Decellularized Apple-Derived Scaffolds for Bone Tissue Engineering In Vitro and In Vivo
Summary
In this study, we detail methods of decellularization, physical characterization, imaging, and in vivo implantation of plant-based biomaterials, as well as methods for cell seeding and differentiation in the scaffolds. The described methods allow the evaluation of plant-based biomaterials for bone tissue engineering applications.
Abstract
Plant-derived cellulose biomaterials have been employed in various tissue engineering applications. In vivo studies have shown the remarkable biocompatibility of scaffolds made of cellulose derived from natural sources. Additionally, these scaffolds possess structural characteristics that are relevant for multiple tissues, and they promote the invasion and proliferation of mammalian cells. Recent research using decellularized apple hypanthium tissue has demonstrated the similarity of its pore size to that of trabecular bone as well as its ability to effectively support osteogenic differentiation. The present study further examined the potential of apple-derived cellulose scaffolds for bone tissue engineering (BTE) applications and evaluated their in vitro and in vivo mechanical properties. MC3T3-E1 preosteoblasts were seeded in apple-derived cellulose scaffolds that were then assessed for their osteogenic potential and mechanical properties. Alkaline phosphatase and alizarin red S staining confirmed osteogenic differentiation in scaffolds cultured in differentiation medium. Histological examination demonstrated widespread cell invasion and mineralization across the scaffolds. Scanning electron microscopy (SEM) revealed mineral aggregates on the surface of the scaffolds, and energy-dispersive spectroscopy (EDS) confirmed the presence of phosphate and calcium elements. However, despite a significant increase in the Young's modulus following cell differentiation, it remained lower than that of healthy bone tissue. In vivo studies showed cell infiltration and deposition of extracellular matrix within the decellularized apple-derived scaffolds after 8 weeks of implantation in rat calvaria. In addition, the force required to remove the scaffolds from the bone defect was similar to the previously reported fracture load of native calvarial bone. Overall, this study confirms that apple-derived cellulose is a promising candidate for BTE applications. However, the dissimilarity between its mechanical properties and those of healthy bone tissue may restrict its application to low load-bearing scenarios. Additional structural re-engineering and optimization may be necessary to enhance the mechanical properties of apple-derived cellulose scaffolds for load-bearing applications.
Introduction
Large bone defects caused by an injury or disease often require biomaterial grafts for complete regeneration1. Current techniques designed to improve bone tissue regeneration regularly use autologous, allogeneic, xenogeneic, or synthetic grafts2. For autologous bone grafting, considered the "gold standard" grafting practice to repair large bone defects, bone is extracted from the patient. However, this grafting procedure has several drawbacks, including size and shape limitations, tissue availability, and sampling site morbidity3. Moreover, autologous grafting procedures are susceptible to surgical site infections, subsequent fractures, hematoma formation at the sampling or reconstructed site, and post-operative pain4. Bone tissue engineering (BTE) offers a potential alternative to conventional bone grafting methods5. It combines structural biomaterials and cells to build new functional bone tissue. When designing biomaterials for BTE, it is critical to combine a macroporous structure, surface chemistry that promotes cell attachment, and mechanical properties that closely resemble those of native bone6. Past research has indicated that the ideal pore size and elastic modulus for biomaterials utilized in BTE are approximately 100-200 µm7 and 0.1-20 GPa, respectively, depending on the grafting site8. Besides, the porosity and pore interconnectivity of the scaffolds are critical factors affecting cell migration, nutrient diffusion, and angiogenesis8.
BTE has shown promising results with various biomaterials developed and evaluated as alternative options to bone grafts. Some of these biomaterials are osteoinductive materials, hybrid materials, and advanced hydrogels8. Osteoinductive materials stimulate the development of newly formed bone structures. Hybrid materials are composed of synthetic and/or natural polymers8. Advanced hydrogels mimic the extracellular matrix (ECM) and are capable of delivering the necessary bioactive factors to promote bone tissue integration8. Hydroxyapatite is a traditional material and a common choice for BTE due to its composition and biocompatibility9. Bioactive glass is another type of biomaterial for BTE, which has been shown to stimulate specific cell responses to activate genes necessary for osteogenesis10,11. Biodegradable polymers, including poly(glycolic acid) and poly(lactic acid), have also been extensively used in BTE applications12. Finally, natural or naturally-derived polymers like chitosan, chitin, and bacterial cellulose have also demonstrated encouraging outcomes for BTE13. However, while both synthetic and natural polymers show potential for BTE, the development of a functional scaffold with the desired macrostructure typically necessitates extensive protocols.
Conversely, native macroscopic cellulose structures can be readily derived from diverse plants and our research group previously demonstrated the applicability of cellulose-based scaffolds derived from plants to different tissue reconstructions. Indeed, following a simple surfactant treatment, we harnessed the inherent structure of the plant material, highlighting its potential as a versatile biomaterial14. Moreover, these cellulose-based scaffolds can be used for in vitro mammalian cell culture applications14, are biocompatible, and support spontaneous subcutaneous vascularization14,15,16,17. Both our research group and others have demonstrated that these scaffolds can be obtained from specific plants based on the intended application14,15,16,17,18,19,20. For example, the vascular structure observed in plant stems and leaves exhibits a striking similarity to the structure found in animal tissues19. Additionally, cellulose scaffolds derived from plants can be readily shaped and subjected to surface biochemical modifications to achieve the desired characteristics16. In a recent study, we incorporated a salt buffer during the decellularization process, leading to enhanced cell attachment observed both in in vitro and in vivo settings16. In the same study, we demonstrated the applicability of plant-derived cellulose scaffolds in composite biomaterials by casting hydrogels onto the surface of the scaffolds. In recent studies, the functionalization of plant-derived scaffolds has been shown to enhance their effectiveness18. For example, a study conducted by Fontana et al. (2017) revealed that the adhesion of human dermal fibroblasts was supported by RGD-coated decellularized stems, whereas non-coated stems did not exhibit the same capability18. Moreover, the authors also demonstrated that modified simulated body fluid could be utilized to artificially mineralize decellularized plant stems. In more recent studies, we explored the concept of mechanosensitive osteogenesis in plant-derived cellulose scaffolds and assessed their potential for BTE17,20. Furthermore, Lee et al. (2019) utilized plant-derived scaffolds to cultivate bone-like tissues in an in vitro setting21. Through comprehensive evaluations of different plant sources, the authors identified apple-derived scaffolds as the most optimal for the culture and differentiation of human induced pluripotent stem cells (hiPSCs). Furthermore, the authors proposed that the structural and mechanical attributes of the apple-derived scaffolds play a pivotal role in their suitability for the intended purpose. Being the initial plant-derived scaffolds implemented in tissue engineering applications, apple-derived scaffolds have been extensively shown to possess a strikingly similar architecture to that of human bone, notably in terms of their interconnected pores ranging from 100 to 200 µm in diameter14,21.
