Draining Lymph Node Metastasis Model for Assessing the Dynamics of Antigen-Specific CD8+ T Cells During Tumorigenesis
Summary
The experimental design presented here provides a useful reproductive model for the studies of antigen-specific CD8+ T cells during lymph node (LN) metastasis, which excludes the perturbation of bystander CD8+ T cells.
Abstract
Tumor antigen-specific CD8+ T cells from draining lymph nodes gain an accumulating importance in mounting anti-tumor immune response during tumorigenesis. However, in many cases, cancer cells form metastatic loci in lymph nodes before further metastasizing to distant organs. To what extent the local and systematic CD8+ T cell responses were influenced by LN metastasis remains obscure. To this end, we set up a murine LN metastasis model combined with a B16F10-GP melanoma cell line expressing the surrogate neoantigen derived from lymphocytic choriomeningitis virus (LCMV), glycoprotein (GP), and P14 transgenic mice harboring T cell receptors (TCRs) specific to GP-derived peptide GP33-41 presented by the class I major histocompatibility complex (MHC) molecule H-2Db. This protocol enables the study of antigen-specific CD8+ T cell responses during LN metastasis. In this protocol, C57BL/6J mice were subcutaneously implanted with B16F10-GP cells, followed by adoptive transfer with naive P14 cells. When the subcutaneous tumor grew to approximately 5 mm in diameter, the primary tumor was excised, and B16F10-GP cells were directly injected into the tumor draining lymph node (TdLN). Then, the dynamics of CD8+ T cells were monitored during the process of LN metastasis. Collectively, this model has provided an approach to precisely investigate the antigen-specific CD8+ T cell immune responses during LN metastasis.
Introduction
Cancer immunotherapy, especially the immune checkpoint blockade (ICB), has revolutionized cancer therapy1. ICB blocks the coinhibitory immunoreceptors (such as PD-1, Tim-3, LAG-3, and TIGIT), which are highly expressed in exhausted CD8+ T cells in the tumor microenvironment (TME), leading to the reinvigoration of exhausted CD8+ T cells2. Considering the heterogeneity of exhausted CD8+ T cells, accumulating evidence revealed that tumor-specific CD8+ T cells derived from the periphery, including draining lymph node (dLN), but not in TME, mediate the efficacy of ICB3,4,5,6,7,8. Recently, TdLN derived TCF-1+TOX– tumor-specific memory CD8+ T cells (TdLN-TTSM) was confirmed to be the genuine responders to ICB which embody several functional properties of conventional memory T cells and could further expand and differentiate into progeny exhausted cells upon ICB treatment9. Altogether, these findings corroborated the importance of LN in mounting anti-tumor immunity.
Lymph node functions as a critical place in facilitating the priming and activation of tumor-specific CD8+ T cells by providing structural basis as well as biological signals10. Several types of cancer cells frequently seed sentinel lymph node (SLN, the first LN draining a primary tumor) before systematic dissemination11. The presence of SLN metastasis is linked with poor outcome in human cancer and preclinical models showed that tumor cells in TdLN could spread to distant organs through both the lymphatic vessels and blood vessels of the node12,13,14,15. SLN biopsy now represents a standard procedure to guide subsequent treatment decisions in many solid tumor types which could avoid unnecessary resection of uninvolved LN16,17. Even to the involved LN, it remains controversial whether and when surgical resection is needed as several studies have demonstrated that the removal of regional LN did not exhibit improved overall survival compared to those that received radiation or systemic therapy without regional LN resection18,19. One interpretation is that metastatic LN (mLN) with microscopic disease may retain some capacity to educate immune cells and provide some therapeutic benefits. So, it is critically important to elucidate how LN metastasis affects the anti-tumor immune response, especially the properties and functions of TdLN-TTSM.
Until now, both preclinical and clinical data have revealed some structural and cellular alterations in mLN20. However, the dynamic changes of tumor-specific CD8+ T cells during LN metastasis have not been delineated. Therefore, developing a compelling model of LN metastasis is needed for further investigation. Indeed, several studies have reported mLN mouse models through different ways14,21,22. For example, spontaneous metastasis in axillary LNs was conducted through the implantation of 4T1 breast cancer cells into the mammary fat pad22. In another study, Reticker-Flynn et al. generated melanoma cell lines with high incidence of spread from subcutaneous primary tumor to LNs through serial inoculation of tumor cells cultured from dissociated mLN tissues (nine rounds)14. Another commonly used model was prepared by the injection of tumor cells into the footpad and the metastatic loci would be formed in popliteal LN22. Notably, it is difficult to evaluate the precise timepoints of intervention because LN metastasis in these models is not always faithful.
