Laparoscopic Posterior Radical Antegrade Modular Pancreatosplenectomy for Distal Pancreatic Carcinoma
Summary
Laparoscopic radical antegrade modular pancreatosplenectomy (L-RAMPS) is currently considered technically safe and feasible. However, due to technical challenges and a lack of supporting evidence for widespread clinical applications, only a limited number of institutions are currently conducting L-RAMPS. This article aims to provide detailed techniques for laparoscopic posterior radical antegrade modular pancreatosplenectomy.
Abstract
Distal pancreatic carcinoma is a highly malignant tumor with strong invasiveness, often growing to the edge of the pancreas and penetrating the pancreatic capsule to infiltrate surrounding tissues. In conventional distal pancreatosplenectomy (DPS), tumor cells are prone to spread along the direction of blood and lymphatic reflux due to surgical compression. Additionally, inflammation makes it challenging to achieve R0 resection, leading to a lower patient survival rate. To address these limitations, radical antegrade modular pancreatosplenectomy (RAMPS) was developed, emphasizing deeper excision, including the left anterior renal fascia, the left anterior renal adipose sac, and even the left adrenal gland, to improve the R0 resection rate. With the advancement of minimally invasive surgical techniques, laparoscopic RAMPS (L-RAMPS) is being considered technically safe and feasible in oncology. However, due to technical difficulties and a lack of supporting evidence for clinical application, only a few institutions are currently conducting L-RAMPS. In this context, this article presents detailed techniques for laparoscopic posterior radical antegrade modular pancreatosplenectomy (L-pRAMPS), offering promise for future clinical applications.
Introduction
Conventional distal pancreatosplenectomy (DPS) has traditionally been the standard surgical procedure for distal pancreatic carcinoma1,2. Distal pancreatic carcinoma is a highly invasive tumor that easily infiltrates into retroperitoneal tissues. Simultaneously, pancreatic cancer is often accompanied by chronic pancreatitis, making the boundary between the pancreas and adjacent tissues unclear. Consequently, during conventional DPS, there is a risk of tumor cells spreading along the direction of blood circulation and lymphatic return. In addition to the inherent inflammation associated with pancreatic cancer, achieving R0 resection is challenging, leading to a relatively low postoperative survival rate for patients3.
With the advancement of surgical techniques and a deeper understanding of the pancreatic lymphatic system, the high positive rate of the surgical margin and incomplete lymph node dissection in conventional DPS have garnered more attention. In response, radical antegrade modular pancreatosplenectomy (RAMPS) has emerged to address these challenges3,4. RAMPS involves the complete removal of tissues around the body and tail of the pancreas, including the tumor, to ensure a negative incisional margin and thorough dissection of lymph nodes in N1 nodes, the superior mesenteric artery (SMA), and the anterior and left of the celiac trunk3,4.
In 2003, Strasberg first reported RAMPS3. With the development of minimally invasive surgical techniques, there have been sporadic reports of laparoscopic RAMPS (L-RAMPS) in recent years. L-RAMPS is preliminarily considered technically safe and feasible in oncology5,6,7,8,9. However, due to technical difficulties and a lack of supporting evidence for clinical application, only a few institutions are currently conducting L-RAMPS. In light of this situation, this article presents the techniques of laparoscopic posterior radical antegrade modular pancreatosplenectomy (L-pRAMPS) in detail, holding great promise for future clinical application.
Protocol
The surgical procedure was approved by the institutional review board at The First Affiliated Hospital of Jinan University. The patient, a 58-year-old female, was admitted with a chief complaint of "abdominal pain for 2 months." A computed tomography (CT) scan conducted at an external hospital revealed a space-occupying lesion in the distal pancreas, raising concerns about the possibility of pancreatic carcinoma. Before the surgery, the patient provided written informed consent. The surgical tools and equipment used for this procedure are listed in the Table of Materials.
1. Preoperative preparation
- Prior to the surgery, conduct preoperative blood tests, including a complete blood count, liver and renal function tests, coagulation function test, and serum tumor biomarkers detection10,11.
NOTE: The blood sample was drawn by the nurse and sent to the laboratory, where various blood tests were performed, and the results were reported. Serum tumor biomarkers detection revealed elevated levels of carcinoembryonic antigen (48.34 ng/mL) and carbohydrate antigen 125 (131.1 U/mL)10,11. - Perform preoperative electrocardiogram, abdominal magnetic resonance imaging (MRI), chest CT, and other related examinations10,11.
NOTE: In this study, the patient underwent abdominal MRI in the Medical Imaging Department. The results of abdominal MRI indicated that the size of the pancreatic body and tail mass is approximately 3.5 cm × 3.0 cm × 3.0 cm, suggesting an epithelial-derived malignant tumor (Figure 1).