In the present study, we further investigated the potential of apple-derived cellulose scaffolds for BTE and conducted an analysis of their mechanical properties both in vitro and in vivo. Although there have been studies on the potential of apple-derived scaffolds for BTE17,20,21, their mechanical properties have been underinvestigated. Results showed wildspread invasion and osteogenic differentiation of MC3T3-E1 preosteoblasts seeded in scaffolds that were cultured in differentiation medium for 4 weeks. The Young's modulus of these scaffolds was 192.0 ± 16.6 kPa, which was significantly higher than those of the blank scaffolds (scaffolds without seeded cells) (31.6 ± 4.8 kPa) and the cell-seeded scaffolds cultured in non-differentiation medium (24.1 ± 8.8 kPa). However, it should be noted that the Young's modulus of healthy human bone tissue typically falls within the range of 0.1-2 GPa for trabecular bone and approximately 15-20 GPa for cortical bone8. Nevertheless, following an 8-week implantation in a rodent calvarial defect, the cell-seeded scaffolds appeared to be well integrated into the surrounding bone, as demonstrated by an average peak force of 113.6 N ± 18.2 N in push-out tests, which is similar to the previously reported fracture load of native calvarial bone22. Overall, results obtained from this study show significant promise, particularly for non-load-bearing applications. However, apple-derived cellulose scaffolds do not currently possess the necessary mechanical properties to precisely match the surrounding bone tissue at an implant site. Consequently, further development is required to unlock the full potential of these scaffolds.
Protocol
The experimental protocols were reviewed and approved by the University of Ottawa Animal Care Committee.
1. Scaffold preparation
- Use a mandolin slicer to cut McIntosh apples (Canada Fancy) into 8 mm-thick slices. Cut the hypanthium tissue of the apple slices into 5 mm x 5 mm squares.
- Place the square samples in 0.1% sodium dodecyl sulfate (SDS) for 2 days.
- Wash the decellularized samples with deionized water and incubate them overnight at room temperature (RT) in 100 mM CaCl2 to remove the remaining surfactant.
- Sterilize the samples (i.e., scaffolds) in 70% ethanol for 30 min, wash them with deionized water, and place them in a 24-well culture plate prior to cell seeding.
2. Cell culture and scaffold seeding
- Maintain MC3T3-E1 Subclone 4 cells in 10-cm diameter cell culture-treated dishes in cell culture conditions (37°C in a humidified atmosphere of 95% air and 5% CO2).
- Prepare cell culture medium made of Eagle's minimum essential medium – alpha modification (α-MEM) supplemented with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin.
- Detach the cells from the culture dishes by trypsinization (0.05% trypsin-EDTA) once they reach 80% confluency.
- Centrifuge the cell suspension at c.a. 200 x g for 3 min. Aspirate the supernatant and resuspend the cells in α-MEM at 2.5 x 107 cells per mL.
- Pipette a 40 µL aliquot of the cell suspension on the surface of the scaffolds, and let the cells adhere for 1 h in cell culture conditions. Subsequently, add 2 mL of culture medium to each culture well of the culture plate.
- Replenish the culture medium every 2-3 days for 14 days.
- Prepare a differentiation medium by adding 50 µg/mL ascorbic acid and 4 mM sodium phosphate to the previously described cell culture medium.
- Induce differentiation of MC3T3-E1 cells by incubating the scaffolds in differentiation medium for 4 weeks. Replenish the medium every 3-4 days. In parallel, incubate scaffolds in non-differentiation culture medium (i.e., medium without the supplements to induce differentiation) for the same duration, with the same medium change schedule to serve as a negative control.
3. Pore size measurements using confocal laser scanning microscopy
- Wash the decellularized apple-derived scaffolds with phosphate-buffered-saline (PBS).
- Incubate the scaffolds in 1 mL of 10% (v/v) calcofluor white stain for 25 min in the dark at RT.
- Wash the scaffolds (n = 3) with PBS and image three randomly selected areas per scaffold with a high-speed resonant confocal laser scanning microscope (CLSM) at 10x magnification, using a DAPI channel, as follows:
- Laser-emission filter configuration: 405 nm (laser); 425-475 nm (emission)
- Adjust the laser power and detector manually to ensure optimal image acquisition. Acquire a z-stack of 20 images with a 5-µm step size.
- Using ImageJ software, process and analyze the confocal images as follows:
- Use the Z-Project to Maximum Intensity function to create an image, and apply the Find Edges function to highlight the edge of the pores.
- Trace the pores manually using the Freehand Selection tool.
- Fit each pore as an ellipse, measure the length of the major axis, compile all the measurements (a total of 54 in the present study – 6 in 3 randomly selected areas for each scaffold), and calculate the average length.
4. Cell distribution analysis using confocal laser scanning microscopy
- Wash the cell-seeded scaffolds cultured in the non-differentiation or differentiation medium three times with PBS. Fix the scaffolds with 4% paraformaldehyde for 10 min.
- Thoroughly wash each scaffold with deionized water, permeabilize the cells with a Triton-X 100 solution for 5 min, and wash again with PBS.
- Incubate the scaffolds in 1 mL of 1% periodic acid for 40 min and rinse with deionized water14,16.
- Incubate the scaffolds in 1 mL of a solution containing 100 mM sodium metabisulphite and 0.15 M hydrochloric acid, supplemented with 100 µg/mL propidium iodide. Completely immerse the scaffolds in the solution.
- Wash the scaffolds with PBS and stain the cell nuclei by incubating the scaffolds in a 5 mg/mL DAPI solution for 10 min in the dark. Thoroughly wash again and store the scaffolds in PBS prior to imaging.
- Image three randomly selected surfaces of three different cell-seeded scaffolds with a high-speed resonant CLSM at 10x magnification, using DAPI and TRITC channels, as follows:
- Laser-emission filter configuration:
DAPI: Laser: 405 nm; Emission: 425-475 nm
TRITC: Laser: 561 nm; Emission: 570-620 nm - Adjust the laser power and detector manually to ensure optimal image acquisition. Acquire a z-stack of 20 images with a 5-µm step size.
- Laser-emission filter configuration:
- Use ImageJ software to process the confocal images and create a maximum projection in the z-axis for image analysis using the Z-Project to Maximum Intensity function.