In the present study, a murine LN metastatic model was established through the intranodal injection of B16F10-GP cells23,24, generated by CRISPR/Cas9-mediated insertion of LCMV virus glycoprotein (GP) gene sequence into the genome of B16F10 cell line9. Then, these mice were transferred with P14 cells which harbor transgenic T cell receptors (TCRs) specifically recognize the H-2Db GP33-41 epitope25,26 and the systemic and local dynamics of antigen-specific CD8+ T cells during LN metastasis could be investigated. Our experimental design provides a useful model for the study of immune responses, especially the antigen-specific CD8+ T cells during the LN metastasis which excludes the perturbation of bystander CD8+ T cells. These results would affect the clinical treatment options of whether to remove or retain the mLN and shed new light on the manipulation of mLN to achieve maximum therapeutic benefits.
Protocol
The C57BL/6J mice (referred to B6 mice) and naive P14 transgenic mice9,27 used were 6-10 weeks of age weighing 18-22 g. Both male and female were included without randomization or blinding. All animal studies were conducted in accordance with the guidelines of the Institutional Animal Care and Use Committee of the Qingdao Agricultural University.
1. Preparation of medium and reagents
- Prepare B16F10-GP melanoma cell culture medium, named D10 (complete DMEM medium) by adding DMEM, 10% fetal bovine serum (FBS), 1% penicillin/streptomycin, 1% L-glutamine with an additional 100 U/mL puromycin. Maintain a sterile D10 and store for up to 2 weeks at 2-4 °C.
- Prepare R2 medium (for termination of red blood cell lysis) by adding RPMI-1640 with 2% FBS, 1% penicillin/streptomycin, and 1% L-glutamine.
- Prepare fluorescence-activated cell sorting (FACS) buffer by adding 1x phosphate-buffered saline (PBS) with 2% FBS and 0.01% of sodium azide. The addition of sodium azide can prolong the storage time of FACS buffer, which can be stored for months at 2-4 ° C.
- Prepare red blood cell lysis (RBL) buffer by adding 155 mM NH4Cl, 10 mM KHCO3, and 0.1 mM ethylenediamine tetraacetic acid (EDTA) into double distilled water and adjust its pH to 7.3. Store RBL buffer at room temperature (RT) and it remains stable for up to 3 months.
- Prepare the anesthetic, 2,2,2-tribromoethanol, as described below.
- Weigh out appropriate-amount of 2,2,2-tribromoethanol crystal in a 50 mL sterile conical tube wrapped with aluminum foil. Add appropriate amount of 2-methyl-2-butanol into the crystal to prepare the stock solution with the concentration of 500 mg/mL.
- Swirl it to mix and warm the tube in a 37 °C water bath until the crystal is dissolved. Make sure all crystals are dissolved.
- Mix the solution thoroughly. Filter the stock solution with a 0.22 µm filter into a sterile container. Store the stock solution frozen, shielded from light, at -20 °C or dilute with PBS into a working solution at the concentration of 12.5 mg/mL and stored at 4 °C.
NOTE: It is best to use the prescribed concentration as higher concentrations of the liquid are irritating.
2. Preparation of B16F10-GP cell suspension
- Thaw and culture a vial of 1 x 106 B16F10-GP cells with 5 mL of D10 in a cell culture incubator at 37 °C and 5% CO2. Subculture cells when they reached 80% to 90% density as described below.
- Discard the original culture medium by aspiration with a pipetting gun and wash the cells 2x with PBS. When cleaning adherent cells with PBS in a cell culture dish, move the PBS to the side wall or drop it into the dish.
- After aspiration of PBS, add 0.5-1 mL of 0.25% trypsin EDTA solution to the cell culture dish or flask. Gently shake to cover the entire cell surface. Place the cell culture dish or flask in an incubator at 37 °C for about 1 min or at RT until the cells are rounded and separated. Observe exfoliation of cells with the help of an inverted microscope.
- Add equal volume of freshly formulated D10 to terminate trypsin digestion. Purge the lower surface of the cell culture flask with a pipetting gun to ensure that all cells were separated.
- Transfer the B16F10-GP cell suspension to a 15 mL centrifuge tube and centrifuge at 163 x g for 4 min at RT.
- Aspirate the supernatant and discard. Resuspend the cells in 1-2 mL of D10 for precipitation. Transfer B16F10-GP cell suspension to a new cell culture dish or flask containing 8-10 mL of D10 and then incubate in a cell incubator at 37 °C at 5% CO2.
- On the day of tumor implantation, collect B16F10-GP cells with a density of approximately 90% as described in steps 2.1.1 to 2.1.4. After centrifugation, aspirate the supernatant and discard. Resuspend the cell precipitate in 1 mL of PBS.
- Count viable cells using a 0.4% trypan blue hemocytometer. Add PBS to dilute the cell to 5 x 105 cells per 100 µL. Place the cells on ice until use.
3. Ectopic inoculation of B16F10-GP cells in the bilateral inguinal region of mice
- Aspirate 100 µL of prepared B16F10-GP cell suspension using a 1 mL syringe. Flick the tube wall to move the bubbles to the top and push the piston to move the top bubble out.
