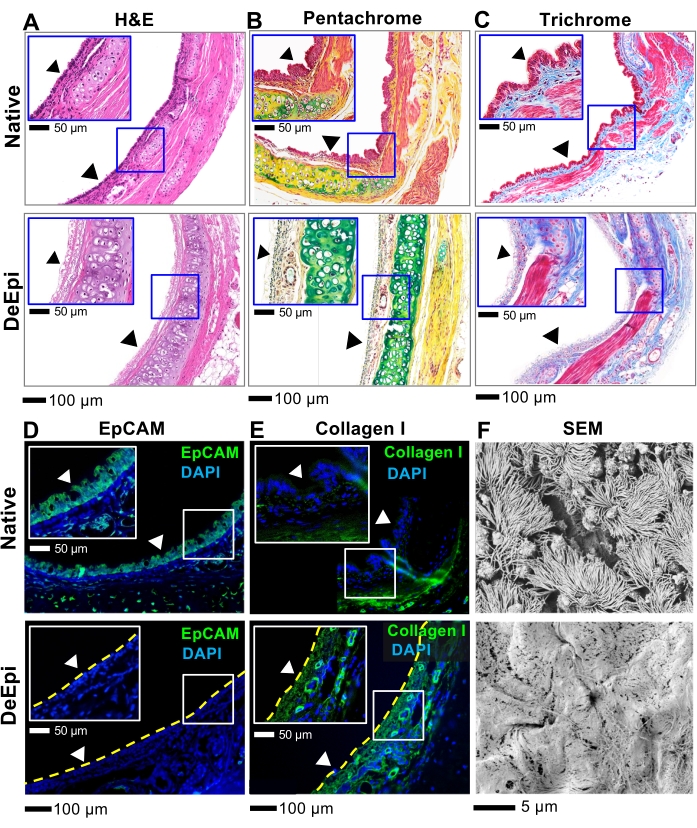
Figure 6: Histology, immunofluorescence, and SEM analyses of native and de-epithelialized rat tracheas. (A) H&E staining. (B) Pentachrome staining where purple is cell cytoplasm, blue is cell nuclei, green is proteoglycans (e.g., mucin), and yellow is collagen fibers. (C) Trichrome staining where pink is cell cytoplasm, dark blue is cell nuclei, and blue is collagen. Fluorescence images of native and de-epithelialized tracheas. (D) EpCAM and (E) collagen I. Arrowheads show the surface of the trachea lumen. (F) SEM micrographs of the trachea lumen. Please click here to view a larger version of this figure.
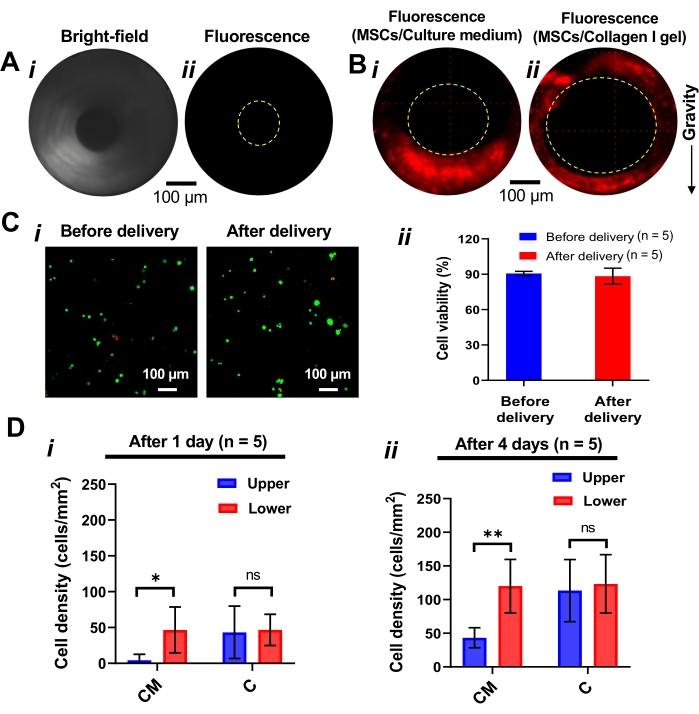
Figure 7: Topical deposition of the exogenous MSCs in the trachea lumen. (A) (i) Bright field and (ii) fluorescence images of the de-epithelialized trachea. (B) Fluorescent images of the de-epithelialized trachea lumen when it is seeded with (i) PBS and (ii) collagen I pre-gel. The homogenous distribution of the cells onto the trachea lumen is achieved when pre-gel collagen is used as a delivery vehicle for cells. (C) (i) Fluorescent images and (ii) quantified viability of the cells before and after 6 h of delivery by pre-gel collagen. n: number of samples. Steps 5.1 and 5.17 of the protocol were followed to assess the viability of the cells. (D) Cell seeding density measured after (i) 1 day and (ii) 4 days after cell seeding and culture. The cell density was calculated by dividing cell numbers by the examined surface area. All values represent mean ± standard deviation. One-way analysis of variance (ANOVA) was used to determine statistically significant differences between groups, with p < 0.05 considered significant. Abbreviations: CM = culture medium, C = collagen. *p < 0.05. **p < 0.01. ns: not significant. No significant difference (ns) between upper and lower surfaces means uniform cell distribution across the circumference of the trachea. Please click here to view a larger version of this figure.
