Neural Stem Cell Transplantation in Experimental Contusive Model of Spinal Cord Injury
Summary
Spinal cord injury is a traumatic condition that causes severe morbidity and high mortality. In this work we describe in detail a contusion model of spinal cord injury in mice followed by a transplantation of neural stem cells.
Abstract
Spinal cord injury is a devastating clinical condition, characterized by a complex of neurological dysfunctions. Animal models of spinal cord injury can be used both to investigate the biological responses to injury and to test potential therapies. Contusion or compression injury delivered to the surgically exposed spinal cord are the most widely used models of the pathology. In this report the experimental contusion is performed by using the Infinite Horizon (IH) Impactor device, which allows the creation of a reproducible injury animal model through definition of specific injury parameters. Stem cell transplantation is commonly considered a potentially useful strategy for curing this debilitating condition. Numerous studies have evaluated the effects of transplanting a variety of stem cells. Here we demonstrate an adapted method for spinal cord injury followed by tail vein injection of cells in CD1 mice. In short, we provide procedures for: i) cell labeling with a vital tracer, ii) pre-operative care of mice, iii) execution of a contusive spinal cord injury, and iv) intravenous administration of post mortem neural precursors. This contusion model can be utilized to evaluate the efficacy and safety of stem cell transplantation in a regenerative medicine approach.
Introduction
A spinal cord injury (SCI) is the most common injury caused by high-energy trauma like motor vehicles accidents, falls, sports and violence 1. In severe SCI, the injury force destroys or damages neural tissue, causing sudden loss of neurological function. Traumatic SCI occurs frequently in young adults between 10 and 40 years of age. It greatly affects the patient’s mental and physical condition and causes enormous economic impact to society 2. The treatment approach in the acute phase is often limited to a high-dose of corticosteroid, surgical stabilization and decompression to possibly attenuate further damage 3-4, but the roles of these methods on locomotor recovery after SCI are still controversial. In addition to acute tissue loss, the traumatic injury and the activation of secondary mechanisms of degeneration cause demyelination and death of multiple cell types 5-6. The degree of recovery of function can often be correlated to the extent of spared white matter at the injury site 7.
Animal models of SCI may be used both to investigate the biological responses of the tissue to injury and to test potential therapies. Moreover, a useful animal model of a human pathology not only has to reproduce some aspects of that condition but also must offer advantages over direct clinical observation and experiment. The most widely used models of spinal cord injury involve contusion or compression injury delivered to the surgically exposed spinal cord 8. The development of a controlled weight-drop contusion injury represent an important milestone in the history of SCI research. The Ohio State University spinal cord research center has pursued the technological challenge of a device that can be used to induce a particular compression of the spinal cord with parameters of impact controlled by a computer 9. This was originally designed for use with rats; later it was modified to apply towards mice 10. The advantages of this kind of approach are that the biomechanics of injury can be studied more in depth and the parameters of injury can be defined in a more complete manner in order to obtain a reproducible experimental model, therefore allowing more precise evaluation of the effects of tested treatments on the functional recovery process.
Many studies have evaluated the transplantation effects of a variety of stem cells in SCI models 11. We have recently isolated adult neural stem cells from the Sub-Ventricular Zone (SVZ) several hours after death of the mouse donor 12-13. This procedure provides a population of neural stem cells, called post mortem neural precursors (PM-NPCs), which seem to be advantageous in a regenerative medicine approach for curing SCI. In this paper we will demonstrate: i) the protocol for cell labeling with the vital tracer PKH26, ii) the surgical procedure to perform on traumatic SCI, and iii) the intravenous (i.v.) administration of labeled cells. Moreover, in this work we demonstrate that transplanted cells migrate to spinal cord lesion sites and differentiate mostly into microtubule associated protein (MAP) 2 positive cells. Furthermore, the differentiation is accompanied by the promotion of a stable recovery of hind limb function.
Protocol
NOTE: All the procedures were approved by the Review Committee of the University of Milan and met the Italian Guidelines for Laboratory Animals in compliance with European Communities Directive dated November 1986 (86/609/EEC).
1. Preparation of Cells for Transplantation
NOTE : Use neural stem cells between the 5th and the 9th passage in culture for these experiments; test the cultures for proliferation and differentiation ability before being labeled for transplantation. Determine the extent of differentiation by immunocytochemistry 12.
