Microdissection and Immunofluorescence Staining of Myocardial Sleeves in Murine Pulmonary Veins
Summary
This protocol demonstrates microscopy-guided isolation and immunofluorescence staining of murine pulmonary veins. We prepare tissue samples containing the left atrium, pulmonary veins, and the corresponding lungs and stain them for cardiac Troponin T and Connexin 43.
Abstract
Pulmonary veins (PVs) are the major source of ectopic beats in atrial arrhythmias and play a crucial role in the development and progression of atrial fibrillation (AF). PVs contain myocardial sleeves (MS) composed of cardiomyocytes. MS are implicated in the initiation and maintenance of AF, as they preserve similarities to the cardiac working myocardium, including the ability to generate ectopic electrical impulses. Rodents are widely used and may represent excellent animal models to study the pulmonary vein myocardium since cardiomyocytes are widely present all over the vessel wall. However, precise microdissection and preparation of murine PVs is challenging due to the small organ size and intricate anatomy.
We demonstrate a microscopy-guided microdissection protocol for isolating the murine left atrium (LA) together with the PVs. Immunofluorescence staining using cardiac Troponin-T (cTNT) and connexin 43 (Cx43) antibodies is performed to visualize the LA and PVs in full length. Imaging at 10x and 40x magnification provides a comprehensive view of the PV structure as well as detailed insights into the myocardial architecture, particularly highlighting the presence of connexin 43 within the MS.
Introduction
Atrial fibrillation (AF) is the most common sustained arrhythmia1. The prevalence of AF is increasing even further with an expected number of ~17.9 million patients in Europe in 20601. AF is clinically highly important since it is an essential risk factor for the development of myocardial infarction, heart failure, or stroke, resulting in an enormous individual, social, and socioeconomic burden1. Even though AF has been known for decades, the pathophysiology of AF is still not fully understood2.
Already in the late 1990s, studies demonstrated the great impact of pulmonary veins (PVs) in initiating and maintaining AF, as they are the main source of AF-triggering ectopic beats3. It has been demonstrated that PVs structurally differ from other blood vessels. While typical blood vessels contain smooth muscle cells, the tunica media of PVs also contains cardiomyocytes4. In rodents, this cardiac musculature is ubiquitously present throughout the whole PVs, including intra- and extrapulmonary parts, as well as the orifice region5. In humans, PVs also contain cardiomyocytes, which can be observed within extensions of the left atrial (LA) myocardium-so-called myocardial sleeves (MS)6,7.
MS have morphological similarities to the atrial myocardium8. The shape and size of atrial and PV cardiomyocytes do not vary significantly between each other and show comparable electrophysiological properties8. Electrophysiologic recordings within the PV have proven the electrical activity of MS, and angiographic imaging has revealed contractions synchronized with the heartbeat9,10.
Gap junctions are pore-forming protein complexes composed of six connexin subunits, which allow the passage of ions and small molecules11. Gap junctions exist in the cell-to-cell appositions, interconnect neighboring cardiomyocytes, and enable an intercellular electrical coupling between cardiomyocytes12,13. Several connexin isoforms are expressed in the heart with connexin 43 (Cx43) being the most common isoform expressed in all regions of the heart14. Previous studies provide evidence for the expression of Cx43 in cardiomyocytes of the PVs15,16.
It remains challenging to investigate MS within intact PVs due to their delicate structure, especially in small animal models. Here, we demonstrate how to identify and isolate PVs together with LA and lung lobes in mice using microscopy-guided microdissection. Additionally, we demonstrate immunofluorescence (IF) staining of PVs to visualize cardiomyocytes and their interconnections within the PVs.
Protocol
Animal care and all experimental procedures were conducted following the guidelines of the Animal Care and Ethics Committee of the Ludwig-Maximilians-University of Munich, and all the procedures using mice were approved by the Regierung von Oberbayern (ROB 55.2-2532. Vet_02-20-215, ROB 55.2-2532. Vet_02-18-46, ROB 55.2-2532. Vet_02-19-86, ROB 55.2-2532. Vet_02-21-178, ROB 55.2-2532. Vet_02-22-170). C57BL6/N mice were commercially obtained.
1. Preparation
- Prepare a 3% agarose gel by mixing 3 mg of agarose and 100 mL of 1x Tris-buffered saline in a 500 mL Erlenmeyer flask. Heat the solution in a microwave at 600 W for 2-3 min until the gel becomes homogeneous.
- Prepare the dissection dish by gently pouring the gel into a 100 mm diameter Petri dish. Fill the Petri dish half with gel. Clear bubbles from the gel to ensure a homogeneous consistency. Leave the dissection dish to cool down until the gel becomes solid.
- Prepare all the buffers and solutions needed, including fixing solution, permeabilization solution, blocking buffer, and washing buffer according to the recipes provided in Table 1.
2. Organ harvest and tissue preparation
NOTE: An extensive protocol detailing the procedure of mouse anesthesia and harvesting the heart has been previously published17,18. Thus, we present only a brief description of that part. Experiments were performed on 12 to 16 weeks old C57BL6 mice (six male, four female). The male's body weight extended from 26 g to 28 g and the female's body weight from 19 g to 22 g. The following steps were performed without prior systemic heparin injection.
- Anesthetize the mouse with isoflurane (5%, 95% oxygen, Flow: 1 L/min) and ensure sufficient anesthesia depth.
- Transfer the mouse to the operation desk positioning it in a supine position. Maintain the narcosis with isoflurane inhalation (2%, 98% oxygen, Flow: 1 L/min) through an anesthesia mask and fix the mouse with tape at its extremities. Inject fentanyl for analgesia (0.1 mg/25 g body weight i.p.).
