Electric Cell-Substrate Sensing for Real-Time Evaluation of Metal-Organic Framework Toxicological Profiles
Summary
The following study evaluates the toxicological profile of a selected metal-organic framework utilizing electric cell-substrate impedance sensing (ECIS), a real-time, high-throughput screening technique.
Abstract
Metal-organic frameworks (MOFs) are hybrids formed through the coordination of metal ions and organic linkers in organic solvents. The implementation of MOFs in biomedical and industrial applications has led to concerns regarding their safety. Herein, the profile of a selected MOF, a zeolitic imidazole framework, was evaluated upon exposure to human lung epithelial cells. The platform for evaluation was a real-time technique (i.e., electric cell-substrate impedance sensing [ECIS]). This study identifies and discusses some of the deleterious effects of the selected MOF on the exposed cells. Furthermore, this study demonstrates the benefits of using the real-time method versus other biochemical assays for comprehensive cell evaluations. The study concludes that observed changes in cell behavior could hint at possible toxicity induced upon exposure to MOFs of different physicochemical characteristics and the dosage of those frameworks being used. By understanding changes in cell behavior, one foresees the ability to improve safe-by-design strategies of MOFs to be used for biomedical applications by specifically tailoring their physicochemical characteristics.
Introduction
Metal-organic frameworks (MOFs) are hybrids formed through the combination of metal ions and organic linkers1,2 in organic solvents. Due to the variety of such combinations, MOFs possess structural diversity3, tunable porosity, high thermal stability, and high surface areas4,5. Such characteristics make them attractive candidates in a variety of applications, from gas storage6,7 to catalysis8,9, and from contrast agents10,11 to drug delivery units12,13. However, the implementation of MOFs into such applications has raised concerns relative to their safety to both the users and the environment. Preliminary studies have shown, for instance, that cellular function and growth change upon the exposure of cells to metal ions or linkers used for MOF synthesis1,14,15. For instance, Tamames-Tabar et al. demonstrated that ZIF-8 MOF, a Zn-based MOF, was leading to more cellular changes in a human cervical cancer cell line (HeLa) and a mouse macrophage cell line (J774) relative to Zr-based and Fe-based MOFs. Such effects were presumably due to the metal component of ZIF-8 (i.e., Zn), which could potentially induce cell apoptosis upon framework disintegration and Zn ion release1. Similarly, Gandara-Loe et al. demonstrated that HKUST-1, a Cu-based MOF, caused the highest reduction in mouse retinoblastoma cell viability when used at concentrations of 10 µg/mL or greater. This was presumably due to the Cu metal ion incorporated during the synthesis of this framework, which, once released, could induce oxidative stress in the exposed cells15.
Moreover, analysis showed that the exposure to MOFs with different physicochemical characteristics could lead to varying responses of exposed cells. For instance, Wagner et al. demonstrated that ZIF-8 and MIL-160 (an Al-based framework), used in the exposure of an immortalized human bronchial epithelial cell, led to cellular responses dependent on frameworks' physicochemical properties, namely hydrophobicity, size, and structural characteristics16. Complementarily, Chen et al. demonstrated that a concentration of 160 µg/mL MIL-100(Fe) exposed to human normal liver cells (HL-7702) caused the largest loss in cellular viability, presumably due to the metal component of this specific framework (i.e., Fe17).
While these studies categorize MOFs' deleterious effects on cellular systems based on their physicochemical characteristics and exposure concentrations, thus raising potential concerns with framework implementation, especially in biomedical fields, most of these evaluations are based on single time point colorimetric assays. For instance, it was shown that when (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) tetrazolium (MTT) and water-soluble tetrazolium salt (WST-1) assays were used, these biochemical reagents could lead to false positives upon their interactions with the particles that the cells were also exposed to18. The tetrazolium salt and neutral red reagents were shown to possess a high adsorption or binding affinity onto the surfaces of the particles, resulting to agent signal interference19. Moreover, for other types of assays, such as flow cytometry, which was previously shown to be used for assessing changes in cells exposed to MOFs20,21, it was shown that major issues have to be circumvented if a viable analysis of particles' deleterious effects is to be considered. In particular, detection ranges of the particles' sizes, especially in mixed populations like the ones offered by MOFs or references of the particles used for calibration before cellular changes, have to be addressed22. It was also shown that the dye used during cell labeling for such cytometry assays could also interfere with the nanoparticles that the cells were exposed to23.
The goal of this study was to use a real-time, high-throughput evaluation assay to assess changes in cell behavior upon exposure to a select MOF. Real-time evaluations can help provide insights into time-dependent effects, as related to the windows of exposures16. Further, they provide information on changes in cell-substrate interactions, cell morphology, and cell-cell interactions, as well as how such changes depend on the physicochemical properties of the materials of interest and exposure times24,25 respectively.
To demonstrate the validity and applicability of the proposed approach, human bronchial epithelial (BEAS-2B) cells, ZIF-8 (a hydrophobic framework of zeolitic imidazolate16), and electric cell-substrate impedance sensing (ECIS) were used. BEAS-2B cells represent a model for lung exposure26 and have been previously used to evaluate changes upon the exposure of cells to nanoclays and their thermally degraded byproducts26,27,28, as well as assess the toxicity of nanomaterials, such as single-walled carbon nanotubes (SWCNTs)18. Furthermore, such cells have been used for more than 30 years as a model for pulmonary epithelial function29. ZIF-8 was chosen due to its wide implementation in catalysis30 and as contrast agents31 for bioimaging and drug delivery32, and thus for the extended potential for lung exposure during such applications. Lastly, ECIS, the noninvasive, real-time technique, was previously used to evaluate changes in cell adherence, proliferation, motility, and morphology16,26 as a result of a variety of interactions between analytes (both materials and drugs) and exposed cells in real-time16,18,28. ECIS uses an alternating current (AC) to measure the impedance of cells immobilized on gold electrodes, with the impedance changes giving insights into changes in resistance and capacitance at the cell-gold substrate interface, barrier function as induced by cell-cell interactions, and over-cell layer coverage of such gold electrodes33,34. Using ECIS allows quantitative measurements at a nanoscale resolution in a noninvasive, real-time manner26,34.
