In Vitro Apical-Out Enteroid Model of Necrotizing Enterocolitis
Summary
This protocol describes an apical-out necrotizing enterocolitis (NEC)-in-a-dish model utilizing small intestinal enteroids with reversed polarity, allowing access to the apical surface. We provide an immunofluorescent staining protocol to detect NEC-related epithelial disruption and a method to determine the viability of apical-out enteroids subjected to the NEC-in-a-dish protocol.
Abstract
Necrotizing enterocolitis (NEC) is a devastating disease affecting preterm infants, characterized by intestinal inflammation and necrosis. Enteroids have recently emerged as a promising system to model gastrointestinal pathologies. However, currently utilized methods for enteroid manipulation either lack access to the apical surface of the epithelium (three-dimensional [3D]) or are time-consuming and resource-intensive (two-dimensional [2D] monolayers). These methods often require additional steps, such as microinjection, for the model to become physiologically translatable. Here, we describe a physiologically relevant and inexpensive protocol for studying NEC in vitro by reversing enteroid polarity, resulting in the apical surface facing outward (apical-out). An immunofluorescent staining protocol to examine enteroid barrier integrity and junctional protein expression following exposure to tumor necrosis factor-alpha (TNF-α) or lipopolysaccharide (LPS) under normoxic or hypoxic conditions is also provided. The viability of 3D apical-out enteroids exposed to normoxic or hypoxic LPS or TNF-α for 24 h is also evaluated. Enteroids exposed to either LPS or TNF-α, in combination with hypoxia, exhibited disruption of epithelial architecture, a loss of adherens junction protein expression, and a reduction in cell viability. This protocol describes a new apical-out NEC-in-a-dish model which presents a physiologically relevant and cost-effective platform to identify potential epithelial targets for NEC therapies and study the preterm intestinal response to therapeutics.
Introduction
Necrotizing enterocolitis (NEC), a severe inflammatory disease of the small intestine occurring in up to 10% of preterm infants, is commonly associated with high morbidity and mortality1,2. Mortality rates approaching 50% in very low birth weight (<1500 g) infants, requiring surgical intervention, are not uncommon3. While the exact etiology of NEC is not currently understood, risk factors, such as formula feeding, are thought to compound with physiological anomalies, such as dysbiosis, an immature intestinal epithelium, and a dysfunctional intestinal barrier, in the development of the disease2,4. Despite significant effort, little progress in the prevention or treatment of NEC has occurred over the last decade5. A novel in vitro method to study NEC and associated intestinal epithelial barrier dysfunction is required to advance understanding of the pathogenesis of the disease, as findings from animal models have, thus far, translated poorly to the bedside6.
A number of in vitro models have been utilized to investigate the mechanisms at play during NEC. The human intestinal epithelial cell line, Caco-2, is among the most commonly utilized in vitro models of NEC7,8. Caco-2 cells emulate brush border morphological features of the small intestine, but, as a cell line, do not differentiate into the wide variety of in vivo cell types, including mucus-producing goblet cells, required for a highly translatable model. HT-29-MTX, human colon adenocarcinoma cells, include a mixed enterocyte and goblet cell phenotype, but still lack crypt-based cell types of the intestinal epithelium9. IEC-6 and IEC-18 are non-transformed cell lines with an immature ileal crypt-like morphology but are not derived from human tissue, limiting their translational capacity. FHs 74-Int and H4 intestinal cell lines are derived from human fetal tissue and do not form tight junctions or polarized monolayers10,11, and thus are immature compared with even the most premature infants susceptible to NEC. Typically, NEC in vitro models utilize lipopolysaccharide (LPS) treatments to induce toll-like receptor 4 (TLR4), a major receptor initiating intestinal inflammation in NEC12. Damage mediated through reactive oxygen species (ROS) treatment, typically via hydrogen peroxide, is often used to induce NEC-like oxidative damage and apoptosis13,14. As the main driver of intestinal inflammation, tumor necrosis factor-alpha (TNF-α), a downstream component of the inflammatory TLR4 signaling, is also commonly utilized in these in vitro models to mimic in vivo pathogenesis15.
Organoids, generated from inducible pluripotent stem cells (iPSCs), have grown in popularity as an in vitro model of the intestine due to their ability to recapitulate the complex in vivo architecture and cell-type composition of the tissue from which they are derived16,17. A related in vitro system, enteroids, are organoids derived from resected intestinal crypts that are more easily established and maintained than iPSC-derived organoids. Enteroids are typically grown in a three-dimensional (3D) extracellular matrix (ECM) with experimental access limited to the basolateral cell surface. Methods, such as microinjection18,19, have been developed to overcome this barrier to the apical surface, but the buildup of sloughed cellular debris and mucus within the lumen renders microinjection both technically difficult and inconsistent. As custom robotic microinjection platforms are not widely accessible20, lab-to-lab variability in technical ability and general technique become significant variables to overcome with microinjection protocols. Two-dimensional (2D) monolayers derived from dissociated 3D enteroids, still comprising all major cell types of the intestinal epithelium, allow access to the apical surface but have traditionally been difficult to maintain without a feeder layer of mesenchymal myofibroblasts21. While cell culture permeable supports can be used to access both the apical and basolateral sides of enteroid monolayers without the use of underlying myofibroblasts, these inserts require excision and mounting of the membrane before use with modalities such as confocal microscopy, resulting in a more technically demanding and difficult process when using traditional microscopy methods22. NEC has been modeled in vitro using traditional 3D enteroids23,24,25 and permeable supports26,27, and intestinal inflammation has recently been replicated with gut-on-a-chip models28,29. While gut-on-a-chip models incorporating microfluidics are, by far, the most advanced and translatable models, this technology is expensive, complex, and inaccessible to most investigators30.
