A Modified Technique for Transverse Aortic Constriction in Mice
Summary
The present protocol describes a modified and simplified technique with a minimally invasive transverse aortic constriction (TAC) procedure using a self-made retractor. This procedure can be conducted without a ventilator or microscope and introduces pressure overload, eventually leading to cardiac hypertrophy or heart failure.
Abstract
Transverse aortic constriction (TAC) is a frequently used surgery in research regarding heart failure and cardiac hypertrophy based on the formation of pressure overload in mouse models. The main challenge of this procedure is to clearly visualize the transverse aortic arch and precisely band the target vessel. Classical approaches perform a partial thoracotomy to expose the transverse aortic arch. However, it is an open-chest model that causes a rather large surgical trauma and requires a ventilator during the surgery. To prevent unnecessary trauma and simplify the operating procedure, the aortic arch is approached via the proximal proportion of the sternum, reaching and binding the target vessel using a small self-made retractor that contains a snare. This procedure can be conducted without entering the pleura cavity and does not need a ventilator or microsurgical operation, which leaves the mice with physiological breathing patterns, simplifies the procedure, and significantly reduces operation time. Due to the less invasive approach and less operation time, mice can undergo fewer stress reactions and recover rapidly.
Introduction
Heart failure is a complex clinical symptom that results from impaired structure and function of ventricular filling or ejecting blood1. The disease stage is mainly defined via the New York Heart Association function classification based on the severity of symptoms and physical activity2. For those patients with an ejection fraction of over 50%, structural and/or functional abnormalities raised natriuretic peptides to support the diagnosis of heart failure with preserved ejection fraction (HFpEF)2. Ischemic heart disease is a leading cause among multiple etiologies of heart failure. Thus, the myocardial infarction model (such as permanent coronary ligation) is often used to study pathophysiology after cardiac hypoperfusion or ischemia-reperfusion injury3,4. Besides acute myocardial injury, other risk factors such as hypertension, diabetes, obesity, and a family history of cardiomyopathy also contribute to the development of heart failure. After patients pass Stage A (at risk for heart failure) and enter Stage B (pre-heart failure), structural modification occurs1. For example, hypertensive patients first go through adaptive left ventricle hypertrophy, and then gradually develop into maladaptive cardiac hypertrophy and transit to heart failure through pathological remodeling5.
As the terminal stage of various cardiovascular diseases, chronic heart failure has been studied for decades6. Multiple mouse models have been widely used in heart failure research, including drug infusion (angiotensin II), metabolic disorders (diabetes or high caloric diet), and aortic constriction7. Among these models, angiotensin II perfusion is accompanied by various organ side effects, such as kidney7. Inducing metabolic disorders usually require a rather long period of time. Ascending aortic constriction has been considered to have limited relevance to human disease7.
TAC is a reliable model that increases afterload and induces cardiac hypertrophy as well as heart failure8. Open-chest TAC model was first described by Rockman et al. and was used in numerous laboratories around the world9. However, this classical TAC procedure causes a rather large trauma to mice and changes their normal behavior, which may take a long recovery time and disturb further treatment10. Other modified closed-chest TAC procedures did reduce some invasive steps but required microsurgical skills or mechanical ventilation10,11.
The present protocol details a step-by-step method with a minimally invasive approach to the aortic arch using a self-made retractor via a 3 mm midline incision of the upper edge of the sternum. This model does not need microsurgical skill, mechanical ventilation, or cutting through the ribs, thereby providing a rapid, surgical trauma-limited, uncomplicated, inexpensive way to perform TAC surgery.
Protocol
The current protocol is approved by the ethics committee of Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China. This procedure is performed on male adult C57/BL6 mice (>10 weeks of age). All surgical instruments were sterilized by autoclaving before the operation.
1. Preparation of surgical instrument
- Prepare a 5 mL syringe and pinch off the tip of the needle with a needle holder to blunt it.
- Prepare a 27 G needle and blunt it with a needle holder. Cutting the tip of the needle with ophthalmic scissors is an alternative way to blunt the needle.
- Bend the tip of the 27 G needle 90°.
NOTE: This needle will be used as a spacer to tie with the aorta for vessel constriction. - Use a commercially obtained iron wire (medical grade, 0.5 mm in diameter, see Table of Materials) and twine around the needle (Figure 1A). Form a small snare or loop at the blunted tip of the needle (Figure 1B).