This study assesses and compares the simplicity and ease of evaluation of MOF-induced changes in cellular behavior in real-time with single-point assay evaluations. Such a study could be further extrapolated for evaluating cell profiles in response to exposure to other particles of interest, thus allowing for safe-by-design particle testing and subsequent helping with implementation. Moreover, this study could complement genetic and cellular assays that are single-point evaluations. This could lead to a more informed analysis of the deleterious effects of particles on the cellular population and could be used for screening such particles' toxicity in a high-throughput manner16,35,36.
Protocol
1. ZIF-8 synthesis
- For the purpose of this example, use a 1:10:100 (metal:linker:solvent) mass ratio to synthesize the ZIF-8. For this, measure out zinc nitrate hexahydrate, and record the measurement. Utilize the example mass ratio to calculate the amount needed for the linker, 2-methylimidazole, and the solvent (i.e., methanol).
- Place the zinc nitrate hexahydrate and linker into two different glass vials. Add half of the calculated amount of methanol to the zinc nitrate hexahydrate and the other half to the linker. Allow each solution to dissolve.
- Combine the two solutions (i.e., solutions containing the metal and linker, respectively). Place the container with the combined solution for the reaction onto a stir plate and stir at 700 rpm for 24 h at room temperature (RT).
2. ZIF-8 collection
- Once the above reaction has concluded (i.e., after 24 h), place the solution into 50 mL centrifuge tubes and centrifuge at 3,075 x g for 5 min (use equal amounts and balance the rotor). Remove any of the supernatants from the centrifuged tubes.
- Add methanol (5-15 mL) to the centrifuge tubes and redisperse the particles.
- Repeat the centrifugation and washing steps at least two to four more times. After the last wash, discharge the supernatant.
NOTE: The washing steps remove any non-precipitated species. - Remove the lids of the tubes and place parafilm tape over the top; subsequently, poke holes in the parafilm. Place the tube/s containing the sample in a vacuum chamber at RT to dry for 24 h.
3. ZIF-8 surface morphology (scanning electron microscopy [SEM])
- Mount the dry powders of ZIF-8 onto carbon tape and sputter for 120 s with gold-palladium to prevent any charging that could occur during the SEM analysis.
- Set the accelerating voltage of the microscope to 15 kV. Bring the particles into focus and adjust the magnification (magnifications of 500x, 1,000x, 1,500x, 4,000x, 10,000x, 20,000x, 30,000x, and 40,000x were used.)
- Image the particles under the magnification that allows both a clear particle size and morphology assessment (recommended to consider ranges for particles of 5 µm, 10 µm, and 20 µm).
- Collect data from at least three different fields of view, and for each, consider different magnifications.
4. ZIF-8 elemental composition
- Follow the steps for SEM imaging.
- Using the energy-dispersive X-ray (EDX) spectroscopy unit of the SEM microscope, analyze the sample elemental composition.
- Make sure the SEM accelerating voltage and magnification match the EDX monitor. Turn off the infrared camera on the SEM microscope.
- On the EDX monitor, go to Collect Spectrum and input a collection time of 200 s. This is recommended to avoid particle charging and disintegration that could lead to false evaluations.
- Collect data from at least three different fields of view. Once the collection is finished, analyze the data, and identify the elements of interest.
5. Cell culture
- Culture immortalized BEAS-2B cells in media supplemented with 5% fetal bovine serum (FBS) and 0.2% penicillin/streptomycin.
NOTE: Passages between 5-10 should be used to ensure that no phenotypical changes have previously taken place in the cell batch being used37 (all reagents are listed in the Table of Materials). - Take a 100 mm cell culture plate and place 10 mL of the media onto the plate. Add 1 mL of the BEAS-2B cell solution to the plate.
- Place the plate into an incubator (37 °C, 5% CO2) and allow the cells to grow.
- Check the plate after 24 h to determine if the cells have reached 80% confluency. Herein, the 80% confluency refers to the percentage of the surface covered by adhered cells. If not, change the media and allow the cells to grow until the 80% confluency level is reached.
6. Cell counting
- Once the cells on the plate have reached a confluency of 80%, remove the media from the plate. Wash the plate with 5 mL of phosphate-buffered saline (PBS).
- Remove the PBS from the plate. Add 2 mL of trypsin to the plate and place it in the incubator (37 °C, 5% CO2) for 2 min.
- Add 2 mL of media to the plate and pipet the media up and down to remove the cells from the plate. Transfer the solution to a 15 mL tube and centrifuge at 123 x g for 5 min.
- Remove the supernatant from the tube and add 2 mL of media. Pipet the media up and down to redisperse the cells.
- Using a 1.5 mL tube, add 20 µL of trypan blue and then 20 µL of the cell solution (1:1 ratio of trypan blue:cell solution).
- Place 10 µL of the cell mixture onto a glass slide and place it onto an automatic cell counter. Wait until the screen comes into focus, then select capture.
- The screen displays the live cell numbers and cell viability. The measurement range extends from 1 x 104-1 x 107 cells.
- Alternatively, count the cells manually using a hemocytometer.
- For this, add 15-20 µL of the trypan blue-treated cell solution to the hemocytometer.
- Use an upright microscope with a 10x objective focus on the grid lines of the hemocytometer.
- Count the number of cells in the four outer squares and divide the number by four. Then, multiply by 10,000 to obtain the live cell count number.
- To calculate cell viability, add together the live and dead cell count to obtain the total cell count, and subsequently divide the live cell count by the total cell count.
7. ZIF-8 dose preparation
- Make a ZIF-8 stock solution of 1 mg/mL. For this, measure the amount of ZIF-8 and add deionized (DI) water to the particles to reach a 1 mg/mL concentration.