- Lift the fur at the xiphoidal process and set a transversal 2 mm cut. Disconnect the fur from the subcutaneous fascia using blunt dissection (backside of scissors) and extend the cut cranially and laterally to expose the thorax.
- Carefully open the abdomen caudal to the costal arch. Lift the xiphoidal process slightly to incise the diaphragm from caudal and make the lungs collapse. Then, cut the diaphragm from left to right without injuring any organs and open the thorax bilaterally to expose the heart.
- Cut the inferior vena cava (IVC) while the heart is still beating to exsanguinate the mouse and proceed to perfuse the heart by penetrating the left ventricle with a 27 G needle and injecting 10 mL of ice-cold 1x phosphate-buffered saline (PBS).
- After clearing the blood from the mouse heart, locate the aortic arch, superior vena cava (SVC), and the trachea. Cut them ~3 mm above the heart base and harvest the heart along with the connected lungs.
- Submerge the harvested organs in a 10 mL conical tube containing 2 mL of sterilized 30% sucrose solution. Allow the samples to dehydrate for 24 h at 4 °C.
3. Microscopy-guided preparation of LA and PVs
- Place the dehydrated heart and lungs into a dissection dish containing 40-50 mL of 1x PBS. Put it under a bright field microscope with 10x magnification and set up the lighting. Place the heart with its dorsal surface on the dissection dish. Identify the transition between the ventricular and atrial wall, and carefully separate the ventricles from the atria using surgical scissors.
NOTE: Cut approximately 1 mm inferior to the atria to keep some ventricular tissue. This will be needed later for pinning the atria to the dissection dish without damaging structures at the heart base. We suggest keeping the ventricles since they can be used as a positive control sample. To embed the ventricular tissue, follow the same procedure outlined for embedding of the PVs, as described in Section 4. - Place the separated atria with the ventricular cutting surface (step 3.1) on the dissection dish so that the heart base is facing up. Orientate the atria so that the lung lobes are posterior, the LA and the left atrial appendage (LAA) are on the right, and the right atrium (RA) and right atrial appendage (RAA) are on the left. Fix the preparation by pinning the remaining left ventricular tissue to the dissection dish. Gently spread out the lung lobes without applying excessive force and pin them to the dissection dish (Figure 1A).
- Get an overview of the anatomic landmarks at the heart base.
- Find the aorta with the aortic arch in the center of the heart base.
- Detect the pulmonary trunk (PT) directly to the right of the aorta and follow its course towards the posterior to distinguish the pulmonary arteries (PAs). Verify their location by following their course up to the left lobe and right superior/middle lobes to find the corresponding lung hilum (LH).
- Determine the position of the trachea and/or the left and right main bronchi (L Br, R Br), which are located posterior to the aorta, and verify their location by following their course towards the LH (Figure 1A).
- Find the SVC to the left of the aorta and verify by following its course into the RA next to the RAA.
- Remove connective and fat tissue around the heart base while preserving all the identified landmarks mentioned above. Additionally, remove the aortic arch and trachea to have a free view of the heart base (Figure 1B).
- Gently separate the PAs from the upper surface of the atrium by blunt dissection using fine forceps. Afterward, cut them off at LH and flip them to the side to expose the left atrial free wall (LAFW) and the PVs in their full dimension. Cut off the main bronchi from the LH and remove the bronchial tissue (Figure 1C).
NOTE: The LH serves both as the common entry point for PAs and airways into the lungs, as well as the exit point for the PVs. It is important to perform every procedure at the LH carefully to avoid any damage to the PVs. - To separate the LA/RA and left and right ventricle (LV, RV), perform a cut starting from the anterior part of the remaining RV upwards through the tricuspid valve (TV) till the anterior side of the SVC to open the RA from the frontal side. Cut the posterior RA free wall (RAFW) next to the interatrial septum to separate the RA and LA. Cut the posterior right ventricular wall next to the interventricular septum to completely sever the LV and RV.
NOTE: An extensive protocol describing the procedure of the preparation and isolation of the right atrium in detail has been published previously19. The separation of the RA is useful to remove unwanted tissue from the LA-PV tissue complex and to collect tissue for additional experiments. - Reduce the size of the lung lobes by cutting off some of the basal lung tissue so that approximately 3-4 mm of lung tissue is left over.
NOTE: Preserve the apices of the lung lobes as they serve as floats during the subsequent embedding steps and allow embedding of the PVs horizontally in the O.C.T compound.
4. Tissue embedding
- Transfer the preparation, including LA and PVs, into a cryomold, ensuring that the heart base and lung apex are facing upwards. Arrange them in a physiological configuration-ensure that the PVs are not twisted or flipped.
- Fill the cryomold with O.C.T compound and gently compress the PVs with fine forceps to remove any remaining air. Maintain their physiological configuration unchanged throughout the process.
- Put the cryomold with the embedded tissue on dry ice to freeze the O.C.T. compound. Store the samples at -80 °C for future use.
NOTE: It is crucial to keep the PVs in a horizontal position to ensure obtaining sections that contain PVs in full length for subsequent procedures.
5. Cutting and collection of frozen tissue sections
NOTE: To cut the tissue blocks, the machine settings were adjusted to a specimen temperature of -18 °C, and blade temperature of -25 °C.
- Install a tissue block on the specimen holder by applying a layer of O.C.T. compound between the tissue block and the specimen holder. Freeze them for 3-4 min.
- Block the specimen head and load the specimen holder with the tissue block on the specimen head by closing the specimen chuck release lever. Finely adjust the position of the tissue block using the fine adjustment knobs.
