Real-Time Monitoring and Modulation of Blood Pressure in a Rabbit Model of Ischemic Stroke
Summary
Continuous arterial blood pressure recording allows the investigation of impacts of various hemodynamic parameters. This report demonstrates the application of continuous arterial blood pressure monitoring in a large animal model of ischemic stroke for determination of stroke pathophysiology, impact of different hemodynamic factors, and the assessment of novel treatment approaches.
Abstract
Control of blood pressure, in terms of both absolute values and its variability, affects outcomes in ischemic stroke patients. However, it remains challenging to identify the mechanisms that lead to poor outcomes or evaluate measures by which these effects can be mitigated because of the prohibitive limitations inherent to human data. In such cases, animal models can be utilized to conduct rigorous and reproducible evaluations of diseases. Here we report refinement of a previously described model of ischemic stroke in rabbits that is augmented with continuous blood pressure recording to assess the impacts of modulation on blood pressure. Under general anesthesia, femoral arteries are exposed through surgical cutdowns to place arterial sheaths bilaterally. Under fluoroscopic visualization and roadmap guidance, a microcatheter is advanced into an artery of the posterior circulation of the brain. An angiogram is performed by injecting the contralateral vertebral artery to confirm occlusion of the target artery. With the occlusive catheter remaining in position for a fixed duration, blood pressure is continuously recorded to allow for tight titration of blood pressure manipulations, whether through mechanical or pharmacological means. At the completion of the occlusion interval, the microcatheter is removed, and the animal is maintained under general anesthesia for a prescribed length of reperfusion. For acute studies, the animal is then euthanized and decapitated. The brain is harvested and processed to measure the infarct volume under light microscopy and further assessed with various histopathological stains or spatial transcriptomic analysis. This protocol provides a reproducible model that can be utilized for more thorough preclinical studies on the effects of blood pressure parameters during ischemic stroke. It also facilitates effective preclinical evaluation of novel neuroprotective interventions that might improve care for ischemic stroke patients.
Introduction
Ischemic stroke (IS) is a leading cause of death and long-term disability worldwide, and its prevalence is projected to increase as society ages1. While substantial advances have been made in acute interventions and secondary prevention strategies, adjunctive neuroprotective treatments have not followed apace2,3,4,5,6,7. Further research is needed into stroke pathobiology because mechanisms by which therapies may or may not prove effective are poorly understood. This is largely due to the heterogeneous nature of the stroke patient population, many of whom have numerous comorbidities that confound analysis1. One driver of limitations in research is the absence of tissue-level data-the gold standard in biomedical research-due to the prohibitive morbidity of sampling tissue from the human central nervous system. Specifically, vascular tissue harvesting in a living human would cause a stroke, so vascular tissue is typically only obtained at autopsy, which is under-representative of the general population and skews toward more advanced disease in elderly patients with concomitant diagnoses.
In such cases, when sufficient human data cannot be utilized, animal models can bridge the data gaps. Large animal models of stroke are limited as most large animals used in research are ungulates having a rete mirabile that prevents direct endovascular access to the cerebral arteries8,9,10,11,12,13,14,15,16,17. Rabbits have a long history of use for the investigation of cardiovascular disease, including intracranial pathologies8,9,10,11,12,13,14,15,16,17. Rabbits present an ideal model for cerebrovascular diseases because they are large enough for endovascular catheterization and lack the rete mirabile that precludes intracranial access in other large mammals9,15,16,17. They have been previously utilized specifically for the investigation of IS through precise and well-controlled occlusion of an intracranial artery with a microcatheter18.