- Sonicate (set the sonicator to sonics) the ZIF-8 stock solution for 2 min at 30 s intervals in a water bath.
- Make different doses of ZIF-8 for cellular exposures by calculating the amount of stock solution and Dulbecco's modified eagle medium (DMEM) needed to reach the desired doses using the equation C1V1 = C2V2. The doses will range from 0-950 µg/mL.
NOTE: In this experiment, doses of 0 µg/mL, 1 µg/mL, 10 µg/mL, 50 µg/mL, 100 µg/mL, 250 µg/mL, 500 µg/mL, 750 µg/mL, and 950 µg/mL were selected.
8. Half-maximal inhibitory concentration (IC 50)
- After cell counting, seed the BEAS-2B cells at a density of 4 x 104 cells/mL in a 96-well plate, with a volume of 100 µL/well.
- To reach a density of 4 x 104 cells/mL, use C1V1 = C2V2 to calculate the amount of cell solution needed to be combined with the media.
- Ensure to maintain blank wells for each dose; leave these wells empty until ZIF-8 exposure.
- Place the 96-well plate into the incubator at 37 °C and 5% CO2 and allow the cells to grow to a confluent monolayer for 24 h.
- Remove the media from the wells and expose the BEAS-2B cells to ZIF-8 doses ranging from 0-950 µg/mL. Add 100 µL of ZIF-8 to their respective blank and cell wells.
- Place the 96-well plates back into the incubator for an exposure time of 48 h.
- After the exposure, add 10 µL of 4-[3-(4-idophenyl)-2-(4-nitrophenyl)-2H-5-tetrazolio]-1,3-benzene disulfonate (WST-1) to each well (blank and cell wells) and incubate for 2 h. The media remains in the wells; the ratio is 1:10 (reagent:media).
- Read the changes in absorbance on a plate reader using a 485 nm absorbance.
- Calculate the cell viability through the software to determine the IC50 value (see section 10).
9. Electrical cell-substrate impedance sensing (ECIS)
- Use a 96-well plate (96W10idf) to perform ECIS analysis. The plate28 contains inter-digitated finger connection electrodes covering an area of about 4 mm2.
- First, test that all the wells are being read by the sensors. For this, place the 96-well plate onto the well station and click on the Setup button.
- If the wells are connected properly, green will appear; any unconnected wells will be identified as red. If such a case arises, remove and reinsert the well plate and click the Setup button again, until all the wells offer viable green readings.
- Stabilize the electrodes for 40 min with 200 µL of media per well to prevent potential drift. Add 200 µL of media to each well being used and then place the 96-well plate onto the ECIS station. Click on Setup/Check to ensure the plate is properly connected. Click on Stabilize.
- Once stabilization is complete, remove the plate from the station and remove the media from each well.
- Seed the BEAS-2B cells at a density of 4 x 104 cells/mL, with a volume of 100 µL per well, into the wells containing the cells and place the plate onto the ECIS station in the incubator.
- On the monitor, click on Check to determine that all the electrodes are read by the sensors on the station.
- Set up the time course measurement by selecting Multiple Freq/Time (MFT). The program will measure each well at seven different frequencies.
- Click Start and allow it to run for 24 h so the cells can form a confluent monolayer.
- After 24 h, the wells containing cells will show increased resistance on the monitor. This is an indicator that cells have attached to the electrodes.
- Press Pause on the monitor to stop the data collection.
- Make the ZIF-8 doses at, above, and below the calculated IC50 value by following the steps outlined in section 7 above.
- Expose the ZIF-8 nanoparticles to their respective blank and cell wells at a volume of 100 µL per well and place the 96-well plate back onto the station.
- Click on Check again on the monitor to ensure the sensors are reading all the electrodes. Click on Resume and allow the system to run for 72 h to monitor the cellular behavior and attachment.
- Once data collection has finished, click on Finish on the monitor. To save the file, click File > Save As. Save the data as a spreadsheet file.
- To get the alpha parameters, click on Model. On the screen pop-up, click on Media-Only Well, then select the well that has only media.
- Click on Set Ref. On the monitor, click on Find.
- If the well shown in the system is not the same as what was chosen, repeat step 9.16.
- Click on Set. Click on Fit T-Range to determine the time range desired for data collection.
- While it is processing, click on Alpha. When the system finishes the analysis, click on Save RbA and save it as a spreadsheet file.
NOTE: Refer to the ECIS manual, which can be found on the manufacturer's website38.
10. Data analysis
- SEM
- Open the imaging software to analyze the SEM images. Click File > Open, and then select the image to analyze.
- Click on Image > Type > 8 Bits to convert the image to grayscale to perform the threshold operation.
- Calibrate the distance measurement of pixels to microns. Click on the Straight-Line tool and trace the length of each particle aimed to be measured.
- Click Analyze and then Set Scale. The known distance will be set to the length of the size of the scale bar on the SEM image. The known unit of length will be set to microns. Check Global and press Ok.
- Click on the Straight-Line tool and trace the diameter of the particle, then select Analyze and Measure. Record the length result.
- Repeat for all the collected images. Average all the diameters from at least 60 particles for a suitable statistics evaluation.
- EDX
- Average all the weight percentages of each element together from all the collected images (see EDX section above).
- Using a spreadsheet, plot a bar graph of each element on the x-axis and their average elemental weight percentage on the y-axis.
- IC50
- Average the blank wells and average the dose wells for each replicate.
- Calculate the adjusted blank by subtracting the control blank average from the dose blank average. Calculate the true cell value for each dose by subtracting the adjusted blank from the average dose value.
- Subtract the adjusted blank value for each dose from the average control value. Calculate the cell viability of each dose utilizing the equation: cell viability = (true cell value/(control value – adjusted blank value)) x 100.
- Repeat this for all other replicates.
- Average the cell viability for each replicate together to get the final cell viability for each dose.
- In the software, choose the XY tab on the screen.
- Enter the concentration (dose) values into the X column and the cell viability (%) from each dose into the Y column.