- Remove the cryomold from the specimen. Unblock the specimen head and the electrical brake of the cryostat.
- Set the cryostat to trim mode with a trim size between 30 µm and 50 µm. Trim the tissue specimen until the PVs become visible.
- Switch to the fine section-cutting mode and set the thickness to 10 µm. Start collecting sections, including PVs and connected lung and atrial tissue. Collect sections using an anti-roll plate or by fixing the free end of each section to the blade holder with a fine cold brush and spread them out.
- Position a slide (kept at room temperature) adjacent to the section. Carefully release the section from the brush, allowing the section to naturally adhere to the slide, as the frozen section and O.C.T. compound melt upon touching the warmer slide surface.
NOTE: Maintain a stable and gentle condition during the sectioning process to prevent any ruptures or folds in the sections. Replace the blade with a new blade if necessary. - Collect up to three sections on each slide. Label the slides and store them at -20 °C.
NOTE: We recommend collecting at least five sections (equivalent to two slides) for each PV.
6. Immunofluorescence staining of cryosections from the PVs
- Arrange the frozen slides on a staining system and allow the sections to thaw for approximately 20 min at room temperature.
NOTE: Fill the staining system with water to keep the samples humidified. Drying of the tissue can damage antigens, causing non-specific antibody binding and overstaining artifacts during the staining procedures. - After 20 min of thawing, cover the sections with a few drops of fixing solution (4% paraformaldehyde [PFA], Table 1) to fix the tissue on the slides for 10 min at room temperature.
- After fixation, transfer the slides to the vertical slide rack and immerse them in a container filled with 1x PBS. Place the container on a shaker at low speed and wash the slides for 5 min. Repeat this wash cycle two more times, using fresh 1x PBS each time.
- After washing, circle every individual section on each slide with a xylol-containing liquid blocker pen. Rearrange the slides on the staining system and apply 1 or 2 drops of permeabilization solution (0.1% Triton X-100, Table 1) onto each sample with a Pasteur pipette. Allow the sections to incubate at room temperature for 10 min to permeabilize the cell and cell organelle membranes.
- After permeabilization, remove the 0.1% Triton X-100 solution by washing the slides for 3 x 5 min in 1x PBS, following the same procedure as described in Step 6.3.
- After washing, rearrange the slides on the staining system and apply 1 or 2 drops of the Blocking Buffer (Table 1) to each section, ensuring they are fully covered with the blocking buffer. Allow the slides to incubate for 1 h at room temperature.
- Prepare 1 mL of the primary antibody mix by pipetting 5 µL of a mouse anti-cTNT antibody (diluted 1:200) and 1 µL of a rabbit anti-Cx43 antibody (diluted 1:1,000) into a 1.5 mL microcentrifuge tube filled with 994 µL of Blocking Buffer. Pipette the mixture up and down to ensure thorough mixing.
NOTE: Adjust the amount of antibody mixture applied according to the size of the section. Ensure that the sections are fully covered by the antibody mixture. The concentration, incubation time, and optimal incubation temperature of antibodies can vary depending on factors such as antibody type, antibody clones, and tissue characteristics. We recommend testing and optimizing the staining conditions individually for each antibody. - After the blocking step (step 6.6), remove the remaining blocking buffer and directly apply 2 or 3 drops of the primary antibody mix onto each section. Allow the slides to incubate overnight at 4 °C.
- After the primary antibody incubation, wash the slides for 3 x 5 min with Washing Buffer under gentle shaking (Table 1).
- Prepare 1 mL of the secondary antibody mix by pipetting 1 µL of an AF488-conjugated goat antimouse secondary antibody (diluted 1:1,000) and 1 µL of an AF568-conjugated goat antirabbit secondary antibody (diluted 1:1,000) into a 1.5 mL microcentrifuge tube filled with 998 µL of Washing Buffer. Pipette the mixture up and down to mix well.
- After the washing step (step 6.9), rearrange the slides in the staining system and apply 2 or 3 drops of the secondary antibody mixture onto each section with a pipette. Allow the antibodies to incubate for 45 min at room temperature while protecting the samples from light to prevent fluorophore quenching. After the secondary antibody incubation, wash the slides for 3 x 5 min with Washing Buffer under gentle shaking (Table 1).
- Rearrange the slides on the staining system. Counterstain the sections with Hoechst-33342 (in a dilution of 1:1,000 in 1x PBS) by applying 2 to 3 drops of the Hoechst-33342 solution onto each section. Allow the slides to incubate for 10 min at room temperature.
- After the incubation, wash the slides 3 x 5 min with Washing Buffer under gentle shaking (Table 1). Mount the slides by applying 1 or 2 drops of fluorescent mounting medium onto each slide. Cover the slides with coverslips.
NOTE: Make sure the coverslip is flat when placing it. If any air bubbles are present, gently apply pressure on the side of the bubble to remove them. - Store the mounted slides in a slide saver at 4 °C.
NOTE: It is recommended to image the slides as early as possible. This protocol is not exclusively valid for the mentioned antibodies. Target antigens and antibodies can be replaced or modified according to the individual experiment requirements or alternative antigen detection strategies.
7. Imaging of the immunofluorescence staining slices
- Set up the microscope.
- Start the microscope with the laser light source, the connected computer, and the accompanying software following the manufacturer's manual.
- Enter the Configuration menu. Click to select the suitable camera type for fluorescence imaging (e.g., DFC365FX monochrome camera).
- Enter the Acquire menu and create three channels.
- Select channel FCr1 for the detection of Hoechst-33342, label it, and select the DAP filter cube in the selection section. Set the exposure time to 200 ms, the electronic gain to 2, and the light intensity to 17%.
