Summary
This paper details the creation of a Psoriasis Thickness Reference Card, enabling psoriasis patients to self-assess their psoriasis plaque thickness without the need for physician assistance. Additionally, the precision of self-assessment for psoriasis redness and scaliness by employing reference images depicting various skin tones is assessed.
Abstract
Psoriasis plaque severity metrics, such as induration (thickness), erythema (redness), and desquamation (scaliness), are associated with the subsequent development of psoriatic arthritis (PsA) among cutaneous-only psoriasis patients (patients with skin or nail psoriasis but no psoriatic arthritis). These metrics can be used for PsA screening. However, a key challenge in PsA screening is to optimize accessibility and minimize costs for patients, while also reducing the burden on healthcare systems. Therefore, an ideal screening tool consists of questions that patients can answer without a physician’s assistance. Although reference images can be used to help a patient self-assess erythema and desquamation severity, a patient would need a tactile induration reference card to self-assess induration severity. This protocol describes how to create an induration reference card, the Psoriasis Thickness Reference Card, as well as how to use it to assess lesion induration severity. Administration of reference images for erythema and desquamation and a Psoriasis Thickness Reference Card for induration to 27 psoriasis patients showed that patients were moderately successful at self-assessing the severity of these three metrics. These findings support the feasibility of a future PsA screening test that patients can complete without the need for physician assistance.
Introduction
Psoriatic arthritis (PsA) is an inflammatory arthritis that develops in approximately 2.9% of patients with psoriasis each year1,2. Early prediction of which patients will develop PsA is crucial for a patient's quality of life as a diagnostic delay of as little as six months may cause irreversible damage to the joints3. A previous study had identified features associated with early-stage PsA. The three components of the Investigator's Global Assessment (IGA)-induration (thickness), erythema (redness), and desquamation (scaliness)-were highly correlated with each other and were nominally associated with subsequent PsA development. Multivariate analysis showed that a history of fingernail psoriasis and untreated induration of psoriasis lesions were independent PsA predictors4. Other studies have also identified psoriasis severity as associated with psoriatic arthritis2,5,6. The collection of these clinical factors is likely to be important in future PsA screening.
Several PsA screening tests exist7,8,9,10, but they require physicians to conduct them. A questionnaire-based screening test that can be completed by patients without physician assistance is an ideal format because it will not burden the dermatology and primary care clinics. Previously, the authors described a questionnaire with images that depict different severities of erythema and desquamation in multiple skin tones11. However, the measurement of induration requires a reference card corresponding to various plaque severities, such as that developed by the National Psoriasis Foundation12. The reference card is characterized by multiple raised areas of varying elevations, each corresponding to a number representing induration severity. To assess a patient's psoriasis severity in terms of induration, the card user compares the thickest psoriasis patch on a patient's body to the elevated areas on the card.
However, distributing a reference card to psoriasis patients may not be the most efficient approach for reaching a broad audience, especially for patients located outside the USA and for online survey studies. For researchers and clinicians gathering induration self-assessments from numerous patients, an alternative option could involve creating a homemade induration card using inexpensive office supplies. For patients, creating a homemade device from inexpensive office supplies can be more cost-effective than purchasing commercially available alternatives. This protocol describes how to create the Psoriasis Thickness Reference Card, an induration reference card that psoriasis patients can feel to assess the severity of their psoriasis induration. The Card can be made with standard office supplies and is inexpensive to make.
Protocol
This study was approved by the University of Utah Institutional Review Board (IRB_00137694). Twenty-seven psoriasis patients at the University of Utah dermatology clinic were enlisted to complete a questionnaire about psoriasis severity at the time of the questionnaire's administration. All patients provided informed consent and completed the questionnaire without instructions from research or medical personnel, either before or immediately after the dermatologist's assessment.
1. Procedure for making a Psoriasis Thickness Reference Card
- Print the Psoriasis Thickness Reference Card template (Supplemental Figure S1) on a standard piece of printer paper. If a printer is not accessible, manually write down the numbers as in the template on any paper and follow the subsequent steps.