Blood pressure (BP) control, both through modulation of absolute BP or BP variability (BPV), the degree to which arterial BP fluctuates around a mean BP, is an emerging potential therapeutic target for IS patients after reports of worse outcomes in those with poorly controlled BP or BPV19,20,21,22. Mechanistic investigation into how changes lead to poor outcomes in IS patients is lacking. This is partly due to the difficulty in obtaining tissue-level data and performing well-controlled analyses in humans. To test interventions that modulate BP or BPV, animal models must be utilized to overcome these limitations. This report describes the successful pairing of a previously validated rabbit model of IS using controlled occlusion of the posterior cerebral artery in conjunction with continuous intra-arterial measurement of BP18. The method presented here improves on the previous approaches to stroke pathophysiology by applying a validated and reproducible stroke model to a system in which precise measurement and control of BP can be achieved. In this refined model, infarct burden can be assessed with post-procedural histopathologic staining of the harvested brain, which is also amenable to various stains and more advanced analyses such as spatial transcriptomics. Additionally, the occluded posterior circulation artery can also be chosen to be evaluated for morbidity analysis following survival procedures.
Protocol
This protocol is approved by the Institutional Animal Care and Use Committee (University of Utah IACUC Protocol Number 21-09021). Mature New Zealand White rabbits are obtained from commercial vendors.
1. Animal acquisition
- Acclimate the animals for the required duration after arrival according to institutional protocol, housing animals socially in a vivarium with standard chow diets. The acclimation period at our institution is 2 weeks.
2. Anesthesia and monitoring
- Induce general endotracheal anesthesia with intramuscular injection of buprenorphine (0.03 mg/kg) followed approximately 30 min later with a ketamine (25-35 mg/kg) and xylazine (3 mg/kg) intramuscular injection. Maintain anesthesia with 1%-5% isoflurane in oxygen administered via an endotracheal tube. During induction, use 100% FiO2, and then titrate down to the lowest FiO2 that maintains a 100% SpO2.
NOTE: Uninterrupted anesthesia is necessary to prevent movement by the animal so that the stroke will be the only disruption of the stroke-induction process. This also prevents spikes in BP that would result from agitation that could arise from inadequate anesthesia. Consistent oxygenation is also important to control for achieving comparable strokes. These measures are all accounted for in the representative results described below. - Confirm adequate depth of anesthesia by applying noxious stimuli to the toe. Apply veterinary eye ointment on the eyes to prevent dryness.
- Monitor oxygen saturation by a pulse oximeter placed on the ear. Obtain intravenous access with an angiocatheter in an auricular vein. Ensure that it is secured with a suture or adhesive transparent film dressing. To mitigate vasospasm, place 0.25 inch of transdermal nitroglycerin on the inside of the ear after induction of anesthesia.
- Provide maintenance fluids with normal saline at a rate of 1 cc/kg/h. Place an esophageal temperature probe to monitor body temperature. Maintain normothermia (33-37 °C) as needed with warming blankets placed under the animal.
3. Surgical preparation
- Place the rabbit in a supine position on a fluoroscopy-compatible operative table. Extend the head as it optimizes positioning for subsequent angiographic views. Rabbits have exquisitely sensitive arteries prone to vasospasm after instrumentation.
- Remove fur from both the inguinal regions using electric clippers. Next, palpate the bilateral femoral arterial pulses to confirm adequate clearance by trimming bilaterally. Prepare the skin with scrubs of chlorhexidine and alcohol, and then drape the skin in the usual sterile fashion.
- Administer local anesthesia by subcutaneously injecting 2 mL of 1% lidocaine in the bilateral inguinal regions. Make a 5 cm surgical incision with a number 10 blade at the site where lidocaine was injected. Use blunt dissection to expose the neurovascular bundle (Figure 1A). If needed, extend the incision to adequately expose an arterial segment large enough for access.
- Upon isolation of the neurovascular bundle, drip several drops of 1% lidocaine on the artery to prevent vasospasm. Separate the artery gently from the vein and the adjacent nerve using forceps. Identify the artery by the characteristic appearance of its muscular wall compared to the thin walls of the vein. The artery will have brighter blood, while the vein will contain darker blood.
4. Arterial access
- After the artery has been isolated, pass the right-angle forceps under the vessel. Grasp two vessel loops with the instrument and gently pass them under the artery. Situate one each at the upstream and downstream ends of the exposed vessel.
- Subject the artery to a gentle traction by pulling the vessel loops. At this point, inspect the vessel for any residual tissue, and remove it with gentle dissection (Figure 1B). This increases the chances of successful access.