Recent advances in apical-out enteroid techniques have allowed easier access to the apical surface of 3D enteroids without risking damage to the structural integrity of the in vitro epithelium31,32,33. Apical-out enteroids share the cell type composition and barrier function of in vivo intestinal epithelium, but, unlike typical 3D enteroids, the apical surfaces of these cells face the culture medium, allowing for more physiologically relevant studies on nutrient absorption, microbial infection, and luminal secretion31. An additional advantage of apical-out enteroids is the ability to homogenously distribute experimental agents to enteroids. Varying treatment volumes based on enteroid size is not required, as it is with microinjection, and the ability to maintain these enteroids in suspension culture negates any ECM interference on experimental agent diffusion32.
Necrotizing enterocolitis is a multifactorial disease involving multiple intestinal epithelial cell types and a variety of environmental and pathophysiological factors34. The varied cell composition of intestinal enteroids is a clear improvement over monocultures in modeling a complex disease such as NEC. Interestingly, while a single inflammatory exposure is often sufficient to induce damage in in vitro monocultures, enteroids, as with mouse models23, appear to require a minimum of two inflammatory components to induce NEC-like damage6. Here, we present an apical-out NEC-in-a-dish model, using apical-out enteroids in combination with hypoxia (an important clinical feature of NEC6) and either LPS or TNF-α, as an improved and more physiologically relevant in vitro model to study epithelial responses to NEC-like inflammation and, potentially, identify therapeutic targets. We describe a protocol for reversing the polarity of small intestinal 3D enteroids, as well as an immunofluorescent staining protocol to identify epithelial barrier disruption and junctional protein expression alterations. Finally, we further demonstrate a simple enteroid viability assay to determine the impact of our dual-hit, apical-out NEC-in-a-dish model.
Protocol
All animal procedures in this study were approved by the University of Oklahoma Health Sciences Center Institutional Animal Care and Use Committee. Small intestine convenience samples from a preterm non-human primate (NHP, 90% gestation, olive baboon, Papio anubis) were obtained following euthanasia for a separate study (Protocol #101523-16-039-I).
1. Establishment of apical-out enteroid NEC-in-a-dish model
- Preparation of media and enteroid treatments
NOTE: Once prepared following standard aseptic technique, the media can be stored at 4 °C for up to 1 week.- Prepare antibiotic-free media (AFM) by mixing 50 mL of human organoid growth media with 50 mL of organoid supplement (see Table of Materials). Prepare 5 mM ethylenediaminetetraacetic acid/1x phosphate-buffered saline (EDTA/PBS) by adding 100 µL of 0.5 M EDTA to 9,900 µL of PBS.
- Chill Dulbecco's modified eagle's medium/nutrient Ham's mixture F12 + 15 mM HEPES Buffer (DMEM/F12) at 2-8 °C.
- Prepare TNF-α stock solution by suspending 100 µg of TNF-α powder in 1 mL of 1x PBS. Further dilute the TNF-α stock to 20 ng/mL and 50 ng/mL concentrations using AFM as the solvent. Prepare LPS stock solution by suspending 2 mg of LPS in 1 mL of 1x PBS. Further dilute stock LPS to 100 µg/mL and 200 µg/mL using AFM as the solvent.
- Reversing the polarity of 3D enteroids
NOTE: The following procedure is intended for the reversal of a single 24-well flat-bottom tissue culture plate into two 24-well ultra-low attachment tissue culture plates. The tissue culture plates should be warmed to 37 °C for at least 30 min prior to use. The derivation of basolateral-out 3D enteroids was achieved as described previously by many groups35,36, with slight modifications for media (detailed above). The general enteroid derivation procedure does not differ from that of humans. In brief, excised tissue is washed, cut into small fragments, and incubated in cell disruption buffer. The cell solution is then run through a 70 µM cell strainer, resulting in crypts that are plated at a high density.- Aspirate media from each well of a 24-well plate of established 3D basolateral-out enteroids.
- Add 500 µL of 5 mM ice-cold EDTA/PBS to each well of the 24-well plate and gently mechanically disrupt the basement membrane extract/ECM dome by pipetting up and down with a p200 pipette a minimum of 5x-6x.
- Transfer the contents of four wells to a 15 mL conical tube and add 8 mL of ice-cold EDTA/PBS to the conical tube. Transfer the remaining 20 wells in the same manner.
- Incubate 15 mL conical tubes at 4 °C for 1 h on a rotating platform or shaker at 330 rpm. After the 1 h incubation, centrifuge the conical tubes at 300 x g for 3 min at 4 °C. Remove and discard the supernatant from each tube.
- Combine and wash the cell pellets with 5 mL of DMEM/F12 and distribute the cell suspension into two 15 mL conical tubes.
- Pellet the cells by centrifuging at 300 x g for 3 min at 4 °C. Remove the supernatant and add 12 mL of AFM to each tube, ensuring the cell pellets are resuspended.
- Pipette 500 µL of the enteroid suspension into each well of two 24-well ultra-low attachment plates.
- Incubate the plates at 37 °C and 5% CO2 for 2-5 days or until enteroids have reversed polarity.
- To check enteroid reversal, first establish the confirmatory immunofluorescent staining and the average time to reversal (~3 days), then confirm the polarity reversal using a basic light microscope visualization. Apical-out enteroids are very cellular in appearance, while basolateral-out enteroids are often dominated by the presence of a large central lumen or budding (see Supplementary Figure 1).