- Select channel FCr2 for the detection of cTNT (stained with AF488-conjugated antibody), label it, and select the L5 filter cube in the selection section. Set the exposure time to 200-300 ms, the electronic gain to 1.8, and the light intensity to 100%.
- Select channel FCr3 for the detection of Cx43 (stained with AF568-conjugated antibody), label it, and select the TXR filter cube in the selection section. Set the exposure time to 150 ms, the electronic gain to 2, and the light intensity to 100%.
NOTE: The spectral characteristics of the filter cubes are listed in Table 2.
- Load the slide on the microscope stage.
- Perform an overview imaging at 10x magnification:
- Select the 10x objective and open the laser shutter by clicking Live. With the FCr1 channel selected, use the navigator function to navigate through the slide until a signal is detected. Roughly focus the sample until the cell nuclei are distinguishable.
- Initiate the spiral scanning mode to capture a complete preview of the sample. Deactivate live by clicking the button again to protect the specimen. Identify the LA, PVs, and lung tissue.
- Define the area of interest for the subsequent tile scan; select multiple focus points within the area of interest. Click Live in the FCr1 channel and adjust the focus for each focus point. Save the corresponding Z-positions, which represent the exact focus position for each focus point. Start the tile scan at 10x magnification to capture a complete overview image of the sample.
- After the scan is complete, open the tile scan and merge the individual images. To enhance the brightness and contrast of single channels, select them in the object bar and adjust their signal range with the sliders as desired.
- Obtain zoomed-in images at 40x magnification:
- Select the 40x objective. Adjust the exposure time and gain settings for each channel as follows: FCr1: DAP filter cube: exposure time: 50 ms, gain: 1.5; FCr2: L5 filter cube: exposure time: 80 ms, gain: 1.8; FCr3: TXR filter cube: exposure time: 20 ms, gain: 2.
- Use the overview image of step 7.3 to locate the PV orifice (PVO), as well as extrapulmonary and intrapulmonary PV (PVex, PVin) regions. Define this area to be scanned.
- Open the submenu Image and click on z to activate Z-Stack.
- Select the FCr1 channel and open the laser shutter to obtain a live preview.
- Go to the Z-Stack submenu and determine the Z-Stack starting and end points by rotating the focus fine adjustment knob clockwise and counterclockwise until the overall signal begins to lose focus.
- After setting up the focus range, select the System Optimized Z-Stack mode, and activate the calculate extended depth of field images (EDF) option. Depending on how flat the tissue is, expect approximately 20 layers with a step interval of approximately 0.5 µm.
- Begin the tile scan. After scanning, open the tile scan and merge only the EDF image. To enhance the brightness and contrast of single channels, select them in the object bar and adjust their signal range with the sliders as desired (see step 7.3.4).
- After generating the images, save the project and export both, the merged version and the raw channels of each image as a TIFF file.
NOTE: Saving the files as ImageJ-readable TIFF allows ImageJ to import the original tile size in µm instead of pixels. Always close the laser shutter whenever a laser signal is not needed to prevent photobleaching of the secondary antibodies.
8. Image editing with ImageJ
- Load the corresponding raw channel files in ImageJ. Click on Image | Color | Merge and choose the desired color for each channel.
- Create a scale bar by selecting Analyze | Tools | Scale Bar and paste it in the bottom right corner. Proceed with further processing and analysis according to the image requirements. Save the merged images when finished.
Representative Results
We performed the microdissection, staining, and imaging of the PVs in 10 12-16-week-old mice. Following the protocol, we successfully microdissected PVs together with the LA in all experimental mice and obtained sections with a comprehensive view of the PVs in eight mice. Overview images were taken at 10x magnification to identify the PV orifice (PVO) region at the LA-PV junction, the extrapulmonary PVs (PVex) (PVs in between the lung hilum and the LA-PV junction), and the intrapulmonary PVs (PVin) (PVs surrounded by lung tissue) (Figure 2). Zoomed-in images of the mentioned regions were obtained at a 40x magnification objective (Figure 3).
We found specific cTNT signals with a typical muscular striation in the PVO, the PVex, and the PVin (Figure 3). Cx43 was found in all three PV regions and was mostly projected between neighboring cardiomyocytes (Figure 3 and Figure 4A). Cx43-related signals were observed at both the polar side (Figure 3, yellow arrows) and the lateral side of the cardiomyocytes in the MS (Figure 3, red arrows).