- Use a 22 G angiocatheter for access. Slightly advance the catheter itself over the inner needle since this often sticks when completely seated and can dislodge the device during access attempts.
- After dissecting the vessel and preparing the angiocatheter, drip lidocaine on the vessel again. The artery will visibly dilate, which increases the chances of successful access and placement of a sheath using the Seldinger technique.
- Apply gentle traction to the downstream vessel loop to engorge the artery by reducing outflow. This also stabilizes the vessel for the access attempt. Slowly advance the needle of the angiocatheter into the middle of the exposed arterial segment (Figure 1C). When a flash of blood is seen in the angiocatheter and the chamber at its hub, advance the catheter over the needle into the arterial lumen.
- If the access attempt is unsuccessful, achieve hemostasis by applying traction on the upstream vessel loop. Flush the angiocatheter with saline and replace it on its introducer needle for additional attempts.
- When the angiocatheter is successfully placed in the vessel to its hub, advance a Cope microwire through the angiocatheter lumen and into the aorta (Figure 1D). Remove the angiocatheter over the wire and replace it with a 5 French slim hydrophilic sheath (Figure 1E).
- Confirm the return of the arterial blood through the sidearm tubing by opening the three-way valve. Flush the sheath with 0.9% saline and lock the valve closed during flushing.
- Secure the sheath hub to the adjacent skin with an additional 3-0 silk suture. Repeat this process for the contralateral femoral artery. For achieving higher efficiencies, two operators can simultaneously work while focusing on one artery each.
5. Cervicocerebral angiography and intracranial access
- Under fluoroscopic visualization, advance a 4 French glide catheter over a 0.035-inch glidewire inserted through the left femoral sheath. Position the tip of the glide catheter in the proximal left vertebral artery. Remove the wire and flush the catheter with heparinized 0.9% saline.
- Perform angiography by hand injecting the left vertebral artery with iodinated contrast under low magnification to visualize the entire head and neck (Figure 2A). Modulate the injection of the contrast solution by starting with a low-pressure injection that crescendos to visualize the entire vasculature.
NOTE: Sufficient injection to visualize the reflux down the right vertebral artery is needed, as this angiographic image will be used for roadmap guidance to select the right vertebral artery efficiently. Gentle injection is needed to minimize vasospasm or more profound injury. Additionally, excessive force or volume can induce transient motion from the animal even under deep anesthesia. - For the left vertebral injection, inject 50% contrast diluted in normal saline with a gentle crescendo from a 3 cc syringe. Injecting 1-2 cc of the dilute contrast is typically sufficient. Determine the adequate amount of injection by checking the reflux down the right vertebral artery and into the right subclavian artery. During this injection, also note the posterior cerebral and superior cerebellar arteries, one of which will be the target to occlude with the microcatheter.
- Prepare a 2.4 French flow-directed microcatheter with a 0.010-inch microwire. Make a c-shape on the tip of the microwire. Under roadmap guidance, advance the microcatheter inside a 4 French glide catheter through the right femoral sheath and over the wire into the right vertebral artery. Due to the propensity for catheter-induced vasospasm, minimize the device manipulation time and the number of catheter attempts performed.
- Advance the microcatheter through the cervical segment of the right vertebral artery. To best pass the sharp turn from the V2 to V3 segment, advance the microcatheter alone while the microwire is back proximal to its tip. Leading with the microwire at this point will often cause selection of small side branches of the vertebral artery and can be the source of substantial vasospasm.
- After passing the sharp turn from V2 to V3, the microcatheter often passes easily to the proximal basilar artery. At this point, advance the microwire and select the desired posterior cerebral or superior cerebellar arteries. Microcatheter injections are not advised given the fragile nature of the intracranial arteries.
- Advance the microcatheter over the microwire into the target artery. Choose a proximal position as it is typically safest in the posterior to communicate due to its angulation at its origin. A deeper position is feasible in the superior cerebellar artery (Figure 2B).
