Generation of Hypoparathyroid Rats via Carbon-Nanoparticle-Assisted Parathyroidectomy
Summary
An animal model of acquired hypoparathyroidism (HypoPT) is crucial to understanding how HypoPT affects mineral ion homeostasis and to verify the effectiveness of novel treatments. Here, a technique is presented to generate an acquired hypoparathyroidism (AHypoPT) rat model by parathyroidectomy (PTX) using carbon nanoparticles.
Abstract
Hypoparathyroidism (HypoPT) is a rare disease involving the parathyroid glands that is characterized by a reduced secretion or potency of the parathyroid hormone (PTH), which leads to high serum phosphorus levels and low serum calcium levels. HypoPT most commonly results from accidental damage to the glands or their removal during thyroid or other anterior neck surgery. Parathyroid/thyroid surgery has become more common in recent years, with a corresponding rise in the occurrence of HypoPT as a postoperative complication. There is a critical need for a HypoPT animal model to better understand the mechanisms underlying the effects of HypoPT on mineral ion homeostasis and to verify the therapeutic effectiveness of novel treatments. Here, a technique is reported to create acquired HypoPT in male rats by performing parathyroidectomy (PTX) using carbon nanoparticles. The rat model shows great promise over the mouse models of hypoparathyroidism. Importantly, the human PTH receptor binding region has an 84.2% sequence similarity with that of the rat, which is higher than the 73.7% similarity shared with mice. Moreover, the effects of estrogen, which can affect the PTH/PTHrP receptor signaling pathway, have not been fully investigated in male rats. Carbon nanoparticles are lymphatic tracers that stain the thyroid lymph nodes black without affecting their function, but they do not stain the parathyroid glands, which makes them easy to identify and remove. In this study, serum PTH levels were undetectable after PTX, and this resulted in significant hypocalcemia and hyperphosphatemia. Thus, the clinical state of postoperative HypoPT can be remarkably represented in the rat model. Carbon-nanoparticle-assisted PTX can, therefore, serve as an extraordinarily effective and readily implementable model for studying the pathogenesis, treatment, and prognosis of HypoPT.
Introduction
Parathyroid hormone (PTH) is secreted by the parathyroid glands. It is a major modulator of the calcium balance, maintains phosphate metabolism, and participates in bone turnover1,2. Hypoparathyroidism (HypoPT) manifests as a decreased secretion or functional loss of PTH. It is a rare endocrine disorder, with a prevalence of approximately 9-37 per 100,000 person-years3,4,5. HypoPT is characterized by decreased serum PTH and calcium levels accompanied by increased serum phosphorus6,7. HypoPT is classified based on its cause: acquired hypoparathyroidism (AHypoPT) or idiopathic hypoparathyroidism (IHypoPT)8. AHypoPT is more commonly encountered in clinical practice; about 75% of AHypoPT cases are caused by resection or accidental injury of the parathyroid glands during thyroid surgery or other head and neck surgeries. Other causes include radiotherapy and chemotherapy for head and neck tumors and drug toxicity1,8. Upgraded diagnostic methods and an increase in screening for thyroid gland-associated diseases have increased the number of thyroid gland surgical operations. This has led to a corresponding increase in the related parathyroid gland complications9,10.
Easily established animal models with stable characteristics are needed to better investigate AHypoPT and verify the therapeutic effectiveness of novel treatments. Parathyroidectomy (PTX) performed on rats and mice has been reported in previous studies6,11; however, due to the extremely small size of the parathyroid glands and the variability in their anatomical distribution, the success rate is relatively low in practice. Thus, thyro-parathyroidectomy (TPTX) (i.e., the total removal of the thyroid and parathyroid glands) is usually performed to ensure the resection of the parathyroid glands12. However, the resulting low thyroxine levels may complicate studies with this animal model13. HypoPT models established by other methods, such as drug stimulation and gene editing, cannot properly represent the most common AHypoPT pathogenesis. Our group previously used knockout mouse models to label the parathyroid glands and allow the removal of the parathyroid glands without damaging the thyroid glands and surrounding anatomical structures14,15. However, this method utilizes transgenic mouse models, which require a longer development time due to the mating and breeding requirements.