2. Surgical technique
- After tracheal intubation and general anesthesia (following institutionally approved protocols)12, place the patient in a supine position with their legs apart. Perform routine disinfection with povidone iodine solution.
NOTE: The chief surgeon is located on the right side of the patient, the assistant is on the left side, and the endoscope holder is positioned between the patient's legs. - Make a longitudinal incision of approximately 1 cm through the umbilicus, insert a pneumoperitoneum needle, inject carbon dioxide gas to maintain an abdominal pressure of 12 mmHg, and place a 10 mm trocar and a 30-degree laparoscope.
- Explore the abdominal cavity to determine if there is any tumor metastasis. Under direct vision, place a 5 mm trocar in the left middle abdomen and right upper abdomen, respectively, and a 10 mm trocar in the left upper abdomen and right middle abdomen, respectively (Figure 2).
- Use non-invasive forceps to lift and pull the stomach toward the cephalic side, and cut the gastrocolic and gastrosplenic ligaments at the upper edge of the transverse colon using an ultrasonic-harmonic scalpel (Figure 3).
- Separate the stomach and spleen by ligating and severing the left gastroepiploic vessels, short gastric vessels, posterior gastric vessels, and left subphrenic vessels near the gastric wall. Enter the lesser omental sac and expose the pancreas. Explore the condition of the pancreatic tumor (Figure 2).
- Separate and expose the common hepatic artery, and remove the lymph nodes adjacent to the common hepatic artery. Suspend the common hepatic artery, separate and expose the celiac trunk and the left gastric artery, and dissect the surrounding lymph nodes (Figure 4).
- Incise the retroperitoneum from the pancreatic neck to the pancreatic tail at the lower margin of the pancreas, and remove the surrounding lymph nodes. Continue to incise the anterior layer of the left Gerota fascia at the lower margin of the pancreas to reveal the left renal vein. Peel the anterior layer of Gerota fascia from the surface of the left kidney along the left renal vein (Figure 5).
- Lift the distal pancreas toward the cephalic side to expose the superior mesenteric artery (SMA) and abdominal aorta, and dissect the surrounding lymph nodes at the same time (Figure 5). Separate and expose the superior mesenteric vein (SMV) at the lower margin of the pancreatic neck, and separate the retropancreatic tunnel along the surface of the SMV and portal vein.
- Sever the pancreas at the neck with a linear stapling device (Figure 6 and Figure 7). Dissect and expose the splenic vein, ligate and sever the root of the splenic vein (Figure 7). Dissect and expose the splenic artery, and remove the surrounding lymph nodes. Then, ligate and cut off the root of the splenic artery (Figure 4).
- Lift the pancreas and the anterior layer of the Gerota fascia toward the cephalic side to expose the left adrenal vessel and sever it (Figure 5). Continue to peel off the anterior layer of the Gerota fascia and the left adrenal gland upwards (Figure 5 and Figure 8).
- Above the pancreas, cut off the posterior peritoneum from right to left along the stomach wall. Clear the connective tissues between the abdominal aorta and the left kidney along the left crus of diaphragm. Sever the Gerota fascia tissues at the upper margin of the left adrenal gland (Figure 8).
- Release the spleen by severing the ligaments and adhesions around the spleen, and ultimately remove the distal pancreas, the anterior layer of the Gerota fascia, the left adrenal gland, and the spleen as a whole (Figure 8). Place the excised specimen into a specimen bag.
- Irrigate the abdominal cavity with sterile distilled water. Carefully examine the surgical field for active bleeding, pancreatic leakage, and gastrointestinal collateral injury. Place a drainage tube beside the incisal margin of the pancreas.
- Extend the umbilical incision to about 5 cm in length, cut the abdominal wall layer by layer, and take out the specimen. Send the specimen for intraoperative rapid frozen pathological examination to see whether there is cancer at the incisal margin of the pancreas (Figure 9).
- Check the number of surgical instruments and gauzes, and remove the trocars under direct vision. Finish the operation after suturing the abdominal wall incisions using 5-0 polypropylene suture.
3. Postoperative procedures
- Send the patient back to the ward safely after resuscitation.
- Administer intravenous antibiotics and somatostatin after the surgery.
- Start a fluid diet on the first day after surgery.
- Send the abdominal drainage fluid to the laboratory every day to check the amylase concentration.
- Remove the abdominal drainage tube on the fourth day after surgery.
Representative Results
The surgery proceeded smoothly, and the intraoperative rapid frozen pathological examination indicated the absence of cancer at the incisal margin of the pancreas. Throughout the surgery, the patient's vital signs remained stable, and the anesthesia was effective. The operation duration was 150 min, with an intraoperative blood loss of 80 mL. Anal exhaust occurred 32 h postoperatively. There were no complications such as abdominal hemorrhage, pancreatic leakage, abdominal infection, or incision infection. The postoperative hospital stay was 6 days.