- Repeat the angiogram by injecting the left vertebral artery catheter with high magnification over the head to confirm occlusion of the target artery (Figure 2B-C). For optimal imaging, inject full strength contrast in the 3 cc syringe. Typically, no more than 1 cc will be required for adequate opacification of all intracranial arteries.
- Gently remove the microwire from the microcatheter under fluoroscopic visualization to confirm a stable position. Place a stopcock on the hub of the microcatheter and close the stopcock to prevent blood loss from retrograde blood flow. Remove the left vertebral catheter to make the left femoral access sheath available.
- During the ensuing occlusion period, acquire intermittent fluoroscopic images to confirm a stable position of the occlusive microcatheter. Results of posterior cerebral artery occlusion periods ranging from 60-240 min have been published previously18.
6. Blood pressure measurement and modulation
- While one femoral access site is utilized for the occlusive intracranial microcatheter, use the contralateral sheath for BP measurement.
- Record continuous arterial BP readings with a 3 French gauge piezoresistive sensor, placed through a femoral sheath and advanced until the sensor tip is in the lower thoracic aorta. Connect this sensor to data acquisition hardware and visualize measured pressures with its associated software. Observe BP in the pressure visualization window. BP recordings can be exported into a spreadsheet for visualization in the statistics software.
- Alternatively, if mechanical manipulation of BP is desired using a balloon catheter, advance a 4 French 5 mm Fogarty balloon catheter through the available femoral sheath. Position the balloon in the infrarenal aorta. Use the 0.025-inch inner lumen for pressure tracing to continuously monitor BP upstream of the balloon and the 4 French diameter of the balloon for a second BP tracing line to be connected to the sheath for continuous monitoring of BP downstream of the balloon.
7. Euthanasia and tissue harvesting
- Remove the occlusive microcatheter after 3 h, and then continue arterial BP measurement and modulation for the additional desired period. A standard recovery period of 3 h is used for visualization of a completed infarct on subsequent histology.
- After completing the prescribed occlusion and recovery times, ensure that the animal is in a surgical plane of anesthesia and perform euthanasia (perfusion fixation with phosphate buffered solution, followed by decapitation after confirmation of absence of cardiac activity). Alternatively, perform perfusion fixation by infusing perfusate through a femoral sheath, and then transecting a jugular vein, the inferior vena cava, or the right atrium.
NOTE: Perfusion may be preferable for some postmortem analyses since gene expression or biomarker values may be affected by the solution. Both the techniques have been performed successfully by our group. - In acute procedures with immediate harvesting of the brain, confirm euthanasia and decapitate the animal. Remove calvarium in a piecemeal fashion with rongeurs, starting at the occipital ridge and working anteriorly until the brain can be harvested intact. Place the brain in formalin or optimal cutting temperature solution and flash freeze, depending on the type of tissue analysis desired.
Representative Results
In the initial experiments with this model, our group successfully achieved the desired outcome of a posterior cerebral or superior cerebellar artery occlusion in 12 out of 14 animals (85.7%). For the experiment, seven males and seven females were studied. The mean animal weight was 3.6 kg (± 0.46 kg). In the two animals in which success was not achieved, profound catheter-induced vasospasm precluded safe access to the intracranial circulation. In one rabbit, intracranial access could not be obtained due to occlusive vasospasm, and in the other animal, intracranial arterial perforation occurred during attempted catheterization, which was likely due to attempting to position the microcatheter too far distally in the posterior cerebral artery.
In all the animals, the brain was successfully harvested and subjected to histopathological analysis with either hematoxylin and eosin (H&E) staining or 2% triphenyltetrazolium chloride (TTC). In keeping with previously published results of the occlusion model, larger infarct volumes occurred with longer occlusion durations, which have been successfully performed from 60 to 240 min18. H&E stain images after 90 min of occlusion with 120 min of reperfusion are provided in Figure 3.
Baseline arterial BPs below normotension (40-60 mmHg systolic BP) were noted in all the animals after induction of anesthesia without use of vasopressors or inflation of an intra-aortic balloon. Partial inflation of the balloon has demonstrated immediate increase in systolic BP, with a sample BP tracing provided in Figure 4. This figure includes tracing of a short duration to visualize both the near-instantaneous change following inflation of the intra-aortic balloon as well as the changes throughout each cardiac cycle.