5. Alkaline phosphatase analysis
- Wash the cell-seeded scaffolds cultured in the non-differentiation or differentiation medium three times with PBS. Fix the scaffolds with 10% neutral buffered formalin for 30 min. Fix blank scaffolds (scaffolds without seeded cells) to serve as a negative control.
- Prepare a 5-bromo-4-chloro-3'-indolyphosphate and nitro-blue tetrazolium (BCIP/NBT) staining solution by dissolving one BCIP/NBT tablet in 10 mL of deionized water.
- Wash the fixed scaffolds with a 0.05% Tween solution and stain with the BCIP/NBT solution for 20 min at RT. Wash the stained scaffolds with a 0.05% Tween solution and store them in PBS prior to imaging.
- Image the stained scaffolds with a 12-megapixel digital camera.
6. Calcium deposition analysis
- Wash the cell-seeded scaffolds cultured in the non-differentiation or differentiation medium three times with PBS. Fix the scaffolds with 10% neutral buffered formalin for 30 min. Fix blank scaffolds (scaffolds without seeded cells) to serve as a negative control.
- Prepare a 2% (w/v) alizarin red S (ARS) staining solution.
- Wash the fixed scaffolds with deionized water and stain them with the ARS solution for 1 h at RT. Wash the stained scaffolds with deionized water and store them in PBS prior to imaging.
- Image the stained scaffolds with a 12-megapixel digital camera.
7. Mineralization analysis
- Wash the cell-seeded scaffolds cultured in the non-differentiation or differentiation medium three times with PBS. Fix the scaffolds with 4% paraformaldehyde for 48 h. Fix blank scaffolds (scaffolds without seeded cells) to serve as a negative control.
- Dehydrate the samples in solutions containing concentrations of ethanol increasing from 50% to 100%, as previously described23.
- Perform scanning electron microscopy (SEM) and energy-dispersive spectroscopy (EDS) to analyze mineral aggregates as follows:
- Dry the samples using a critical point dryer, following the manufacturer's protocol24.
- Apply a 5-nm gold coat on the scaffolds using a gold sputter coater, following the manufacturer's protocol25.
- Image the surface of the scaffolds with a scanning electron microscope set at 3 kV, at 85x magnification.
- Perform EDS by setting the scanning electron microscope at 15 kV. On three randomly selected areas per scaffold, acquire EDS spectra for mineral aggregate composition analysis.
8. Young's modulus measurements
- Remove the cell-seeded scaffolds from their respective incubation medium and immediately test the samples.
- Using a custom-built uniaxial compression apparatus equipped with a 1.5 N load cell, compress the scaffolds (n = 3 per condition) at a constant rate of 3 mm·min-1 to a maximum compressive strain of 10% of the scaffold height.
- Determine the Young's modulus from the slope of the linear portion of the stress-strain curves. In the present study, the modulus was determined between 9% and 10% strain.
9. Cell infiltration and mineralization analysis by histology: In vitro scaffolds
- Wash the cell-seeded scaffolds cultured in non-differentiation or differentiation medium three times with PBS.
- Fix the cell-seeded scaffolds with 4% paraformaldehyde for 48 h before resuspension in 70% ethanol for storage.
- Histology:
NOTE: In the present study, the entire histological preparation (embedding, sectioning, and staining) described in the next steps was performed by the Louise Pelletier Histology Core Facility (University of Ottawa).- After dehydration and embedding in paraffin, cut the samples into 5-µm thick serial sections, starting 1 mm inside the scaffolds, and mount the sections on microscope slides.
- Stain the sections with hematoxylin and eosin (H&E) or von Kossa (VK) stains.
- Image the sections with a slide scanner microscope at 40x magnification (n = 1 scaffold in non-differentiation medium and n = 2 scaffolds in differentiation medium in the present study).
- Using ImageJ software, visually evaluate cell infiltration (H&E staining) and mineralization (VK staining).
10. Rat calvarial defect model
- Get experimental protocols reviewed and approved by the local animal care committee.
- Prepare circular (5-mm diameter and 1-mm thickness) decellularized scaffolds following Section 1 described above and using a 5-mm biopsy punch.
- Perform bilateral craniotomy following an established protocol26, as follows:
- Anesthetize male Sprague-Dawley rats with isoflurane, first at 3% until they are unconscious, and then at 2-3% throughout the procedure.
- Expose the periosteum and cranium by cutting the overlying skin using a scalpel blade. Remove the periosteum.
- Create bilateral defects in both parietal bones on each side of the sagittal suture using a dental drill equipped with a 5-mm diameter trephine under constant irrigation of 0.9% NaCl.
- Clean the surrounding bone with 0.9% NaCl to remove any bone fragments.
- Place the circular, decellularized scaffolds in the defects.
- Close the overlying skin with 4-0 sutures.
- Give the rats unlimited access to food and water and monitor them daily.
- After 8 weeks post-implantation, euthanize the rats by CO2 inhalation and thoracic perforation as a secondary euthanasia measure.
- To expose the cranium and retrieve the implants, remove the skin covering the skull using a scalpel blade.
- Cut the skull at the frontal and occipital bones and on the side of both parietal bones using a dental drill to remove the top section of the skull completely.
11. Push-out test
- Connect a uniaxial compression device (with a 445 N load cell) to the USB-data acquisition module.
- Connect the data acquisition module to a computer equipped with a data acquisition software application.
- Immediately after cranial extraction, place each sample (n = 7 implants from 4 animals in the present study) on the sample holder of the uniaxial compression device so that the dorsal side of the bone is facing up.
- Lower the plunger at 0.5 mm/min until slightly touching the extracted implant.
- Initiate the test by lowering the plunger through the implant until complete push-out while applying compression at a constant speed of 0.5 mm/min using the data acquisition software.
- Record the peak force on the force vs. displacement curve using the data acquisition software.
12. Cell infiltration and mineralization analysis by histology: In vivo scaffolds
- Fix the extracted calvaria and implants in 10% neutral buffered formalin for 72 h before resuspension in 70% ethanol for storage.
- Histology:
NOTE: In the present study, all histological preparation (embedding, sectioning, and staining) described in the next steps was performed by Accel Labs (Montreal, QC, Canada).- Cut the samples (embedded in methyl methacrylate) into 6-µm thick sections at three different levels (top, bottom, and towards the center) and mount them on microscope slides.
- Stain the sections with either H&E or Masson-Goldner's trichrome (MGT).
- Image the sections with a slide scanner microscope at 40x magnification (4 explants from 2 animals in the present study).
- Using ImageJ software, visually evaluate cell infiltration (H&E staining) and collagen deposition (MGT staining).