The histopathological analysis of postoperative paraffin sections revealed moderately differentiated pancreatic ductal adenocarcinoma (Figure 10). No tumor tissues were identified at the incisal margin of the pancreas. Fourteen lymph nodes were removed, and one exhibited metastasis. The tumor stage was determined to be pT3N1M0, IIB (Table 1). A postoperative CT scan demonstrated the successful removal of the tumor without significant recurrence or metastasis (Figure 11).
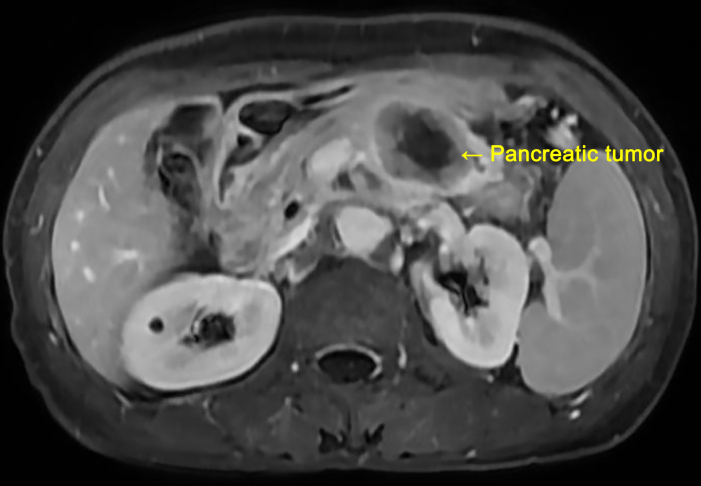
Figure 1: Presurgical abdominal MRI image of the patient. The abdominal MRI indicates that the size of the pancreatic body and tail mass is approximately 3.5 cm × 3.0 cm × 3.0 cm, suggesting an epithelial-derived malignant tumor. Please click here to view a larger version of this figure.
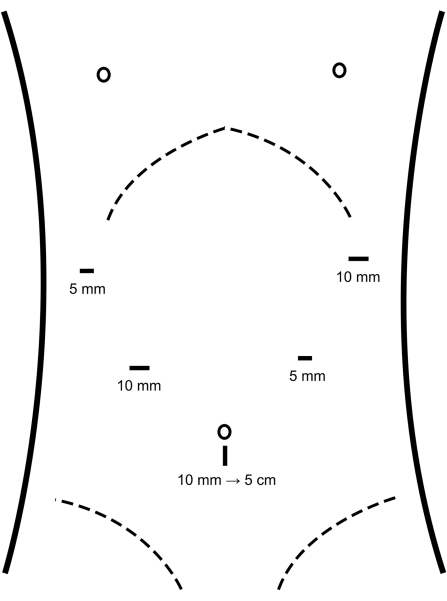
Figure 2: Location of the trocars. Trocars are placed into the abdominal cavity at the locations depicted in the figure. Please click here to view a larger version of this figure.
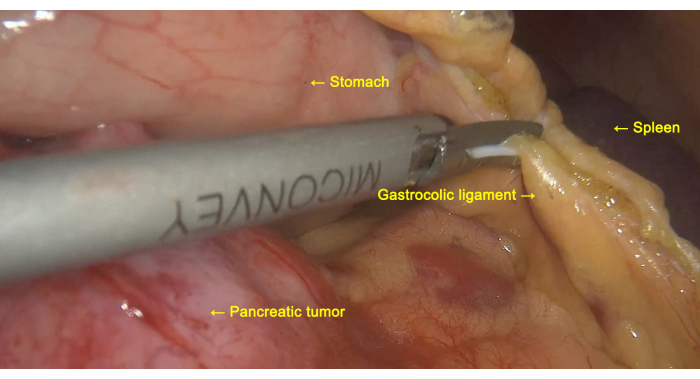
Figure 3: Exploration of the pancreatic tumor. The gastrocolic ligament is cut to facilitate the exploration of the pancreatic tumor. Please click here to view a larger version of this figure.
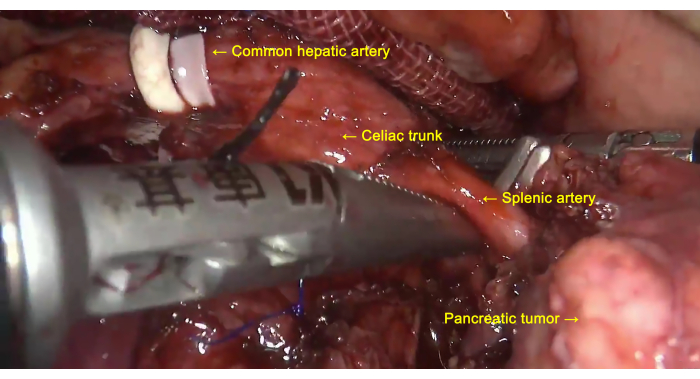
Figure 4: Lymph node dissection. Lymph nodes around the common hepatic artery, celiac trunk, and splenic artery are dissected. Please click here to view a larger version of this figure.