Therefore, we aimed to establish an easily generated model of AHypoPT. This study describes a rat model for PTX using carbon nanoparticle labeling. A carbon nanoparticle suspension of 50 mg/mL, which is commonly used in thyroid surgery, evenly distributes in the thyroid glands after local injection16. The thyroid glands turn black, but the parathyroid glands are left unstained17, thus clearly distinguishing the parathyroid glands from the thyroid glands and allowing the PTX to be performed without affecting the thyroid glands. This method is suitable for rats of different ages. The injection of the carbon nanoparticle suspension is safe and has a negligible effect on thyroid function18. The carbon nanoparticle-labeled PTX rat model generated in this study showed significant hypocalcemia and hyperphosphatemia phenotypes during the 4 week observation period. Thus, this AHypoPT model is easy to establish and has a reproducible phenotype.
Protocol
This study was approved by the Institutional Animal Care and Use Committee at the State Key Laboratory of Oral Diseases, Sichuan University. Permission was obtained from relevant local agencies before the experiment. Eight 8-10 week old male Sprague-Dawley (SD) rats, with an average weight of 200-250 g, were used for the present study. The animals were obtained from a commercial source (see Table of Materials). Food and water were provided ad libitum throughout the experimental period.
1. Preoperative preparation for the generation of carbon-nanoparticle-assisted PTX rats
- Anesthetize the 8-10 week old rats using 2.0%-2.5% isoflurane inhalation, followed by an intraperitoneal (i.p.) injection of tribromoethanol at 10 mL/kg body weight. Guarantee a sufficient depth of anesthesia by testing the absence of the pupillary light reflex. Use vet ointment on the eyes to prevent dryness while under anesthesia.
NOTE: Rats that are 8-10 weeks old are recognized as adult rats. However, this modeling method can be used on rats as young as 7 days old. - Prepare the anesthetized SD rats for surgery by shaving the fur of the ventral neck region in the supine position. Disinfect the operative area using povidone-iodine cotton balls (see Table of Materials).
- Cover the animal with surgical drapes (see Table of Materials), and expose the surgical region, aiming to minimize microbial contamination.
2. Parathyroidectomy (PTX)
- Start at the midpoint between the two ears, and cut a 2 cm incision longitudinally toward the tail using a surgical scalpel. Dissect the fascia and fat layer successively with sharp, curved, serrated forceps.
- Separate the paratracheal muscles, and expose the trachea using relatively blunt forceps under a stereo microscope at 4x-5x magnification.
- Locate the left and right lobes of the butterfly-shaped thyroid glands by the side of the trachea.
- Inject 1 µL of the carbon nanoparticle suspension (see Table of Materials) beneath the membrane of the thyroid glands using a 10 µL syringe with a 30 G beveled needle. After 5 min, irrigate the operative region with saline to clean the extra carbon nanoparticle suspension covering the thyroid gland membrane.
NOTE: The recommended injection point is the mesial portion of the thyroid gland lobe, which has fewer blood vessels. A carbon nanoparticle suspension is commonly used in thyroid surgery due to its ability to detect the lymph nodes. Staining the thyroid glands and leaving the parathyroid glands unstained eases the identification of the latter. - Check to ensure that the thyroid glands turn black while the parathyroid glands remain unstained in ~5 min. Observe the highlighted parathyroid glands under ordinary light using either the light of a stereo microscope or a table lamp.
NOTE: Typically, rodents have two drop-shaped parathyroid glands located on the left and right surfaces of the thyroid glands. Occasionally, additional parathyroid glands may be located further away. - Precisely cut the unstained parathyroid glands with microsurgery forceps and scissors. Use sterile cotton balls for hemostasis or a gelatin sponge if there is more bleeding.
- Close the muscles, fat layers, and skin, layer by layer, with an interrupted horizontal mattress suture using 6-0 polyglactin 910 sutures (see Table of Materials).
- For the sham group, perform all the steps of the preoperative preparation and PTX except step 2.6. Anesthetize the rats, and separate the tissues above the trachea. Locate the parathyroid glands but do not remove them. Conduct postoperative recovery and observation together with the rats of the PTX group.