NOTE: Once the procedure has been standardized, the conversion rate to apical-out typically exceeds 95%. The use of apical-out enteroids is intended as a terminal experiment, though it is theoretically possible to passage apical-out enteroids into a small number of basolateral-out enteroids.
- Apical-out NEC-in-a-dish
NOTE: Once the enteroids have reversed polarity, use the cultures in subsequent experiments quickly, as their longevity in apical-out conformation has not been confirmed beyond 3-4 days. For the following NEC-in-a-dish model, the enteroids were treated for 24 h, then collected immediately afterward and preserved for downstream experiments.- Once the enteroids have visually reversed polarity with at least 95% efficiency, remove the plates from the incubator and collect eight wells of a 24-well plate into a 15 mL conical tube. As the apical-out enteroids are in suspension, simply pipette the enteroid/media solution. Centrifuge the 15 mL tube at 300 x g for 5 min at 4 °C.
- Once the cells are pelleted, remove the supernatant and resuspend the cell pellet in 4 mL of AFM mixed with the desired volume (10 µL here to match the highest treatment volume added) of 1x PBS (control).
- Pipette 500 µL of enteroid/PBS/AFM suspension into eight wells of a 24-well ultra-low attachment plate and incubate the plate at 37 °C and 5% CO2 for 24 h.
- Repeat Steps 1.3.1.-1.3.3. for TNF-α (20 ng/mL and 50 ng/mL) and LPS (100 µg/mL and 200 µg/mL) treatments at normoxia. For all hypoxia treatments, repeat Steps 1.3.1.-1.3.3., but place the plate in a separate incubator at 37 °C, 5% CO2, and 1% O2 for 24 h.
NOTE: Step 1.3.4. can be done simultaneously with Steps 1.3.1.-1.3.3, but, for clarity, the procedure above was separated by treatment in order to reduce complexity.
2. Immunofluorescent staining and confocal microscopy
- Preparation of staining solutions
- Prepare 0.1% Triton X-100 by mixing 7 µL of Triton X-100 in 1x PBS to a final volume of 7 mL. Prepare PBST (PBS-Tween) by adding 30 µL of Tween-20 to 1x PBS to a final volume of 30 mL.
- Prepare 10% normal donkey serum (NDS)/PBST by adding 700 µL of NDS to 6.3 mL of PBST. Prepare 1% NDS/PBST by adding 70 µL of NDS to PBST to a final volume of 7 mL.
NOTE: Donkey serum was used here to correlate with the secondary antibody host. However, the blocking serum species should match the secondary antibody species in order to properly block non-specific binding.
- Staining (Day 1)
- Transfer the contents of four wells of a 24-well plate to a 1.5 mL microcentrifuge tube. Repeat for all desired wells/treatments, with each treatment in a separate tube. Centrifuge the tubes at 300 x g for 4 min at 4 °C. Once the enteroids are pelleted, remove the supernatant.
- Resuspend the cell pellets in 300 µL of 4% formaldehyde fixative for 30 min at room temperature (RT). After 30 min, centrifuge the tubes at 300 x g for 4 min at 4 °C, remove the supernatant, and wash the pellet with 500 µL of sterile, RT 1x PBS.
- Centrifuge the tubes at 300 x g for 4 min at 4 °C. Remove the supernatant and add 500 µL of 0.1% Triton X-100, and let sit for 1 h at RT.
- After 1 h, centrifuge the tubes at 300 x g for 4 min at 4 °C and remove the supernatant. Wash with 500 µL of 1x PBS and place the tubes on a rotator or shaker at 200 rpm for 15 min at 2-8 °C. Repeat this a total of 4x.
- Centrifuge the tubes at 300 x g for 4 min at 4 °C and remove the supernatant. Add 500 µL of 10% NDS/PBST and incubate at RT for 45 min. During the incubation, prepare primary antibody solutions by combining 20 µL of recombinant anti-villin antibody, 10 µL of E-cadherin antibody, and 1970 µL of 1% NDS/PBST for eight wells of a 24-well plate.
- After the 45 min incubation, centrifuge the tubes at 300 x g for 4 min at 4 °C. Place the tubes on ice. Remove the supernatant and replace with 250 µL of primary antibody solution. Incubate the tubes overnight at 2-8 °C.
- Staining (Day 2)
- Centrifuge the tubes at 300 x g for 4 min at 4 °C and remove the supernatant. Add 250-500 µL of PBST to each tube, depending on the size of the pellet, and place on the rotator or shaker at 200 rpm for 1 h at 2-8 °C. Repeat the PBST wash 4x.
- Prepare secondary antibody solution by adding 52 µL of Cy3 donkey anti-rabbit IgG (H + L) to 50% glycerol and 5.2 µL of donkey anti-mouse fluorescent green 488 secondary antibodies to 5142.8 µL of 1% NDS/PBST.
- Following the last PBST wash and centrifugation, remove the supernatant from the tubes. Dispense 200 µL of secondary antibody solution to each tube and incubate in the dark overnight at 2-8 °C.
- Mounting stained apical-out enteroids (Day 1)
- Bring glycerol-based mountant containing blue nuclear dye to RT over 1 h.
- Centrifuge the tubes at 300 x g for 4 min at 4 °C. Remove 100 µL of the supernatant and resuspend the cells in the remaining ~100 µL of supernatant. Transfer the cell suspension to 0.5 mL tubes.
- Centrifuge the tubes in a mini centrifuge for 20 s to pellet the cells. Remove the supernatant and resuspend the cell pellets in 100 µL of far-red nucleic acid stain. Incubate the tubes at RT for 10 min.