- Resuspend cells to a concentration of 1 x 106 cells/150 μl (transplant 1 x 106 cells per mouse). Prepare at least 1.2 x 106 cells per mouse because an excess of cells is required for syringe loading purposes.
- Wash cells 3 times using neural stem cell medium 13 in a 10 ml conical vial. In each washing step, precipitate cells by centrifugation (500 x g for 5 min, RT).
- Count the cells before the final spin.
- Centrifuge the cells (500 x g for 5 min), and then aspirate the supernatant, being careful not to remove any cells but leaving no more than 25 μl of supernatant.
- Prepare a 2x cell suspension by adding 1 ml of Diluent C to the cell pellet and resuspend with gentle pipetting.
- Immediately before staining, prepare a 2x Dye Solution (4 x 10–6 M) in Diluent C by adding 4 µl of the PKH26 ethanol dye solution to 1 ml of Diluent C in a tube and mix well to disperse.
- Rapidly add the 1 ml of 2x cell suspension to 1 ml of 2x Dye Solution and immediately mix the sample by pipetting (final cell density will be 1.2 x 107 cells/ml and 2 x 10–6 M PKH26).
- Incubate the cell/dye suspension for 1-5 min.
- Stop the staining by adding an equal volume (2 ml) of 1% BSA solution in HBSS and incubate for 1 min.
- Centrifuge cells (500 x g for 10 min) and carefully remove the supernatant.
- Resuspend cell pellet in 10 ml of HBSS and centrifuge (500 x g for 5 min).
- Wash the cell pellet 2 more times with 10 ml of complete medium to ensure removal of unbound dye.
- Resuspend the cell pellet in 10 ml of complete medium for assessment of cell recovery, cell viability and fluorescence intensity. If cells are needed for transplantation, wash and resuspend them in a sterile physiological solution at a concentration of 3.3 x 105 cells/50 μl.
2. Preparation for Surgery
- Clean and sterilize surgery equipment.
- Prepare the surgery area by wiping with an aseptic agent. Set up the IH Impactor device.
- Prepare the anesthesia equipment: the Active Gas Scavenger with VetScav Filter Weighing Device, continuous flow Induction Chamber, Oxygen Generator, Low Flow O2 Meter for Mice. Clean the induction chamber and the mask used during surgery.
- Anesthetize animals with 2.5% (v/v) isoflurane in oxygen (1 L/min), and wait 5 min after the flow induction for the drug to take effect. Check if the whiskers are twitching or if there is slow hind limb withdrawal in response to pinching the footpad. During the surgery, reduce the isoflurane concentration to 2.0% (v/v) isoflurane in oxygen (1 L/min).
3. Preparation of Mice for Surgery and Transplantation
- Keep the male adult CD1 mice (25–30 g) for at least 3 days before the experiments in standard conditions (22 ± 2 °C, 65% humidity, and artificial light between 08:00 a.m. to 08:00 p.m.).
NOTE: The entire procedure from surgical preparation to suturing will take about 40 min.- To identify the animal, mark the tail by means of water resistant colored ink.
- Use an electric clipper to cut the dorsal hair of the mouse from the neck, about at T2 level, to the lumbar area.
- Sterilize the prepared area with an iodide solution and ethanol (70% in sterile water).
- Treat the animal with a sub cutaneous (s.c.) injection of 200 μl gentamycin (1 mg/ml in sterile saline solution).
- Apply ophthalmic lubricant to both animal eyes to prevent desiccation and inject buprenorphine (subcutaneously, 0.03 mg/kg) to alleviate pain.
4. Laminectomy
- Place the mouse on a slide warmer to avoid the problem of hypothermia during the surgery. Position the animal with the dorsal side up.
- Make a vertical incision with a scalpel over the region of interest, from T7 to T12.
- Hold the skin and superficial fat pad by using standard forceps (usually found in the space between the 5th and 6th thoracic dorsal processes).
- Count the process under the vessel as T6 then move to T7.
- Place a little bearing under the ventral side of the mouse to increase the curvature of the spine. Immobilize the spinal cord by blocking it with the Graefe forceps.