- Transform the X values by clicking Analyze > Transform > Ok, and Transform X = Log(X).
- Normalize the Y values by selecting the Transformed Data sheet, clicking on Analyze, and then selecting Normalize.
- Select the Normalize of Transform data sheet, click Analyze, and select Nonlinear Regression (Curve Fit). Select Dose-Response-Inhibition and click Log(Inhibitor) Vs. Response-Variable Slope (Four Parameters). Click on OK to view the results.
- ECIS
- From the spreadsheet, take the resistance (ohm) columns for each replicate (blank wells and dose wells) and average them together from the 4,000 Hz frequency. This frequency allows cell-cell contact evaluation38.
- Calculate the corrected resistance by subtracting the averaged blank from the averaged dose for each replicate. Average the corrected resistance for the replicates for each dose.
- Repeat the same steps for the alpha values to calculate the average alpha parameter. Plot the x-axis as time (h) and the y-axis as the average resistance/alpha values for each dose value.
11. Statistical analysis
- SEM
- Perform a standard deviation in the spreadsheet file where the average diameters of the ZIF-8 particles were calculated.
- Utilizing the STDEV function in the spreadsheet, highlight the data of the 60 particles and click Enter. Graph the standard deviation for each average diameter calculated.
- EDX
- Similar to SEM (steps 11.1.1-11.1.2), calculate the standard deviation for each average elemental composition using the STDEV function in the spreadsheet. Graph the standard deviations for each average elemental composition.
- IC50
- Open the software utilized to calculate the IC50 and click on Grouped Data Sheet.
- Enter the ZIF-8 doses in the rows labeled Group A, Group B, etc. Enter the average cell viability for each dose and replicate in the column corresponding with the ZIF-8 dose.
- Click on Analyze, and under Column Analyses, select One-way ANOVA (and nonparametric or mixed).
- In the Experimental Design tab, select No Matching Or Pairing. Under Gaussian Distribution Of Residuals, select Yes use ANOVA. Finally, select Yes Use Ordinary ANOVA Test under Assume Equal SDs.
- Under the Multiple Comparisons tab, select Compare the Mean of Each Column with the Mean of a Control Column. Click on OK to view the results.
Representative Results
Using a common in vitro model cell line39 (BEAS-2B), this study aimed to demonstrate the feasibility and applicability of ECIS to assess changes in cell behavior upon exposure to a lab-synthesized MOF. These changes assessment was complemented by analysis through conventional colorimetric assays.
The physicochemical characteristics of the framework were first evaluated to ensure the reproducibility of the methods employed, the validity of the obtained results, and the pertinent discussions of such results. The analysis of the surface morphology of ZIF-8, for instance, was performed through SEM; images showed that the frameworks displayed a rhombic dodecahedron morphology40 and had an average size of 62.87 nm ± 9.61 nm (Figure 1A). The elemental composition of the MOFs was determined viaEDX spectroscopy. The results showed that the main composition of ZIF-8 consisted of C, N, and Zn (i.e., the makeup of the imidazolate linker and the metal ion, Zn16) (Figure 1B). Previous X-ray diffraction showed that ZIF-8 crystalline phases have identified specific peaks at 10.33°, 12.8°, 14.7°, 16.5°, and 18°, with such peaks being attributed to planes (002), (112), (022), (013), and (222), respectively, 16,41.
For the proposed cell behavior screening, the IC50 (concentration where ZIF-8 inhibits cell growth by 50%) was determined29. For this, cells were exposed to ZIF-8 for 48 h at doses ranging from 0-950 µg/mL (Figure 2A). The dose-response graph of exposed cells is displayed in Figure 2B, with the subsequent recorded IC50 of 35.7 µg/mL. In addition, the analysis revealed a dose-dependent trend in the viability of cells upon exposure to the ZIF-8.
Upon determining the IC50, ECIS analysis was performed. Briefly, ECIS was used as a real-time screening strategy of BEAS-2B exposed to ZIF-8 MOFs at IC50, as well as below and above this concentration, namely 15.7 µg/mL (C1), 35.7 µg/mL (C2), 55.7 µg/mL (C3), and 75.7 µg/mL (C4) respectively, with the cells being exposed for a total period of 72 h. The inclusion of ECIS was envisioned to allow insight into determining changes to cell coverage, morphology, and viability, all in real-time. The ECIS results were recorded as changes in resistance and changes in cell-substrate interactions, namely the alpha parameter42.
The resistance trends are displayed in Figure 3A as changes in the profiles of the cells treated with ZIF-8 compared to the control (black line) (i.e., unexposed cells). Specifically, the analysis showed that the wells containing cells on the gold electrodes exposed to C2-C4 doses displayed an initial slight increase in resistance immediately after cell exposure to the frameworks (all relative to the control [i.e., unexposed cells]). These initial changes were subsequently followed by sharp decreases in recorded resistances, with such decreases being prevalent between 6-8 h from the exposure (Figure 3A). A complete loss in resistance was observed after 14-18 h from the exposure time. Cells exposed to doses below the IC50 value displayed resistances, like the wells with control cells, until about 16-18 h from the exposure when resistance losses appear. Also, during the cellular exposure, a continuous signal perturbation of the signal of the wells used in the experiments was observed. These perturbances persisted during the analysis of the alpha parameter (Figure 3B), with this analysis further showing that exposed cells change their cell-substrate interactions with profiles similar to the resistance ones. Moreover, such changes were dependent of the time from the exposure and the dose used in such exposure.
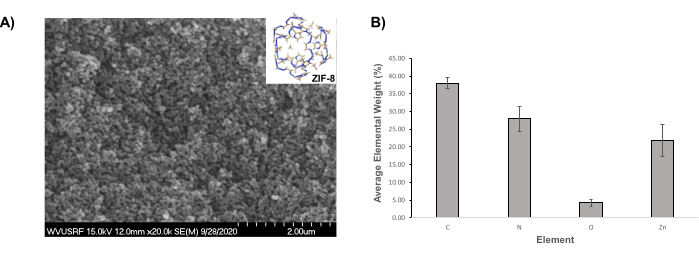
Figure 1: SEM image and average elemental composition ZIF-8 particles. (A) Representative SEM image of ZIF-8 particles. (B) Average elemental composition of ZIF-8 particles (± standard deviation [SD] bars). Please click here to view a larger version of this figure.