- Centrifuge the tubes in a mini centrifuge for 20 s to pellet the cells. Remove the supernatant and resuspend the cell pellets in 100 µL of 1x PBS. Repeat for a second wash.
- Centrifuge the tubes in a mini centrifuge for 20 s to pellet the cells and remove 70 µL of the supernatant. Resuspend the cells in the remaining volume of PBS and transfer the contents to a labeled coverslip (24 mm x 60 mm).
- Apply 75 µL of mountant directly onto the specimen and remove any bubbles with a pipette tip. Allow the coverslips to cure overnight (18-24 h) at RT in the dark.
- Mounting stained apical-out enteroids (Day 2)
- After curing overnight, apply a thin layer (~15 µL) of glycerol to the coverslips. Mount the coverslip to the glass slide and gently press into place. Tap to remove any bubbles between the coverslip and the slide.
- Allow the coverslip to set at RT in the dark for a minimum of 2 h, before imaging at 20x magnification with a confocal microscope. For microscopic visualization of enteroids using various confocal acquisition methods, refer to Lallemant et al.37.
- Immunofluorescent quantification
NOTE: This method determines the corrected total fluorescence by removing the background signal using ImageJ software (https://imagej.nih.gov/ij/).- Open the confocal images in ImageJ and outline the desired area with the region of interest (ROI) tool.
- Set user-defined parameters by navigating to Analyze > Set Measurements. Select Area > Integrated Density > Mean Grey Value under the settings tab.
- Navigate to Analyze > Measure. A pop-up window will appear containing the measurements. Copy and paste these measurements into a spreadsheet.
- To subtract the background signal, select a small non-fluorescent area of the image. Navigate to Analyze > Measure for that region. A pop-up window will appear containing the measurements. Copy and paste the output into a spreadsheet.
- Multiply the area of the selected cell by the average fluorescence of background readings, and subtract the sum from the integrated density to calculate the corrected total cell fluorescence (CTCF). Repeat the steps above for all images of interest, while maintaining records in a spreadsheet or statistical software package (see Table of Materials).
3. Apical-out NEC-in-a-dish cell viability
- Enteroid treatments
- Establish apical-out NEC-in-a-dish model for 24 h, as in Step 1.
- Thaw cell viability assay reagent overnight at 4 °C. On the day of assay, bring the reagent to RT 30 min before use. Invert the reagent to mix.
- Before performing the assay, remove 100 µL of media from each well, leaving the remaining 400 µL. Add 400 µL of cell viability assay reagent to each well.
- Vigorously mix well the contents on a plate shaker at RT for 5 min at 200 rpm to induce cell lysis. Following shaking, incubate the plate at RT for an additional 25 min.
- After 25 min incubation, mix the contents of one well via pipetting and transfer 200 µL of the 800 µL to a single well of a 96-well, opaque, polystyrene, clear-bottomed plate. Repeat for the remaining 600 µL, creating four technical replicates per well. Transfer the remaining contents of each well of the 24-well plate into a 96-well plate in this manner.
- Using a plate reader capable of luminescence, record values at 0.25 ms integration and compare the relative values among treatments. Alternatively, compare treatment luminescence to an adenosine triphosphate (ATP) standard.
Representative Results
The use of enteroids to model intestinal inflammation, even within the context of necrotizing enterocolitis, is now common. However, most methods currently utilized either lack access to the apical surface of enteroids, negating the physiological relevance of compounds intended for eventual use as oral therapeutics, or are technically difficult and time-consuming, as with enteroid-derived monolayers. To increase the utility of current in vitro enteroid models of NEC, we reversed the polarity of enteroids, and, in combination with 24 h hypoxia and LPS or TNF-α, generated an apical-out NEC-in-a-dish model. Immunofluorescent staining confirmed apical-out localization of nuclei (blue) toward the lumen and the detection of villin32 (red, apical marker) at the outer edge of the epithelium (Figure 1A). Exposure to 200 µg/mL LPS, 50 ng/mL TNF-α, or hypoxia alone did not induce overt changes in gross cell morphology, E-cadherin38 (green), or villin localization or fluorescent intensity compared to control (Figure 1A–D; Figure 2). Treatment with LPS or TNF-α, in combination with hypoxia, disrupted epithelial architecture and caused a significant loss of adherens junction and brush border protein expression, indicated by the loss of E-cadherin and villin staining (Figure 1E-F; Figure 2). These reductions in enterocyte brush border and adherens junction protein expressions likely indicate a reduction in epithelial barrier integrity and are a common feature of both human surgical NEC and in vitro modeling of the disease23.
To determine the impact of NEC-in-a-dish components on the viability of 3D apical-out enteroids, cell viability was evaluated using a cell viability assay designed for 3D cultures, centered on ATP quantification following cell lysis. Enteroids were treated with LPS (200 µg/mL) or TNF-α (50 ng/mL) for 24 h under normoxic or hypoxic conditions. LPS or TNF-α alone did not significantly affect cell viability compared to control (Figure 3A). However, significant reductions in viability occurred when apical-out enteroids were treated with 200 µg/mL LPS or 50 ng/mL TNF-α in combination with hypoxia (****p < 0.0001) compared to hypoxia alone (Figure 3B). These experiments demonstrate the versatility and physiological relevance of utilizing apical-out enteroids in a cost-effective and straightforward NEC-in-a-dish model.