Representative Results
Pore size measurement, cell distribution, and in vitro mineralization (Figure 1 and Figure 2)
Complete removal of native cellular components of the apple tissue scaffolds was achieved after treating the scaffolds with SDS and CaCl2 (Figure 1A). The scaffolds exhibited a highly porous structure, which was confirmed using confocal microscopy. The quantification of the images demonstrated an average pore size of 154 µm ± 40 µm. The pore size distribution ranged between 73 µm and 288 µm. However, the majority of the pores ranged between 100 µm and 200 µm (Figure 1C).
Following a 4-week culture period in differentiation medium, the cell-seeded scaffolds exhibited widespread white mineral deposits (Figure 1A). The scaffolds containing cells displayed an opaque white coloration, suggesting mineralization, which was not observed in the blank scaffolds (scaffolds without seeded cells). Furthermore, analysis using confocal laser scanning microscopy revealed a homogeneous cell distribution within the scaffolds (Figure 1B).
Scaffolds seeded or not with cells were stained with BCIP/NBT and ARS to analyze ALP activity and mineralization, respectively (Figure 1D). The BCIP/NBT staining revealed a substantial increase in ALP activity (depicted by a strong purple color) within the cell-seeded scaffolds cultured in differentiation medium, in contrast to the blank scaffolds or the cell-seeded scaffolds cultured in non-differentiation medium. Likewise, the cell-seeded scaffolds cultured in differentiation medium exhibited a more intense red color upon staining with ARS, indicating greater mineralization compared to the blank scaffolds or the cell-seeded scaffolds cultured in non-differentiation medium. Background staining was observed in the blank scaffolds, potentially due to the presence of CaCl2 in the decellularization protocol.
Staining (H&E and VK) was performed on the scaffolds to analyze cell infiltration and mineralization, and SEM and EDS were used to further evaluate mineralization (Figure 2). H&E staining (Figure 2A) showed good cell infiltration in the cell-seeded scaffolds cultured in non-differentiation or differentiation medium. Multiple nuclei were visible in the periphery and throughout the scaffolds. The presence of collagen was also observed in the scaffolds in pale pink. In addition, VK staining performed on the scaffolds after 4 weeks of culture in differentiation medium revealed that the pore walls were stained whereas calcium deposits were solely detected along the outer edges of the pore walls in the scaffolds cultured in non-differentiation medium and may have resulted from calcium absorption during the decellularization treatment. Localized mineralization on the surface of the cell-seeded scaffolds cultured in differentiation medium for 4 weeks was observed by SEM analysis (Figure 2B). More specifically, mineral deposits resembling spheroid aggregates were observed on the periphery of the pores. In contrast, no mineral aggregates were observed on the blank scaffolds or the cell-seeded scaffolds cultured for 4 weeks in non-differentiation medium. Distinct characteristic peaks corresponding to phosphorous (P) and calcium (Ca) were observed in the EDS spectra of the selected regions of interest, specifically on the mineral deposits observed on the cell-seeded scaffolds cultured for 4 weeks in differentiation medium (Figure 2B).
In vitro biomechanical analysis (Figure 3)
The Young's modulus of the cell-seeded scaffolds was measured after 4 weeks of culture in either non-differentiation or differentiation medium (n = 3 for each experimental condition). It was compared to Young's modulus of the blank scaffolds (scaffolds without seeded cells) (Figure 3). No significant difference was observed in the modulus between the blank scaffolds (31.6 kPa ± 4.8 kPa) and the cell-seeded scaffolds cultured in non-differentiation medium (24.1 kPa ± 8.8 kPa; p = 0.88). In contrast, a significant difference was noted between the modulus of the blank scaffolds (31.6 kPa ± 4.8 kPa) and that of the cell-seeded scaffolds cultured in differentiation medium (192.0 kPa ± 16.6 kPa; p < 0.001). Additionally, a significant difference (p < 0.001) was observed between the Young's moduli of the cell-seeded scaffolds cultured in non-differentiation and differentiation media. Supplementary Figure 1 shows a typical stress-strain curve for the Young's modulus calculation.
In vivo biomechanical performance and bone regeneration (Figure 4 and Figure 5)
Surgical craniotomies were conducted on a total of 6 Sprague-Dawley rats. Bilateral 5-mm diameter defects were created in both parietal bones of the skull using a trephine bur, and apple-derived cellulose scaffolds without seeded cells were implanted in the calvarial defects (Figure 4A). After 8 weeks of implantation, the animals were euthanized, and the upper part of their skulls was collected and processed for either mechanical testing or histological analysis.
Based on visual assessment, the scaffolds seemed well integrated in the skull surrounding tissues. Mechanical push-out tests were performed to quantitatively assess the integration of the scaffolds (n = 7) in the host calvaria. The measurements were conducted using a uniaxial compression device (Figure 4B) immediately after the euthanasia of the animals. Results revealed that the peak force was 113.6 N ± 18.2 N (Table 1).
Histological analysis was conducted to assess cell infiltration and deposition of extracellular matrix within the implanted scaffolds (Figure 5). H&E staining revealed cellular infiltration within the scaffold pores and evidence of vascularization, as shown by the presence of blood vessels within the scaffolds. Additionally, MGT staining demonstrated the presence of collagen within the scaffolds.