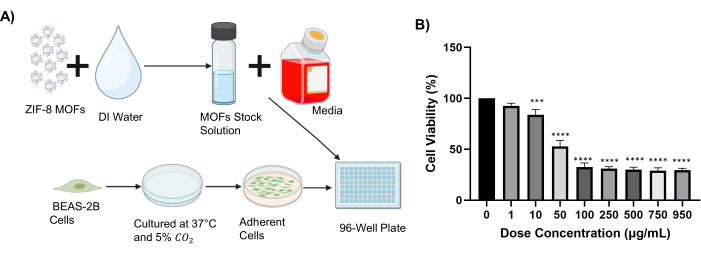
Figure 2: Cell viability. (A) Cell treatment schematic (created with Biorender.com). (B) Viability of BEAS-2B cells exposed to ZIF-8 particles at doses ranging from 0-950 µg/mL; this analysis was used to determine the IC50 (± SD bars; ***p = 0.0001 and **** p < 0.0001 relative to the control). Please click here to view a larger version of this figure.
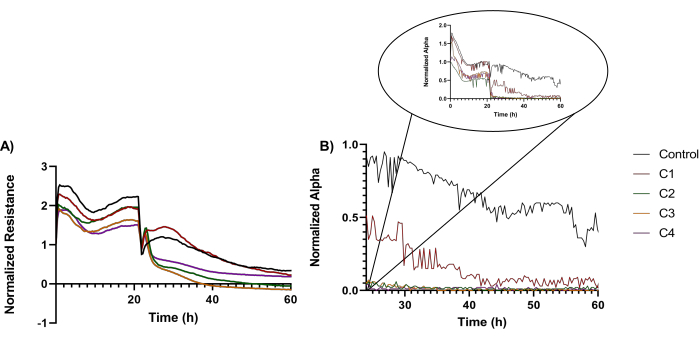
Figure 3: Representative cellular resistance and changes in cellular attachment. (A) Representative cellular resistance of BEAS-2B cells exposed to ZIF-8 particles below, at, and above the IC50 concentration. (B) Changes in the cellular attachment (i.e., changes in the alpha parameter) of BEAS-2B cells when exposed to ZIF-8 particles below and above the IC50 concentration. Please click here to view a larger version of this figure.
Discussion
Previous analysis showed that ECIS could be used to assess the behavior of cells exposed to analytes (i.e., carbon nanotubes35, drugs43, or nanoclays16). Furthermore, Stueckle et al. used ECIS to evaluate the toxicity of BEAS-2B cells exposed to nanoclays and their byproducts and found that the cellular behavior and attachment were dependent on the physicochemical characteristics of such materials42. Herein, we proposed to determine the possible changes of BEAS-2B cells in response to exposure to MOFs, to thus contribute to the body of knowledge aimed to differentiate any deleterious effects of ZIF-8 on cellular systems in vitro, all in a high-throughput and real-time manner.
The analysis first allowed for the evaluation of framework properties to thus help correlate the changes in cellular behavior to the physicochemical properties of the tested MOFs44. Specifically, upon cellular exposure to MOFs with regular geometries of similar elemental composition (see results above), the analysis showed changes in cellular behavior, presumably resulting from the uptake of the frameworks and their profile cellular degradation. Specifically in terms of the uptake, previous analysis of toxicity on hydrophobic materials, such as this framework, showed that when such materials are exposed to cells, their uptake is more disruptive to a cellular membrane than the uptake of their hydrophilic counterparts presumably due to their ability to remove lipids from the membrane's structural bilayer16,45 during their cellular translocation thus leading to membrane dysfunctions or plasma membrane perturbations46,47. In particular, lipid removal was shown to lead to changes in membrane integrity, cell signaling48, and an increase in cellular uptake49, with a downside of cell function effects. For instance, Farcal et al. demonstrated that a hydrophobic TiO2 nanomaterial had an increased cytotoxic effect when compared to its hydrophilic counterpart in exposed alveolar macrophages50. In addition, Wagner et al., demonstrated that hydrophobic ZIF-8 caused more disruption to BEAS-2B cells when compared to hydrophilic MIL-16016.
The recorded changes in cell behavior are presumably due to disturbances in cell-substrate interactions26 and/or cell viability and proliferation16 onto the electrodes as a result of cell exposure to the selected framework. For instance, Wagner et al. evaluated similar parameters in cells exposed to nanoclays. The authors found that a complete loss in cell-substrate interaction was observed over the course of the 72 h experiments. In addition, the authors demonstrated a time-dependent effect on cell viability and proliferation when the BEAS-2Bs were exposed to their selected nanoclays26. While the particles being tested herein are MOFs, the possible extended association with the effects seen for nanoclays is feasible, as some of the physicochemical characteristics of these particles (namely size, hydrophobicity, and composition) are similar to those of the MOFs . Furthermore, Stueckle et al. also demonstrated time- and treatment-dependent cellular behavior when nanoclays were exposed to immortalized BEAS-2B cells. Others have demonstrated that ZIF-8, at a dose of 100 µg/mL, cause BEAS-2B cells to have a complete loss in the cell monolayer and cell viability, presumably due to changes in cell-substrate interactions16. It is, however, noted that in such studies, the ZIF-8 particles were obtained under different synthesis conditions (i.e., 10 min versus 24 h used in this study), with such synthesis conditions subsequently resulting in different shapes/morphologies of the frameworks to thus support previous conclusions. This shows that the reaction time6, temperature2, and solvent44 used during particle synthesis influence their shape, size, and composition profiles, and subsequently lead to differential effects and behaviors in exposed cells.