Discussion
In this work, we created an imaging-guided bioreactor that can allow (i) monitoring of the trachea lumen in situ after the cell removal and exogenous cell delivery and (ii) long-term in vitro culture of the cell-seeded trachea tissue. Using this custom-built bioreactor, we demonstrated (i) selective removal of the endogenous epithelial cells from the trachea lumen using detergent and vibration-assisted airway wash and (ii) uniform distribution of exogenous cells onto the luminal surface of the denuded trachea using cell-loaded collagen I pre-gel. Moreover, the GRIN lens-based imaging modality confirmed the removal of the epithelium in situ. Furthermore, with further analyses, including H&E, trichrome, pentachrome, immunofluorescence staining, and SEM, we confirmed the removal of the epithelium and maintenance of the architecture and components of underlying tissue layers. In addition, the GRIN lens-based imaging of newly implanted exogenous MSCs showed the uniform distribution of the cells when collagen I pre-gel was used as a delivery vehicle. Lastly, the implanted cells survived and proliferated on the de-epithelialized trachea, highlighting the efficacy of the bioreactor in the long-term culture of the cell-seeded airway tissues.
The critical step in the de-epithelialization protocol is creating a thin detergent solution layer in the trachea lumen. In current decellularization protocols, multiple cycles of chemicals and enzymes are being used over a long period, reducing the number of collagen fibers, glycosaminoglycans (GAGs), proteoglycans, and chondrocytes, compromising the biological and biomechanical properties of the airway scaffold19,20,21. On the other hand, the ECM provides biochemical and mechanical cues and suitable physical environments for survival, proliferation, and differentiation of implanted exogenous cells22,23. The thin-film deposition method described in this study uses a small amount of detergent (less than 50 μL) for the epithelium ablation while allowing preservation of the underlying tissue layer and subepithelial cellular components. Mechanical vibration of the detergent-exposed tissue demonstrated in this study is an important procedure as it permits detachment and clearance of the lysed cells. The cell debris and partially disrupted cells resulting from exposure of the epithelium to the detergent are challenging to be washed only by flooding the trachea with the washing solution. Therefore, mechanically vibrating the trachea during washing provides sufficient shear forces to clear the cell debris from the tracheal lumen.
In general, the pre-gel-based cell seeding approach can be influenced by delivery speed, the viscosity of the pre-gel (that is directly related to the pre-gel concentration), surface tension, and gelation time. In particular, during pre-gel preparation, it is essential to maintain the pre-gel concentration low enough to transport the cell-suspended pre-gel in the tubing and high enough to maintain the cells on the upper surface of the trachea. Therefore, it is strongly recommended to find the optimum concentration for each pre-gel by testing various concentrations. In addition, practice the delivery technique such as the delivery speed with less precious cells to ensure the successful delivery of the cell-loaded pre-gel. Moreover, while we used MSCs as a cell model to demonstrate (i) the distribution of the cells with pre-gel as a delivery vehicle and (ii) the capability of the bioreactor system for culturing the newly seeded cells on denuded trachea lumen, the use of stem cells (e.g., basal cells) and primary airway epithelial cells in future studies could allow studying cell differentiation and functional epithelial cell regeneration24,25. In addition, future studies can include adding an air-liquid interface (ALI) to the current platform to generate in vivo-like shear stress on seeded cells for the generation of functional epithelium. To add ALI to the current device, a small animal ventilator can be connected to the trachea inlet to ventilate the trachea. Moreover, to achieve the ALI, the thin-film deposition method can be utilized to instill a culture medium liquid plug into the cell-seeded trachea. The thickness of the liquid film can be modulated by changing the liquid plug velocity26,27. The plug can be initiated after the newly seeded cells adhere and engraft firmly onto the trachea lumen.