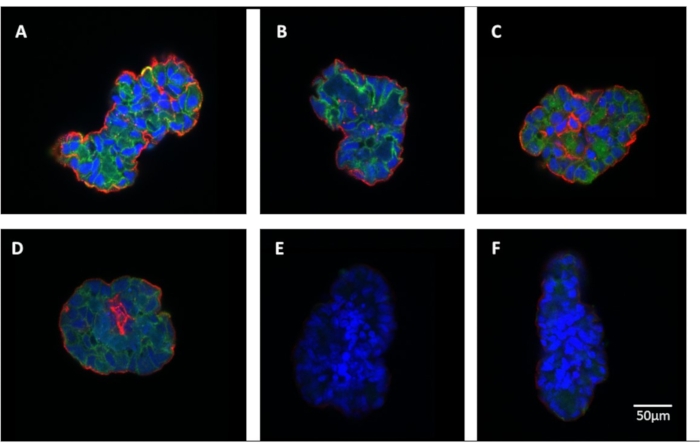
Figure 1: Confirmation of apical-out phenotype and morphological effects of lipopolysaccharide (LPS), tumor necrosis factor alpha (TNF-α), and hypoxia. Representative immunohistochemical staining of 3D apical-out enteroids for enterocyte brush border and adherens junction proteins after 24 h. (A) Control, (B) hypoxia, (C) LPS (200 µg/mL), (D) TNF-α (50 ng/mL), (E) LPS (200 µg/mL) + hypoxia, (F) TNF-α (50 ng/mL) + hypoxia. Scale bar = 50 µm for each image. Please click here to view a larger version of this figure.
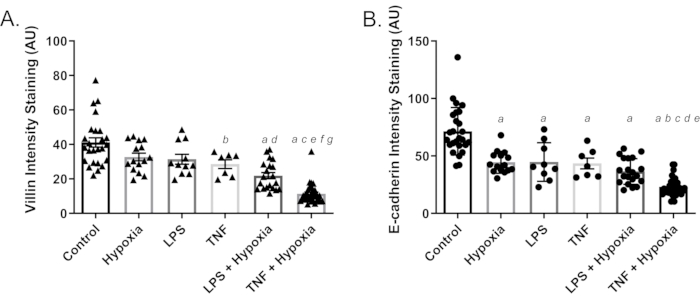
Figure 2: Quantification of staining intensity for brush border and adherens junction proteins following 24 h lipopolysaccharide (LPS), tumor necrosis factor alpha (TNF-α), and hypoxia treatments. (A) Villin (red) levels compared among treatments; a = p < 0.0001 compared to control; b = p < 0.05 compared to control; c = p < 0.0001 compared to hypoxia; d = p < 0.05 compared to hypoxia; e = p < 0.001 compared to TNF-α; f = p < 0.01 compared to LPS + hypoxia; g = p < 0.0001 compared to LPS; and (B) E-cadherin (green) levels compared among treatments; a = p < 0.0001 compared to control; b = p < 0.0001 compared to hypoxia; c = p < 0.01 compared to LPS; d = p < 0.01 compared to TNF-α; e = p < 0.05 compared to LPS + hypoxia. Significance was assessed using one-way analysis of variance (ANOVA) with Tukey's test for post-hoc analysis, presented as mean ± standard error of the mean (SEM) (n ≥ 7/group). Control represents normoxia with PBS as vehicle control. Please click here to view a larger version of this figure.
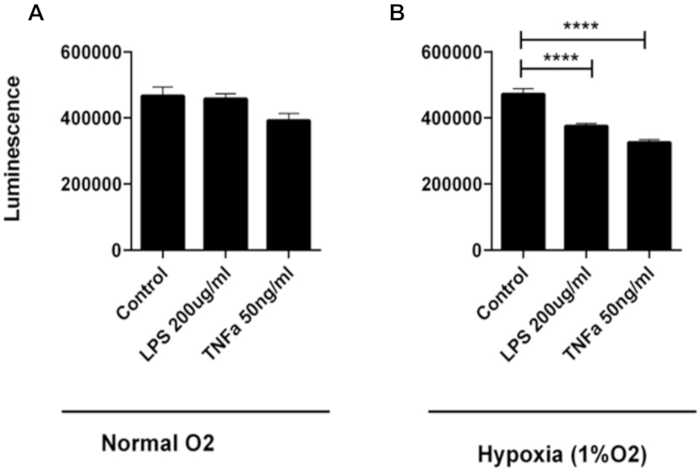
Figure 3: Effect of lipopolysaccharide (LPS), tumor necrosis factor alpha (TNF-α), and hypoxia on apical-out cell viability. The viability of 3D apical-out enteroids treated with LPS (200 µg/mL) or TNF-α (50 ng/mL) for 24 h under (A) normoxic or (B) hypoxic conditions was measured via luminescence. Statistical significance was calculated using one-way analysis of variance (ANOVA) with Tukey's test for post-hoc analysis. Enteroids treated with LPS (200 µg/mL) and TNF-α (50 ng/mL), in combination with hypoxia, were significantly less viable compared to hypoxia alone, seen here with an increase in luminescence. LPS (200 µg/mL) or TNF-α (50 ng/mL), under normoxic conditions, did not differ from control. Data are presented as mean ± standard error of the mean (SEM), n = 8/group, ****p < 0.0001. Control represents normoxia with PBS as vehicle control. Please click here to view a larger version of this figure.
Supplementary Figure 1: Morphology of three-dimensional enteroids. (A) While basolateral-out enteroids are dominated by the appearance of a large and open central lumen, with or without budding, (B) apical-out enteroids are characterized by a dense, cellular appearance. Scale bar = 100 µm for each image. Please click here to download this File.