3. Postoperative recovery and observation
- After surgery, place the rats on a thermostatic electric blanket (37 °C) to maintain their body temperature. Inject buprenorphine hydrochloride 0.01 mg/kg subcutaneously (s.c) every 12 h as postoperative analgesia. Transfer the rats to a sterile cage when they begin to move and try to crawl.
- Closely observe the rats postoperatively for 2 h. Return the rats to the rearing room, observe them routinely, and record their state.
- Draw 10 µL of blood from the tail vein 7 days after the surgery. Measure the serum Ca2+, serum Pi, and serum PTH using appropriate commercial kits (see Table of Materials). A successful parathyroidectomy produces a reduced serum ionized Ca2+ level 2 SD lower than that of sham-operated rats (9.00 mmol/L, n = 16).
NOTE: A statistics and graphing software (see Table of Materials) was used for the statistical analysis. A Student's t-test was used to compare the serum and urinary parameters between the sham and PTX groups. p < 0.05 was considered statistically significant. Serum and urinary Ca2+ and Pi and serum urea and creatinine were measured with commercial kits according to the manufacturers' instructions (see Table of Materials). Serum C-telopeptide of type I collagen and osteocalcin were measured with commercially available ELISA kits (see Table of Materials).
Representative Results
The locations and number of parathyroid glands were initially observed in rats under a dissection microscope. Before the carbon nanoparticle injection, the thyroid glands were a translucent red color, and the parathyroid glands were hardly distinguishable under the microscope (Figure 1A). After the nanoparticle injection, the thyroid glands were stained black, while the parathyroid glands remained unstained (Figure 1B). The careful dissection of the light-colored parathyroid glands left the thyroid glands untouched (Figure 1C). Generally, the parathyroid glands were distributed over the lateral or posterior edges of the thyroid glands.
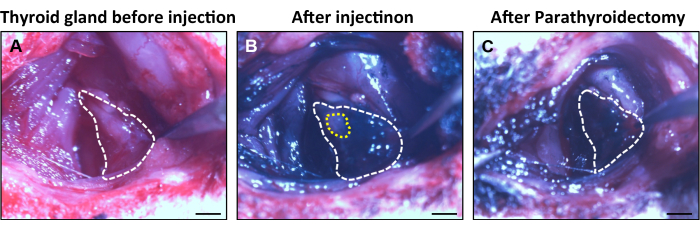
Figure 1: The appearance of the thyroid and parathyroid glands during the surgical procedures. (A) The thyroid glands (white dotted line) are located lateral to the trachea. (B,C) The thyroid glands showed black staining (white dotted line) after the injection of the carbon nanoparticles, while the parathyroid glands (yellow dotted line) exhibited a light color. Scale bars = 2 mm. Please click here to view a larger version of this figure.
The operation time from preoperative preparation to PTX completion was approximately 20 min. The 4 week survival rate of the postoperative rats was 90.9% (60/66). The PTX rats were observed to be hunch-backed 1 week after surgery. A sham-operated control group was simultaneously established by conducting all the steps in the protocol except for step 2.6. All the surviving carbon nanoparticle-labeled PTX rats had a lower mean ionized Ca2+ level, which was 2 SD lower than that of the sham-operated group. The hypoparathyroidism phenotype in the carbon nanoparticle-labeled PTX rats, evidenced by reduced serum calcium, elevated serum phosphate, and undetected PTH, remained steady during the 4 week monitoring period.
At 7 days after surgery, the serum Ca2+ and PTH levels were significantly reduced in the PTX rats compared to the sham group (Ca2+ = 4.97 mmol/L ± 0.99 mmol/L vs. 8.98 mmol/L ± 0.58 mmol/L, p < 0.05; PTH = 13.13 pg/mL ± 6.58 v pg/mL s. 313.06 pg/mL ± 75.24 pg/mL, p < 0.05). Serum Pi was significantly increased after the PTX surgery (Pi = 13.90 mmol/L ± 1.77 mmol/L vs. 7.46 mmol/L ± 1.28 mmol/L). The serum levels of urea and creatinine were comparable between the sham and PTX groups 7 days after the PTX surgery (urea = 8.71 mmol/L ± 0.81 mmol/L vs. 8.84 mmol/L ± 0.89 mmol/L, p > 0.05; creatinine = 49.03 µmol/L ± 13.14 µmol/L vs. 53.15 µmol/L ± 18.28 µmol/L, p > 0.05). At 14 days after the PTX surgery, the urinary Ca2+ and Pi levels were significantly reduced (Ca2+ = 2.33 mmol/L ± 0.53 mmol/L vs. 7.18 mmol/L ± 4.27 mmol/L, p < 0.05; Pi = 2.40 mmol/L ± 1.90 mmol/L vs. 5.29 mmol/L ± 1.52 mmol/L, p < 0.05) (Figure 2).