The analysis also showed that cells exposed to ZIF-8 doses below the IC50 value displayed resistances similar to those of the controls, presumably since such doses were not significant enough to induce ECIS-observable cell transformation in the conditions and timeframe these experiments were undertaken in. However, at doses above IC50, a complete loss in cellular resistance was observed and only within a few hours after the exposure, most likely because of changes in cell viability and induced possible changes in cell-substrate interactions16 (Figure 3A). These claims are supported by previous studies that showed that non-viable cells change their shape and detach from the substrates. For instance, Verma et al. used a real-time impedance sensing technique to assess the cytotoxicity of nanoclays in a human alveolar epithelial cell line (A549). It was shown that the platelet-type nanoclays caused an increase in the number of detached and deformed cells when compared to the tubular type nanoclays51. In addition, Xiao et al. demonstrated that the cytoskeleton of fibroblastic V79 cells was compromised when exposed to cadmium chloride, due to an influx of water, extracellular ions, and cytotoxic chemicals from the culture medium. The changes in cell cytoskeleton led to the loss of cell-substrate interactions. Furthermore, when benzalkonium chloride was exposed to V79 cells, there was a significant decrease in cellular resistance, due to damage to the cellular membrane that the exposure caused52.
Also, changes in resistance can be due to the cell reactions to the physicochemical characteristics of ZIF-8. In particular, ZIF-8 is a hydrophobic framework; previous analysis has shown that hydrophobicity can cause an increase in toxicity16. Metal ions, such as Zn, have also been previously shown to induce a higher degree of toxicity when compared to other metals like Zr and Fe1 namely, possibly contributing to higher cellular death.
It was further shown that all cells recorded an initial decrease in resistance right after exposure, followed by a subsequent increase. Eldawud et al. demonstrated similar effects when SWCNTs were exposed to BEAS-2B cells. The authors interpreted the increase in cellular resistance as being due to the BEAS-2B cells overcoming the disturbances caused by materials' exposure and subsequently regaining their integrity18 all while eliminating electrode drift28, to thus ensure reproducibility of the data analysis (see ECIS handbook).
Even though this study showed that ECIS could be a valuable tool to use in cell behavior screening, especially due to the high-throughput feature, it is recommended that the technique does not replace single time point assays when a full picture of a material's deleterious assessment is desired. In particular, the single point assay could further allow for the evaluation of cell transformation at their genetic level45,53, as shown by Eldawud et al., for instance for BEAS-2B cells exposed to nanodiamonds and through fluorescence-activated cell sorting (FACS) 45. Yu et al., also used FACS to evaluate human colon cancer cells (HCT0116) exposed to hyaluronic acid-modified mesoporous silica nanoparticles (HA-MSNs)54.
The results presented suggest that the route of change is dependent on the MOFs' physicochemical characteristics, as well as the time and dosing of such frameworks used in exposures. The results and methods described herein further emphasize the importance of real-time measurements and their advantages in understanding the changes in cellular profiles. These can potentially be supplemented with additional biochemical assays to evaluate the mechanism of toxicity, thus leading to safe-by-design strategies of MOFs that have no deleterious effects on cell behavior, recorded as cell attachment, cell-cell interactions, or cell-substrate interactions.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work was funded in part by the National Institute of General Medical Sciences (NIGMS) T32 program (T32 GM133369) and the National Science Foundation (NSF 1454230). Additionally, WVU Shared Research Facilities and Applied Biophysics assistance and support are acknowledged.
Materials
| 4-[3-(4-idophenyl)-2-(4-nitrophenyl)-2H-5-tetrazolio]-1,3-benzene disulfonate (WST-1 assay) | Roche | 5015944001 | |
| 0.25% Trypsin-EDTA (1x) | Gibco | 25255-056 | |
| 100 mm plates | Corning | 430167 | |
| 1300 Series A2 biofume hood | Thermo Scientific | 323TS | |
| 2510 Branson bath sonicator | Process Equipment & Supply, Inc. | 251OR-DTH | |
| 2-methylimidazole, 97% | Alfa Aesar | 693-98-1 | |
| 5 mL sterile microtube | Argos Technologies | T2076S-CA | |
| 50 mL tubes | Falcon | 352098 | |
| 96W10idf well plates | Applied Biophysics | 96W10idf PET | |
| 96-well plates | Fisherbrand | FB012931 | |
| Biorender | Biorender | N/A | |
| Countess cell counting chamber slides | Invitrogen | C10283 | |
| Countess II FL automated cell counter | Life Technologies | C0916-186A-0303 | |
| Denton Desk V sputter and carbon coater | Denton Vacuum | N/A | |
| Dimethly sulfoxide | Corning | 25-950-CQC | |
| DPBS/Modified | Cytiva | SH30028.02 | |
| Dulbecco's modified Eagle medium | Corning | 10-014-CV | |
| ECIS-ZΘ | Applied Biophysics | ABP 1129 | |
| Excel | Microsoft | Version 2301 | |
| Falcon tubes (15 mL) | Corning | 352196 | |
| Fetal bovine serum | Gibco | 16140-071 | |
| FLUOstar OPTIMA plate reader | BMG LABTECH | 413-2132 | |
| GraphPad Prism Software (9.0.0) | GraphPad Software, LLC | Version 9.0.0 | |
| HERAcell 150i CO2 Incubator | Thermo Scientific | 50116047 | |
| Hitachi S-4700 Field emission scanning electron microscope equipped with energy dispersive X-ray | Hitachi High-Technologies Corporation | S4700 and EDAX TEAM analysis software | |
| ImageJ software | National Institutes of Health | N/A | |
| Immortalized human bronchial epithelial cells | American Type Culture Collection | CRL-9609 | |
| Isotemp freezer | Fisher Scientific | ||
| Methanol, 99% | Fisher Chemical | 67-56-1 | |
| Parafilm sealing film | The Lab Depot | HS234526A | |
| Penicillin/Steptomycin | Gibco | 15140-122 | |
| Sorvall Legend X1R Centrifuge | Thermo Scientific | 75004220 | |
| Sorvall T 6000B | DU PONT | T6000B | |
| Trypan blue, 0.4% solution in PBS | MP Biomedicals, LLC | 1691049 | |
| Vacuum Chamber | Belart | 999320237 | |
| Zinc Nitrate Hexahydrate, 98% extra pure | Acros Organic | 101-96-18-9 |
Riferimenti
- Tamames-Tabar, C., et al. Cytotoxicity of nanoscaled metal-organic frameworks. Journal of Materials Chemistry. B. 2 (3), 262-271 (2014).