- Cut the paravertebral muscles bilaterally from T7 and T10 vertebral level by using the scalpel until the dorsal surface of the lamina contacts the scalpel tip.
- Use the scalpel to tick off the junction from T7 until T10. Stop in the space between T8 and T9 spiny protrusions. Cut the tissues between T8-T9 and T9-T10 with the micro scissors.
- Use the Rongeur to remove the T9 process. Expose the junction by carefully scraping away the muscle layer with the micro scissors. Continue until bone is exposed.
- Use the forceps to remove muscles from the lamina and open a small space between the vertebrae. Gently insert the micro scissors under the bone, cut the lamina on both sides and remove this part with forceps.
- Remove the lamina with the forceps to expose the cord. Be sure not to leave any free or jagged bone fragments behind; remove them with the Rongeur.
- Use the small tip forceps to remove the periosteum as well as any bone fragments or muscles that may be near the incision.
- Remove the top half of the T9 dorsal process and proceed to the IH Impactor device protocol.
5. IH Impactor Device Protocol (Contusion)
- Place the mouse in the middle of the stabilization platform of the IH Impactor device.
- Block the animal with the two teeth forceps connected with the stabilization platform by two joint positioning arms (left arm for the thoracic vertebra, right arm for cervical vertebra).
- Use the rostral arm to grasp the lateral edge of the rostral vertebral body (T8).
- Manipulate the caudal arm in the same manner to grasp the T10 vertebral body.
- Place the stabilization platform in the device and lower the tip (diameter 0.75 mm) as close to the cord as possible without touching it.
- Lift the tip three complete turns before initiating the impact.
- Perform the contusion with the impactor set to deliver a force of 60 kdyn at 100 mm/sec.
6. Sutures and Post-care
- Suture the incision with a 4/0 absorbable suture thread. Cover the exposed spinal cord at the site of removed lamina; suture the tissue at the extremities of the cut by means of a small needle. Put the two stitches immediately above and under the site of spinal cord exposure.
- Close the skin using two or three Reflex clips without pinching off the underlying muscles.
- Hydrate the mouse post-surgery with 2 ml of saline solution subcutaneously injected in the lower back.
- Place the mouse back in a pre-warmed cage to avoid hypothermia during surgical recovery. Place cages on heating pads.
- Monitor the mice during the acute phase post-injury by checking the size bladder, the suture and the animal weight twice a day. Gently squeeze bladder (twice a day for 7 days) to avoid urinary tract’s infections (urine could be cloudy, bloody or containing any precipitates).
- Treat mice once a day with a saline solution injection (2 ml for two days post-surgery s.c. injection) and antibiotic (gentamycin 0.2 ml; s.c. injection) for five days post injury.
- Treat mice for post-operative analgesia with buprenorphine (0.1 mg/kg; twice a day) for 3 days post injury.
7. Tail Vein Injection of Cells
NOTE: In the following step the procedure for injecting the cells into the tail vein is demonstrated. Cells could be also administered with an intraspinal transplant by using a stereotaxic frame 15-16, or into the cisterna magna 17. It is important to consider that other cell types could be transplanted with this method, such as mesenchymal stromal cells (e.g., bone marrow mesenchymal stem cells, adipose derived stem cells, amniotic fluid cells). Furthermore, other treatment options such as nanoparticles can be injected via the tail vein after the spinal cord injury.
- Use 70% ethanol and PBS to clean the needle before use. Handle the needle and the syringe only with sterile gloves.
- Resuspend the cells in the test tube and load 75 μl of cells into a 0.3 ml syringe (29 G needle and 0.33 cc syringe).
- Make sure that no bubbles are present within the cell suspension. Keep the syringe in a horizontal position to avoid cell sedimentation.
- Place the mouse underneath the heat lamp to dilate the tail veins.
- Grab the mouse and gently pull it into the mouse restrainer to visualize the lateral tail vein as a narrow blue line.
- Clean the tail with an alcohol swab. Once the vein is visualized, grab the tail vein between the middle finger and thumb of the left hand.
- Bring the needle to the surface at a 15° angle from the horizon and make sure the bevel is up.
- Inject 50 μl of cells at a rate of .33 μl/sec. After the injection, delay the retraction of the needle by 10 sec. Retract the needle slowly and pay attention to the possible efflux of cell suspension.