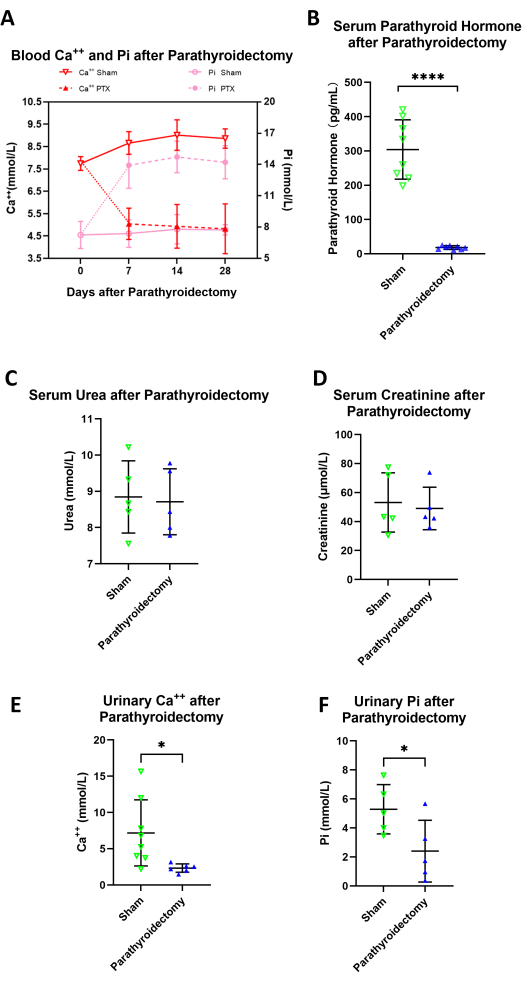
Figure 2: Serum Ca2+, Pi, PTH, urea, and creatinine levels and urinary Ca2+ and Pi levels after carbon-nanoparticle-assisted parathyroidectomy. (A) The PTX rats exhibited stable hypocalcemia and hyperphosphatemia over the 4 week observation period (N = 4). (B) Serum PTH was undetectable in the PTX rats 7 days after the operation (N = 8). (C,D) The serum levels of urea and creatinine were comparable between the sham and PTX groups 7 days after the surgery (N = 5). (E,F) The urinary Ca2+ and Pi levels were significantly reduced 14 days after PTX surgery (N = 8). The error bars indicate the standard deviation. Abbreviations: PTX = parathyroidectomy; Ca++ = ionized calcium in serum; PTH = parathyroid hormone; Pi = ionized phosphorous in serum. Please click here to view a larger version of this figure.
There were no significant differences in body weight between the PTX and sham groups on postoperative day 7 (POD7), POD14, and POD28 (body weight on POD0 = 256.40 g ± 4.76 g vs. 252.56 g ± 6.69 g, p > 0.05; body weight on POD7 = 266.00 g ± 6.93 g vs. 257.44 g ± 30.56 g, p > 0.05; body weight on POD14 = 294.80 g ± 25.90 g vs. 288.22 g ± 37.35 g, p > 0.05; body weight on POD28 = 327.75 g ± 24.82 g vs. 324.17 g ± 57.97 g, p > 0.05). Moreover, serum C-telopeptide of type I collagen (CTX-1) was statistically decreased on POD28 (CTX-1 = 82.03 pg/mL ± 8.98 pg/mL vs. 100.33 pg/mL ± 6.36 pg/mL, p < 0.05). Serum osteocalcin showed no significant difference on POD28 (osteocalcin = 913.66 pg/mL ± 378.03 pg/mL vs. 1066.17 pg/mL ± 549.80 pg/mL, p > 0.05) (Figure 3).