NOTE: This reusable instrument acts as a special self-made retractor and allows one to pass silk suture under the aorta in the latter steps. - Autoclave all the surgical tools before surgery (including the special retractor made above) to sterilize them.
2. Animal preparation
- Anesthetize the mouse via intraperitoneal injection of pentobarbital sodium solution (50 mg/kg, see Table of Materials) according to the body weight. Subcutaneously inject 100 μL of 0.5% lidocaine locally into the surgical area.
- Confirm that the mouse is fully anesthetized by pinching the mouse's toe with the tip of the forceps.
- Apply ophthalmic ointment to both the eyes to prevent dryness while under anesthesia.
- Remove the hair from the throat and upper chest with depilatory cream or a clipper.
- Disinfect the unhaired area with three alternating scrubs of iodine and 70% ethanol in concentric circles moving outward.
- Place the mouse in the supine position on a heating pad covered with a sterile drape with its head pointing to the operator. Fix the extremities with adhesive tape and incisors with a 4-0 suture (Figure 2A).
- Insert a rectal probe (see Table of Materials) to monitor body temperature during the operation. Maintain the body temperature at 37 °C during the whole operation.
- Put on a pair of sterile gloves.
NOTE: Once the sterile gloves are put on, do not touch other unsterilized areas or objects. - Cut a hole (1-1.5 cm in diameter) in a new disposable sterile drape with sterilized ophthalmic scissors. Place the sterile drape over the mouse and ensure the unhaired operating field is visible through the hole.
NOTE: This step allows the operator to avoid touching other unsterilized areas, which helps maintain sterile conditions during surgery.
3. Constriction of transverse aorta
- Make a 1.5 cm vertical skin incision at the level of the suprasternal notch (Figure 2B) with scalpel blade. Expand the skin incision down to the level of the sternal angle.
NOTE: The suprasternal notch is a notch located on the upper edge of the manubrium sterni. It is a surface symbol that can be detected when mouse hair is removed (Figure 2B). The sternal angle is the joint between manubrium sterni and corpora sterni. When the sternum is exposed, it presents as a horizontal white line on the sternum at the same level of costa secunda (Figure 2C). - Carefully separate the skin and superficial fascia and expose the trachea and proximal sternal. Carefully make a 3-4 mm longitudinal midline incision to the sternum starting from the upper edge to the sternal angle (Figure 2D).
NOTE: The cut must not exceed the sternal angle, for there is a risk of causing acute pneumothorax in the mouse. - Gently lift the sternum with forceps and the thymus and arcus aortae beneath it. Carefully use straight forceps and bluntly dissect the target vessel and tissues around it.
- Hold the previously self-made retractor (step 1.4) that contained a wire snare on the end of the user's right hand.
- Carefully pass the snare of the self-made retractor under the aorta between the left common carotid artery and the origin of the right innominate artery (Figure 2E).
- Pass the self-made retractor to the left hand to spare the right hand for other instruments.
- With microscopic tweezers in the right hand, pass a 7-0 silk suture through the loop of the snare at the end and pull out the retractor. This will allow to leave the silk suture around the artery for further ligation (Figure 2F).
NOTE: Be gentle and do not drag the aortic arch too much from its original position. - Place a 27 G needle parallel to the aorta and tie the suture around the needle and the aorta (Figure 2G,H).
NOTE: The sham procedure should be the same, except that there is no aorta ligation. - Hold on to the 27 G needle with ophthalmic forceps and another forceps holding the knot tied to the 27 G needle and aorta. Quickly but gently pull out the 27 G needle from the ligation (Figure 2H).
NOTE: The knot must not be too tight, or it won't be easy to pull out the needle. Be careful when pulling out the needle in case of rupturing the aorta. - Suture the skin with 4-0 synthetic monofilament suture in an interrupted suture pattern (Figure 2I).
4. Postoperative care
- Place the mouse on a warm pad (see Table of Materials) and wait for it to revive. Generally, the mouse revives in about 60 min.
NOTE: The animal must not be left unattended until it regains sufficient consciousness to the main sternal recumbency. - Subcutaneously inject 0.5 mL of warm saline and monitor the animals' fluid homeostasis.
NOTE: This step prevents animals from dehydration and hypovolemic circulation status. - Apply erythromycin ointment to the wound surface every day for 3 days.