- Clean the syringe as in step 7.1 between loads.
8. Behavioral Tests and Hind Limb Function
- Place the mouse in the open field.
- Evaluate locomotor function and hind limb recovery after contusion with the open field test according to the Basso mouse scale (BMS) 18 .
NOTE: Neurological function must be evaluated periodically, from day 3 after injury for 4 weeks 18.
9. Perfusion
- At the end of the experimental period, anesthetize animals by an intraperitoneal injection of sodium pentobarbital (65 mg/kg). Use toe pinches to evaluate the anesthesia level and proceed only after mouse is unresponsive to the noxious stimuli.
- Restrain the animal in a supine position on a surgery plane.
- Cut through the integument and abdominal wall just beneath the rib cage. Separate the diaphragm from the liver.
- Use the scissors to cut the diaphragm to expose the pleural cavity.
- Cut the sides of the rib cage until the collar bones.
- Use the hemostats to clamp the xiphoid cartilage and place the hemostat over the head.
- Hold the bottom third of the heart on a transversal plane with the forceps. Insert the needle into the left ventricle.
- Use a hemostat to clamp the heart. This secures the needle and prevents leakage.
- Use the scissors to cut the right atrium.
- Allow the PBS to pump (18 ml/min) through the animal. Maintain this pressure throughout the buffer infusion period (3-4 min; corresponding to about 70 ml PBS). Continue until the heart is clean.
- Switch the stopcock to allow the fixative (4% Paraformaldehyde in distilled water, 4%PFA) through the pump. Allow the 4% PFA to pump through the animal for approximately 8-10 min (300 ml). Gradually increase the pressure to reach a maximum of 30 ml/min. A hardened liver is the best indication of a successful perfusion.
10. Tissue Collection and Processing, Histology and Iimmunohistochemistry
- Dissect spinal cords from T5 to L1. Then post-fix the tissue in 6 ml in 4% PFA (O/N at 4 °C).
- Put the same tissue in a solution with 30% sucrose for 72 hr at 4 °C in order to cryo protect it and to prevent the formation of crystals during freezing.
- Quick freeze the cord using dry ice and store it at -80 °C.
- Section by means of a cryostat with 15 µm thickness and collect sections onto glass slides and continue for immunocytochemistry.
- Rinse sections with 200 μl of PBS for each slide (3 times; 5 min each, RT).
- Permeabilize each slide with 200 μl of 10% NGS and 0.2% Triton X-100 in PBS for 1 hr at RT.
- Rinse sections with 200 μl of PBS for each slide (3 times; 5 min each, RT).
- Block non-specific sites with 200 μl of blocking solution for slide (5% NGS; 0.1% Triton X-100 in PBS) for 30 min (RT).
- Incubate each slide with 200 μl of primary antibodies O/N at 4 °C (dilute antibody in 5% NGS; 0.1% Triton X-100 in PBS).
- Wash sections with 200 μl of PBS for each slide (3 times; 5 min each; RT).
- Incubate with appropriate secondary antibodies for 2 hr at RT.
- Wash sections with 200 μl of PBS for each slide (3 times; 5 min each; RT).
- Stain nuclei with 200 μl of 4',6-diamidin-2-phenilindole (DAPI) (1 µg/ml final concentration, 10 min at RT).
- Mount by using the FluorSave Reagent and analyse by confocal microscopy.
NOTE: In control determinations, primary antibodies must be omitted and replaced with equivalent concentrations of unrelated IgG of the same subclass. Microtubule-Associated Protein 2 primary antibody was used.
Representative Results
The total number of transplanted cells is 1 x 106 cells and was divided into three consecutive injections in the tail vein. We administered 3.3 x 105 cells in 50 µl of phosphate buffer solution (PBS). The first injection was performed within 30 min after injury, the second 6 hr later and the last 18 hr after the lesion. The choice of a time limit of 18 hr after SCI for administering PM-NPCs was determined by the optimal permeability of the blood brain barrier at this time 14. To evaluate the effect of stem cells injection it would be useful to have positive control laminectomies animals (n = 14) and PBS injected animals as a negative control (n = 14).