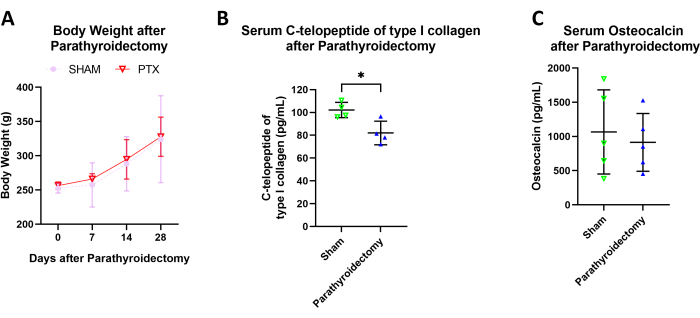
Figure 3: Body weight, blood C-telopeptide of type I collagen, and osteocalcin levels after carbon-nanoparticle-assisted parathyroidectomy. (A) There were no significant differences in body weight between the PTX and sham groups on POD7, POD14, and POD28 (N = 14).(B) The PTX rats exhibited a statistical decrease in serum C-telopeptide of type I collagen (N = 4). (C) There were no significant differences in the serum osteocalcin levels (N = 5). The error bars indicate the standard deviation. Please click here to view a larger version of this figure.
Discussion
Epidemiological reports indicate that the detection of thyroid diseases has increased significantly, and the number of related surgeries performed has increased accordingly19,20. The incidence rate of postsurgical hypoparathyroidism is approximately 7.6%8,21, while the increased morbidity of acquired hypoparathyroidism has caused this rare disease to gain greater research attention. It is, therefore, particularly important to establish a suitable animal model to investigate the pathogenesis of the disease, as well as to test the outcomes of novel therapeutic treatments. However, at present, there are limited animal models available. Moreover, the success rate, survival rate, and difficulty of surgical procedures in producing such models remain problematic. Our group has previously reported two HypoPT models in mice. In PTHcre+/Rosa-mTmG mice, the parathyroid glands were fluorescently labeled to help accurately dissect the parathyroid glands, and this method was also helpful for finding parathyroid glands with abnormal anatomical distribution to improve the success rate of surgery14. Another modeling approach used transgenic mice, in which parathyroid gland cells could be targeted by diphtheria toxin. The parathyroid glands could then be destroyed by the systemic administration of the diphtheria toxin without requiring surgery14,15. However, the abovementioned methods require extensive crossbreeding of transgenic mice, resulting in relatively high time and cost requirements. Moreover, the systemic administration of diphtheria toxin may have widespread side effects. Currently, thyro-parathyroidectomy (TPTX) is the usual procedure performed to ensure the resection of the parathyroid glands12. Although the technique is easily performed and has a high success rate, the damage to the thyroid glands cannot be ignored. The potential impact of injury to or destruction of the thyroid glands on the experimental results might be significant, meaning this is a major limitation of all studies in this field21,22.
In the current study, a carbon nanoparticle suspension, commonly used to visualize the thyroid glands in clinical practice, was injected to enhance the PTX surgery. This method is safe, fast, and highly feasible. It can effectively label the thyroid glands with a black stain and leave the parathyroid glands unstained, which enables the precise identification and dissection of the parathyroid glands while avoiding injury to the thyroid glands. This labeling method has the same effect as that achieved using the fluorescent labeling of transgenic mice but is not limited by the genotype. Furthermore, the surgery time of carbon-nanoparticle-assisted PTX is around 20 min, which saves time compared with the 2 h surgery required for 5-ALA fluorescence identification23. In addition, due to the biosecurity of the carbon nanoparticles24, this modeling method can be used on rats as young as 7 days old. One critical step to be noted during the surgery is that the dosage of the carbon nanoparticle suspension can be adjusted according to the weight of the rats. The volume of carbon nanoparticle suspension used in this study (1 µL) is enough for surgery on adult rats, even if some amount is lost in the syringe. The distribution of all the parathyroid glands is difficult for beginners to identify, and plenty of practice is recommended.