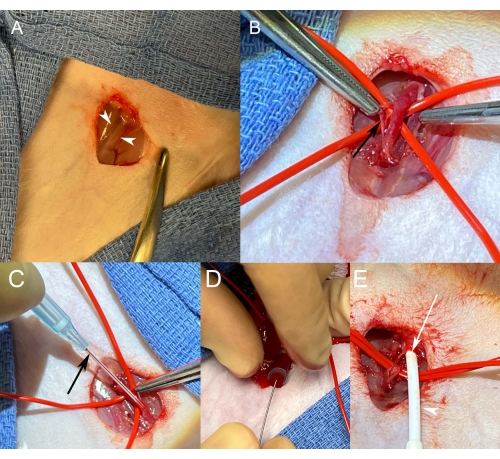
Figure 1: Femoral artery access. (A) Surgical exposure of the right femoral neurovascular bundle before blunt dissection. White arrowheads indicate the medial and lateral borders of the bundle to be exposed with dissection. (B) After isolation, the artery becomes engorged when dripping with lidocaine solution and applying gentle traction to the downstream vessel loop. The vessel can be cleaned by gentle dissection of tissue (black arrow) off the adventitia. (C) Maintaining gentle tension on the vessel, a 22 G angiocatheter is advanced into the vessel. After seeing blood flash in the angiocatheter (black arrow) and its chamber, the angiocatheter is gently advanced into the artery. (D) With the angiocatheter advanced in the artery to its hub, a Cope wire is advanced into the artery through the angiocatheter. (E) After removing the angiocatheter over a Cope microwire, a vascular sheath (white arrowhead) is advanced along with its inner introducer over the wire. The sheath is seen entering the artery, the wall of which can be seen at the arteriotomy site (white arrow). Please click here to view a larger version of this figure.
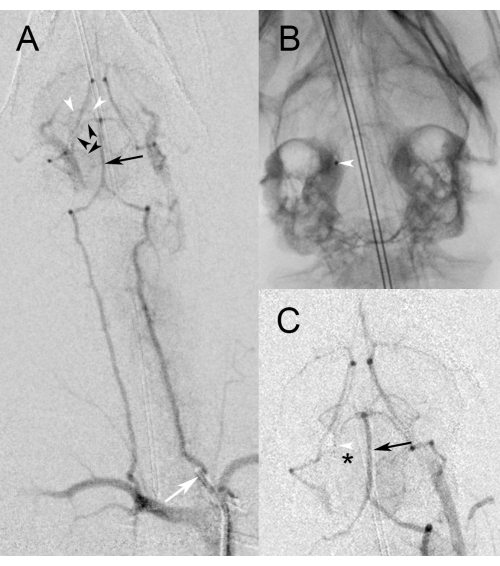
Figure 2: Angiographic images. (A) Low magnification view of digital subtraction angiography during injection of the proximal left vertebral artery (white arrow) demonstrates filling of the basilar artery (black arrow). Note the reflux back down the right vertebral artery into the subclavian artery, which can be used as a roadmap to guide catheterization. Black arrowheads delineate the course of the right superior cerebral artery that will be targeted for occlusion. White arrowheads identify the posterior cerebellar artery, which can also be targeted. (B) High magnification spot fluoroscopic image demonstrates the microcatheter in the right superior cerebellar artery from a right vertebral approach. The white arrowhead indicates the radiopaque marker at the microcatheter tip. (C) High magnification digital subtraction angiography during injection of the left vertebral artery demonstrates persistent filling of the basilar artery (black arrow) while the microcatheter runs through it. No filling is noted beyond the mid-right superior cerebellar artery, where the tip of the microcatheter is indicated by the white arrowhead. The black asterisk identifies non-perfused territory downstream to the occlusion in the superior cerebellar artery. Please click here to view a larger version of this figure.