- Lin, W. X., et al. Low cytotoxic metal-organic frameworks as temperature-responsive drug carriers. ChemPlusChem. 81 (8), 804-810 (2016).
- Vasconcelos, I. B., et al. Cytotoxicity and slow release of the anti-cancer drug doxorubicin from ZIF-8. RSC Advances. 2 (25), 9437-9442 (2012).
- Yang, B. C., Shen, M., Liu, J. Q., Ren, F. Post-synthetic modification nanoscale metal-organic frameworks for targeted drug delivery in cancer cells. Pharmaceutical Research. 34 (11), 2440-2450 (2017).
- Lucena, M. A. M., et al. Application of the metal-organic framework Eu(BTC) as a luminescent marker for gunshot residues: A synthesis, characterization, and toxicity study. ACS Applied Materials & Interfaces. 9 (5), 4684-4691 (2017).
- Kayal, S., Sun, B. C., Chakraborty, A. Study of metal-organic framework MIL-101(Cr) for natural gas (methane) storage and compare with other MOFs (metal-organic frameworks). Energy. 91, 772-781 (2015).
- Gutov, O. V., et al. Water-stable zirconium-based metal-organic framework material with high-surface area and gas-storage capacities. Chimica. 20 (39), 12389-12393 (2014).
- Ghorbanloo, M., Safarifard, V., Morsali, A. Heterogeneous catalysis with a coordination modulation synthesized MOF: morphology-dependent catalytic activity. New Journal of Chemistry. 41 (10), 3957-3965 (2017).
- Valvekens, P., et al. Base catalytic activity of alkaline earth MOFs: a (micro) spectroscopic study of active site formation by the controlled transformation of structural anions. Chemical Science. 5 (11), 4517-4524 (2014).
- Taylor, K. M. L., Rieter, W. J., Lin, W. B. Manganese-based nanoscale metal-organic frameworks for magnetic resonance imaging. Journal of the American Chemical Society. 130 (44), 14358-14359 (2008).
- Taylor-Pashow, K. M. L., Della Rocca, J., Xie, Z. G., Tran, S., Lin, W. B. Postsynthetic modifications of iron-carboxylate nanoscale metal-organic frameworks for imaging and drug delivery. Journal of the American Chemical Society. 131 (40), 14261-14263 (2009).
- Kundu, T., et al. Mechanical downsizing of a gadolinium(III)-based metal-organic framework for anticancer drug delivery. Chimica. 20 (33), 10514-10518 (2014).
- Orellana-Tavra, C., et al. Drug delivery and controlled release from biocompatible metal-organic frameworks using mechanical amorphization. Journal of Materials Chemistry. B. 4 (47), 7697-7707 (2016).
- Su, H. M., et al. A highly porous medical metal-organic framework constructed from bioactive curcumin. Chemical Communications. 51 (26), 5774-5777 (2015).
- Gandara-Loe, J., et al. Metal-organic frameworks as drug delivery platforms for ocular therapeutics. ACS Applied Materials & Interfaces. 11 (2), 1924-1931 (2019).
- Wagner, A., et al. Toxicity screening of two prevalent metal organic frameworks for therapeutic use in human lung epithelial cells. International Journal of Nanomedicine. 14, 7583-7591 (2019).
- Chen, G. S., et al. In vitro toxicity study of a porous iron(III) metal-organic framework. Molecules. 24 (7), 1211 (2019).
- Eldawud, R., Wagner, A., Dong, C. B., Rojansakul, Y., Dinu, C. Z. Electronic platform for real-time multi-parametric analysis of cellular behavior post-exposure to single-walled carbon nanotubes. Biosensors & Bioelectronics. 71, 269-277 (2015).
- Kroll, A., Pillukat, M. H., Hahn, D., Schnekenburger, J. Current in vitro methods in nanoparticle risk assessment: Limitations and challenges. European Journal of Pharmaceutics and Biopharmaceutics. 72 (2), 370-377 (2009).
- Lucena, F. R. S., et al. Induction of cancer cell death by apoptosis and slow release of 5-fluoracil from metal-organic frameworks Cu-BTC. Biomedicine & Pharmacotherapy. 67 (8), 707-713 (2013).
- Orellana-Tavra, C., et al. Tuning the endocytosis mechanism of Zr-based metal-organic frameworks through linker functionalization. ACS Applied Materials & Interfaces. 9 (41), 35516-35525 (2017).
- Zucker, R. M., Ortenzio, J. N. R., Boyes, W. K. Characterization, detection, and counting of metal nanoparticles using flow cytometry. Cytometry. Part A. 89 (2), 169-183 (2016).
- Robson, A. L., et al. Advantages and limitations of current imaging techniques for characterizing liposome morphology. Frontiers in Pharmacology. 9, 80 (2018).
- Giaever, I., Keese, C. R. Micromotion of mammalian cells measured electrically. Proceedings of the National Academy of Sciences. 88 (17), 7896-7900 (1991).
- Wegener, J., Keese, C. R., Giaever, I. Electric cell-substrate impedance sensing (ECIS) as a noninvasive means to monitor the kinetics of cell spreading to artificial surfaces. Experimental Cell Research. 259 (1), 158-166 (2000).
- Wagner, A., et al. Toxicity evaluations of nanoclays and thermally degraded byproducts through spectroscopical and microscopical approaches. Biochimica et Biophysica Acta. General Subjects. 1861, 3406-3415 (2017).