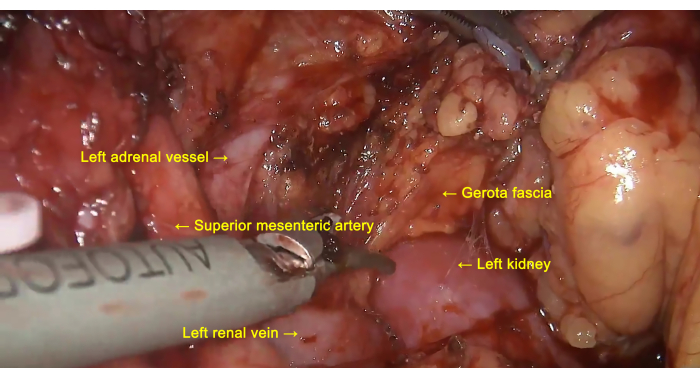
Figure 5: Removal of the anterior layer of Gerota fascia. Lymph nodes around the superior mesenteric artery (SMA) are dissected, and the anterior layer of Gerota fascia is peeled from the surface of the left kidney along the left renal vein. Please click here to view a larger version of this figure.
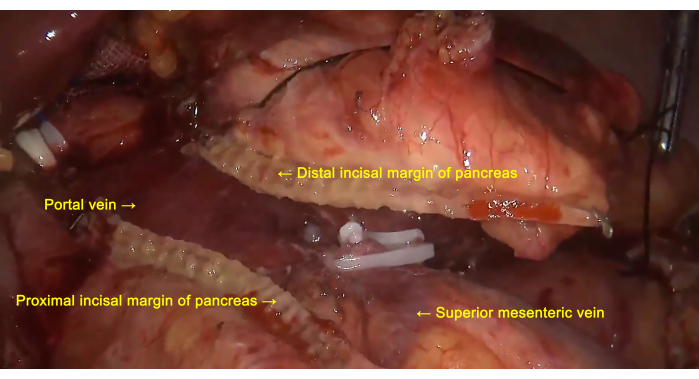
Figure 6: Pancreatic transection. The pancreas is severed at the neck using a linear stapling device. Please click here to view a larger version of this figure.
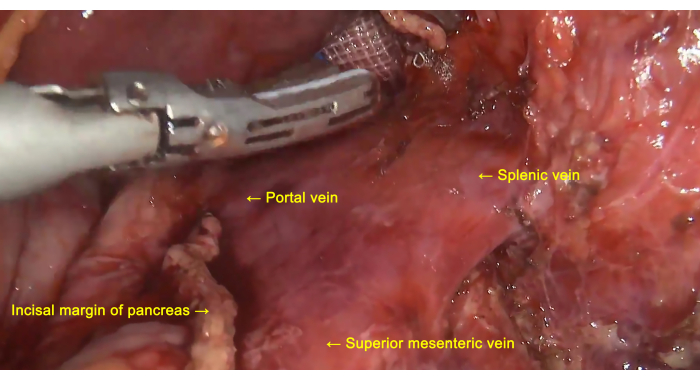
Figure 7: Treatment of the splenic vein. The splenic vein is dissected, exposed, ligated, and severed at the root. Please click here to view a larger version of this figure.
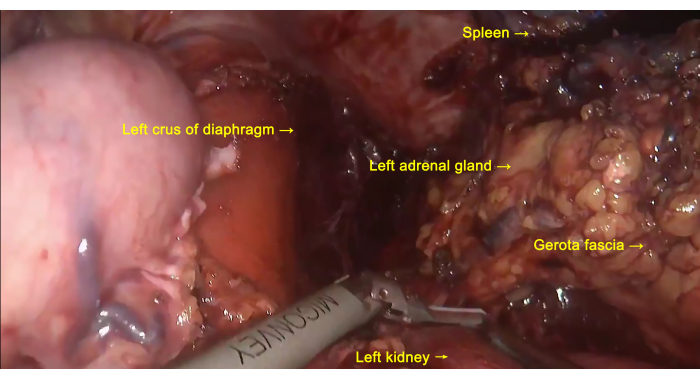
Figure 8: En Bloc resection. The distal pancreas, the anterior layer of Gerota fascia, the left adrenal gland, and the spleen are removed as a single entity. Please click here to view a larger version of this figure.