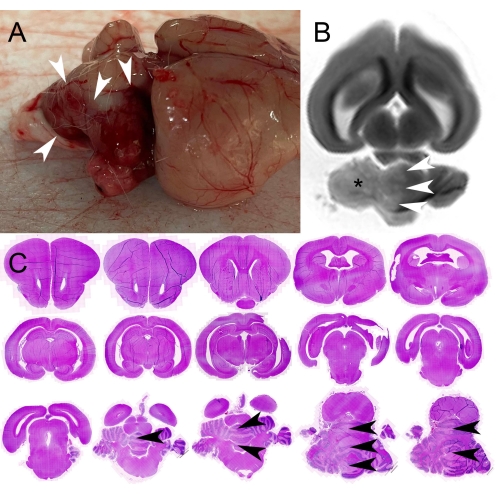
Figure 3: Pathology images. (A) Photograph of intact harvested brain showing the surface of the brain from the animal's right. Note the darkened appearance of the superior cerebellum indicating petechial hemorrhage in the acutely infarcted tissue. White arrowheads demarcate the margin of the infarction. (B) Long-axis T2-weighted magnetic resonance image of the intact brain in formalin. Note the increased signal in the right cerebellum (asterisk), consistent with the infarct, the border of which is delineated by white arrowheads. (C) Bright field images of 1.5 mm thick serial coronal sections following hematoxylin and eosin (H&E) staining demonstrates infarction in the right cerebellum, the margin of which is indicated by black arrowheads on multiple slices. These sections were sliced from blocks of a harvested rabbit brain cut in the coronal plane with a cutting matrix. Please click here to view a larger version of this figure.
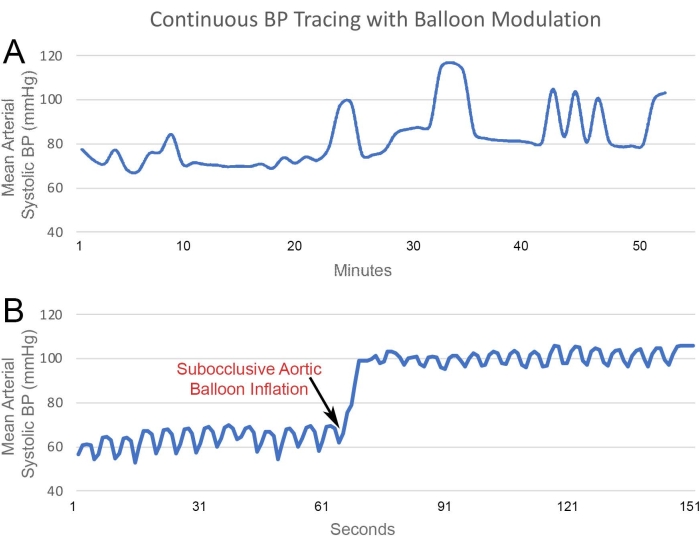
Figure 4: BP Monitoring. BP pressure tracing from a Fogarty balloon catheter positioned in the infrarenal aorta. (A) Data from approximately 1 h of BP monitoring demonstrates real-time arterial pressure changes with changes in balloon inflation. (B) Short-term tracing demonstrates the pressure changes throughout the cardiac cycle. Additionally, small, rapid changes are noted from respiratory variability, which is physiologically normal. An immediate near-doubling of measured BP is noted following the inflation of the balloon. Please click here to view a larger version of this figure.
Discussion
Substantial progress has been made in the management of IS, particularly considering advances in acute intervention and secondary prevention strategies. However, more work can be done to improve care for IS patients. Limited progress in other aspects of IS treatment, particularly in the realm of neuroprotection, likely results from the limitations in pathophysiological understanding of mechanistic processes at the tissue and molecular level. Impactful data from humans is unrealistic and likely impossible to acquire. In such circumstances, tissue-level data from animal models can bridge knowledge gaps and affect meaningful change.