- For post-surgical pain treatment, administer 1 mg/L of buprenorphine local anesthetic parenterally to the incision site (see Table of Materials).
- Postoperatively, monitor the mouse for signs of infection and impairment (malnourishment, hunched posture, and ruffled fur).
NOTE: Animals that underwent surgery are not returned to the company of other animals until fully recovered.
5. Ultrasound imaging
- Confirm successful ligation of the transverse aorta by calculating blood flow velocity via an ultra-sound imaging system12,13 (24 MHz, see Table of Materials) 28 days after surgery.
- Anesthetize the animal with 4.5% isoflurane in 20% oxygen-enriched air and maintain with 0.5% isoflurane in 20% oxygen-enriched air.
- Place the mouse in a supine position and fix its extremities to the electrode with adhesive tape.
- Using depilatory cream, remove the chest hair and apply an ultrasonic coupling agent to the chest.
- Tilt the platform to the left as far as possible. While keeping the probe in the vertical position, gradually lower it along the right parasternal line on the chest with the notch pointing to the animal's chin.
- Under the B mode of the instrument, adjust the X and Y-axis until the aortic arch and constriction site are clearly visible.
- Click on the Pulse Wave Doppler button to switch to pulse wave mode.
- Adjust the dashed cursor sample volume box distal to the constricted site and find the stenotic jet with the highest velocity.
- Click on the Pulse Wave Doppler button again to get the waveform of aortic flow and measure the peak velocity. Calculate the blood flow velocity according to the peak of the wave patterns (Figure 3A,B).
- Set the imager to B mode. Rest the platform in the horizontal position. Rotate the probe 30° to the left parasternal line.
- Adjust X and Y-axis to obtain a short axis view. Press M mode to show the indicator line and click on Cine Store to save the image for later measurement of cardiac wall thickness, chamber dimension, and ejection fraction.
NOTE: The formula for calculating ejection fraction: 100% * ((LV Vol;d – LV Vol;s) / LV Vol;d). The formula for calculating cardiac output: Stroke Volume * Heart rate (at the first frame drawn) / 1000. Most indexes such as ejection fraction and cardiac output can be measured automatically via the ultrasonic imaging system's software. LV Vol;d: diastolic volume of the left ventricle. LV Vol;s: systolic volume of the left ventricle.
Representative Results
After successful TAC surgery, pressure overload was detected using an ultrasound imaging system. Four weeks after surgery, mice develop decreased heart function. In the present study, the efficacy of TAC surgery was validated via ejection fraction (EF), fractional shortening (FS), left ventricular mass (LV mass) and left ventricular internal diameter (LVID) of mice who underwent TAC surgery after 4 weeks. EF was significantly reduced in TAC mice after 4 weeks compared to sham mice (47% ± 10% vs. 78% ± 4%, p < 0.0001) (Figure 4A). LV mass was significantly elevated in TAC mice (158.1 ± 50.5 vs. 91.8 ± 21.7 mg, p = 0.0226) (Figure 4B). FS was significantly reduced in TAC mice (23 ± 5 vs. 46% ± 3%, p < 0.0001) (Figure 4C). LVID was significantly elevated in TAC mice (2.88 ± 0.39 vs. 1.81 ± 0.52 mm, p = 0.0044) (Figure 4D). Data represents six mice each for TAC and Sham groups. Due to the small invasive procedures, the survival rate is rather high and mainly dependent on bleeding, which can be reduced to less than 5% for a skilled performer. When fully mastered, the general survival rate presented in C57BL/6J mice after 4 weeks is over 95%. An unpaired t-test was performed to compare the sham and TAC groups. All data are presented as mean ± SEM (error bars).

Figure 1: Self-made snare containing retractor for passing silk suture around the aortic arch. (A) Overall view of the retractor.(B) Detail of the retractor. Arrow indicates the snare for the silk to pass through. Please click here to view a larger version of this figure.