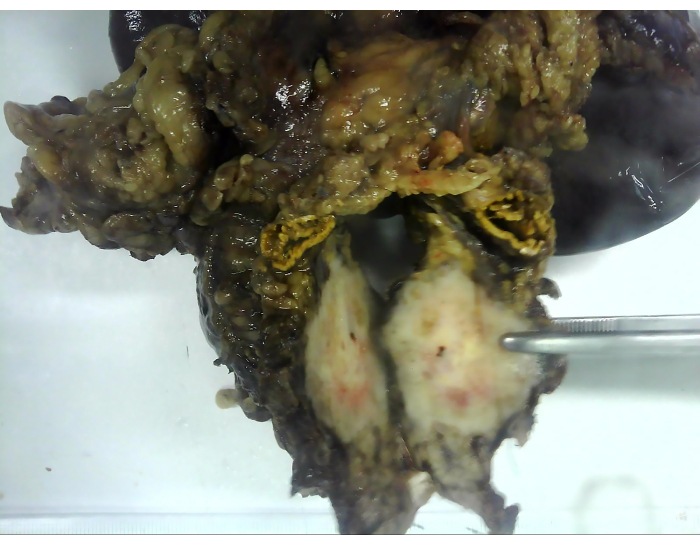
Figure 9: The excised specimen. The excised specimen includes the distal pancreas, the anterior layer of Gerota fascia, the left adrenal gland, and the spleen. Please click here to view a larger version of this figure.
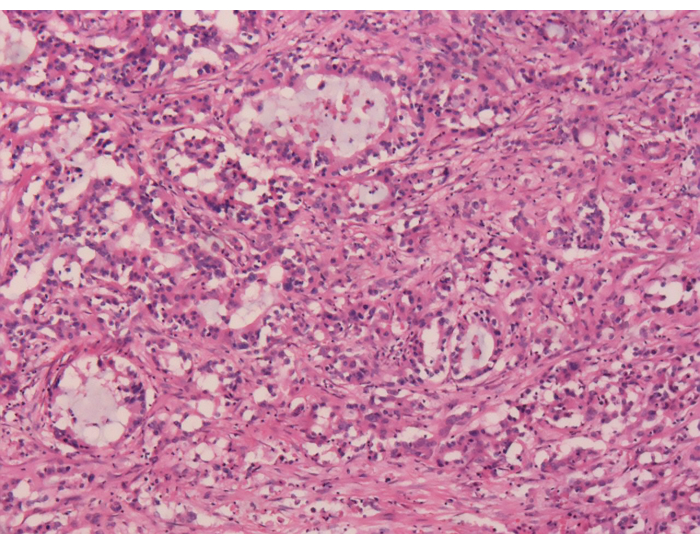
Figure 10: Postoperative histopathological examination. Postoperative paraffin sections reveal moderately differentiated pancreatic ductal adenocarcinoma. Magnification: 40x. Please click here to view a larger version of this figure.
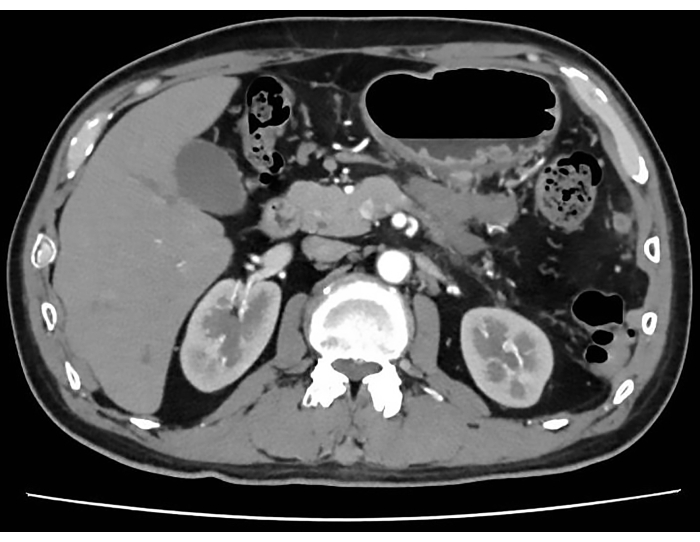
Figure 11: Postoperative CT scan. The postoperative CT scan indicates successful tumor removal without significant recurrence or metastasis. Please click here to view a larger version of this figure.
| Operation time (min) | 150 |
| Intraoperative blood loss (mL) | 80 |
| First flatus (h) | 32 |
| First postoperative liquid diet (days) | 1 |
| Postoperative hospital stay (days) | 6 |
| Postoperative complications (yes/no) | no |
| Bleeding (yes/no) | no |
| Pancreatic leakage (yes/no) | no |
| Abdominal infection (yes/no) | no |
| Incision infection (yes/no) | no |
| Pathological result | Pancreatic ductal adenocarcinoma |
| Differentiation | Moderate |
| TNM stage | pT3N1M0 |
| AJCC stage | IIB |
Table 1: The surgical outcomes of the patient.