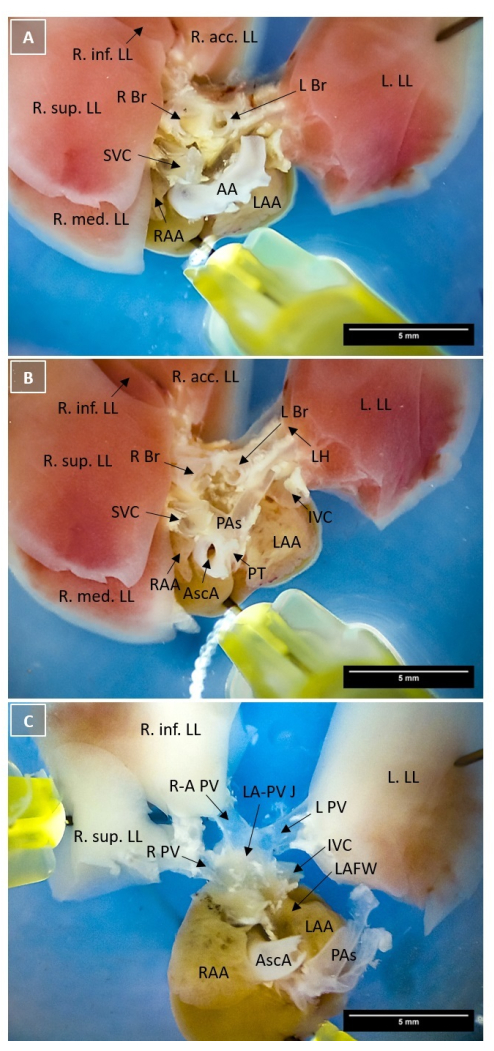
Figure 1: Identification of murine pulmonary veins under the microscope. Brightfield microscopy (10x magnification). The heart is pinned to the dissection dish with four pins at the atrium, the left lung lobe, the right middle lung lobe, and the right inferior lung lobe. (A) Overview of the heart base after separating the ventricular tissue from the atrium with the aortic arch in the middle. The atrium is located at the bottom; the lung lobes are spread out in the upper half of the picture. PVs are concealed by the AA, the main bronchi, and the PAs. (B) Top view image of the heart base after removing trachea, parts of the main bronchi, the aortic arch, as well as connective and fat tissue. The PAs in the middle of the image show their full course from the PT to the LH and cover the PVs. (C) Top view image of the PVs after cutting the PAs from the lung lobes and removing the extrapulmonary bronchial tissue. Scale bars = 5 mm. Abbreviations: AA = aortic arch; AscA = ascending aorta; IVC = inferior vena cava; L Br = left main bronchus; L PV = left pulmonary vein; LA-PV J = left atrium-pulmonary vein junction; LAA = left atrial appendage; LAFW = left atrial free wall; LH = lung hilum; L. LL = left lung lobe; PAs = pulmonary arteries; PT = pulmonary trunk; R Br = right main bronchus; R PV = right pulmonary vein; R-A PV = right ascending pulmonary vein; RAA = right atrial appendage; R. acc. LL = right accessory lung lobe; R. inf. LL = right inferior lung lobe; R. mid. LL = right middle lung lobe; R. sup. LL = right superior lung lobe; SVC = superior vena cava. Please click here to view a larger version of this figure.
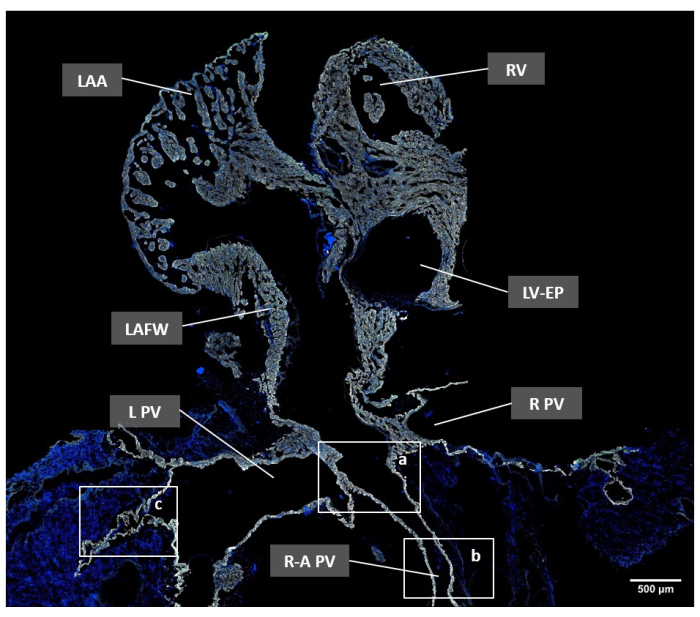
Figure 2: Overview of the adult mouse pulmonary veins and the left atrium in a transversal section image by immunofluorescence microscopy. Cell nuclei were stained with Hoechst-33342 (blue); cardiomyocytes were labeled with anti-cTNT antibody (white); and gap junctions were detected using anti-Cx43 antibody (green). The white rectangles highlight the following regions: a, PV orifice (PVO); b, extrapulmonary PV (PVex); and c, intrapulmonary PV (PVin). Scale bar = 500 µm. Abbreviations: L PV = left pulmonary vein; LAA = left atrial appendage; LAFW = left atrial free wall; LV-EP = left ventricle – ejection path; R PV = right pulmonary vein; R-A PV = right ascending pulmonary vein; RV = Right ventricle. Please click here to view a larger version of this figure.
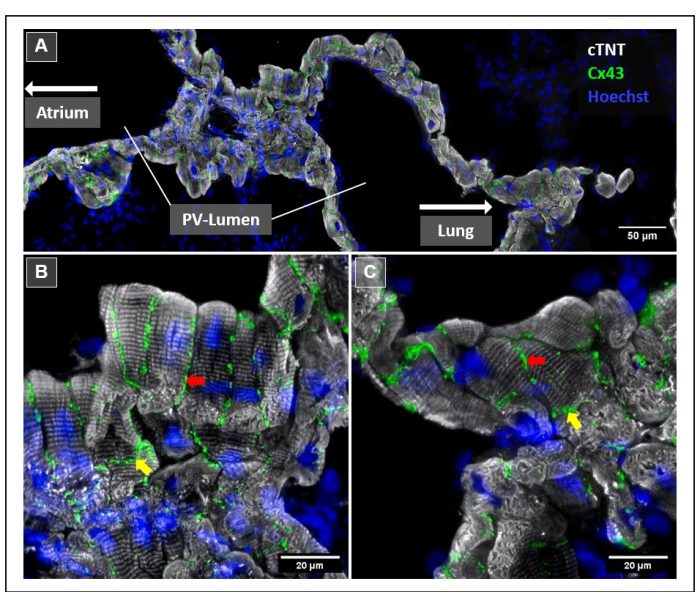
Figure 3: Zoomed-in view of the right PV myocardium using immunofluorescence microscopy. Cell nuclei were stained with Hoechst-33342 (blue); cardiomyocytes were labeled with anti-cTNT antibody (white); and Cx43+ gap junctions were detected by anti-Cx43 antibody (green). (A) EDF tile scans showing a longitudinal section through the right extrapulmonary PV. The LA and PV orifice are not shown in the image and would be located to the left, while intrapulmonary PV and corresponding lung parenchyma (also not shown) would be located to the right. (B,C) EDF single tile scans of the right extrapulmonary PV. Arrows highlight Cx43+ gap junctions at the cellular borders of cardiomyocytes. The yellow arrows indicate gap junctions at the polar sides of cardiomyocytes, colocalized with intercalated discs, while the red arrows indicate gap junctions at the lateral side of cardiomyocytes. Scale bars = 50 µm (A), 20 µm (B,C). Abbreviations: PV = pulmonary vein; EDF = extended depth of field; LA = left atrium. Please click here to view a larger version of this figure.