- Place sticky notes.
- Purchase a package of sticky notes. The smallest-sized sticky notes (5.08 cm height x 5.08 cm width or 5.08 cm height x 3.81 cm width) will work. The thickness of each sticky note should be between 0.09 and 0.1 mm.
NOTE: The authors have tested multiple brands of sticky notes (see Table of Materials) in the market and found that they all fulfill this requirement. If in doubt, one can calculate the thickness of each page by dividing the total thickness of a stack of sticky notes by the number of pages in the stack. - Place a stack of three sticky notes on the box labeled "3."
- Place a stack of 5 sticky notes on the box labeled "5."
- Place a stack of 8 sticky notes on the box labeled "8."
- Place a stack of 10 sticky notes on the box labeled "10."
- Place a stack of 13 sticky notes on the box labeled "13."
- Purchase a package of sticky notes. The smallest-sized sticky notes (5.08 cm height x 5.08 cm width or 5.08 cm height x 3.81 cm width) will work. The thickness of each sticky note should be between 0.09 and 0.1 mm.
- Align the adhesive side of the sticky notes on the left side of each corresponding box and press them down to adhere each stack firmly to the sheet of paper.
- Apply a strip of tape along the right side of each stack of sticky notes (Figure 1). Ensure the tape adheres smoothly, creating a gentle slope between the top of the stack and the paper.
NOTE: If the left side of each stack is not adhered securely to the paper via the sticky note adhesive, tape the left side in the same way as the right side.
2. Usage of the Psoriasis Thickness Reference Card
- Slide a finger across the taped side of each stack of sticky notes to get a sense of its thickness or elevation from the paper it is adhered to.
- In a similar manner, slide a finger across the edge of one's thickest psoriasis patch, feeling its elevation from the surrounding, unaffected skin.
- Identify the stack that is most similar in elevation to one's thickest psoriasis patch and record the corresponding score of 0 to 13.
Representative Results
To validate whether patient assessments of desquamation, erythema, and induration are comparable to those of a dermatologist, 27 psoriasis patients at the University of Utah dermatology clinic were enlisted to complete a questionnaire about psoriasis severity at the time of the questionnaire's administration. Each participant was provided with a Psoriasis Thickness Reference Card along with the questionnaire. The dermatologist provided their independent assessment of the current status of each patient using the same measuring scales.
Psoriasis severity was categorized into two groups, mild versus moderate to severe. Grouping moderate and severe psoriasis aligns with the clinical management of psoriasis patients. For induration, moderate corresponds to 5-8 sticky notes and severe corresponds to 10-13 sticky notes. The accuracy of patients' assessments of desquamation, erythema, and induration was evaluated by sensitivity (the test's ability to correctly identify an individual as having the disease) and specificity (the test's ability to correctly identify an individual as not having the disease) using the physician's assessments as the gold standard. The Wilcoxon signed-rank test was used to examine whether patient assessments of psoriasis severity differed significantly from those of the attending dermatologist. Cohen's kappa was used to evaluate whether the two assessments were comparable.
The results demonstrated that the patients' and physician's answers to some questions regarding psoriasis severity are moderately to highly concordant (Figure 2 and Table 1). The numbers of physician-patient severity assessments with matching values were 23 (85.2%), 25 (92.6%), and 22 (84.6%) for desquamation, erythema, and induration (which was answered by 26 out of 27 participants), respectively. There was no significant difference between patient and physician reports of desquamation (p = 0.42), erythema (p > 0.99), and induration (p > 0.99) by the Wilcoxon signed-rank test. Sensitivity ranged from 0.83 to 0.93 and specificity from 0.77 to 0.92. Cohen's kappa values revealed at least some level of agreement between the physician and patient assessments of all three variables (kappa scores > 0.69, p-values < 0.000002). Using Cohen's kappa interpretations suggested by McHugh13, moderate agreement was observed between physician and patient measurements of desquamation and induration; physician and patient measurements of erythema exhibited strong agreement.