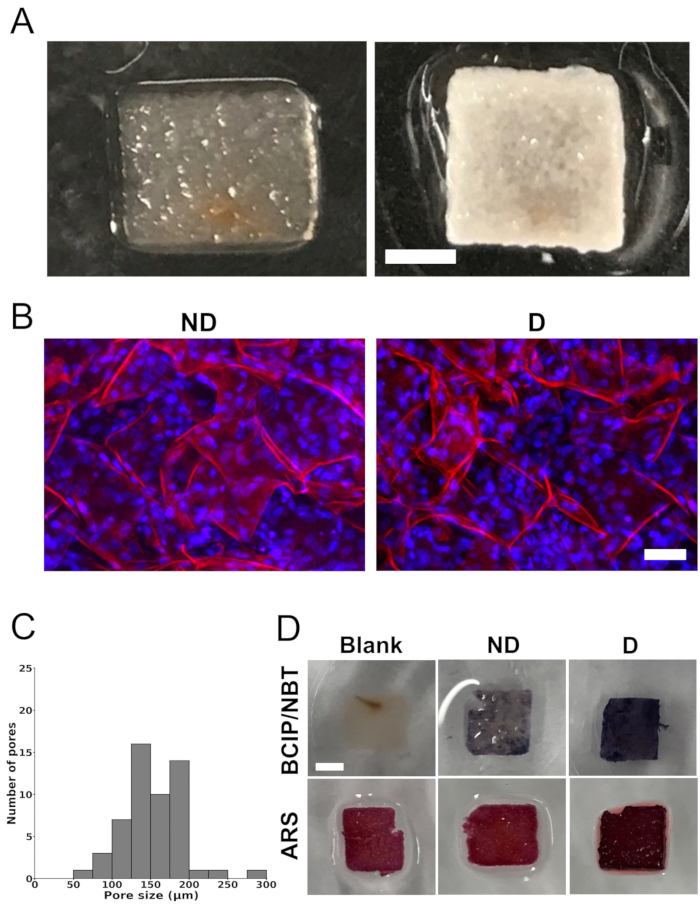
Figure 1: Scaffold images, pore-size distribution, and in vitro mineralization. (A) Representative photographs of an apple-derived cellulose scaffold after removal of the native cells and surfactant (left) and a scaffold seeded with MC3T3-E1 cells after 4 weeks of culture in osteogenic differentiation medium (right). Scale bar represents 2 mm. (B) Representative confocal laser scanning microscope images showing seeded cells in apple-derived cellulose scaffolds after 4 weeks of culture in non-differentiation medium ("ND") or osteogenic differentiation medium ("D"). Scale bar represents 50 µm. Staining was performed on the scaffolds for cellulose (red) using propidium iodide and for cell nuclei (blue) using DAPI. (C) Pore size distribution of decellularized apple-derived cellulose scaffolds, before being seeded with MC3T3-E1 cells, from maximum projections in the z-axis of confocal images. The analysis was performed on a total of 54 pores in 3 different scaffolds (6 pores in 3 randomly selected regions of interest per scaffold). (D) Representative images of scaffolds stained with 5-bromo-4-chloro-3'-indolyphosphate and nitro-blue tetrazolium (BCIP/NBT) to assess alkaline phosphatase (ALP) activity and with alizarin red S (ARS) to visualize calcium deposition, indicating mineralization (scale bar = 2 mm – applies to all). The scaffolds labeled as "blank" (scaffolds without seeded cells) showed no staining with BCIP/NBT, indicating the absence of ALP activity. On the other hand, the cell-seeded scaffolds cultured in differentiation medium ("D") displayed higher ALP activity, indicated by a more intense blue color, compared to the cell-seeded scaffolds cultured in non-differentiation medium ("ND"). For ARS staining, both the blank scaffolds and the scaffolds cultured in non-differentiation medium ("ND") exhibited a lighter shade of red compared to the scaffolds cultured in differentiation medium ("D"). The presence of calcium deposition in the scaffolds cultured in differentiation medium ("D") was illustrated by an intense deep red color. Each analysis was performed on three different scaffolds (n = 3). This figure was adapted with permission from Leblanc Latour (2023)27. Please click here to view a larger version of this figure.
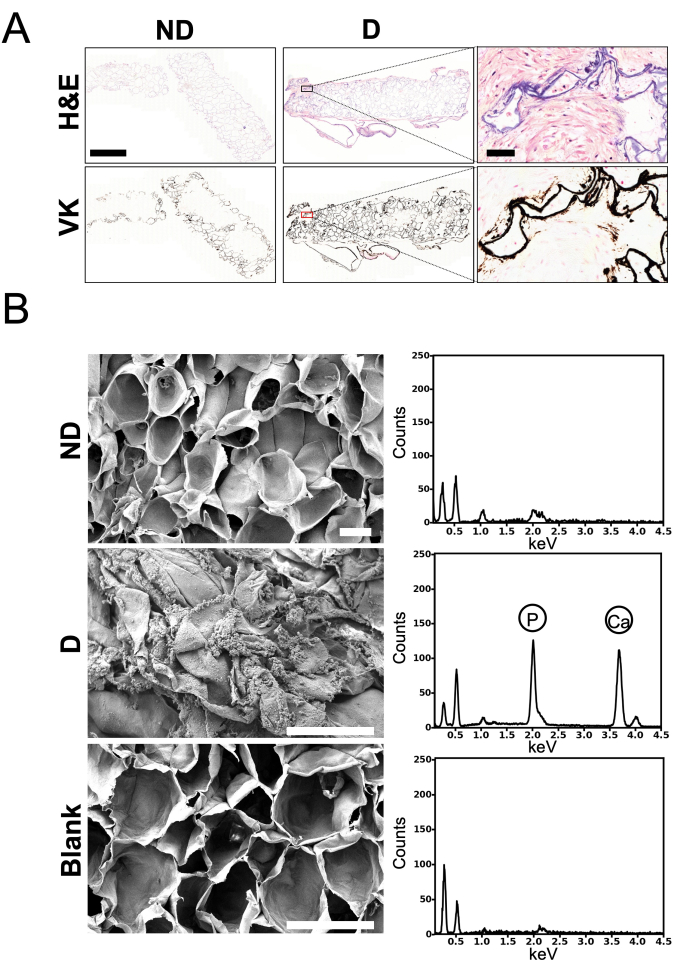
Figure 2: Histology, scanning electron microscopy (SEM), and energy-dispersive spectroscopy (EDS) analysis of in vitro scaffolds. (A) Representative images of top histological cross-sections of the scaffolds. Paraffin-embedded scaffolds were sliced into 5-µm thick sections that were stained with hematoxylin and eosin (H&E) to visualize cell infiltration, or with von Kossa (VK) to visualize mineralization within the scaffolds. Scaffolds were infiltrated with MC3T3-E1 cells, as shown by blue (nuclei) and pink (cytoplasm) staining visible at the periphery and throughout the scaffolds. Collagen (pale pink) was also visible (zoomed-in inset of "H&E – D"). Mineralization was observed only at the periphery of the pore walls in the scaffolds cultured in non-differentiation medium ("ND"). The pore walls in the scaffolds cultured in differentiation medium ("D") were entirely stained in black. The analysis was performed on one scaffold cultured in non-differentiation medium ("ND") and on two scaffolds cultured in differentiation medium ("D") (Scale bar for the lower magnification pictures = 1 mm, scale bar for the higher magnification pictures = 50 µm). (B) Representative micrographs obtained by SEM as well as EDS spectra. The scaffolds underwent sputter coating with gold and were imaged using a field-emission scanning electron microscope at a voltage of 3.0 kV (scale bar = 100 μm – applies to all). EDS spectra were acquired on each scaffold. Phosphorus (2.013 keV) and calcium (3.69 keV) peaks are denoted on each EDS spectrum. Both SEM and EDS were performed on three different scaffolds. Blank: scaffolds without seeded cells. This figure was adapted with permission from Leblanc Latour (2023)27. Please click here to view a larger version of this figure.