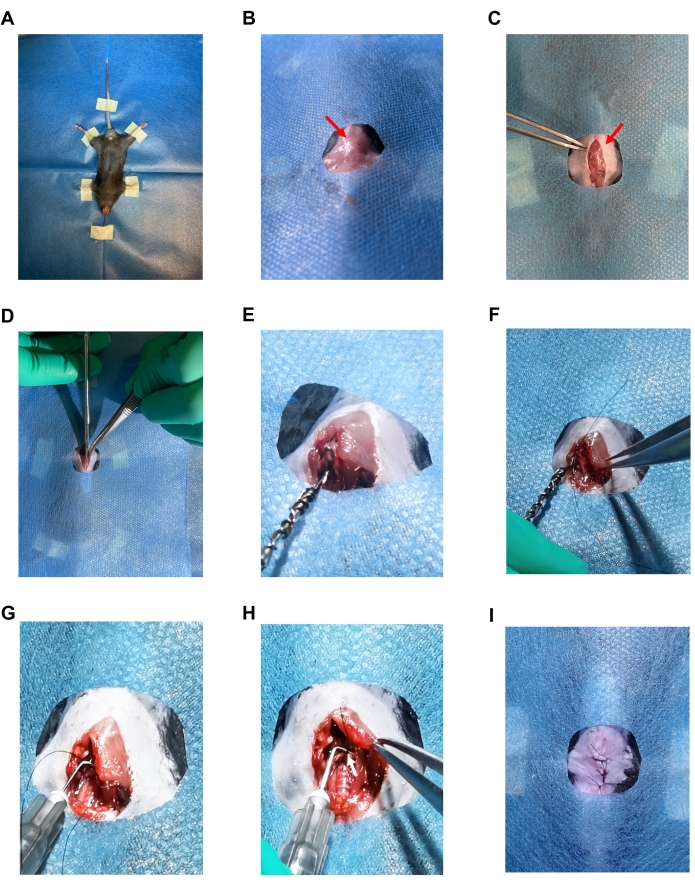
Figure 2: Images of the TAC surgery. (A) Supine position mouse fixed with tape and suture. (B) Sterile drape showing only the surgical area. (C) 1.5 cm vertical skin incision. The red arrow indicates a sternal angel. (D) Longitudinal midline incision that was made to the sternum. (E) Image showing the snare of the self-made retractor passing under the aorta. (F) Image showing the 7-0 silk suture passing through the snare loop. (G) A 27 G needle that was placed parallel to the aorta. (H) Ligation of the aorta with the 27 G needle. The white arrow indicates a ligation knot. (I) Sutured skin with 4-0 silk suture. Please click here to view a larger version of this figure.
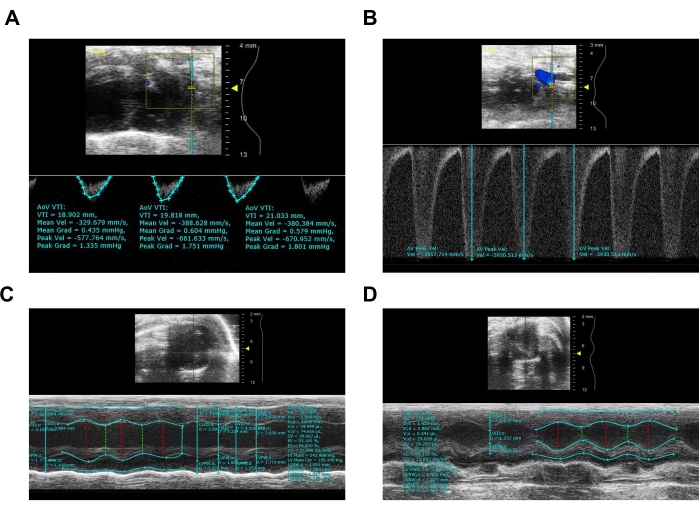
Figure 3: Representative image from ultrasound imaging system of sham and TAC mice after 4 weeks. (A) Pulsed-wave Doppler imaging of sham aortic arch. (B) Pulsed-wave Doppler imaging of aortic arch after TAC. (C) M-mode image of sham mouse calculating EF, LV Mass, wall thickness, and LVID. (D) M-mode image of TAC mouse calculating EF, LV Mass, wall thickness, and LVID. Please click here to view a larger version of this figure.