Discussion
DPS has been widely used as the standard radical surgery for distal pancreatic carcinoma1,2. However, due to the highly invasive nature of pancreatic cancer, it is easy for the tumor to grow to the edge of the pancreas and even break through the surface of the pancreas. Meanwhile, accompanying chronic pancreatitis makes the boundary between the pancreas and the surrounding tissues unclear. Therefore, it is difficult to achieve R0 resection during conventional surgical procedures3,4. In conventional DPS, due to the incomplete understanding of the pancreatic lymphatic system and limited surgical techniques, a surgical mode has been developed that neither completely removes the lymphatic reflux loop nor cleans up any lymph nodes around the celiac trunk or SMA. Therefore, the survival rate of patients is low3,4.
With the development of surgical techniques and the deepening understanding of the pancreatic lymphatic system, the high positive rate of surgical margin and incomplete lymph node dissection in DPS have received attention. RAMPS is a new technique that differs from DPS, and its concept stems from the more commonly mentioned radical resection of pancreatic head carcinoma. In 2003, Strasberg applied the concept of Whipple surgery to radical resection of pancreatic body and tail carcinoma and proposed RAMPS, which pioneered the whole resection of pancreatic body and tail combined with the spleen from right to left3. RAMPS aims at the complete resection of tissues around pancreatic body and tail, including the tumor, to ensure a negative incisal margin and thorough dissection of lymph nodes. In 2007, Strasberg's team reviewed the clinical data from 23 cases of RAMPS and found that the negative margin rate was as high as 91%, with an average survival time of 21 months and a 5-year overall survival rate of 26%, which was very similar to the results of Whipple surgery4. Reviewing the literature since RAMPS was proposed, it has been found that RAMPS can achieve a significantly improved R0 resection rate, N1 lymph nodes dissection rate, and postoperative survival rate4.
The basic procedure for RAMPS is to first cut off the neck of the pancreas, ligate and detach the splenic artery and vein from the root, remove the lymph nodes surrounding the celiac trunk and the SMA, and then completely remove the distal pancreas, the spleen, and the anterior layer of the Gerota fascia13,14. The Gerota fascia, also known as the renal fascia, is derived from extraperitoneal tissue and has a relatively tough texture15. The Gerota fascia is divided into two layers, including the prerenal fascia and the retrorenal fascia. Two layers of fascia wrap around the kidney and adrenal gland. At the superior and lateral borders, the two layers of fascia fuse with each other and are continuous with the subphrenic fascia and the fascia transversalis, respectively16,17,18. RAMPS accords with the concept of surgical oncology. RAMPS first cuts off the pancreatic neck and disconnects the splenic blood vessels, then follows the lymphatic reflux direction of the distal pancreas and resects it as a whole (en bloc). By using this operation to pre-block the tumor cell proliferation pathway, which is in line with the "no-touch" concept of surgical oncology. Because of the theoretical rationality and perioperative safety, RAMPS helps to improve the R0 resection rate of distal pancreatic carcinoma and has become increasingly widespread in recent years19,20,21.
There are two surgical approaches for RAMPS, including the anterior radical antegrade modular pancreatosplenectomy (aRAMPS) and the pRAMPS. If the preoperative CT scan indicates that the tumor has not penetrated the posterior pancreatic capsule, aRAMPS is feasible, and the resection boundary is located behind the prerenal fascia. On the contrary, the posterior pathway (pRAMPS) can be used to remove the left adrenal gland and prerenal fascia. The aRAMPS enters the lesser omentous sac by cutting off the gastrocolic ligament, dissociates pancreatic tissues adjacent to SMV to establish a tunnel behind the pancreatic neck, dissects relevant lymph nodes, and severs the splenic artery and vein after ligation. The posterior boundary of aRAMPS is the abdominal aorta and the upper margin of the left renal vein. During this procedure, the prerenal fascia is removed, but the left adrenal gland is preserved. The posterior dissection of pRAMPS is deeper and needs to sweep to the diaphragm and retroperitoneal muscles, exposing the left renal vein throughout. During this procedure, the left adrenal gland is excised along with the prerenal fascia22. There is currently no clear definition of the choice between aRAMPS and pRAMPS. In general, pRAMPS may be more appropriate for patients with periadrenal adipose tissue invasion, and the removal of a unilateral adrenal gland will not cause complications such as adrenal insufficiency23. A retrospective study found no statistically significant difference between aRAMPS and pRAMPS in the incidence of pancreatic leakage, mean length of stay, and 90-day mortality24. For this surgical patient, the possibility of periadrenal adipose tissue invasion could not be ruled out through preoperative imaging examination. In order to achieve more thorough lymph node dissection, we performed pRAMPS for the patient. A thorough dissection of the surrounding lymph nodes, complete resection of the distal pancreas, spleen, prerenal fascia, and adrenal gland as a whole (en bloc), are critical steps within the protocol.