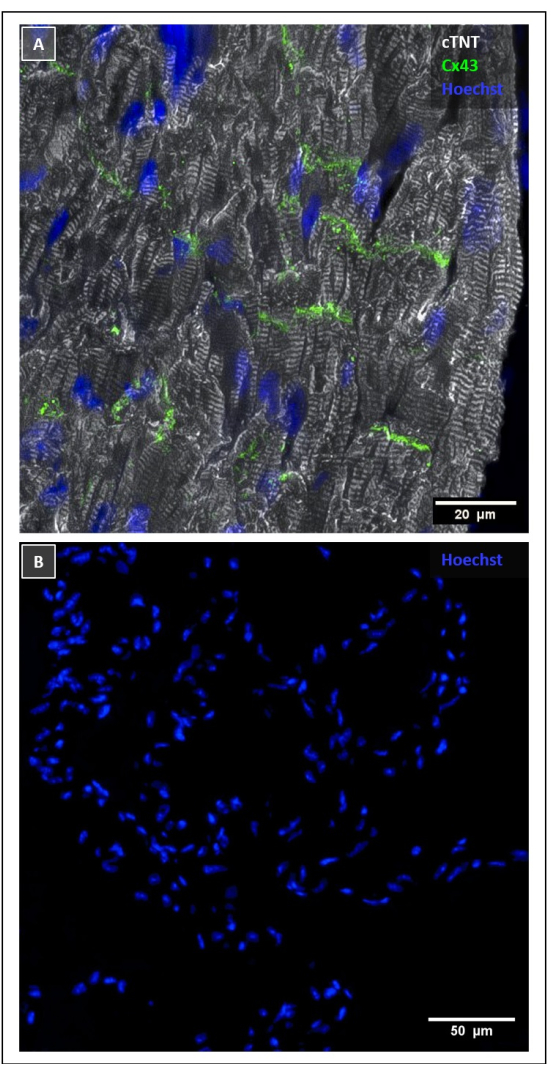
Figure 4: Immunofluorescence staining of control tissue. (A) Positive control staining of the murine LV. Cell nuclei were stained with Hoechst 33342 (blue); cardiomyocytes were labeled with anti-cTNT antibody (white); and Cx43+ gap junctions were detected with anti-Cx43 antibody (green). (B) Negative control staining of the right extrapulmonary PV by using secondary antibodies only. Cell nuclei are stained with Hoechst 33342 (blue). Scale bars = 20 µm (A), 50 µm (B). Abbreviations: LV = left ventricle; PV = pulmonary vein. Please click here to view a larger version of this figure.
| Filter-Cube | Excitation Filter | Emission Filter |
| DAP | 350 / 50 | 460 / 50 |
| L5 | 480 / 40 | 527 / 30 |
| TXR | 560 / 40 | 630 / 75 |
Table 1: Excitation and emission filter characteristics of the Leica DM6 B filter cubes.
| Compound | Final concentration | g or mL / 100 mL required |
| Fixing solution | ||
| Paraformaldehyde (PFA) 16% | 4% | 25 mL |
| Phosphate Buffered Saline (PBS) 1x concentrated | 75 mL | |
| Permeabilization Solution | ||
| Triton X-100 | 0.1% | 0.1 mL |
| Phosphate Buffered Saline (PBS) 1x concentrated | 99.9 mL | |
| Blocking Buffer | ||
| Normal goes serum (NGS) | 10% | 10 mL |
| Bovine Serum Albumin (BSA) | 0.5% | 0.5 mg |
| Tween 20 | 0.1% | 0.1 mL |
| Phosphate Buffered Saline (PBS) 1x concentrated | 89.9 mL | |
| Washing Buffer | ||
| Bovine Serum Albumin (BSA) | 0.5% | 0.5 mg |
| Tween 20 | 0.1 mL | |
| Phosphate Buffered Saline (PBS) 1x concentrated | 99.9 mL | |
Table 2: Buffer Recipes.
Discussion
With this protocol, we share a method to distinguish and isolate the PVs of the mouse heart and perform immunofluorescence staining on them. After the organ harvest, the heart and lungs were dehydrated in sterilized sucrose solution, followed by separating the ventricles from the atrium and lung lobes under microscopic guidance. Afterwards, the heart base was prepared to visualize the PVs followed by cutting them from the lungs at the hilum. The subsequent immunofluorescence staining was performed using a cryotechnique by embedding the tissue in O.C.T. compound, cutting it with a cryotome, and performing immunofluorescence staining for cTNT and Cx43.