- Wagner, A., et al. Early assessment and correlations of nanoclay’s toxicity to their physical and chemical properties. ACS Applied Materials & Interfaces. 9 (37), 32323-32335 (2017).
- Wagner, A., et al. Incineration of nanoclay composites leads to byproducts with reduced cellular reactivity. Scientific Reports. 8 (1), 10709 (2018).
- Zhao, F., Klimecki, W. T. Culture conditions profoundly impact phenotype in BEAS-2B, a human pulmonary epithelial model. Journal of Applied Toxicology. 35 (8), 945-951 (2015).
- Wu, S. X., Wang, W. H., Fang, Y. Z., Kong, X. J., Liu, J. H. Efficient Friedel-Crafts acylation of anisole over silicotungstic acid modified ZIF-8. Reaction Kinetics Mechanisms and Catalysis. 122, 357-367 (2017).
- Shu, F. P., et al. Fabrication of a hyaluronic acid conjugated metal organic framework for targeted drug delivery and magnetic resonance imaging. RSC Advances. 8 (12), 6581-6589 (2018).
- Shi, Z. Q., et al. FA-PEG decorated MOF nanoparticles as a targeted drug delivery system for controlled release of an autophagy inhibitor. Biomaterials Science. 6 (10), 2582-2590 (2018).
- Ebrahim, A. S., et al. Functional optimization of electric cell-substrate impedance sensing (ECIS) using human corneal epithelial cells. Scientific Reports. 12 (1), 14126 (2022).
- Szulcek, R., Bogaard, H. J., van Nieuw Amerongen, G. P. Electric cell-substrate impedance sensing for the quantification of endothelial proliferation, barrier function, and motility. Journal of Visualized Experiments. (85), e51300 (2014).
- Eldawud, R., et al. Carbon nanotubes physicochemical properties influence the overall cellular behavior and fate. Nanoimpact. 9, 72-84 (2018).
- An, Y., Jin, T. Y., Zhang, F., He, P. Electric cell-substrate impedance sensing (ECIS) for profiling cytotoxicity of cigarette smoke. Journal of Electroanalytical Chemistry. 834, 180-186 (2019).
- Kaur, G., Dufour, J. M. Cell lines. Spermatogenesis. 2 (1), 1-5 (2012).
- . Allied Biophysics Available from: https://biophysics.com (2023)
- Park, Y. H., Kim, D., Dai, J., Zhang, Z. Human bronchial epithelial BEAS-2B cells, an appropriate in vitro model to study heavy metals induced carcinogenesis. Toxicology and Applied Pharmacology. 287 (3), 240-245 (2015).
- Yang, F., et al. Morphological map of ZIF-8 crystals with five distinctive shapes: Feature of filler in mixed-matrix membranes on C3H6/C3H8 separation. Chemistry of Materials. 30 (10), 3467-3473 (2018).
- Rose, O. L., et al. Thin films of metal-organic framework interfaces obtained by laser evaporation. Nanomaterials. 11 (6), 1367 (2021).
- Stueckle, T. A., et al. Impacts of organomodified nanoclays and their incinerated byproducts on bronchial cell monolayer integrity. Chemical Research in Toxicology. 32 (12), 2445-2458 (2019).
- Eldawud, R., et al. Potential antitumor activity of digitoxin and user-designed analog administered to human lung cancer cells. Biochimica et Biophysica Acta. General Subjects. 1864 (11), 129683 (2020).
- Zhuang, J., et al. Optimized metal-organic-framework nanospheres for drug delivery: Evaluation of small-molecule encapsulation. ACS Nano. 8 (3), 2812-2819 (2014).
- Eldawud, R., et al. Combinatorial approaches to evaluate nanodiamond uptake and induced cellular fate. Nanotechnology. 27 (8), 085107 (2016).
- Houthaeve, G., De Smedt, S. C., Braeckmans, K., De Vos, W. H. The cellular response to plasma membrane disruption for nanomaterial delivery. Nano Convergence. 9 (1), 6 (2022).
- Otero-Gonzalez, L., Sierra-Alvarez, R., Boitano, S., Field, J. A. Application and validation of an impedance-based real time cell analyzer to measure the toxicity of nanoparticles impacting human bronchial epithelial cells. Environmental Science & Technology. 46 (18), 10271-10278 (2012).
- Ibarguren, M., Lopez, D. J., Escriba, P. V. The effect of natural and synthetic fatty acids on membrane structure, microdomain organization, cellular functions and human health. Biochimica et Biophysica Acta. 1838 (6), 1518-1528 (2014).
- Nor, Y. A., et al. Shaping nanoparticles with hydrophilic compositions and hydrophobic properties as nanocarriers for antibiotic delivery. ACS Central Science. 1 (6), 328-334 (2015).
- Farcal, L., et al. Comprehensive in vitro toxicity testing of a panel of representative oxide nanomaterials: First steps towards an intelligent testing strategy. PLoS One. 10 (5), e0127174 (2015).
- Verma, N. K., Moore, E., Blau, W., Volkov, Y., Babu, P. R. Cytotoxicity evaluation of nanoclays in human epithelial cell line A549 using high content screening and real-time impedance analysis. Journal of Nanoparticle Research. 14, 1137 (2012).
- Xiao, C. D., Lachance, B., Sunahara, G., Luong, J. H. T. Assessment of cytotoxicity using electric cell-substrate impedance sensing: Concentration and time response function approach. Analytical Chemistry. 74 (22), 5748-5753 (2002).
- Coyle, J. P., et al. Carbon nanotube filler enhances incinerated thermoplastics-induced cytotoxicity and metabolic disruption in vitro. Particle and Fibre Toxicology. 17 (1), 40 (2020).
- Yu, M. H., et al. Hyaluronic acid modified mesoporous silica nanoparticles for targeted drug delivery to CD44-overexpressing cancer cells. Nanoscale. 5 (1), 178-183 (2013).