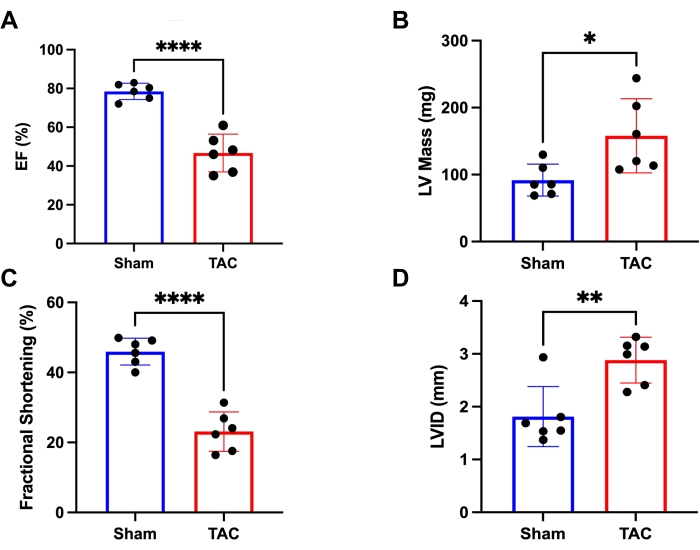
Figure 4: Heart function measured via Ultrasound Imaging System. (A) Ejection fraction (EF) of the mice in two groups. (B) The left ventricular mass (LV mass) of the mice was in two groups. (C) Fractional shortening (FS) of the mice in two groups. (D) The left ventricular internal diameter (LVID) of the mice in two groups. *p < 0.05, **p < 0.005, ***p < 0.0005. Data represents six mice per group. Please click here to view a larger version of this figure.
Discussion
The induction of sustained pressure overload can gradually cause cardiac hypertrophy and heart failure. This model has been used in numerous laboratories around the world14,15,16. The protocol provided an improved TAC method that does not need microsurgical skills or mechanical ventilation.
The most important step in this protocol is passing silk suture under the aortic arch. When the snare has hooked the aortic arch, all moves must be gentle to reduce unnecessary traction to the artery. Also, the suture around the aorta must not be too tight in case of difficulty when pulling the spacer out. After the operation, abundant food and water are also important for the mouse to recover rapidly.
Previous manuscripts have provided other methods for TAC. Eichhorn et al. published a closed chest method that ligates the transverse aorta10. The whole procedure allows the ribs to remain intact, thus causing very small trauma. Zaw et al. provided a TAC method without entering the pleural cavity17. Tavakoli et al. presented a minimally invasive transverse aortic constriction that does not need intubating and ventilation11. All the above techniques require microsurgical skills. In addition, Lao et al. provided a method to produce TAC models with absorbable sutures18. The protocol in this study offers an alternative way to rapidly (within 10 min) conduct TAC surgery that does not need to operate under a microscope. Minimizing surgical trauma benefits the mice and reduces confounding factors during the experiment. Unlike the open-chest model, this model is minimally invasive and does not affect the normal breathing dynamics of the mouse. When fully mastered, the survival rate of this technique is over 95%. Also, it does not need mechanical ventilation and microscopic surgery skills; a self-made reusable retractor will do all the trick, avoiding systemic inflammatory effects induced by ventilation19. All these together significantly simplify the operating procedure.
There are some limitations of this technique. The acute increase of afterload does not fully reflect the gradual progression of arterial hypertension. The discrepancies in pathophysiology between multifactorial heart failure mouse models and clinical heart failure patients have raised concerns among researchers20. The pathophysiology presented in mice cannot be completely applied to humans.
In conclusion, this protocol provides an alternative procedure to conduct TAC, which can facilitate investigators when inducing heart failure or cardiac hypertrophy in mice.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work is funded by the National Natural Science Foundation of China (NSFC 81822002). We thank all the members who took part in this work.
Materials
| 4-0 nonabsorbable suture | Jinhuan | HM403 | Used for suturing the skin |
| 5 mL syringe | Haifuda Technology Co., Ltd. | BD-309628 | Used for making snare containing retractor |
| 7-0 nonabsorbable suture | Jinhuan | HM701 | Used for aorta ligation |
| Animal temperature monitor | Kaerwen | FT3400 | Used for monitoring body temperature |
| Buprenorphine | Sigma | B-044 | Used for post-surgical pain treatment |
| Depilatory cream | Veet | N/A | Used for remove body hair from the surgical area |
| Heating Pad | Xiaochuangxin | N/A | Used for maintaining body temperature |
| Ibuprofen | MCE | HY-78131 | Used for post-surgical pain treatment |
| Iron wire (0.5 mm) | Qing Yuan | Iron wire #26 | Used for making snare containing retractor |
| Microscopic tweezers | RWD | F12006-10 | Used for penetrating and separating the tissue to open operation space |
| Needle holder | RWD | F12005-10 | Used for pinching off the tip of gauge needle and blunting it |
| Ophthalmic forceps | RWD | F14012-10 | Used for holding skin and other tissues |
| Ophthalmic scissors | RWD | S11001-08 | Used for making sking incision of mouse |
| Pentobarbital sodium | Sigma | P3761 | Used for mouse anesthesia |
| Sterile operating mat | Hale & hearty | 211002 | Used for placing animal during surgery |
| Ultra-sound imaging system | Fujifilm visualsonics | vevo1100 | Used for measure the blood flow velocity, left ventricular wall thickness and ejection fraction, https://www.visualsonics.com/product/imaging-systems/vevo-1100 |
Riferimenti
- Heidenreich, P. A., et al. AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 145 (18), 895 (2022).