Isolating the PVs can be useful for studying their role in the arrhythmogenesis of AF. However, PV isolation in mice is challenging. In addition to their small size, PVs are hidden behind the main bronchi and the prominent vessels at the heart base, convoluting their detection massively. This protocol provides a simple and transparent way to investigate the murine PVs and permits an economic approach to study their morphological characteristics in detail. The proposed harvesting and preparation approach preserves the anatomical relations between PVs, the corresponding lung lobes, and the LA, facilitating investigations of the entire LA-PV tissue complex.
MS have a heterogeneous arrangement. Significant variability in continuity and fiber orientation has been described in multiple species, including rats and humans, and are mostly found around the PV orifices4,20. Those diversities result in anisotropic electrical conduction and can induce the initiation of reentry excitations and autonomic activity in PVs21. Furthermore, the electrical interconnection between MS and atrial myocardium at the LA-PV junction can lead to the transition of desynchronized electrical impulses from the PVs to the left atrium, resulting in AF2. Preserving the morphological proportions at the LA-PV junction is, therefore, crucial for studying the interaction between the atrial myocardium and PVs. To achieve this, the accurate identification of the main bronchi and PAs is essential. We identified the landmarks according to their anatomic positions and relations using a microscope with 10x magnification and by systematically following their course from the heart base to the LH.
We performed IF staining of the murine PVs. Staining approaches based on immunoreactive dyes are widely established in different species and allow the direct detection of target antigens, providing high-quality visual contrast in microscopy. This approach enables both qualitative and quantitative characterization of intact tissue slices on a protein level. We use the cardiac isotype of TNT to distinguish between myocardium and non-cardiac musculature. The presence of cardiomyocytes in PVs, as well as the SVC and IVC has been described previously7. However, other tubular structures of the lungs, including the PAs, bronchi, and private lung vessels, do not contain heart muscle cells, which makes them easy to distinguish by IF staining7. By labeling structure proteins such as cTNT, cardiomyocytes show their characteristic cross striation depending on their orientation towards the beam path. The direction of the striation can be mapped in ImageJ with plugins such as OrientationJ or Directionality Plugin. This aids in quantifying and characterizing the directionality of cardiomyocytes and might be useful for identifying regions with anisotropic electrical conduction. Furthermore, this study supports previous findings regarding the abundant presence of Cx43 in the murine PV myocardium, which is organized in a similar manner as in the LV (Figure 4A)15,16. Remarkably, we observed gap junctions at the lateral membrane of cardiomyocytes in murine PVs. This condition, known as lateralization, has been demonstrated as a pathophysiologic mechanism in AF, especially in male mice22. Thus, this protocol highlights the potential for similar studies in mouse MS cardiomyocytes.
PV isolation is an important technique for arrhythmia research since it allows a number of investigations, including IF-staining, flow cytometry, fluorescence-activated cell sorting, optical mapping, and patch clamp. However, since the current protocol was established for IF imaging of fixed and embedded tissue, direct proof that cardiomyocytes can be isolated from PV preparations in sufficient quality to allow experiments such as patch clamp is currently missing. The success of the microdissection depends on the experience of the researcher due to the small size and fragility of the murine cardiac tissue. The quality of IF imaging is also largely dependent on the embedding step. Horizontal arrangements are difficult to achieve, the tissue integrity of the sections can suffer, and PVs can rupture if they are not filled with O.C.T. compound. Furthermore, the validity of the protocol is limited to 2D imaging, as the method does not allow the 3D reconstruction of the PVs.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the German Centre for Cardiovascular Research (DZHK; 81X3600221to H.V., 81X2600255 to S.C.), the China Scholarship Council (CSC201808130158 to R.X.), the German Research Foundation (DFG; Clinician Scientist Program in Vascular Medicine (PRIME), MA 2186/14-1 to P. T.), and the Corona Foundation (S199/10079/2019 to S. C.).
Materials
| Adhesion slides | Epredia | 10149870 | |
| AF568-secondary antibody | Invitrogen | A11036 | Host: Goat, Reactivity: Rabbit |
| Agarose | Biozym LE | 840104 | |
| Alexa Fluor 488-secondary antibody | Cell Signaling Technology | 4408S | Host: Goat, Reactivity: Mouse |
| Anti-Connexin 43 /GJA1 antibody | Abcam | ab11370 | Polyclonal Antibody, Clone: GJA1, Host: Rabbit |
| Anti-cTNT antibody | Invitrogen | MA5-12960 | Monoclonal Antibody, Clone: 13-11, Host: Mouse |
| Bovine serum albumin | Sigma-Aldrich | A2153 | |
| Brush | Lukas | 5486 | size 6 |
| Cover slips | Epredia | 24 mm x 50 mm | |
| Cryotome Cryo Star NX70 | Epredia | Settings: Specimen temperature: -18 °C, Blade Temperature: -25 °C | |
| DFC365FX camera | Leica | ||
| DM6 B fluorescence microscope | Leica | ||
| Dry ice | |||
| Dubecco's phosphate-buffered saline (DPBS) 1x conc. | Gibco | 14040133 | 500 mL |
| Dumont #5FS Forceps | F.S.T. | 91150-20 | 2 pieces needed |
| Fine Scissors | F.S.T. | 14090-09 | |
| Fluorescence mounting medium | DAKO | S3023 | |
| Graefe Forceps | F.S.T. | 11052-10 | |
| Hoechst 33342 | Invitrogen | H3570 | Cell nuclei counterstaining |
| ImageJ | FIJI | analysis and processing software | |
| LAS X | Leica | Imaging software for Leica DM6 B | |
| Microtome blades S35 | Feather | 207500000 | |
| Microwave | |||
| Normal goat serum | Sigma-Aldrich | S26-M | |
| O.C.T. compound | Tissue-Tek | 4583 | |
| Paraformaldehyde 16% | Pierce | 28908 | methanol-free |
| Pasteur pipettes | VWR | 612-1681 | |
| Petri dish | TPP | 93100 | 100 mm diameter |
| Rocker 3D digital | IKA Schüttler | 00040010000 | |
| Slide staining jars | EasyDip | M900-12 | |
| Specimen Molds | Tissue-Tek Cryomold | 4557 | 25 mm x 20 mm x 5 mm |
| StainTray M920 staining system | StainTray | 631-1923 | Staining system for 20 slides |
| Sterican Needle | Braun | 4657705 | G 27 – used for injection (step 2) and pinning (step 3 and 4) in the protocol |
| Student Vannas Spring Scissors | F.S.T. | 91500-09 | |
| Super PAP Pen Liquid Blocker | Super PAP Pen | N71310-N | |
| Syringes | Braun | 4606108V | 10 mL |
| Tris base | Roche | TRIS-RO | component for 1x Tris-Buffered Saline (TBS) |
| Triton X-100 | Sigma-Aldrich | T8787 | |
| Tween 20 | Sigma-Aldrich | P2287 |
References
- Lippi, G., Sanchis-Gomar, F., Cervellin, G. Global epidemiology of atrial fibrillation: An increasing epidemic and public health challenge. International Journal of Stroke. 16 (2), 217-221 (2021).