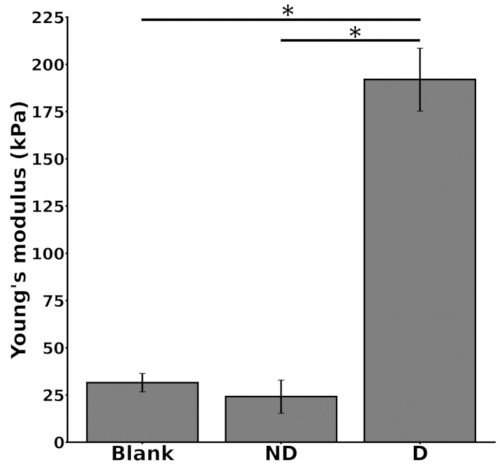
Figure 3: Young's moduli of in vitro scaffolds after 4 weeks of culture in either non-differentiation medium ("ND") or differentiation medium ("D"). Data are presented as mean ± standard error of the mean (SEM) of three replicate samples for each condition. Statistical significance (* indicates p<0.05) was determined using a one-way analysis of variance (ANOVA) and the Tukey post-hoc test. Blank: scaffolds without seeded cells. This figure was adapted with permission from Leblanc Latour (2023)27. Please click here to view a larger version of this figure.

Figure 4: Scaffold photograph prior to implantation and push-out test after 8 weeks of implantation: (A) Representative photograph of a scaffold prior to implantation; (B) Uniaxial compression device used for the push-out tests, with the load cell indicated by an asterisk (*) and the sample indicated by an arrow. This figure was adapted with permission from Leblanc Latour (2023)27. Please click here to view a larger version of this figure.
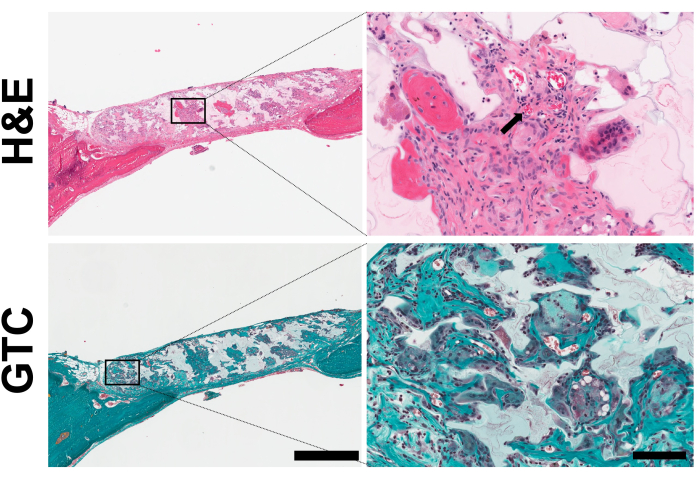
Figure 5: Histology analysis of in vitro scaffolds. Representative images of histological cross-sections from non-seeded scaffolds after 8 weeks of implantation. The sections were stained with either hematoxylin and eosin (H&E) to visualize cells or Masson-Goldner's trichrome (MGT) to visualize collagen. The arrow indicates red blood cells. The presence of collagen is visible (scale bar = 1 mm and 200 μm for the left and right insets, respectively). This figure was adapted with permission from Leblanc Latour (2023)27. Please click here to view a larger version of this figure.
| Sample number | Peak force (N) |
| 1 | 92.8 |
| 2 | 162.7 |
| 3 | 140.3 |
| 4 | 135.7 |
| 5 | 37.7 |
| 6 | 157.8 |
| 7 | 67.9 |
| Mean | 113.6 |
| SEM | 18.2 |
Table 1: Measured peak force from push-out tests.
Supplementary Figure 1: Typical stress-strain curve for Young's modulus calculation. Please click here to download this File.
Discussion
Several in vitro and in vivo studies have demonstrated the biocompatibility of plant-derived cellulose and its potential use in tissue engineering14,15,16,18,19,20, more specifically for hosting osteogenic differentiation20,21. The objectives of the present study were to further investigate the potential of apple-derived cellulose scaffolds for BTE and to assess the mechanical properties of these scaffolds both in vitro and in vivo.
For in vitro studies, preosteoblast cells (MC3T3-E1) were seeded in the scaffolds after eliminating the native cells from the apple tissue. MC3T3-E1 cells are commonly used to investigate the biomineralization of extracellular matrix28,29,30. Scaffolds were then cultured for 4 weeks in osteogenic differentiation or non-differentiation medium. The findings indicated that the cells proliferated and underwent differentiation within the scaffolds, especially when cultured in differentiation medium, thus highlighting the potential of cellulose scaffolds derived from apples to support the development of bone tissue. Cell nuclei were observed in large numbers in the scaffold pores, corroborating observations reported in earlier studies14,15,16,17,20,21. Moreover, similar to our previous findings14 and those of another group21, the average diameter of the scaffold pores was ~154 µm, with most of the pores with diameters between 100 µm and 200 µm (Figure 1C). These dimensions are in agreement with the ideal pore size range (100–200 µm) known to facilitate bone growth7.
The staining analysis showed higher ALP activity following a 4-week culture period in differentiation medium. More calcium deposits were observed on the surface of the cell-seeded scaffolds cultured in differentiation medium compared to both the blank scaffolds and the cell-seeded scaffolds cultured in non-differentiation medium. Similar results have been observed with differentiated hiPSCs in apple-derived scaffolds21. While the presence of ALP was qualitatively assessed in the present study, additional assays could be performed for a quantitative analysis. The histological evaluation of the scaffolds further confirmed that the infiltrated osteoblasts mineralized the scaffolds after differentiation. Notably, the periphery of the constructs cultured in non-differentiation medium was also stained with VK. Residual CaCl2 in the scaffolds may have caused this non-specific staining observed in the periphery post-decellularization. A qualitative analysis of SEM pictures was performed to further assess mineralization. After culture in differentiation medium, cell-seeded scaffolds exhibited signs of ECM mineralization. Mineral aggregates were visible on the scaffold surface, specifically at the edges of the pores. These observations are consistent with previous studies using ECM30 and plant-derived scaffolds21. These aggregates were absent on the surface of the scaffolds without seeded cells. EDS analysis of the aggregates revealed the presence of high levels of P and Ca, thereby suggesting the presence of apatite. Of note is that while the protocol described in the present study allows visualizing cellular infiltration and the presence of mineralization, it does not allow the assessment of their progression over time. Histological analysis at multiple time points would be necessary to fully evaluate this progression. In addition, further analysis would be required to quantify cellular infiltration and mineralization on the surface as well as throughout the whole scaffold. Finally, while the elemental composition of the mineral deposits was determined by EDS, other characterization techniques, such as X-ray diffraction (XRD), may be required to provide information on the crystal structure of the observed deposits.