With the development and progress of minimally invasive surgical techniques, various surgical fields have seen the application of minimally invasive methods, including pancreatic surgery. In 2007, the first case of L-RAMPS was reported by Fernández-Cruz25. In 2011, Choi reported a case of modified L-aRAMPS26. In 2014, Lee proposed the Yonsei criteria for minimally invasive RAMPS, specifying that the tumor should be confined to the pancreas and located more than 1 to 2 cm from the abdominal axis, and the fascia layer between the distal pancreas and the left adrenal gland and kidney should remain intact27. This study demonstrated the feasibility and safety of minimally invasive RAMPS in both techniques and oncology outcomes for patients selected by Yonsei criteria. In 2014, Han reported a case of robot-assisted aRAMPS that met Yonsei criteria, and the patient survived for 5 years without recurrence28. L-RAMPS is preliminarily considered technically safe and feasible in oncology.
However, due to technical difficulties and a lack of supporting evidence for clinical applications, only a few institutions are currently conducting L-RAMPS. Moreover, in the early stages of developing this technique, it may require a longer operative time and a longer learning curve, which are the limitations of this technique.
According to our experience, L-RAMPS can be preliminarily considered feasible and safe in both techniques and oncology outcomes. However, due to the widespread technical difficulties and the lack of supporting evidence for clinical application in the academic community, further evaluation remains to be validated by high-quality multi-center, large-sample clinical studies. We recommend that laparoscopic or robotic RAMPS should be performed selectively by experienced surgeons. In general, L-pRAMPS is technically challenging to treat distal pancreatic carcinoma that invades the retroperitoneal space, and L-aRAMPS may be a priority for surgeons who are in the early stages of the learning curve.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work was supported by grants from the Basic and Applied Basic Research Project of Guangzhou Basic Research Program (No. 2023A04J1917), the Fundamental Research Funds for the Central Universities (No. 21622312), the Special Foundation for Scientific Research Development of the Affiliated Shunde Hospital of Jinan University (No. 202101004), and Guangdong Basic and Applied Research Foundation (No. 2022A1515012581).
Materials
| 10-mm trocar | Xiamen Surgaid Medical Device Co., LTD | NGCS 100-1-10 | Sterile, ethylene oxide sterilized, disposable |
| 12-mm trocar | Xiamen Surgaid Medical Device Co., LTD | NGCS 100-1-12 | Sterile, ethylene oxide sterilized, disposable |
| 5-mm trocar | Xiamen Surgaid Medical Device Co., LTD | NGCS 100-1-5 | Sterile, ethylene oxide sterilized, disposable |
| Hem-o-lok | America Teleflex Medical Technology Co., LTD | 544240 | Sterile, ethylene oxide sterilized, disposable |
| Linear stapling device | America Ethicon Medical Technology Co., LTD | PSEE60A | Sterile, ethylene oxide sterilized, disposable |
| Pneumoperitoneum needle | Xiamen Surgaid Medical Device Co., LTD | NGCS 100-1 | Sterile, ethylene oxide sterilized, disposable |
| Suction and irrigation tube | Tonglu Hengfeng Medical Device Co., LTD | HF6518.035 | Sterile,dry heat sterilized, reusable |
| Ultrasounic-harmonic scalpel | Chongqing Maikewei Medical Technology Co., LTD | QUHS36S | Sterile, ethylene oxide sterilized, disposable |
Riferimenti
- Mayo, W. J. I. The surgery of the pancreas: I. Injuries to the pancreas in the course of operations on the stomach. II. Injuries to the pancreas in the course of operations on the spleen. III. Resection of half the pancreas for tumor. Ann Surg. 58 (2), 145-150 (1913).
- Andrén-Sandberg, A., Wagner, M., Tihanyi, T., Löfgren, P., Friess, H. Technical aspects of left-sided pancreatic resection for cancer. Dig Surg. 16 (4), 305-312 (1999).
- Strasberg, S. M., Drebin, J. A., Linehan, D. Radical antegrade modular pancreatosplenectomy. Surgery. 133 (5), 521-527 (2003).
- Strasberg, S. M., Linehan, D. C., Hawkins, W. G. Radical antegrade modular pancreatosplenectomy procedure for adenocarcinoma of the body and tail of the pancreas: ability to obtain negative tangential margins. J Am Coll Surg. 204 (2), 244-249 (2007).
- Sato, S., et al. Feasibility of laparoscopic radical antegrade modular pancreatosplenectomy (RAMPS) as a standard treatment for distal resectable pancreatic cancer. Langenbecks Arch Surg. 408 (1), 217 (2023).
- Shen, Z., et al. Laparoscopic radical antegrade modular pancreatosplenectomy via dorsal-caudal artery approach for pancreatic neck-body cancer. J Vis Exp. (189), e63235 (2022).
- Wu, S., Cai, H., Peng, B., Cai, Y. 34;Plane first" approach for laparoscopic radical antegrade modular pancreatosplenectomy. Surg Endosc. 36 (10), 7471-7476 (2022).