- Hold the mouse in a supine position, exposing its abdomen. Press the right hind limb of the mouse in the supine position with a finger so that the skin in the right inguinal region is fully exposed (Same with the left inguinal region).
- Remove the fur on the abdomen with depilatory cream, then clean the bilateral abdomen area with cotton containing 75% ethanol.
- Before inserting the needle, make sure that the skin in the inguinal region is tightened. Insert the needle at a 45° angle in the subcutaneous space of the inguinal region of the upper thigh. Make sure the needle is beveled upward and the depth of the needle to 0.5-1 cm.
- Apply gentle suction before injection, if there is no blood, inject the cells slowly injected into the subcutaneous tissue. At the same time, observe a small bolus (formation of fluid pocket) in the subcutaneous region.
- After injection, remove the needle, put it in the sharps box, and return the mouse to its cage.
4. Adoptive transfer of P14 T cells into tumor-bearing mice
- Perform adoptive transfer in tumor-bearing mice 6-8 days after tumor implantation, when the tumors are palpable (approximately 3-5 mm in diameter). Intraperitoneally inject 4 mg cyclophosphamide the day before transfer28,29,30.
NOTE: The aim of CTX injection is to create space in the lymphatic compartment for adoptively-transferred T cells by transiently eliminating proliferating lymphocytes. - Isolate lymphocytes from the spleen and LNs of naïve P14 transgenic mice as described below31,32 (6-10 weeks old, the same sex as tumor-bearing mice to avoid rejection issues).
NOTE: Perform the following procedures in a biosafety cabinet to ensure strictly sterile conditions.- Prepare two 6 cm dishes and add 3 mL of R2 medium to one of the dishes and 3 mL of RBL buffer to the other dish. Place a 70 µm cell filter in the Petri dish containing RBL buffer.
- Euthanize P14 mice in airtight containers containing isoflurane followed by cervical dislocation. Adjust the number of P14 mice according to the number of recipient mice.
- Harvest the spleen, inguinal, and axillary lymph nodes from the euthanized mice and transfer them to 6 cm dishes containing 4 mL of R2 medium and placed on ice.
- Place the spleen in a strainer soaked with 3 mL of RBL buffer, grind the spleen with the putter inside of the 1 mL syringe. Incubate the cells at RT for 1-2 min, then terminate the reaction with 3 mL of cold R2 medium.
- Grind the LNs with the putter inside of the 1 mL syringe until only connective tissue remains in the strainer with a 70 µm cell filter. Rinse the filter with cold R2 medium and transfer the cell suspension to a new 15 mL centrifuge tube. Centrifuge the samples at 500 x g for 6 min at 4 °C.
- Discard the supernatant and resuspend the cells with 3 mL of PBS. Use a 70 µm cell filter to remove flocculent material from the cell suspension.
- Centrifuge the cell suspension at 500 x g for 6 min at 4 °C. Resuspend the cells with 3 mL of PBS and place the tube on ice. Take a small sample, mix with trypan blue, and count cells using a blood cell counting plate.
- Determine the percentage of P14 (live/dead–CD45.1+CD8+Vα2+) cells by flow cytometry. Ensure the transferred donor cells exhibit distinct congenic marker with recipient mice (tumor-bearing B6 mice is CD45.2+). Perform staining prior to transfer to verify the correct phenotype of the transferred cells.
- Add 5 x 104– 1 x 105 cells to a 1.5 mL centrifuge tube containing 1 mL of FACS buffer. Centrifuge the cell suspension at 500 x g at 4 °C for 3 min.
- Discard the supernatant, flick the bottom of the tube to disperse the cells, and place the tube on ice.
- Prepare the following conjugated antibody mixture9,32 diluted in 100 µL of FACS buffer: anti-CD8, 1:200; anti-TCR Vα2, 1:100; anti-CD45.1, 1:200; Live/dead stain, 1:400. (Table of Materials).
- Centrifuge the antibodies mixture at 15,000 x g for 3 min to aggregate the particles. Place the mixture on ice and shield it from light by wrapping it in foil. Only take the supernatant to avoid the aspiration of particles.
- Resuspend the cells in 100 µL of the antibody mixture and thoroughly mix by flicking the tube wall. After wrapping in tin foil, incubate the tube for 30 min on ice.
- Wash the cells 2x with FACS buffer. Centrifuge the tube at 500 x g at 4 °C for 3 min. Resuspend the cells with 200 µL of FACS buffer and transfer the cell suspension to a flow tube.
- Run the flow tube with stained cells on a flow cytometer to determine the percentage of live/dead–CD45.1+CD8+Vα2+ cells.
NOTE: Generally, over 90% of CD8+ T cells from P14 transgenic mice harbored Vα2+TCRs, which are specific to H-2Db GP33-41 epitope of LCMV (lymphocytic choriomeningitis virus).