Discussion
The recent development of enteroid models derived from intestinal epithelial crypts allows for a more physiologically relevant in vitro tissue in which to study necrotizing enterocolitis pathogenesis. Despite including all major differentiated cell types of the intestinal epithelium, 3D enteroids are still subject to several significant limitations. The conventional, basolateral-out enteroids are suspended in 3D ECM hydrogel domes, the composition and density of which may limit normal diffusion within the tissue culture environment39. Importantly, these enteroids supply limited access to the apical surface of cells, which is the surface that would, in vivo, interact with luminal bacteria and antigens, such as enterocyte TLR4-binding of LPS.
While the pathophysiology of NEC remains unclear, increased expression and activation of apical TLR4 in the preterm intestinal epithelium are thought to play a prominent role40. Downstream of TLR4 activation, increased intestinal permeability, reduced epithelial repair, and bacterial translocation result in a dysfunctional intestinal barrier, leading to necrosis of the gut2. This inflammatory cycle in the distal intestine of premature infants is thought to originate in the intestinal lumen, initiating at the apical surface of enterocytes. Thus, models utilizing basolateral-out 3D enteroids in mechanistic studies involving bacterial translocation of the intestinal epithelium do not recapitulate in vivo pathogenesis as is currently understood and are likely to yield suboptimal information.
Here, we describe the establishment of an apical-out NEC-in-a-dish model, allowing easy access to the apical surface of enteroid cells and, presumably, resulting in a more physiologically relevant model compared with conventional 3D basolateral-out enteroids. The polarity reversal in these enteroids allows for potential orally-administered therapeutics to be simply added to the media and taken up naturally by enterocytes, as would occur in vivo. This approach does not require expensive and technically challenging techniques, such as microinjection, and is more resource-friendly than the establishment of 2D monolayers or gut-on-a-chip models.
Our representative results indicate apical-out enteroids subjected to LPS or TNF-α, in combination with hypoxia, suffer reduced viability, disrupted epithelial architecture, and a reduction in adherens junction protein expression compared to control, or compared to hypoxia or inflammatory mediator treatment alone, mimicking the known features of in vivo NEC. Disruption of adherens junctions, critical to intestinal epithelial barrier function41, has been reported in NEC42,43. Tight junction architectural disruption, which is also important in NEC44, likely also occurred, though these changes will be evaluated in future studies. In a physiologically translatable tissue, this dual-hit model allows for the study of preterm intestinal responses to factors inherent to NEC, while providing a platform through which potentially novel therapeutic targets can be identified. Future studies can extend the application of apical-out enteroids beyond NEC to studies focused on parasitic, bacterial, or viral infections31,32, host-microbiome interactions45, and gastrointestinal absorptive function46, but within the physiological context of the preterm infant.
This protocol for apical-out NEC-in-a-dish includes several critical steps warranting emphasis. The composition and freshness of the cell culture media are important in avoiding excessive longitudinal variability in experiments and ensuring enteroids are healthy before experimental treatment. Similarly, stock solutions of TNF-α and LPS should be aliquoted and stored at -80 °C to avoid excessive freeze/thaw cycles and preserve optimal activity. Finally, care should be taken to ensure all ECM is solubilized during the EDTA/PBS incubation. Incomplete removal of ECM could result in enteroids failing to reverse polarity. If necessary, the incubation period can be extended to 1.5 h to ensure complete removal of the matrix.
While this apical-out NEC-in-a-dish model is cost-effective and straightforward, the method is subject to several limitations. Apical-out NEC-in-a-dish is not as physiologically relevant as gut-on-a-chip models28,29 but represents an intermediate option where and when chip options are not accessible. In addition, as a suspension culture, enteroids must be collected, pelleted, and resuspended for media changes, adding a centrifugation step to an otherwise simple process. Another limitation is the longevity of apical-out enteroids, as they are generally considered suitable only for end-point analyses. As basolateral growth factor receptors are no longer in constant contact with media growth factors, apical-out enteroids are characterized by reduced proliferation compared with basolateral-out enteroids, resulting in the relative inability to passage apical-out cultures31,32. Additionally, whether the exact composition, including the relative proportions, of cell types are represented in enteroids compared to in vivo tissue is not known, but studies generally suggest minor differences31,32. Finally, the 24 h development of this apical-out NEC-in-a-dish model, while convenient, may be relatively brief in comparison with in vivo formula-based NEC models47, potentially requiring additional standardization if longer periods of intervention are desired.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. HC is supported by grant P20GM134973 from the National Institutes of Health. KB is supported by a Children's Hospital Foundation (CHF) and Presbyterian Health Foundation (PHF) grant. We thank the Laboratory for Molecular Biology and Cytometry Research at OUHSC for the use of the Core Facility, which provided confocal imaging.