As detailed above, rabbits provide an optimal combination of size, physiology, and anatomy for the investigation of cerebrovascular pathologies18. Lacking a rete mirabile, there are no structural barriers to the intracranial arteries. Additionally, the intracranial vessels are large enough to accommodate endovascular devices, which is not similarly feasible in rodent models. Data from the intracranial tissues can be analyzed in multiple ways, whether through established histopathological and immunohistochemical stains or cutting-edge methods such as endovascular biopsy samples analyzed with single-cell RNA sequencing or spatial transcriptomics of intact tissues9,15,16,17,18. This reported protocol improves upon previous reports of the rabbit occlusion model given its application of multiple posterior circulation arteries and emphasis on the practical steps to mitigate vasospasm or arterial injury18. This protocol is also an improvement on the existing reports given the feasible and reproducible methods for continuous BP monitoring.
While rabbits present immense potential for advances in the pathobiological understanding of cerebrovascular diseases, they also present technical challenges. According to anecdotal reports from veterinary collaborators, rabbits have a well-earned reputation for being hemodynamically unstable. Hypotension during anesthesia induction is inevitable. To mitigate effects, prompt intubation after sedation is needed. Efficient exposure and prompt access of a femoral artery allow early hemodynamic monitoring by virtue of BP measurement. However, this must be balanced with meticulous techniques to limit blood loss during access. Limiting blood loss must also be a priority throughout all the steps of the endovascular procedure, which can be achieved with concerted observation during device exchanges and using rotating hemostatic valves on all catheters. As the entire protocol occurs over several hours, replacement intravenous fluids are also needed to counteract blood loss and insensible losses. Finally, rabbit arteries are profoundly sensitive and prone to vasospasm, which can be prepared for with topical nitroglycerine, as described above. Minimal instrumentation can limit vasospasm, and this is best achieved by concerted planning to minimize arterial exposure to mechanical stressors. Lidocaine dripped on the artery can counteract this reaction, and verapamil (1 mg/mL) can be similarly dripped on the vessel or infused into the artery through a catheter. Finally, pausing for few minutes can allow vasospasm to resolve.
Despite the challenges, the similarity of rabbit anatomy and physiology to humans can be useful in modeling human diseases and the ability to minimize these challenges make them suitable for experimentation. Coupled with cutting-edge sequencing and imaging, rabbits offer a remarkable opportunity for investigating cerebrovascular disease. In particular, the methods described above allow the well-controlled study of IS and the effects of various hemodynamic parameters on its pathophysiology, diagnosis, and management.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
The research reported in this publication was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health under Award Numbers UL1TR002538 and KL2TR002539 and by Transformational Grant 19TPA34910194 from the American Heart Association.
Materials
| 3-0 Silk Suture | Ethicon | A184H | |
| Buprenorphine | Sigma-Aldrich | B9275 | |
| Catheter | Terumo | CG415 | 4F glide catheter |
| Endovascular Pressure Sensor | Millar | SPR-524 | |
| Euthasol | Virbac | PVS111 | |
| Guidewire | Terumo | GR1804 | |
| Iohexol | ThermoFisher | 466651000 | Iodinated Contrast |
| Ketamine | Biorbyt | orb61131 | |
| LabChart Software | ADInstruments | ||
| Lidocaine | Spectrum | LI102 | |
| Microcatheter | Medtronic | EV3 105-5056 | Marathon Microcatheter |
| Microwire | Medtronic | EV3 103-0608 | Mirage Microwire |
| PowerLab | ADInstruments | ||
| Rabbit Brain 2mm Coronal Cutting Matrix | Ted Pella | 15026 | |
| Saline | FisherScientific | 23-535435 | |
| Sheath | Merit Medical | PSI-5F-11 | |
| Xylazine | ThermoFisher | J61430.14 |
Riferimenti
- the American Heart Association. Heart Disease and Stroke Statistics-2022 Update: A Report From the American Heart Association. Circulation. 145 (8), 153 (2022).
- Jadhav, A. P., Campbell, B. C. V. Ongoing advances in medical and interventional treatments of large vessel occlusion stroke. Stroke. 52 (3), 1115-1117 (2021).
- Caprio, F. Z., Sorond, F. A. Cerebrovascular disease: Primary and secondary stroke prevention. The Medical Clinics of North America. 103 (2), 295-308 (2019).