In addition, in situ GRIN lens-based imaging approach used in this protocol is critical for confirming the efficacy of the de-epithelialization and assessing the quality of distribution of newly seeded cells. Unlike conventional tissue analyses methods, such as histology and immunofluorescence, which require dissecting the samples from the trachea tissue for analysis, the imaging platform developed in our study allows rapid and non-destructive monitoring of the airway lumen during de-epithelialization and cell delivery. To keep the trachea sterilized during in situ imaging, make sure to sterilize the GRIN lens by wiping it with 70% IPA or ethanol before introducing it into the trachea lumen. Moreover, to image the cells using the GRIN lens, the cells (either endogenous tracheal epithelial cells or newly implanted cells) can be labeled with various fluorescent dyes with different excitation/emission wavelengths. To do this, make sure to use the appropriate laser light generator to excite the fluorescently labeled cells and integrate the right filter lenses to the dual-edge super-resolution dichroic mirror.
Despite the novelty and innovativeness of the de-epithelialization, cell delivery, and in situ imaging methods, this study has several limitations. The de-epithelialization and cell delivery methods described in this study are applicable to small airways (diameter: less than 3-4 mm) where the surface tension is dominant. Forming a liquid plug and creating a thin film of the de-epithelialization solution or cell-loaded pre-gel suspension in larger airways, such as swine and human trachea, or bronchi, can be challenging as the surface tension becomes negligible compared with other forces, gravity in particular. Moreover, by using the thin-film deposition approach and modulating time and concentration, we were able to ablate the epithelium with a strong detergent (SDS) and preserve the underlying tissue layer. In future studies, however, milder decellularization detergents, such as 3-[(3-cholamidopropyl)-dimethylammonio]-1-propanesulfonate (CHAPS)28, or non-detergent solution, such as sodium hydroxide (NaOH)29, can be tested for the preservation of ECM components and support of newly implanted cells. Furthermore, the fluorescence intensity of CFSE-labeled cells reduces over time due to the division of the cells. In our study, the fluorescent images of MSCs cultured on the de-epithelialized tracheas for 4 days showed a substantial decrease in their fluorescence intensity. For prolonged monitoring and tracking of exogenous cells seeded onto the tissue scaffolds, different cell labeling methods that can allow long-term or permanent fluorescent labeling of the cells would be necessary.
We envision that the imaging-guided bioreactor platform and cell removal and delivery protocols described in this report can allow the creation of de-epithelialized airway tissue scaffolds that can be used to generate the bioengineered airway tissues. Such in vitro airways can offer high-fidelity in vitro modeling of human airway diseases and drug screening. For example, to establish functional airway epithelium, airway basal stem cells can be first implanted onto the de-epithelialized airway tissue30. Then, the air-liquid interface (ALI), which is critical for the differentiation of basal stem cells into various epithelial cells, can be generated within the trachea. Moreover, the in situ imaging system can be modified and used to fluorescently visualize the respiratory tract of patients with lung airway disorders. To be used clinically to evaluate airway tissues in situ, the rigid GRIN lens can be replaced with flexible optical fiber imaging probes to navigate the airways. Furthermore, the ability to homogeneously distribute the cells inside the respiratory tract using hydrogel-based cell seeding can provide an unprecedented opportunity to replace damaged or injured epithelium in patients. In particular, the cell replacement approach developed and used in this study can accelerate cell-based treatment for treating respiratory disorders, such as cystic fibrosis and primary ciliary dyskinesia, and repairing donor lungs that are refused for transplantation due to severely injured airway tissues31,32,33,34.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This research has been supported in part by the American Thoracic Society Foundation Research Program, the New Jersey Health Foundation, and the National Science Foundation (CAREER Award 2143620) to J.K.; and the National Institutes of Health (P41 EB027062) to G.V.N.