Materials
| 0.5 M EDTA, pH 8.0 | Fisher Scientific | 15575-020 | |
| 1.5 mL microcentrifuge tubes | Fisher Scientific | 05-408-129 | |
| 15 mL Conical tube | VWR | 89039-666 | |
| CellTiter-Glo 3D Cell Viability Assay | Promega | G9681 | |
| Corning Costar Ultra-Low Attachment 24-Well Microplates | Fisher Scientific | 07-200-602 | |
| Cover Glass 24 mm x 60 mm | Thermo Scientific | 102460 | |
| Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor 488 | Thermo Scientific | A-21202 | |
| Donkey Anti-Rabbit IgG Antibody, Cy3 conjugate | Sigma-Aldrich | AP182C | |
| Dulbecco's Modified Eagle's Medium/Nutrient Ham's Mixture F-12 (DMEM-F12) with 15 mM HEPES buffer | STEMCELL Technologies | 36254 | |
| E-cadherin antibody (7H12) | Novus Biologicals | NBP2-19051 | |
| Formaldehyde solution 4%, buffered, pH 6.9 | Millipore Sigma | 1004960700 | |
| Glycerol | Sigma-Aldrich | 56-81-5 | |
| ImageJ | Fiji | N/A | |
| IntestiCult Organoid Growth Medium (Human) | STEMCELL Technologies | 06010 | |
| Leica SP8 Confocal Microscope | Leica Biosystems | ||
| Lipopolysaccharides from Escherichia coli O111:B4, purified by gel filtration chromatography | Millipore Sigma | L3012-10MG | |
| Microscope Slides | Fisher Scientific | 12-544-7 | |
| Normal Donkey Serum | Sigma-Aldrich | 566460 | |
| Nunc MicroWell 96-Well, Nunclon Delta-Treated, Flat-Bottom Microplate | Thermo Scientific | 136101 | |
| PBS (Phosphate Buffered Saline), 1x [-] calcium, magnesium, pH 7.4 | Corning | 21-040-CM | |
| Prolong Glass Antifade Mountant with NucBlue | Fisher Scientific | P36983 | |
| Recombinant Anti-Villin antibody [SP145] | Abcam | ab130751 | |
| Recombinant Human TNF-α protein 100 µg | Bio-Techne | 210-TA-100/CF | |
| SpectraMax iD3 Multi-Mode Microplate Reader | Molecular Devices | ||
| Thermo Forma Series II Water-Jacketed Tri-Gas Incubator, 184L | Fisher Scientific | 3140 | |
| TO-PRO-3 Iodide (642/661) | Thermo Scientific | T3605 | |
| Triton X-100 | Sigma-Aldrich | 9002-93-1 | |
| Tubes, 0.5 mL, flat cap | Thermo Scientific | AB0350 | |
| Tween-20 | Sigma-Aldrich | 9005-64-5 |
Riferimenti
- Nanthakumar, N., et al. The mechanism of excessive intestinal inflammation in necrotizing enterocolitis: an immature innate immune response. PLoS One. 6 (3), 17776 (2011).
- Neu, J., Walker, W. A. Necrotizing enterocolitis. The New England Journal of Medicine. 364 (3), 255-264 (2011).
- Bazacliu, C., Neu, J. Necrotizing enterocolitis: long term complications. Current Pediatric Reviews. 15 (2), 115-124 (2019).
- Lim, J. C., Golden, J. M., Ford, H. R. Pathogenesis of neonatal necrotizing enterocolitis. Pediatric Surgery International. 31 (6), 509-518 (2015).
- Neu, J. Necrotizing enterocolitis: the future. Neonatology. 117 (2), 240-244 (2020).
- Kovler, M. L., Sodhi, C. P., Hackam, D. J. Precision-based modeling approaches for necrotizing enterocolitis. Disease Models & Mechanisms. 13 (6), (2020).
- Emami, C. N., Mittal, R., Wang, L., Ford, H. R., Prasadarao, N. V. Recruitment of dendritic cells is responsible for intestinal epithelial damage in the pathogenesis of necrotizing enterocolitis by Cronobacter sakazakii. Journal of Immunology. 186 (12), 7067-7079 (2011).
- Chen, L., et al. Human β-defensin-3 reduces excessive autophagy in intestinal epithelial cells and in experimental necrotizing enterocolitis. Scientific Reports. 9 (1), 19890 (2019).
- De Fazio, L., et al. Necrotizing enterocolitis: overview on in vitro models. International Journal of Molecular Sciences. 22 (13), 6761 (2021).
- Gimeno-Alcañiz, J. V., Collado, M. C. Impact of human milk on the transcriptomic response of fetal intestinal epithelial cells reveals expression changes of immune-related genes. Food & Function. 10 (1), 140-150 (2019).
- Claud, E. C., Savidge, T., Walker, W. A. Modulation of human intestinal epithelial cell IL-8 secretion by human milk factors. Pediatric Research. 53 (3), 419-425 (2003).
- De Plaen, I. G. Inflammatory signaling in necrotizing enterocolitis. Clinics in Perinatology. 40 (1), 109-124 (2013).
- Subramanian, S., Geng, H., Tan, X. D. Cell death of intestinal epithelial cells in intestinal diseases. Sheng Li Xue Ba : [Acta Physiologica Sinica]. 72 (3), 308-324 (2020).
- Li, B., et al. Intestinal epithelial cell injury is rescued by hydrogen sulfide. Journal of Pediatric Surgery. 51 (5), 775-778 (2016).
- Khailova, L., et al. Bifidobacterium bifidum reduces apoptosis in the intestinal epithelium in necrotizing enterocolitis. American Journal of Physiology. Gastrointestinal and Liver Physiology. 299 (5), 1118-1127 (2010).
- Aguilar, C., et al. Organoids as host models for infection biology – a review of methods. Experimental & Molecular Medicine. 53 (10), 1471-1482 (2021).
- Stelzner, M., et al. A nomenclature for intestinal in vitro cultures. American Journal of Physiology. Gastrointestinal and Liver Physiology. 302 (12), 1359-1363 (2012).
- Bartfeld, S., Clevers, H. Organoids as model for infectious diseases: culture of human and murine stomach organoids and microinjection of helicobacter pylori. JoVE: Journal of Visualized Experiments. (105), e53359 (2015).
- Bartfeld, S., et al. In vitro expansion of human gastric epithelial stem cells and their responses to bacterial infection. Gastroenterology. 148 (1), 126-136 (2015).
- Williamson, I. A., et al. A high-throughput organoid microinjection platform to study gastrointestinal microbiota and luminal physiology. Cellular and Molecular Gastroenterology and Hepatology. 6 (3), 301-319 (2018).