- Calculate the absolute number of live/dead–CD45.1+CD8+Vα2+ cells by multiplying its percentage and the live cell number obtained in steps 4.2 and 4.3.
- Aspirate 200 µL of P14 cell (5 x 105 cells) suspension with a 100 U insulin syringe and remove bubbles. The initial number of transferred naive P14 cells (5 x 103 – 5 x 105) did not affect the phenotype of transferred cells9.
- Place the mice in a cage and warm with an infrared lamp for 5-10 min to expand the tail vein. Hold in place with an appropriately sized mouse fixator, straighten the tail, and wipe it with a cotton ball containing 75% ethanol to make the veins visible.
- Insert the needle parallel to the tail vein and gently pull back the plunger. If there is blood flowing into the syringe, slowly push the cell suspension into the vein.
NOTE: If there is resistance or swelling of the tail during injection, the injection position needs to be adjusted. The injection site should start at the distal end. - After the injection is completed, pull out the needle quickly and press the injection site gently with a cotton ball. Return the mouse to a new clean cage and closely observe it for several minutes for any adverse effects.
5. Resection of the primary tumor
NOTE: Ensure all surgical instruments are autoclaved before use. Sterilize the operating area inside the biosafety cabinet with 75% ethanol, followed by UV irradiation for at least 30 min. Wear clean gowns, hats, masks, and sterile gloves during surgery.
- When the tumor is palpable (approximately 5 mm in diameter), resect the primary tumor.
- Anesthetize the mice with intraperitoneal injection of Ketamine (75 mg/kg). Pinch foot pad to evaluate the degree of anesthesia, if there is no pain reflex, it indicates the right timing for surgery. If the hind limbs were withdrawn, provide another dose of 10-30 µL.
NOTE: Alternatively, intraperitoneal medetomidine (1 mg/kg body weight; Domitor) is recommended to anesthetize mice. - Apply the drying preventive ointment on mouse eyes. Remove the fur on the abdomen with depilatory cream to fully expose the surgical field.
- Place the mouse in a biosafety cabinet and place it on an anatomical board covered with clean absorbent paper in a supine position so that the longitudinal axis of the mouse is parallel to the experimenter.
NOTE: To maintain a strict sterile environment, the following procedures must be performed in a biosafety cabinet. - Disinfect the abdomen of the mice with a cotton ball soaked in povidone-iodine. Incise the skin near the tumor-bearing site with a sterile scalpel or opthalmic scissors. Insert closed scissors tip into the incision to clearly expose the tumor. When incising the skin, make sure not to damage the inguinal lymph nodes.
- During tumor removal, keep the capsule as intact as possible. Carefully and gently remove the connective tissue adjacent to the tumor with sterile scissors. Remove the tumor in situ completely, otherwise it may recur.
6. Intranodal injection of B16F10-GP cells in the inguinal lymph node
NOTE: After bilateral tumor clearance, B16F10-GP cells were injected into unilateral inguinal lymph node and PBS was injected into the other side.
- Aspirate 20 µL (5 x 104 cells) of B16F10-GP cell suspension with a 100 U insulin syringe, remove bubbles and then inject into one inguinal lymph node. Inject an equal volume of PBS into the inguinal lymph node on the other side.
- During the injection, insert the needle from the distal end of the lymph node, and then slowly insert the needle to the center of the lymph node. At this time, if the fluid is accurately injected into the lymph node, the lymph node can be seen to swell significantly.
NOTE: When the needle is inserted from the distal end of the lymph node, make sure not to puncture the node.
- During the injection, insert the needle from the distal end of the lymph node, and then slowly insert the needle to the center of the lymph node. At this time, if the fluid is accurately injected into the lymph node, the lymph node can be seen to swell significantly.
- Suture the incision with 2-3 stitches using a 3-0 suture. Disinfect the skin surrounding the wound with cotton impregnated with povidone iodine. Take care to bypass lymph nodes during suturing.
- Place the mouse in a clean cage and keep warm using infrared light in the lateral decubitus position. Monitor continuously until consciousness is recovered. Ensure that the mice that has undergone surgery is not returned to the company of other animals until fully recovered.
- Give buprenorphine every 4-6 h for 3 consecutive days after the operation to relieve postoperative pain (at a dose of 0.1-0.5 mg/kg, SQ or IP). Monitor the feeding, drinking, movement, and activity areas of the mice. Typically, mice recover from surgical trauma within 3 days.
NOTE: If mice are unable to resume normal feeding and activities and show any signs of infection, consult a veterinarian for intervention or euthanize it. - Sacrifice mice at different time points: day 8 and day 18 after intranodal injection of tumor cells. Recover activated donor cells using flow cytometry analysis.