- McDonagh, T. A., et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. European Heart Journal. 42 (36), 3599 (2021).
- Lv, B., et al. Induction of myocardial infarction and myocardial ischemia-reperfusion injury in mice. Journal of Visualized Experiments. (179), e63257 (2022).
- Curaj, A., Simsekyilmaz, S., Staudt, M., Liehn, E. Minimal invasive surgical procedure of inducing myocardial infarction in mice. Journal of Visualized Experiments. (99), e52197 (2015).
- Nakamura, M., Sadoshima, J. Mechanisms of physiological and pathological cardiac hypertrophy. Nature Reviews Cardiology. 15 (7), 387-407 (2018).
- Wang, H., et al. Bibliometric analysis on the progress of chronic heart failure. Current Problems in Cardiology. 47 (9), 101213 (2022).
- Riehle, C., Bauersachs, J. Small animal models of heart failure. Cardiovascular Research. 115 (13), 1838-1849 (2019).
- Melleby, A. O., et al. A novel method for high precision aortic constriction that allows for generation of specific cardiac phenotypes in mice. Cardiovascular Research. 114 (12), 1680-1690 (2018).
- Rockman, H. A., Wachhorst, S. P., Mao, L., Ross, J. ANG II receptor blockade prevents ventricular hypertrophy and ANF gene expression with pressure overload in mice. The American Journal of Physiology. 266, 2468-2475 (1994).
- Eichhorn, L., et al. A closed-chest model to induce transverse aortic constriction in mice. Journal of Visualized Experiments. (134), e57397 (2018).
- Tavakoli, R., Nemska, S., Jamshidi, P., Gassmann, M., Frossard, N. Technique of minimally invasive transverse aortic constriction in mice for induction of left ventricular hypertrophy. Journal of Visualized Experiments. (127), e56231 (2017).
- Lang, R. M., et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Journal of the American Society of Echocardiography. 28 (1), 1-39 (2015).
- Li, L., et al. Assessment of cardiac morphological and functional changes in mouse model of transverse aortic constriction by echocardiographic imaging. Journal of Visualized Experiments. (112), e54101 (2016).
- Wang, X., et al. ATF4 protects the heart from failure by antagonizing oxidative stress. Circulation Research. 131 (1), 91-105 (2022).
- Li, J., et al. GCN5-mediated regulation of pathological cardiac hypertrophy via activation of the TAK1-JNK/p38 signaling pathway. Cell Death & Disease. 13 (4), 421 (2022).
- Syed, A. M., et al. Up-regulation of Nrf2/HO-1 and inhibition of TGF-beta1/Smad2/3 signaling axis by daphnetin alleviates transverse aortic constriction-induced cardiac remodeling in mice. Free Radical Biology and Medicine. 186, 17-30 (2022).
- Zaw, A. M., Williams, C. M., Law, H. K., Chow, B. K. Minimally invasive transverse aortic constriction in mice. Journal of Visualized Experiments. (121), e55293 (2017).
- Lao, Y., et al. Operating transverse aortic constriction with absorbable suture to obtain transient myocardial hypertrophy. Journal of Visualized Experiments. (163), e61686 (2020).
- Veldhuizen, R. A., Slutsky, A. S., Joseph, M., McCaig, L. Effects of mechanical ventilation of isolated mouse lungs on surfactant and inflammatory cytokines. European Respiratory Journal. 17 (3), 488-494 (2001).
- Withaar, C., Lam, C. S. P., Schiattarella, G. G., de Boer, R. A., Meems, L. M. G. Heart failure with preserved ejection fraction in humans and mice: embracing clinical complexity in mouse models. European Heart Journal. 42 (43), 4420-4430 (2021).