- Moorefield, E. C., Blue, R. E., Quinney, N. L., Gentzsch, M., Ding, S. Generation of renewable mouse intestinal epithelial cell monolayers and organoids for functional analyses. BMC Cell Biology. 19 (1), 15 (2018).
- VanDussen, K. L., et al. Development of an enhanced human gastrointestinal epithelial culture system to facilitate patient-based assays. Gut. 64 (6), 911-920 (2015).
- Werts, A. D., et al. A novel role for necroptosis in the pathogenesis of necrotizing enterocolitis. Cellular and Molecular Gastroenterology and Hepatology. 9 (3), 403-423 (2020).
- Li, B., et al. Intestinal epithelial tight junctions and permeability can be rescued through the regulation of endoplasmic reticulum stress by amniotic fluid stem cells during necrotizing enterocolitis. FASEB Journal: Official Publication of the Federation of American Societies for Experimental Biology. 35 (1), 21265 (2021).
- Ares, G. J., Buonpane, C., Yuan, C., Wood, D., Hunter, C. J. A novel human epithelial enteroid model of necrotizing enterocolitis. JoVE:Journal of Visualized Experiments. (146), e59194 (2019).
- Senger, S., et al. Human fetal-derived enterospheres provide insights on intestinal development and a novel model to study necrotizing enterocolitis (NEC). Cellular and Molecular Gastroenterology and Hepatology. 5 (4), 549-568 (2018).
- Li, B., et al. Activation of Wnt signaling by amniotic fluid stem cell-derived extracellular vesicles attenuates intestinal injury in experimental necrotizing enterocolitis. Cell Death & Disease. 11 (9), 750 (2020).
- Beaurivage, C., et al. Development of a human primary gut-on-a-chip to model inflammatory processes. Scientific Reports. 10 (1), 21475 (2020).
- Jeon, M. S., et al. Contributions of the microbiome to intestinal inflammation in a gut-on-a-chip. Nano Convergence. 9 (1), 8 (2022).
- Bein, A., et al. Microfluidic organ-on-a-chip models of human intestine. Cellular and Molecular Gastroenterology and Hepatology. 5 (4), 659-668 (2018).
- Co, J. Y., et al. Controlling epithelial polarity: a human enteroid model for host-pathogen interactions. Cell Reports. 26 (9), 2509-2520 (2019).
- Co, J. Y., Margalef-Català, M., Monack, D. M., Amieva, M. R. Controlling the polarity of human gastrointestinal organoids to investigate epithelial biology and infectious diseases. Nature Protocols. 16 (11), 5171-5192 (2021).
- Li, Y., et al. Next-generation porcine intestinal organoids: an apical-out organoid model for swine enteric virus infection and immune response investigations. Journal of Virology. 94 (21), 01006-01020 (2020).
- Li, B., et al. Neonatal intestinal organoids as an ex vivo approach to study early intestinal epithelial disorders. Pediatric Surgery International. 35 (1), 3-7 (2019).
- Stewart, C. J., Estes, M. K., Ramani, S. Establishing human intestinal enteroid/organoid lines from preterm infant and adult tissue. Methods in Molecular Biology. 2121, 185-198 (2020).
- Mahe, M. M., Sundaram, N., Watson, C. L., Shroyer, N. F., Helmrath, M. A. Establishment of human epithelial enteroids and colonoids from whole tissue and biopsy. JoVE: Journal of Visualized Experiments. (97), e52483 (2015).
- Lallemant, L., Lebreton, C., Garfa-Traoré, M. Comparison of different clearing and acquisition methods for 3D imaging of murine intestinal organoids. Journal of Biological Methods. 7 (4), 141 (2020).
- Buonpane, C., et al. ROCK1 inhibitor stabilizes E-cadherin and improves barrier function in experimental necrotizing enterocolitis. American Journal of Physiology. Gastrointestinal and Liver Physiology. 318 (4), 781-792 (2020).
- Shin, W., et al. Spatiotemporal gradient and instability of Wnt induce heterogeneous growth and differentiation of human intestinal organoids. iScience. 23 (8), 101372 (2020).
- Egan, C. E., et al. Toll-like receptor 4-mediated lymphocyte influx induces neonatal necrotizing enterocolitis. The Journal of Clinical Investigation. 126 (2), 495-508 (2016).
- Schneider, M. R., et al. A key role for E-cadherin in intestinal homeostasis and Paneth cell maturation. PLoS One. 5 (12), 14325 (2010).
- Khailova, L., et al. Bifidobacterium bifidum improves intestinal integrity in a rat model of necrotizing enterocolitis. American Journal of Physiology. Gastrointestinal and Liver Physiology. 297 (5), 940-949 (2009).
- Khailova, L., et al. Changes in hepatic cell junctions structure during experimental necrotizing enterocolitis: effect of EGF treatment. Pediatric Research. 66 (2), 140-144 (2009).
- Ravisankar, S., et al. Necrotizing enterocolitis leads to disruption of tight junctions and increase in gut permeability in a mouse model. BMC Pediatrics. 18 (1), 372 (2018).
- Han, X., et al. Creating a more perfect union: modeling intestinal bacteria-epithelial interactions using organoids. Cellular and Molecular Gastroenterology and Hepatology. 12 (2), 769-782 (2021).
- Joo, S. S., et al. Porcine intestinal apical-out organoid model for gut function study. Animals: An Open Access Journal from MDPI. 12 (3), 372 (2022).
- Nolan, L. S., Gong, Q., Hofmeister, H. N., Good, M. A protocol for the induction of experimental necrotizing enterocolitis in neonatal mice. STAR Protocols. 2 (4), 100951 (2021).