Representative Results
The schematic diagram of this experimental design is shown in Figure 1A. A total of 5 x 105 B16F10-GP cells in 100 µL of PBS were subcutaneously (s.c.) implanted into the bilateral inguinal region of CD45.2 C57BL/6J mice. After 7 days, these tumor-bearing mice were intraperitoneally (i.p.) injected with 4 mg CTX, followed by the adoptive transfer of 5 x 105 CD45.1+P14 cells through tail intravenous (i.v.) injection. When tumors grew to approximately 3-5 mm in diameter (about 7 days after P14 cells transfer), primary tumors were resected, and 5 x 104 B16F10-GP cells in 20 µL of PBS were directly injected into unilateral inguinal lymph node. The inguinal lymph node on the other side was injected with equal volumes of PBS. Representative hematoxylin and eosin (H&E, 100x) staining of the non-metastatic lymph node (nLN) and metastatic lymph node (mLN) at indicated time points are shown in Figure 1B. The structure of nLN was intact. At the early stage of LN metastasis (D8), mLN was partially occupied with tumor cells (black arrow), and there is still some remaining area with lymphocytes that have not been invaded by tumor cells (red arrow). While at the late stage of LN metastasis (D18), mLN is filled with tumor cells accompanied by tumor angiogenesis and little lymphocytes. Activated P14 cells recovered in TdLN produced high level of IFN-γ after GP33-41 peptide stimulation9,31. Here, the percentages of activated P14 cells were analyzed through flow cytometry at different time points and the gating strategy is shown in Figure 2. The frequency of antigen-specific CD8+ T cells in peripheral blood at the early stage (D8) and late stage (D18) is 2.81% and 1.48%, respectively (Figure 3A). Tumor-specific CD8+ T cells have been reported to strictly reside in dLN during tumorigenesis and non-draining LN recovered limited donor cells33. The percentage of antigen-specific P14 cells in nLN was stable during LN metastasis. Intriguingly, antigen-specific CD8+ T cells in mLN were transiently boosted at the early stage, evidenced by the higher frequency of P14 cells compared to nLN, while it sharply decreased at the late stage (Figure 3B).
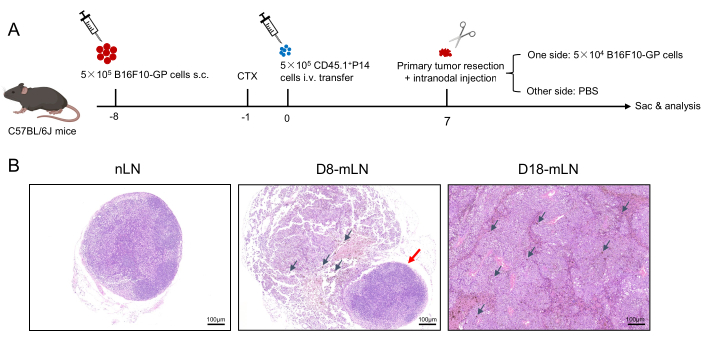
Figure 1: Schematic diagram of the experimental design. (A) C57BL/6J mice (CD45.2+) are implanted with 5 x 105 B16F10-GP tumor cells on the bilateral inguinal region. After 7 days, these mice are intraperitoneally injected with 4 mg CTX, and followed by the adoptive transfer of different congenically marked (CD45.1+) P14 cells the next day. When tumors grow to approximately 3-5 mm in diameter (about 7 days after P14 cells transfer), primary tumors are resected, then 5 x 104 B16F10-GP cells in 20 µL of PBS are directly injected into unilateral inguinal lymph node, and the inguinal lymph node on the other side is injected with equal volumes of PBS. (B) Representative hematoxylin and eosin (H&E,100x) staining of the LNs. Abbreviations: s.c. = subcutaneous; CTX= cyclophosphamide; i.v. = intravenous; Sac = sacrifice; nLN = non-metastatic LN; mLN = metastatic LN. Please click here to view a larger version of this figure.
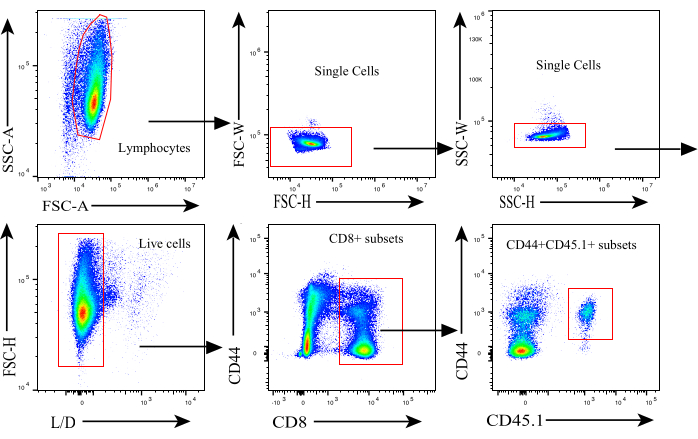
Figure 2: Gating strategy for flow cytometry analysis. Gating strategy used to identify donor-derived activated antigen-specific CD8+ T cells. Abbreviations: L/D = live/dead. Please click here to view a larger version of this figure.