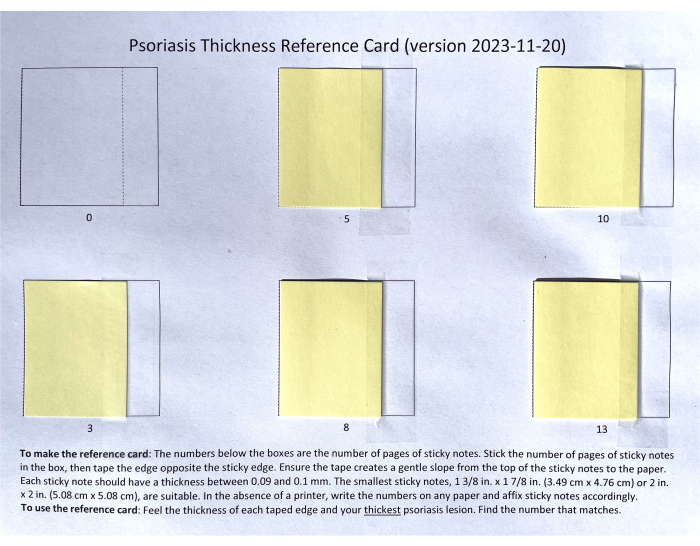
Figure 1: Completed Psoriasis Thickness Reference Card. The number under each square in the card indicates the number of sticky notes in each stack. The stacks of sticky notes are attached to the paper using the adhesive side of the notes on the left and a piece of tape on the right. Please click here to view a larger version of this figure.
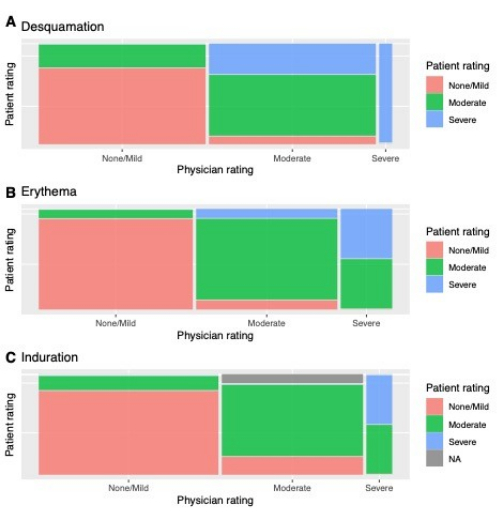
Figure 2: Proportions of patient assessments that matched or did not match physician assessments. Each patient rated the severity of their current psoriasis lesions based on three psoriatic features; the attending dermatologist also rated each patient's psoriasis severity on the same scale. Each of the three columns represents physician ratings; the width of each column represents the proportion of patients given that rating by the physician. Within each column, the different colors represent the proportion of those patients given a specific physician rating who were also given a specific patient rating, e.g. approximately 50% of the patients who were assessed as having severe erythema by a physician were self-assessed as having moderate erythema. (A) Desquamation, (B) erythema, and (C) induration. Abbreviation: NA = not applicable (patient did not provide an assessment). Please click here to view a larger version of this figure.
| Desquamation | Erythema | Induration | |
| True positive* | 13 | 14 | 10 |
| False positive* | 3 | 1 | 2 |
| False negative* | 1 | 1 | 2 |
| True negative* | 10 | 11 | 12 |
| Sensitivity | 0.93 | 0.93 | 0.83 |
| Specificity | 0.77 | 0.92 | 0.86 |
| Accuracy | 0.85 | 0.93 | 0.85 |
| Wilcoxon signed rank test p-value | 0.42 | >0.99 | >0.99 |
| Cohen’s kappa value [95% CI] | 0.70 [0.43,0.97] | 0.85 [0.65,1.00] | 0.69 [0.41,0.97] |
| Cohen’s kappa category | Moderate | Strong | Moderate |
Table 1: Predictive value, sensitivity, and specificity of patient psoriasis severity ratings. Physician ratings were treated as the true values to which the patient ratings were compared. *Positive is moderate to severe psoriasis; negative is mild to no psoriasis. Abbreviation: CI = confidence interval.