- Kleindorfer, D. O., et al. Guideline for the prevention of stroke in patients with stroke and transient ischemic attack: A guideline from the American Heart Association/American Stroke Association. Stroke. 52 (7), 364 (2021).
- Kitagawa, K. Blood pressure management for secondary stroke prevention. Hypertension Research: Official Journal of the Japanese Society of Hypertension. 45 (6), 936-943 (2022).
- Buchan, A. M., Pelz, D. M. Neuroprotection in acute ischemic stroke: A brief review. The Canadian Journal of Neurological Sciences. 49 (6), 741-745 (2021).
- Paul, S., Candelario-Jalil, E. Emerging neuroprotective strategies for the treatment of ischemic stroke: An overview of clinical and preclinical studies. Experimental Neurology. 335, 113518 (2021).
- Zabriskie, M., et al. New Zealand White rabbits fed high cholesterol diets develop morbid systemic diseases before intracranial atherosclerosis is detected. Journal of Veterinary Science & Medical Diagnosis. 8 (3), (2019).
- McNally, J. S., et al. Rabbit models of intracranial atherosclerotic disease for pathological validation of vessel wall MRI. The Neuroradiology Journal. 34 (3), 193-199 (2020).
- Brousseau, M. E., Hoeg, J. M. Transgenic rabbits as models for atherosclerosis research. Journal of Lipid Research. 40 (3), 365-375 (1999).
- Ji, D., Zhao, G., Songstad, A., Cui, X., Weinstein, E. J. Efficient creation of an APOE knockout rabbit. Transgenic Research. 24 (2), 227-235 (2015).
- Abela, G. S., et al. Triggering of plaque disruption and arterial thrombosis in an atherosclerotic rabbit model. Circulation. 91 (3), 776-784 (1995).
- Aliev, G., Burnstock, G. Watanabe rabbits with heritable hypercholesterolaemia: a model of atherosclerosis. Histology and Histopathology. 13 (3), 797-817 (1998).
- Brinjikji, W., Ding, Y. H., Kallmes, D. F., Kadirvel, R. From bench to bedside: Utility of the rabbit elastase aneurysm model in pre-clinical studies of intracranial aneurysm treatment. Journal of Neurointerventional Surgery. 8 (5), 521-525 (2016).
- Zabriskie, M. S., Wang, C., Wang, S., Alexander, M. D. Apolipoprotein E knockout rabbit model of intracranial atherosclerotic disease. Animal Models and Experimental Medicine. 3 (2), 208-213 (2020).
- Zabriskie, M. S., Cooke, D. L., Wang, C., Alexander, M. D. Spatially resolved transcriptomics for evaluation of intracranial vessels in a rabbit model: Proof of concept. bioRxiv. , (2022).
- Alexander, M. D., Darflinger, R. D., Sun, Z., Cooke, D. L. Assessment of cell yield among different devices for endovascular biopsy to harvest endothelial cells. Biotechniques. 66 (1), 34-36 (2017).
- English, J. D., et al. A novel model of large vessel ischemic stroke in rabbits: microcatheter occlusion of the posterior cerebral artery. Journal of Neurointerventional Surgery. 7 (5), 363-366 (2015).
- Peng, T. J., Ortega-Gutiérrez, S., de Havenon, A., Petersen, N. H. Blood pressure management after endovascular thrombectomy. Frontiers in Neurology. 12, 723461 (2021).
- Nepal, G., Shrestha, G. S., Shing, Y. K., Muha, A., Bhagat, R. Systolic blood pressure variability following endovascular thrombectomy and clinical outcome in acute ischemic stroke: A meta-analysis. Acta Neurologica Scandinavica. 144 (4), 343-354 (2021).
- Bennett, A. E., et al. Increased blood pressure variability after endovascular thrombectomy for acute stroke is associated with worse clinical outcome. Journal of Neurointerventional Surgery. 10 (9), 823-827 (2018).
- de Havenon, A., et al. Increased blood pressure variability contributes to worse outcome after intracerebral hemorrhage. Stroke. 49 (8), 1981-1984 (2018).