- Kato, T., et al. Laparoscopic radical antegrade modular pancreatosplenectomy with anterocranial splenic artery-first approach for left-sided resectable pancreatic cancer (with Videos). Ann Surg Oncol. 29 (6), 3505-3514 (2022).
- Zhang, R. C., et al. Laparoscopic radical antegrade modular pancreatosplenectomy: preliminary experience with 10 cases. BMC Surg. 21 (1), 78 (2021).
- Tempero, M. A., et al. Pancreatic adenocarcinoma, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 19 (4), 439-457 (2021).
- Conroy, T., et al. Pancreatic cancer: ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann Oncol. 34 (11), 987-1002 (2023).
- Disma, N., et al. Airway management in neonates and infants: European Society of Anaesthesiology and Intensive Care and British Journal of Anaesthesia joint guidelines. Eur J Anaesthesiol. 14 (1), 3-23 (2024).
- Watanabe, G., et al. Left kidney mobilization technique during radical antegrade modular pancreatosplenectomy (RAMPS). Langenbecks Arch Surg. 404 (2), 247-252 (2019).
- Rosso, E., et al. Laparoscopic radical antegrade modular pancreatosplenectomy with vascular resection for pancreatic cancer: tips and tricks. J Gastrointest Surg. 24 (12), 2896-2902 (2020).
- Gao, Z., et al. An anatomical, histopathological, and molecular biological function study of the fascias posterior to the interperitoneal colon and its associated mesocolon: their relevance to colonic surgery. J Anat. 223 (2), 123-132 (2013).
- Molmenti, E. P., Balfe, D. M., Kanterman, R. Y., Bennett, H. F. Anatomy of the retroperitoneum: observations of the distribution of pathologic fluid collections. Radiology. 200 (1), 95-103 (1996).
- Marks, S. C., Raptopoulos, V., Kleinman, P., Snyder, M. The anatomical basis for retrorenal extensions of pancreatic effusions: the role of the renal fasciae. Surg Radiol Anat. 8 (2), 89-97 (1986).
- O’Connell, A. M., Duddy, L., Lee, C., Lee, M. J. CT of pelvic extraperitoneal spaces: an anatomical study in cadavers. Clin Radiol. 62 (5), 432-438 (2007).
- Takahashi, A., et al. Radical antegrade modular pancreatosplenectomy enhances local control of the disease in patients with left-sided pancreatic cancer. HPB (Oxford). 25 (1), 37-44 (2023).
- Li, J., et al. Radical antegrade modular pancreatosplenectomy (RAMPS) versus standard retrograde pancreatosplenectomy (SRPS) for resectable body and tail pancreatic adenocarcinoma: protocol of a multicenter, prospective, randomized phase III control trial (CSPAC-3). Trials. 24 (1), 541 (2023).
- Kiritani, S., et al. Radical antegrade modular pancreatosplenectomy for left-sided pancreatic ductal adenocarcinoma may reduce the local recurrence rate. Dig Surg. 39 (4), 191-200 (2022).
- Kuriyama, N., et al. Anterior versus posterior radical antegrade modular pancreatosplenectomy for pancreatic body and tail cancer: an inverse probability of treatment weighting with survival analysis. Surg Today. 53 (8), 917-929 (2023).
- Sato, A., Yazawa, T., Yamamoto, G., Yamamoto, H. Assessment of safety and benefits of posterior radical antegrade modular pancreatosplenectomy in patients without invasion to left adrenal gland. Asian J Surg. 46 (1), 587-589 (2023).
- Kwon, J., et al. Clinical outcome of RAMPS for left-sided pancreatic ductal adenocarcinoma: a comparison of anterior ramps versus posterior ramps for patients without periadrenal infiltration. Biomedicines. 9 (10), 1291 (2021).
- Fernández-Cruz, L., et al. Curative laparoscopic resection for pancreatic neoplasms: a critical analysis from a single institution. J Gastrointest Surg. 11 (12), 1607-1622 (2007).
- Choi, S. H., Kang, C. M., Lee, W. J., Chi, H. S. Multimedia article. Laparoscopic modified anterior RAMPS in well-selected left-sided pancreatic cancer: technical feasibility and interim results. Surg Endosc. 25 (7), 2360-2361 (2011).
- Lee, S. H., et al. Minimally invasive RAMPS in well-selected left-sided pancreatic cancer within Yonsei criteria: long-term (>median 3 years) oncologic outcomes. Surg Endosc. 28 (10), 2848-2855 (2014).
- Han, D. H., Kang, C. M., Lee, W. J., Chi, H. S. A five-year survivor without recurrence following robotic anterior radical antegrade modular pancreatosplenectomy for a well-selected left-sided pancreatic cancer. Yonsei Med J. 55 (1), 276-279 (2014).