Supplemental Figure S1: Psoriasis thickness Reference Card template. The template may be printed and used to directly place the sticky notes on, or it may be used as a guide for numbering and placing sticky notes on a sheet of paper if no printer is available. Please click here to download this File.
Discussion
Protocol step 1.4 is a crucial aspect of the proper construction of the Psoriasis Thickness Reference Card. To achieve an accurate representation of plaque thickness, it is imperative to avoid placing the tape vertically or flush against the vertical edge of the stack of sticky notes. If the tape is positioned in such a manner, the sharp edge of each stack will not mimic the feel of a psoriasis plaque. The tape should be applied in a way that creates a smooth slope connecting the stack to the paper.
Since the method is relatively simple, there are limited instances where modifications and troubleshooting might be necessary. If the sheet of printer paper is too flimsy, a piece of cardstock can be used as an alternative. Furthermore, in case the left sides of the stacks of sticky notes lose their adhesiveness, the left side of each stack may be taped to the paper using the same instructions as described in protocol step 1.4.
The main alternative method for induration scoring involves the physician assessing the patients' induration severity. While this approach benefits from the expertise of a trained professional, it does require additional time during the visit, and it may not be practical for patients in remote locations. To address these challenges, the Psoriasis Thickness Reference Card offers a solution by empowering patients to conduct the induration assessment themselves before their clinic visit or even from the comfort of their home. This patient-driven approach provides a convenient and efficient means of monitoring induration severity. Moreover, research indicates that induration severity at its worst holds a greater predictive value for PsA than induration severity at the time of a clinical visit4. Therefore, frequent monitoring of psoriasis thickness by patients is more likely to yield data that are relevant for PsA prediction, enhancing the accuracy of the assessment and potentially leading to early detection and intervention.
Patients can utilize the card to assess the severity of induration in their psoriasis lesions, but it demonstrates only moderate concordance with the doctor's assessment. The cause of the discordance between patient and doctor assessments is not entirely clear.
A 2022 study found that a Patient-oriented Psoriasis Area and Severity Index had good interrater reliability and was positively correlated with the Psoriasis Area and Severity Index (PASI) as calculated by physicians. The present study did not include the percentage of body surface affected (BSA) and PASI. While BSA has been shown to be associated with PsA5,14, previous research demonstrates that induration is a stronger predictor4. Furthermore, both BSA and PASI are more challenging to measure, making them less suitable for patient self-assessment. As a result, BSA and PASI were not incorporated into the study.
In conclusion, the use of the Psoriasis Thickness Reference Card empowers psoriasis patients to independently measure the thickness of their psoriasis lesions. This approach can be utilized in a screening test to predict PsA development in individuals with cutaneous-only psoriasis. Advantages include simplicity, cost-effectiveness, and self-reliance, enabling convenient administration even for patients situated in remote locations. This approach has the potential to expand PsA risk assessment accessibility and reduce research costs related to psoriasis and PsA.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This study has been supported by the Discovery Research Grant (B.-J.F.) and the PsA Diagnostic Test Grant (B.-J.F. and J.A.W.) from the National Psoriasis Foundation, and the Utah Genome Project Partnership Grant (B.-J.F. and J.A.W.) and the Immunology, Inflammation, & Infectious Disease (3i) Initiative Seed Grant (B.-J.F. and J.A.W.) from the University of Utah. The author would like to thank Tyler S. Nelson (Physician Assistant, University of Utah Department of Dermatology) and Angela Contreras (Study Coordinator, University of Utah Department of Dermatology) for recruiting patients for the study.