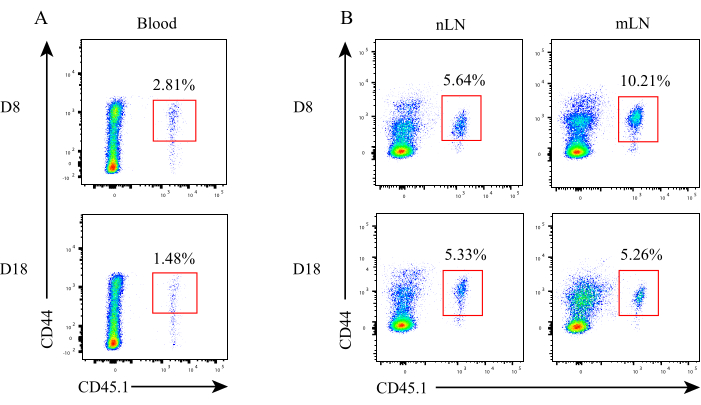
Figure 3: Dynamics of antigen-specific CD8+ T cells during LN metastasis. The proportion of antigen-specific CD8+ T cells in (A) peripheral blood, (B) nLN and mLN at different time points. Abbreviations: nLN = non-metastatic LN; mLN = metastatic LN. Please click here to view a larger version of this figure.
Discussion
During tumorigenesis, antigen-presenting cells (APCs) engulf tumor antigens and migrate to TdLN where they prime CD8+ T cells. After priming and activation, CD8+ T cells leave the TdLN and infiltrate the tumor to kill tumor cells10. Through TdLN resection and the administration of FTY720 which block the exit of immune cells from the lymphoid organs, several studies have demonstrated the pivotal role of TdLN in ensuring the efficacy of PD-1/PD-L1 checkpoint therapy34,35. Consistent with this, we recently found that tumor-specific memory CD8+ T cells (TTSM) predominantly reside in TdLN, these TdLN-TTSM cells serve as bona fide responders to ICB9. Unfortunately, several types of cancer cells often spread to TdLNs from primary tumor sites, which lead to structural reconfirmation and immune cell dysfunction in TdLNs. The impaired quantity and quality of CD8+ T cells in mLNs has already been described20,36. However, the dynamic changes of CD8+ T cells, especially the tumor antigen-specific CD8+ T cells during the LN metastatic cascade have not been elucidated.
Here, we developed a convenient mouse model to monitor the dynamics of systemic and local antigen-specific CD8+ T cells during the process of LN metastasis. B16F10-GP melanoma cells were subcutaneously implanted into the bilateral inguinal region, followed by adoptively transferring with P14 cells which could be specifically activated by the GP-derived peptide GP33-41. At day 7 post cell transfer when P14 cells in inguinal LNs were fully activated, primary tumors in both sides were resected and B16F10-GP cells were directly injected into unilateral inguinal LN, sham operation on the other side was performed through the injection with equal volumes of PBS. This ingenious design enables the comparison of P14 cells in mLN and non-metastatic LN (nLN) within the same mice. Simultaneously, P14 cells in periphery blood within the same mice were detected at different time points during LN metastasis by bleeding the orbital vein. Apart from the frequencies of P14 cells in periphery blood and TdLNs at different stages during LN metastasis, the transcriptional and epigenetic properties of these antigen-specific CD8+ T cells could be further examined with other techniques.
Of note, several steps should be performed cautiously. Firstly, the primary tumors should be excised thoroughly, as the residual tumor cells would regrow rapidly. Secondly, the operation should be performed gently, as tumor tissues contain abundant new blood vessels which are usually inevitably broken during primary tumor resection and lead to the death of surgical mice due to massive hemorrhage. Therefore, the timing of primary tumor resection is critically important. Generally, it is relatively safe to remove it when the tumor grows to the size about 5 x 5 mm, and tumor cells should be precisely injected into LN rather than the bottom or adjacent tissues. Moreover, the volume of tumor cell suspensions should be controlled below 20 µL, otherwise spill would form new tumor outside the LN. Lastly, a limitation of this protocol is that the operation is traumatic, and mice may experience infection during healing process, which would affect the properties of antigen-specific CD8+ T cells. Therefore, it is critically important to maintain strict asepsis during the surgery and sham-operated PBS injection should be done to exclude the impact of injection-induced physical damage to the antigen-specific CD8+ T cells.
Overall, we provide a convenient model to investigate the antigen-specific CD8+ T cells during LN metastasis and the interactions between antigen-specific CD8+ T cells and other immune cells or stroma cells during LN metastasis could also be elucidated. In addition, it could be easily extended to several other tumor models. Collectively, this protocol offers a useful reproductive model for the study of cancer immunology.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Science Foundation for Outstanding Young Scholars of China (No. 82122028 to LX), the National Natural Science Foundation of China (No. 82173094 to LX), Natural Science Foundation of Chong Qing (No. 2023NSCQ-BHX0087 to SW).