- Wijesurendra, R. S., Casadei, B. Mechanisms of atrial fibrillation. Heart. 105 (24), 1860-1867 (2019).
- Haïssaguerre, M., et al. Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. The New England Journal of Medicine. 339 (10), 659-666 (1998).
- Mueller-Hoecker, J., et al. Of rodents and humans: a light microscopic and ultrastructural study on cardiomyocytes in pulmonary veins. International Journal of Medical Sciences. 5 (3), 152-158 (2008).
- Kramer, A. W., Marks, L. S. The occurrence of cardiac muscle in the pulmonary veins of Rodenita. Journal of Morphology. 117 (2), 135-149 (1965).
- Nathan, H., Eliakim, M. The junction between the left atrium and the pulmonary veins. Circulation. 34 (3), 412-422 (1966).
- Nathan, H., Gloobe, H. Myocardial atrio-venous junctions and extensions (sleeves) over the pulmonary and caval veins: Anatomical observations in various mammals. Thorax. 25 (3), 317-324 (1970).
- Bond, R. C., Choisy, S. C., Bryant, S. M., Hancox, J. C., James, A. F. Ion currents, action potentials, and noradrenergic responses in rat pulmonary vein and left atrial cardiomyocytes. Physiological Reports. 8 (9), 14432 (2020).
- Thiagalingam, A., et al. Pulmonary vein contraction: Characterization of dynamic changes in pulmonary vein morphology using multiphase multislice computed tomography scanning. Heart Rhythm. 5 (12), 1645-1650 (2008).
- Spach, M. S., Barr, R. C., Jewett, P. H. Spread of excitation from the atrium into thoracic veins in human beings and dogs. The American Journal of Cardiology. 30 (8), 844-854 (1972).
- Scott McNutt, N., Weinstein, R. S. Membrane ultrastructure at mammalian intercellular junctions. Progress in Biophysics and Molecular Biology. 26, 45-101 (1973).
- Barr, L., Dewey, M. M., Berger, W. Propagation of action potentials and the structure of the nexus in cardiac muscle. Journal of General Physiology. 48 (5), 797-823 (1965).
- Kawamura, K., Konishi, T. Ultrastructure of the cell junction of heart muscle with special reference to its functional significance in excitation conduction and to the concept of "disease of intercalated disc". Japanese Circulation Journal. 31 (11), 1533-1543 (1967).
- Van Kempen, M. J., Fromaget, C., Gros, D., Moorman, A. F., Lamers, W. H. Spatial distribution of connexin43, the major cardiac gap junction protein, in the developing and adult rat heart. Circulation Research. 68 (6), 1638-1651 (1991).
- Verheule, S. Tissue structure and connexin expression of canine pulmonary veins. Cardiovascular Research. 55 (4), 727-738 (2002).
- Xiao, Y., et al. Expression of connexin 43, ion channels and Ca2+-handling proteins in rat pulmonary vein cardiomyocytes. Experimental and Therapeutic Medicine. 12 (5), 3233-3241 (2016).
- Xia, R., et al. Whole-mount immunofluorescence staining, confocal imaging and 3D reconstruction of the sinoatrial and atrioventricular node in the mouse. Journal of Visualized Experiments. (166), (2020).
- Tomsits, P., et al. Medetomidine/midazolam/fentanyl narcosis alters cardiac autonomic tone leading to conduction disorders and arrhythmias in mice. Lab Animal. 52 (4), 85-92 (2023).
- Xia, R., et al. Isolation and culture of resident cardiac macrophages from the murine sinoatrial and atrioventricular node. Journal of Visualized Experiments. (171), (2021).
- Bredeloux, P., Pasqualin, C., Bordy, R., Maupoil, V., Findlay, I. Automatic activity arising in cardiac muscle sleeves of the pulmonary vein. Biomolecules. 12 (1), 23 (2021).
- Chen, P. S., et al. The mechanisms of atrial fibrillation. Journal of Cardiovascular Electrophysiology. 17, S2-S7 (2006).
- Thibault, S., Ton, A. -. T., Huynh, F., Fiset, C. Connexin lateralization contributes to male susceptibility to atrial fibrillation. International Journal of Molecular Sciences. 23 (18), 10696 (2022).