Materials
| Post-it Notes, 1 3/8 in x 1 7/8 in, Canary Yellow, 6 Pads/Pack | 3M | 00021200569029 | The smallest sized sticky notes (5.08 cm height x 5.08 cm width or 5.08 cm height x 3.81 cm width) work best with the printed template. The thickness of each sticky note should be between 0.09 and 0.1 mm; sticky notes from the brands Post-it, Pen+Gear, and Office Works meet these requirements. |
| Printworks Printer and Copy Paper – 500 Pack – White | Paris Corporation | 0009014600006 | Any brand of printer paper is acceptable; if recreating the template manually rather than printing, any brand of paper is acceptable. |
| R | http://www.r-project.org | The R programming language version 4.3.1 was used to analyze the data in this study. | |
| Scotch Magic Tape Dispensered Rolls | 3M | 00021200000041 | Any brand of smooth adhesive tape is acceptable; do not use tape with a coarse texture. |
Riferimenti
- Merola, J. F., et al. Incidence and prevalence of psoriatic arthritis in patients with psoriasis stratified by psoriasis disease severity: Retrospective analysis of an electronic health records database in the United States. Journal of the American Academy of Dermatology. 86 (4), 748-757 (2022).
- Eder, L., et al. The incidence and risk factors for psoriatic arthritis in patients with psoriasis: a prospective cohort study. Arthritis & Rheumatology. 68 (4), 915-923 (2016).
- Haroon, M., Gallagher, P., FitzGerald, O. Diagnostic delay of more than 6 months contributes to poor radiographic and functional outcome in psoriatic arthritis. Annals of the Rheumatic Diseases. 74 (6), 1045-1050 (2015).
- Belman, S., et al. Psoriasis characteristics for the early detection of psoriatic arthritis. The Journal of Rheumatology. 48 (10), 1559-1565 (2021).
- Tey, H. L., et al. Risk factors associated with having psoriatic arthritis in patients with cutaneous psoriasis. The Journal of Dermatology. 37 (5), 426-430 (2010).
- Rouzaud, M., et al. Is there a psoriasis skin phenotype associated with psoriatic arthritis? Systematic literature review. Journal of the European Academy of Dermatology and Venereology JEADV. 28, 17-26 (2014).
- Gladman, D. D., et al. Development and initial validation of a screening questionnaire for psoriatic arthritis: the Toronto Psoriatic Arthritis Screen (ToPAS). Annals of the Rheumatic Diseases. 68 (4), 497-501 (2009).
- Tom, B. D. M., Chandran, V., Farewell, V. T., Rosen, C. F., Gladman, D. D. Validation of the Toronto Psoriatic Arthritis Screen Version 2 (ToPAS 2). The Journal of Rheumatology. 42 (5), 841-846 (2015).
- Husni, M. E., Meyer, K. H., Cohen, D. S., Mody, E., Qureshi, A. A. The PASE questionnaire: pilot-testing a psoriatic arthritis screening and evaluation tool. Journal of the American Academy of Dermatology. 57 (4), 581-587 (2007).
- Ibrahim, G. H., Buch, M. H., Lawson, C., Waxman, R., Helliwell, P. S. Evaluation of an existing screening tool for psoriatic arthritis in people with psoriasis and the development of a new instrument: the Psoriasis Epidemiology Screening Tool (PEST) questionnaire. Clinical and Experimental Rheumatology. 27 (3), 469-474 (2009).
- Walsh, J. A., et al. PAPRIKA: A question bank for assessing psoriatic arthritis risk in individuals of diverse ancestries. Arthritis Care & Research. , (2023).
- Feldman, S. R., Krueger, G. G. Psoriasis assessment tools in clinical trials. Annals of the Rheumatic Diseases. 64, 65-68 (2005).
- McHugh, M. L. Interrater reliability: the kappa statistic. Biochemia Medica. 22 (3), 276-282 (2012).
- Montes de Oca Pedrosa, A., Oakley, A., Rogers, J., Rangaitaha Epiha, M. PASI vs PO-PASI: Patient-Oriented PASI (PO-PASI) is comparable to clinician score PASI. The Australasian Journal of Dermatology. 63 (1), e67-e70 (2022).

.