The current study has some limitations. For instance, it is impossible to identify remote parathyroid glands unattached to the thyroids using carbon nanoparticles. If the serum parameters remain unchanged after surgery, it may indicate that some remote parathyroid glands were present and not removed. The staining period required for the optimal differentiation and identification of the parathyroid glands was not measured; however, the thyroid glands were stained properly within 5 min of nanoparticle administration and retained the stain during the entire surgical procedure. The functioning of the thyroid glands during the follow-up period was not recorded in this study. However, in our previous study, which involved utilizing a transgenic mouse model to identify and remove the parathyroid glands, the thyroid gland function was shown to be preserved15. The tolerance of the rats to the carbon nanoparticles was also not tested in this study; however, these nanoparticles have been commercially used as pharmaceuticals in clinical surgeries16. Generally, this method allows researchers to choose an animal with a desired genotype and operation time point. Ultimately, this approach is expected to provide useful rat models for acquired hypoparathyroidism.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work was supported by NSFC grant 81800928, Research Funding from the West China School/Hospital of Stomatology Sichuan University (No. RCDWJS2021-1), and the State Key Laboratory of Oral Diseases Open Funding grant SKLOD-R013.
Materials
| 0.9% Sodium Chloride Solution | Kelun Co. Sichuan, China | ||
| 10 µL 30G NanoFil Syringe | WPI | ||
| 6-0 polyglactin 910 suture with needle | Ethicon, Inc | J510G | |
| Calcium LiquiColor test | EKF | 0155-225 | For Ca2+ analysis |
| Carbon Nanoparticles Suspension Injection | Lummy, Chongqing, China | H20073246 | 1 mL : 50 mg |
| Creatinine (Cr) Assay kit ( sarcosine oxidase ) | Jiancheng, Nanjing, China | C011-2-1 | For creatinine analysis |
| Disposable Scalpel | Shinva, China | ||
| Dumstar Biology forceps | Shinva, China | ||
| Micro Dissecting Spring Scissors | Shinva, China | ||
| MicroVue Rat intact PTH ELISA | Immunotopics | 30-2531 | For the measurement of PTH in rat serum |
| Needle Holder | Shinva, China | ||
| Phosphorus Liqui-UV test | EKF | 0830-125 | For Pi analysis |
| Ply gauze | Weian Co. Henan, China | ||
| Povidone-Iodine | Yongan pharmaceutical Co.Ltd. Chengdu, China | ||
| Prism 9.0 (statistics and graphing software) | GraphPad Software, Inc., San Diego, CA, USA | https://www.graphpad.com/scientific-software/prism/ | |
| Rat C-telopeptide of type I collagen (CTX-I) ELISA Kit | CUSABIO, Wuhan, China | CSB-E12776r | For CTX-I analysis |
| Rat Osteocalcin/Bone Gla Protein (OT/BGP) ELISA Kit | CUSABIO, Wuhan, China | CSB-E05129r | For osteocalcin analysis |
| Safety Single Edge Razor Blades | American Safety Razor Company | 66-0089 | |
| Sprague-Dawley Rats | 8 to 10 weeks old | ||
| Surgical Incise Drapes | Liangyou Co. Sichuan, China | ||
| Urea Assay Kit | Jiancheng, Nanjing, China | C013-2-1 | For urea analysis |
Riferimenti
- Bilezikian, J. P., et al. Hypoparathyroidism in the adult: Epidemiology, diagnosis, pathophysiology, target-organ involvement, treatment, and challenges for future research. Journal of Bone and Mineral Research. 26 (10), 2317-2337 (2011).
- Bilezikian, J. P. Hypoparathyroidism. The Journal of Clinical Endocrinology and Metabolism. 105 (6), 1722-1736 (2020).
- Underbjerg, L., Sikjaer, T., Mosekilde, L., Rejnmark, L. Postsurgical hypoparathyroidism-Risk of fractures, psychiatric diseases, cancer, cataract, and infections. Journal of Bone and Mineral Research. 29 (11), 2504-2510 (2014).
- Underbjerg, L., Sikjaer, T., Mosekilde, L., Rejnmark, L. The epidemiology of nonsurgical hypoparathyroidism in Denmark: A nationwide case finding study. Journal of Bone and Mineral Research. 30 (9), 1738-1744 (2015).