Materials
| 1× PBS | Gibco, Thermo Fisher Scientific | 10-010-031 | |
| 3-port connector | World Precision Instruments | 14048-20 | |
| 4-port connector | World Precision Instruments | 14047-10 | |
| Accelerometer | STMicroelectronics | IIS3DWBTR | |
| Achromatic doublet | Thorlabs | AC254-150-A-ML | |
| Aluminum pin stub | TED PELLA | 16111 | |
| Antibiotic-antimycotic | Thermo Fisher Scientific | 15240062 | |
| Assembly rod | Thorlabs | ER1 | |
| Button head screws | McMaster-Carr | 91255A274 | |
| Cage cube | Thorlabs | C4W | |
| Carbon double-sided conductive tape | TED PELLA | 16073 | |
| CFSE labelling kit | Abcam | ab113853 | |
| Citrisolv (clearing agent) | Decon | 1061 | |
| C-mount adapter | Thorlabs | SM1A9 | |
| Collagen I | Advanced BioMatrix | 5153 | |
| Conductive liquid silver paint | TED PELLA | 16034 | |
| Dichroic mirror | Semrock | DI03-R488 | Reflected laser wavelengths: 473.0 +- 2 nm 488.0 +3/-2 nm |
| Dulbecco's modified Eagle’s medium | Gibco, Thermo Fisher Scientific | 11965118 | |
| Female luer bulkhead to hose barb adapter | Cole-Parmer | EW-45501-30 | |
| Female luer to tubing barb | Cole-Parmer | EW-45508-03 | |
| Female to male luer connector | Cole-Parmer | ZY-45508-80 | |
| Fetal bovine serum | Gibco, Thermo Fisher Scientific | 10082147 | |
| Filter lens | Chroma Technology Corp | ET535/50m | |
| Fluorescent microscope | Nikon | Eclipse E1000 – D | |
| Fusion 360 | Autodesk | ||
| Hex nut | McMaster-Carr | 91813A160 | |
| Hexamethyldisilazane (HMDS) | Fisher Scientifc | AC120585000 | |
| Imaging fiber | SELFOC, NSG group | GRIN lens | |
| Laser | Opto Engine | MDL-D-488-150mW | |
| Lens tubes | Thorlabs | SM1L40 | |
| LIVE/DEAD Viability/Cytotoxicity Kit (Invitrogen) | Thermo Fisher Scientific | L3224 | |
| MACH 3 CNC Control Software | Newfangled Solutions | ||
| Objective lens | Olympus | UCPLFLN20X | |
| Peristaltic Pump | Cole Parmer | L/S standard digital pump system | |
| Recombinant human FGF-basic | PeproTech | 100-18B | |
| Retaining ring | Thorlabs | SM1RR | |
| Scientific CMOS camera | PCO Panda | PCO Panda 4.2 | |
| Sodium dodecyl sulfate | VWR | 97064-472 | |
| Solidworks (2019) | Dassault Systèmes | ||
| Stackable lens tube | Thorlabs | SM1L10 | |
| Subwoofer plate amplifier | Dayton Audio | SPA250DSP | |
| Subwoofer speaker | Dayton Audio | RSS21OHO-4 | Diaphragm diameter: 21 cm |
| Syringe Pump | World Precision Instruments | AL-4000 | |
| Threaded cage plate | Thorlabs | CP33 | |
| Threaded luer adapter | Cole-Parmer | EW-45513-81 | |
| Tube lens | Thorlabs | AC254-150-A-ML | |
| Tygon Tubing | Cole-Parmer | 13-200-110 | |
| XY Translator | Thorlabs | CXY1 |
Riferimenti
- Rackley, C. R., Stripp, B. R. Building and maintaining the epithelium of the lung. The Journal of Clinical Investigation. 122 (8), 2724-2730 (2012).
- Rayner, R. E., Makena, P., Prasad, G. L., Cormet-Boyaka, E. Optimization of Normal Human Bronchial Epithelial (NHBE) cell 3D cultures for in vitro lung model studies. Scientific Reports. 9 (1), 500 (2019).
- Gohy, S., Hupin, C., Ladjemi, M. Z., Hox, V., Pilette, C. Key role of the epithelium in chronic upper airways diseases. Clinical and Experimental Allergy. 50 (2), 135-146 (2020).
- Ganesan, S., Comstock, A. T., Sajjan, U. S. Barrier function of airway tract epithelium. Tissue Barriers. 1 (4), 24997 (2013).
- De Rose, V., Molloy, K., Gohy, S., Pilette, C., Greene, C. M. Airway epithelium dysfunction in cystic fibrosis and COPD. Mediators of Inflammation. 2018, 1309746 (2018).
- Horani, A., Ferkol, T. W. Advances in the genetics of primary ciliary dyskinesia: Clinical implications. Chest. 154 (3), 645-652 (2018).
- Berical, A., Lee, R. E., Randell, S. H., Hawkins, F. Challenges facing airway epithelial cell-based therapy for cystic fibrosis. Frontiers in Pharmacology. 10, 74 (2019).
- Shrestha, J., et al. Lung-on-a-chip: the future of respiratory disease models and pharmacological studies. Critical Reviews in Biotechnology. 40 (2), 213-230 (2020).
- Benam, K. H., et al. Small airway-on-a-chip enables analysis of human lung inflammation and drug responses in vitro. Nature Methods. 13 (2), 151-157 (2016).