PM-NPCs Improve Recovery of Hind Limb Function, migrate to lesion site and differentiate in MAP-2 positive cells
The T9 contusion caused the transient loss of hind limb function followed by a progressive gradual recovery (Figure 1). Within 2-3 weeks, PBS-treated injured mice improved and hind limb function reached 3 points of BMS (corresponding to plantar placing of the paw with or without weight support or occasional, frequent, or consistent dorsal stepping, but not plantar stepping 18). Instead, within the same observational period, injured mice treated with PM-NPCs showed a higher recovery, reaching 4.5 points of BMS (corresponding to frequent or consistent plantar stepping without coordination, or frequent or consistent plantar stepping with some coordination). The behavioral improvement was particularly evident in the period between day 7 and day 14 after SCI. No signs of allodynia-like forelimb hyper sensibility 19 were recorded at any time in any experimental group throughout the observational period of 30 days.
Most engrafted PM-NPCs, labeled with PKH26 (Figure 2), accumulated at the edges of the lesion forming clusters (Figure 3) from the early days of their administration. Then the transplanted cells migrated along the lesion edges and in a more diffused fashion where they differentiated, gradually assuming the asymmetric cellular conformation of neurons. At 30 days after lesion and transplantation, the cell body of PM-NPCS increased in size and in most cells dendritic-like processes were obvious and fully immunostained by the specific antibodies to MAP-2 (Figure 4). The achievement of morphological complexity and the positivity to MAP2 by transplanted PM-NPCs is likely not due to fusion with surviving host spinal cord neurons, which is evident in their clearly differentiated morphology and the absence of two nuclei in any single labeled cell.
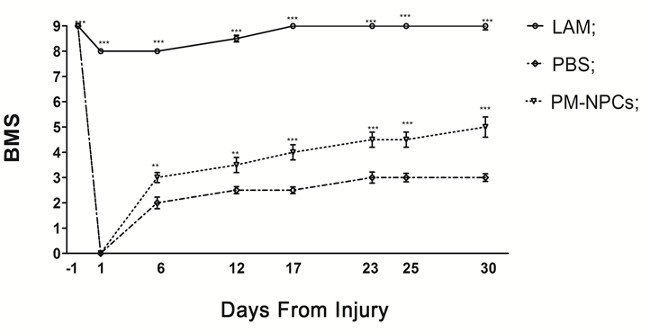
Figure 1. PM-NPCs improve functional recovery in injured animals. The open field locomotion was the test employed for the determination of motor function recovery 18. Animals were tested the day before the contusion and scored 9 points in the BMS scale. On the first day post injury in the lesioned animals, the BMS score decreased to zero. The recovery of hind limb function of lesioned mice showed a remarkable and long lasting improvement when animals were treated with PM-NPCs. The analysis was performed in double blind, and each group was composed of 14 animals. Values represent average ± SEM. We determined the statistical differences by means of ANOVA test followed by Tukey’s post-test. ***P <0.001; **P <0.01 vs PBS.
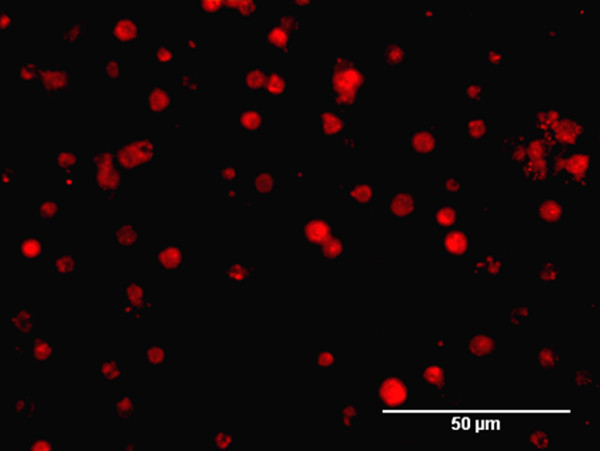
Figure 2. PKH26 labeling of PM-NPCS. After labeling procedure with PKH26, PMNPCS are visualized with the live image microscope EVOS fl and photomicrograph was taken with the same instrument (scale bar = 50 µm). Please click here to view a larger version of this figure.