The Young’s modulus of the scaffolds was significantly higher after culture in differentiation medium (by approximately 8-fold). In contrast, the modulus of the scaffolds cultured in non-differentiation medium resembled that of the blank scaffolds (scaffolds without seeded cells), similar to findings reported in a previous study with apple-derived scaffolds20. Despite the higher Young’s modulus of the scaffolds cultured in differentiation medium, it remained much lower than that of natural bone (0.1 to 2 GPa for trabecular bone and 15 to 20 GPa for cortical bone)8, cancellous allograft (3.78 GPa)31, alloplastic grafts made of poly-ether-ether-ketone (3.84 GPa)31, titanium (50.20 GPa)31, and cobalt-chromium alloy (53.15 GPa)31 implants. Therefore, the scaffolds in their current formulation may not be suitable for load-bearing applications. Of note is that the Young’s modulus provides information on the stiffness of a material. However, the yield strength could also be considered to provide a broader understanding of the scaffold mechanical properties.
Decellularized apple scaffolds were then implanted in 5-mm bilateral critical-sized calvarial defects in rats (n = 6 animals). Push-out tests (on 7 explants), performed to evaluate the integration of the scaffolds within the host calvaria, indicated that the average peak force was 113.6 N ± 18.2 N, which is similar to the fracture load previously reported for calvarial bone (127.1 ± 9.6 N)22, suggesting that the scaffolds integrated well in the surrounding bone tissue. It should, however, be noted that the push-out test only provides information on the interface between the scaffold and the surrounding tissue. Previous studies have combined push-out tests with indentation tests to provide a broader understanding of the mechanical properties of the bone-implant interface and the scaffold itself22. Additionally, bilateral defects are more prone to misalignment with the plunger32. Proper placement of the cranial explant, single defect model, or additional clamping system on the universal testing device could potentially avoid misalignment32.
In summary, this study confirmed that apple-derived cellulose scaffolds could promote preosteoblast adhesion and proliferation. In addition, mineralization occurred within the cell-seeded scaffolds cultured in differentiation medium, leading to an increase in the scaffold Young’s modulus. However, the modulus remained significantly lower than that of natural bone. Interestingly, the force needed to displace the implanted apple-derived cellulose scaffolds was comparable to the fracture load of calvarial bone and other types of scaffolds previously used for BTE22. Finally, similarly to results illustrated in previous reports, cells infiltrated the implanted scaffolds and deposited collagen. Overall, this study demonstrates the potential of cellulose scaffolds derived from plants for BTE applications. These findings are in agreement with a previous study conducted by a different research group, further substantiating the suitability of plant-derived cellulose scaffolds for BTE purposes21. Nevertheless, the disparity in the stiffness of the scaffolds compared to trabecular or cortical bone highlights the importance of developing composite biomaterials that can more closely match the mechanical properties of natural bone. Therefore, despite the increasing interest in utilizing plant-derived scaffolds for BTE purposes, their use may remain limited to non-load-bearing applications. As we previously demonstrated16, re-engineering plant-derived cellulose scaffolds via chemical modification or developing composites with other biological/synthetic polymers may be essential for load-bearing applications.
Disclosures
The authors have nothing to disclose.
Acknowledgements
Funding for this project was provided by the Natural Sciences and Engineering Research Council of Canada (NSERC) (Discovery Grant) and by the Li Ka Shing Foundation. M.L.L. received support from the Ontario Centers of Excellence TalentEdge program, and R.J.H. was supported by an NSERC postgraduate scholarship and an Ontario Graduate Scholarship (OGS).
Materials
| 4′,6-diamidino-2-phenylindole | ThermoFisher | D1306 | DAPI |
| 5-bromo-4-chloro-3'-indolyphosphate and nitro-blue tetrazolium | Sigma-Aldrich | B5655 | BCIP/NBT |
| Alizarin red S | Sigma-Aldrich | A5533 | ARS |
| Ascorbic acid | Sigma-Aldrich | A4403 | Cell Culture |
| Calcium Chloride | ThermoFisher | AA12316 | CaCl2 |
| Calcofluor White | Sigma-Aldrich | 18909 | |
| Dental drill | Surgical tool | ||
| Ethanol | ThermoFisher | 615095000 | |
| Fetal bovine serum | Hyclone Laboratories | SH30396 | FBS |
| Formalin | Sigma-Aldrich | HT501128 | 10% Formalin |
| Goldner's trichrome stain | Sigma-Aldrich | 1.00485 | GTC |
| Hematoxylin and eosin stain | Fisher Scientific | NC1470670 | H&E |
| High-speed resonant confocal laser scanning microscope | Nikon | Nikon Ti-E A1-R | |
| Hydrochloric acid | Sigma-Aldrich | 258148 | |
| ImageJ software | National Institutes of Health | ||
| Irrigation saline | Baxter | JF7123 | 0.9% NaCl |
| MC3T3-E1 Subclone 4 cells | ATCC | CRL-2593 | Pre-osteoblast cells |
| McIntosh apples | Canada Fancy grade | ||
| Methyl methacrylate | Sigma-Aldrich | M55909 | Histological embedding |
| Minimum Essential Medium | ThermoFisher | M0894 | α-MEM |
| Paraformaldehyde | Fisher Scientific | O4042 | 4%; PFA |
| Penicillin/Streptomycin | Hyclone Laboratories | SV30010 | Cell Culture |
| Periodic acid | Sigma-Aldrich | 375810 | |
| Phosphate buffered saline | Hyclone Laboratories | 2810305 | PBS; without Ca2+ and Mg2+ |
| Propidium iodide | Invitrogen | p3566 | |
| Scanning electron microscope | JEOL | JSM-7500F FESEM | SEM and EDS |
| Slide scanner microscope | Zeiss | AXIOVERT 40 CFL | |
| Sodium dodecyl sulfate | Fisher Scientific | BP166 | SDS |
| Sodium metabisulphite | Sigma-Aldrich | 31448 | |
| Sodium phosphate | ThermoFisher | BP329 | |
| Sprague-Dawley rats | Charles-River Laboratories | 400 | Male |
| Sutures | Ethicon | J494G | 4-0 |
| Trephine | ACE Surgical Supply Co | 583-0182 | 5-mm diameter |
| Triton-X 100 | ThermoFisher | 807423 | |
| Trypsin | Hyclone Laboratories | SH30236.02 | Cell Culture |
| Tween | Fisher Scientific | BP337 | |
| Universal compression Device | CellScale | UniVert | |
| Von Kossa stain | Sigma-Aldrich | 1.00362 | Histology |
References
- Schmitz, J. P., Hollinger, J. O. The critical size defect as an experimental model for craniomandibulofacial nonunions. Clinical Orthopaedics and Related Research. 205, 299-308 (1986).