- Plebani, R., et al. Modeling pulmonary cystic fibrosis in a human lung airway-on-a-chip. Journal of Cystic Fibrosis. , (2021).
- Griffith, L. G., Swartz, M. A. Capturing complex 3D tissue physiology in vitro. Nature Reviews Molecular Cell Biology. 7 (3), 211-224 (2006).
- Gilpin, S. E., Wagner, D. E. Acellular human lung scaffolds to model lung disease and tissue regeneration. European Respiratory Review. 27 (148), 180021 (2018).
- Badylak, S. F., Taylor, D., Uygun, K. Whole-organ tissue engineering: decellularization and recellularization of three-dimensional matrix scaffolds. Annual Review of Biomedical Engineering. 13, 27-53 (2011).
- Gilpin, S. E., Charest, J. M., Ren, X., Ott, H. C. Bioengineering lungs for transplantation. Thoracic Surgery Clinics. 26 (2), 163-171 (2016).
- Calle, E. A., Leiby, K. L., Raredon, M. B., Niklason, L. E. Lung regeneration: steps toward clinical implementation and use. Current Opinion in Anaesthesiology. 30 (1), 23-29 (2017).
- Planchard, D. . Engineering Design with SOLIDWORKS 2022: A Step-by-Step Project Based Approach Utilizing 3D Solid Modeling. , (2022).
- Coward, C. . A Beginner’s Guide to 3D Modeling: A Guide to Autodesk Fusion 360. , (2019).
- Meza, G., Carpio, C. D., Vinces, N., Klusmann, M. . 2018 IEEE XXV International Conference on Electronics, Electrical Engineering and Computing (INTERCON. , 1-4 (2018).
- Crapo, P. M., Gilbert, T. W., Badylak, S. F. An overview of tissue and whole organ decellularization processes. Biomaterials. 32 (12), 3233-3243 (2011).
- Tchoukalova, Y. D., Hintze, J. M., Hayden, R. E., Lott, D. G. Tracheal decellularization using a combination of chemical, physical and bioreactor methods. The International Journal of Artificial Organs. 41 (2), 100-107 (2017).
- Partington, L., et al. Biochemical changes caused by decellularization may compromise mechanical integrity of tracheal scaffolds. Acta Biomaterialia. 9 (2), 5251-5261 (2013).
- Balestrini, J. L., et al. Production of decellularized porcine lung scaffolds for use in tissue engineering. Integrative Biology. 7 (12), 1598-1610 (2015).
- Taylor, D. A., Sampaio, L. C., Ferdous, Z., Gobin, A. S., Taite, L. J. Decellularized matrices in regenerative medicine. Acta Biomaterialia. 74, 74-89 (2018).
- Huang, S. X., et al. Efficient generation of lung and airway epithelial cells from human pluripotent stem cells. Nature Biotechnology. 32 (1), 84-91 (2014).
- Huang, S. X. L., et al. The in vitro generation of lung and airway progenitor cells from human pluripotent stem cells. Nature Protocols. 10 (3), 413-425 (2015).
- Kim, J., O’Neill, J. D., Dorrello, N. V., Bacchetta, M., Vunjak-Novakovic, G. Targeted delivery of liquid microvolumes into the lung. Proceedings of the National Academy of Sciences of the United States of America. 112 (37), 11530-11535 (2015).
- Kim, J., O’Neill, J. D., Vunjak-Novakovic, G. Rapid retraction of microvolume aqueous plugs traveling in a wettable capillary. Applied Physics Letters. 107 (14), 144101 (2015).
- O’Neill, J. D., et al. Decellularization of human and porcine lung tissues for pulmonary tissue engineering. The Annals of Thoracic Surgery. 96 (3), 1046-1056 (2013).
- Sengyoku, H., et al. Sodium hydroxide based non-detergent decellularizing solution for rat lung. Organogenesis. 14 (2), 94-106 (2018).
- Walters, M. S., et al. Generation of a human airway epithelium derived basal cell line with multipotent differentiation capacity. Respiratory Research. 14 (1), 135 (2013).
- O’Neill, J. D., et al. Cross-circulation for extracorporeal support and recovery of the lung. Nature Biomedical Engineering. 1 (3), 0037 (2017).
- Guenthart, B. A., et al. Regeneration of severely damaged lungs using an interventional cross-circulation platform. Nature Communications. 10 (1), 1985 (2019).
- Chen, J., et al. Non-destructive vacuum-assisted measurement of lung elastic modulus. Acta Biomaterialia. 131, 370-380 (2021).
- Dorrello, N. V., et al. Functional vascularized lung grafts for lung bioengineering. Science Advances. 3 (8), 1700521 (2017).