Materials
| 1.5 mL centrifuge tube | KIRGEN | KG2211 | |
| 100 U insulin syringe | BD Biosciences | 320310 | |
| 15 mL conical tube | BEAVER | 43008 | |
| 2,2,2-Tribromoethanol (Avertin) | Sigma | T48402-25G | |
| 2-Methyl-2-butanol | Sigma | 240486-100ML | |
| 70 μm nylon cell strainer | BD Falcon | 352350 | |
| APC anti-mouse CD45.1 | BioLegend | 110714 | Clone:A20 |
| B16-GP cell line | Beijing Biocytogen Co.Ltd, China | Custom | |
| BSA-V (bovine serum albumin) | Bioss | bs-0292P | |
| cell culture dish | BEAVER | 43701/43702/43703 | |
| centrifuge | Eppendorf | 5810R-A462/5424R | |
| cyclophosphamide | Sigma | C0768-25G | |
| Cyclophosphamide (CTX) | Sigma | PHR1404 | |
| Dulbecco's Modified Eagle Medium | Gibco | C11995500BT | |
| EDTA | Sigma | EDS-500g | |
| FACS tubes | BD Falcon | 352052 | |
| fetal bovine serum | Gibco | 10270-106 | |
| flow cytometer | BD | FACSCanto II | |
| hemocytometer | PorLab Scientific | HM330 | |
| isoflurane | RWD life science | R510-22-16 | |
| KHCO3 | Sangon Biotech | A501195-0500 | |
| LIVE/DEAD Fixable Near-IR Dead Cell Stain Kit, for 633 or 635 nm excitation | Life Technologies | L10199 | |
| needle carrier | RWD Life Science | F31034-14 | |
| NH4Cl | Sangon Biotech | A501569-0500 | |
| paraformaldehyde | Beyotime | P0099-500ml | |
| PE anti-mouse TCR Vα2 | BioLegend | 127808 | Clone:B20.1 |
| Pen Strep Glutamine (100x) | Gibco | 10378-016 | |
| PerCP/Cy5.5 anti-mouse CD8a | BioLegend | 100734 | Clone:53-6.7 |
| RPMI-1640 | Sigma | R8758-500ML | |
| sodium azide | Sigma | S2002 | |
| surgical forceps | RWD Life Science | F12005-10 | |
| surgical scissors | RWD Life Science | S12003-09 | |
| suture thread | RWD Life Science | F34004-30 | |
| trypsin-EDTA | Sigma | T4049-100ml |
Riferimenti
- Morad, G., Helmink, B. A., Sharma, P., Wargo, J. A. Hallmarks of response, resistance, and toxicity to immune checkpoint blockade. Cell. 184 (21), 5309-5337 (2021).
- Korman, A. J., Garrett-Thomson, S. C., Lonberg, N. The foundations of immune checkpoint blockade and the ipilimumab approval decennial. Nat Rev Drug Discov. 21 (7), 509-528 (2022).
- Chamoto, K., et al. Mitochondrial activation chemicals synergize with surface receptor PD-1 blockade for T cell-dependent antitumor activity. Proc Natl Acad Sci U S A. 114 (5), E761-E770 (2017).
- Spitzer, M. H., et al. Systemic immunity is required for effective cancer immunotherapy. Cell. 168 (3), 487-502 (2017).
- Yost, K. E., et al. Clonal replacement of tumor-specific T cells following PD-1 blockade. Nat Med. 25 (8), 1251-1259 (2019).
- Wu, T. D., et al. Peripheral T cell expansion predicts tumour infiltration and clinical response. Nature. 579 (7798), 274-278 (2020).
- Connolly, K. A., et al. A reservoir of stem-like cd8(+) t cells in the tumor-draining lymph node preserves the ongoing antitumor immune response. Sci Immunol. 6 (64), eabg7836 (2021).
- Schenkel, J. M., et al. Conventional type I dendritic cells maintain a reservoir of proliferative tumor-antigen specific Tcf-1+ CD8+ T cells in tumor-draining lymph nodes. Immunity. 54 (10), 2338-2353 (2021).
- Huang, Q., et al. The primordial differentiation of tumor-specific memory cd8(+) t cells as bona fide responders to pd-1/pd-l1 blockade in draining lymph nodes. Cell. 185 (22), 4049-4066 (2022).
- Kanda, Y., Okazaki, T., Katakai, T. Motility dynamics of T cells in tumor-draining lymph nodes: A rational indicator of antitumor response and immune checkpoint blockade. Cancers (Basel). 13 (18), 4616 (2021).
- Karaman, S., Detmar, M. Mechanisms of lymphatic metastasis. J Clin Invest. 124 (3), 922-928 (2014).