- Yu, X., Tang, X., Gohil, S. V., Laurencin, C. T. Biomaterials for bone regenerative engineering. Advanced Healthcare Materials. 4 (9), 1268-1285 (2015).
- Parikh, S. N. Bone graft substitutes: Past, present, future. Journal of Postgraduate Medicine. 48 (2), 142-148 (2002).
- Silber, J. S., et al. Donor site morbidity after anterior iliac crest bone harvest for single-level anterior cervical discectomy and fusion. Spine (Phila Pa 1976). 28 (2), 134-139 (2003).
- Amini, A. R., Laurencin, C. T., Nukavarapu, S. P. Bone tissue engineering: recent advances and challenges. Critical Reviews in Biomedical Engineering. 40 (5), 363-408 (2012).
- Butler, D. L., Goldstein, S. A., Guilak, F. Functional tissue engineering: the role of biomechanics. Journal of Biomechanical Engineering. 122 (6), 570-575 (2000).
- Karageorgiou, V., Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials. 26 (27), 5474-5491 (2005).
- Bose, S., Roy, M., Bandyopadhyay, A. Recent advances in bone tissue engineering scaffolds. Trends in Biotechnology. 30 (10), 546-554 (2012).
- Yoshikawa, H., Myoui, A. Bone tissue engineering with porous hydroxyapatite ceramics. Journal of Artificial Organs. 8 (3), 131-136 (2005).
- Fu, Q., Saiz, E., Rahaman, M. N., Tomsia, A. P. Bioactive glass scaffolds for bone tissue engineering: state of the art and future perspectives. Materials Science & Engineering. C, Materials for Biological Applications. 31 (7), 1245-1256 (2011).
- Xynos, I. D., Edgar, A. J., Buttery, L. D. K., Hench, L. L., Polak, J. M. Ionic products of bioactive glass dissolution increase proliferation of human osteoblasts and induce insulin-like growth factor II mRNA expression and protein synthesis. Biochemical and Biophysical Research Communications. 276 (2), 461-465 (2000).
- Kroeze, R., Helder, M., Govaert, L., Smit, T. Biodegradable polymers in bone tissue engineering. Materials. 2 (3), 833-856 (2009).
- Venkatesan, J., Vinodhini, P. A., Sudha, P. N. Chitin and chitosan composites for bone tissue regeneration. Advances in Food and Nutrition Research. 73, 59-81 (2014).
- Modulevsky, D. J., Lefebvre, C., Haase, K., Al-Rekabi, Z., Pelling, A. E. Apple derived cellulose scaffolds for 3D mammalian cell culture. PLoS One. 9 (5), e97835 (2014).
- Modulevsky, D. J., Cuerrier, C. M., Pelling, A. E. Biocompatibility of subcutaneously implanted plant-derived cellulose biomaterials. PLoS One. 11 (6), e0157894 (2016).
- Hickey, R. J., Modulevsky, D. J., Cuerrier, C. M., Pelling, A. E. Customizing the shape and microenvironment biochemistry of biocompatible macroscopic plant-derived cellulose scaffolds. ACS Biomaterials Science & Engineering. 4 (11), 3726-3736 (2018).
- Hickey, R. J., Leblanc Latour, M., Harden, J. L., Pelling, A. E. Designer scaffolds for interfacial bioengineering. Advanced Engineering Materials. 25, 2201415 (2023).
- Fontana, G., et al. Biofunctionalized plants as diverse biomaterials for human cell culture. Advanced Healthcare Materials. 6 (8), 1601225 (2017).
- Gershlak, J. R., et al. Crossing kingdoms: Using decellularized plants as perfusable tissue engineering scaffolds. Biomaterials. 125, 13-22 (2017).
- Leblanc Latour, M., Pelling, A. E. Mechanosensitive osteogenesis on native cellulose scaffolds for bone tissue engineering. Journal of Biomechanics. 135, 111030 (2022).
- Lee, J., Jung, H., Park, N., Park, S. H., Ju, J. H. Induced osteogenesis in plants decellularized scaffolds. Scientific Reports. 9 (1), 20194 (2019).
- Zhao, J., et al. Enhanced healing of rat calvarial defects with sulfated chitosan-coated calcium-deficient hydroxyapatite/bone morphogenetic protein 2 scaffolds. Tissue Engineering. Part A. 18 (1-2), 185-197 (2012).
- Murtey, M. D., Ramasamy, P. . Sample Preparations for Scanning Electron Microscopy – Life Sciences. In: Modern Electron Microscopy in Physical and Life Sciences. , 161-186 (2016).
- . . tousimis Critical Point Dryers – Samdri®-PVT-3D. , (2022).
- . . Leica EM ACE200 Vacuum Coater. , (2023).
- Spicer, P. P. Evaluation of bone regeneration using the rat critical size calvarial defect. Nature Protocols. 7 (10), 1918-1929 (2012).
- Leblanc Latour, M. . Cellulose biomaterials for bone tissue engineering [dissertation]. , (2023).
- Kodama, H. -. A., Amagai, Y., Sudo, H., Kasai, S., Yamamoto, S. Establishment of a clonal osteogenic cell line from newborn mouse calvaria. Japanese Journal of Oral Biology. 23 (4), 899-901 (1981).
- Wang, D., et al. Isolation and characterization of MC3T3-E1 preosteoblast subclones with distinct in vitro and in vivo differentiation/mineralization potential. Journal of Bone and Mineral Research. 14 (6), 893-903 (1999).
- Addison, W. N., et al. Extracellular matrix mineralization in murine MC3T3-E1 osteoblast cultures: An ultrastructural, compositional and comparative analysis with mouse bone. Bone. 71, 244-256 (2015).
- Heary, R. F., Parvathreddy, N., Sampath, S., Agarwal, N. Elastic modulus in the selection of interbody implants. Journal of Spine Surgery. 3 (2), 163-167 (2017).
- Lawson, Z. T., et al. Methodology for performing biomechanical push-out tests for evaluating the osseointegration of calvarial defect repair in small animal models. MethodsX. 8, 101541 (2021).