- Astor, M. C., et al. Epidemiology and health-related quality of life in hypoparathyroidism in Norway. The Journal of Clinical Endocrinology and Metabolism. 101 (8), 3045-3053 (2016).
- Rodriguez-Ortiz, M. E., et al. Calcium deficiency reduces circulating levels of FGF23. Journal of the American Society of Nephrology. 23 (7), 1190-1197 (2012).
- Davies, B. M., Gordon, A. H., Mussett, M. V. A plasma calcium assay for parathyroid hormone, using parathyroidectomized rats. The Journal of Physiology. 125 (2), 383-395 (1954).
- Clarke, B. L., et al. Epidemiology and diagnosis of hypoparathyroidism. The Journal of Clinical Endocrinology and Metabolism. 101 (6), 2284-2299 (2016).
- Liu, Y., Shan, Z. Expert consensus on diagnosis and treatment for elderly with thyroid diseases in China. Aging Medicine. 4 (2), 70-92 (2021).
- Sulejmanovic, M., Cickusic, A. J., Salkic, S., Bousbija, F. M. Annual incidence of thyroid disease in patients who first time visit department for thyroid diseases in Tuzla Canton. Materia Socio-Medica. 31 (2), 130-134 (2019).
- Liao, H. W., et al. Relationship between fibroblast growth factor 23 and biochemical and bone histomorphometric alterations in a chronic kidney disease rat model undergoing parathyroidectomy. PloS One. 10 (7), 0133278 (2015).
- Russell, P. S., Gittes, R. F. Parathyroid transplants in rats: A comparison of their survival time with that of skin grafts. The Journal of Experimental Medicine. 109 (6), 571-588 (1959).
- Sakai, A., et al. Osteoclast development in immobilized bone is suppressed by parathyroidectomy in mice. Journal of Bone and Mineral Metabolism. 23 (1), 8-14 (2005).
- Bi, R., Fan, Y., Luo, E., Yuan, Q., Mannstadt, M. Two techniques to create hypoparathyroid mice: parathyroidectomy using GFP glands and diphtheria-toxin-mediated parathyroid ablation. Journal of Visualized Experiments. (121), e55010 (2017).
- Bi, R., et al. Diphtheria toxin- and GFP-based mouse models of acquired hypoparathyroidism and treatment with a long-acting parathyroid hormone analog. Journal of Bone and Mineral Research. 31 (5), 975-984 (2016).
- Huang, Y., et al. Carbon nanoparticles suspension injection for photothermal therapy of xenografted human thyroid carcinoma in vivo. MedComm. 1 (2), 202-210 (2020).
- Zhang, R. J., Chen, Y. L., Deng, X., Yang, H. Carbon nanoparticles for thyroidectomy and central lymph node dissection for thyroid cancer. The American Surgeon. , (2022).
- Long, M., et al. A carbon nanoparticle lymphatic tracer protected parathyroid glands during radical thyroidectomy for papillary thyroid non-microcarcinoma. Surgical Innovation. 24 (1), 29-34 (2017).
- Li, Y., et al. Efficacy and safety of long-term universal salt iodization on thyroid disorders: Epidemiological evidence from 31 provinces of mainland China. Thyroid. 30 (4), 568-579 (2020).
- Powers, J., Joy, K., Ruscio, A., Lagast, H. Prevalence and incidence of hypoparathyroidism in the United States using a large claims database. Journal of Bone and Mineral Research. 28 (12), 2570-2576 (2013).
- Zihao, N., et al. Promotion of allogeneic parathyroid cell transplantation in rats with hypoparathyroidism. Gland Surgery. 10 (12), 3403-3414 (2021).
- Goncu, B., et al. Xenotransplantation of microencapsulated parathyroid cells as a potential treatment for autoimmune-related hypoparathyroidism. Experimental and Clinical Transplantation. , (2021).
- Jung, S. Y., et al. Standardization of a physiologic hypoparathyroidism animal model. PLoS One. 11 (10), 0163911 (2016).
- Chen, W., Lv, Y., Xie, R., Xu, D., Yu, J. Application of lymphatic mapping to recognize and protect parathyroid in thyroid carcinoma surgery by using carbon nanoparticles. Journal of Clinical Otorhinolaryngology, Head, and Neck Surgery. 28 (24), 1918-1920 (1924).