- Pereira, E. R., et al. Lymph node metastases can invade local blood vessels, exit the node, and colonize distant organs in mice. Science. 359 (6382), 1403-1407 (2018).
- Brown, M., et al. Lymph node blood vessels provide exit routes for metastatic tumor cell dissemination in mice. Science. 359 (6382), 1408-1411 (2018).
- Reticker-Flynn, N. E., et al. Lymph node colonization induces tumor-immune tolerance to promote distant metastasis. Cell. 185 (11), 1924-1942 (2022).
- Leong, S. P., et al. Impact of nodal status and tumor burden in sentinel lymph nodes on the clinical outcomes of cancer patients. J Surg Oncol. 103 (6), 518-530 (2011).
- Lyman, G. H., et al. Sentinel lymph node biopsy for patients with early-stage breast cancer: American society of clinical oncology clinical practice guideline update. J Clin Oncol. 35 (5), 561-564 (2017).
- Wong, S. L., et al. Sentinel lymph node biopsy and management of regional lymph nodes in melanoma: American society of clinical oncology and society of surgical oncology clinical practice guideline update. Ann Surg Oncol. 25 (2), 356-377 (2018).
- Faries, M. B., et al. Completion dissection or observation for sentinel-node metastasis in melanoma. N Engl J Med. 376 (23), 2211-2222 (2017).
- Giuliano, A. E., et al. Effect of axillary dissection vs no axillary dissection on 10-year overall survival among women with invasive breast cancer and sentinel node metastasis: The ACOSOG Z0011 (alliance) randomized clinical trial. JAMA. 318 (10), 918-926 (2017).
- du Bois, H., Heim, T. A., Lund, A. W. Tumor-draining lymph nodes: At the crossroads of metastasis and immunity. Sci Immunol. 6 (63), eabg3551 (2021).
- An, S., et al. Locally trapping the c-c chemokine receptor type 7 by gene delivery nanoparticle inhibits lymphatic metastasis prior to tumor resection. Small. 15 (9), e1805182 (2019).
- Lee, C. K., et al. Tumor metastasis to lymph nodes requires yap-dependent metabolic adaptation. Science. 363 (6427), 644-649 (2019).
- Buchwald, Z. S., et al. Tumor-draining lymph node is important for a robust abscopal effect stimulated by radiotherapy. J ImmunoTher Cancer. 8 (2), e000867 (2020).
- Siddiqui, I., et al. Intratumoral Tcf1+PD-1+CD8+ T cells with stem-like properties promote tumor control in response to vaccination and checkpoint blockade immunotherapy. Immunity. 50 (1), 195.e10-211.e10 (2019).
- Ashton-Rickardt, P. G., et al. Evidence for a differential avidity model of T cell selection in the thymus. Cell. 76 (4), 651-663 (1994).
- Manjunath, N., et al. Effector differentiation is not prerequisite for generation of memory cytotoxic T lymphocytes. J Clin Invest. 108 (6), 871-878 (2001).
- Khan, O., et al. TOX transcriptionally and epigenetically programs CD8+ T cell exhaustion. Nature. 571 (7764), 211-218 (2019).
- North, R. J. Cyclophosphamide-facilitated adoptive immunotherapy of an established tumor depends on elimination of tumor-induced suppressor T cells. J Exp Med. 155 (4), 1063-1074 (1982).
- Maine, G. N., Mule, J. J. Making room for T cells. J Clin Invest. 110 (2), 157-159 (2002).
- Xue, G., et al. Adoptive cell therapy with tumor-specific th9 cells induces viral mimicry to eliminate antigen-loss-variant tumor cells. Cancer Cell. 39 (12), 1610.e9-1622.e9 (2021).
- Prokhnevska, N., et al. CD8+ T cell activation in cancer comprises an initial activation phase in lymph nodes followed by effector differentiation within the tumor. Immunity. 56 (1), 107.e5-124.e5 (2023).
- Wang, L., et al. Tumor transplantation for assessing the dynamics of tumor-infiltrating CD8+ T cells in mice. J Vis Exp. (172), e62442 (2021).
- Liu, Q., et al. Tumor-specific memory cd8(+) t cells are strictly resident in draining lymph nodes during tumorigenesis. Cell Mol Immunol. 20 (4), 423-426 (2023).
- Fransen, M. F., et al. Tumor-draining lymph nodes are pivotal in pd-1/pd-l1 checkpoint therapy. JCI Insight. 3 (23), e124507 (2018).
- Francis, D. M., et al. Blockade of immune checkpoints in lymph nodes through locoregional delivery augments cancer immunotherapy. Sci Transl Med. 12 (563), eaay3575 (2020).
- Garner, H., de Visser, K. E. Immune crosstalk in cancer progression and metastatic spread: A complex conversation. Nat Rev Immunol. 20 (8), 483-497 (2020).