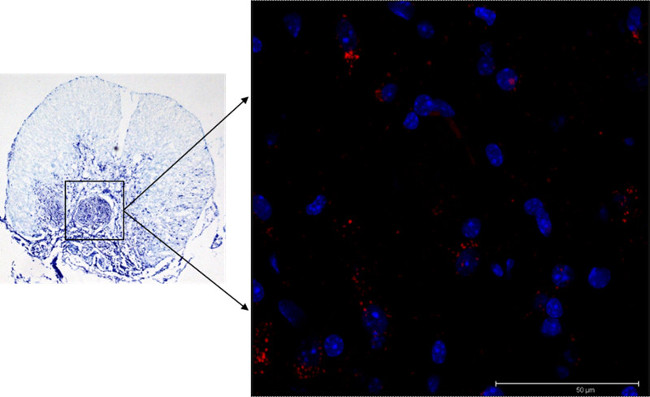
Figure 3. Localization of PM-NPCs in the lesion site. PKH26-labelled PM-NPCs (red) are present throughout the edges of the lesion site at 30 days after their i.v. injection. The image is representative for 1 mouse, but similar images were obtained for at least 5 animals (scale bar = 50 µm). Please click here to view a larger version of this figure.
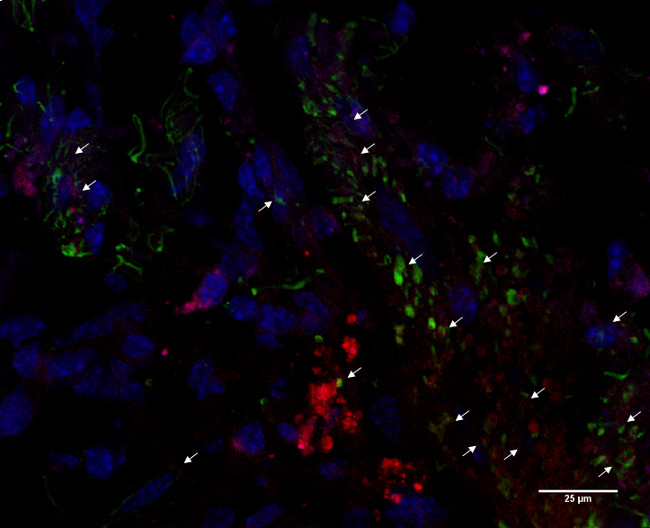
Figure 4. MAP2 expression in transplanted PM-NPCs. Most PKH26-labeled PM-NPCs (red) acquired a neuronal-like shape with dendritic-like processes and had differentiated MAP-2 positive cells (green). Nuclei are stained in blue (DAPI). The image is representative for 1 mouse, but similar images were obtained for at least 5 animals (scale bar = 25 µm). Please click here to view a larger version of this figure.
Discussion
In this paper we described a method to obtain a reproducible model of traumatic spinal cord injury using an Infinite Horizon Impactor at a force of 70 kdyne (severe). Using a larger force paradigm (80 kdyne), we can cause a more severe injury that unfortunately is associated with higher mice mortality. In order to avoid this problem, we commonly choose a moderate force paradigm (70 kdyne) that is associated to a repeatable lesion with a gradual recovery of function and lower mortality. To produce such a stable injury it is very important to pay particular attention to the correct fixation of the animal on the impactor platform; in particular the spinal cord must be centered at the impactor tip, and the two arms of the impactor must be parallel to each other. Moreover, attention must be taken in blocking the animal with the impactor forceps, when the vertebrae may be crushed and the cord may be damaged by the tips of the forceps. The positioning of the animal after the laminectomy is critical, and the animal handling during this procedure is also very important. Special attention must also be given to the laminectomy procedure, which must always be performed at the same site and for the same extension. When these procedures are performed during laminectomy, another methodological issue to monitor is the reduction of the risk of damaging the cord when using micro-scissors, Rongeur, or forceps to cut bones and free the cord through the removal of the lamina, to eliminate the remaining lateral bone protrusions and fragments, and to remove the periosteum. The use of micro-scissors and Rongeur with tips pointing upwards will reduce the risk of encountering the aforesaid problems.
An important limitation of this method is the high probability that the animals may develop internal and external severe urinary infections in the post injury period. The external infection is due to the inability of the lesioned mice to move with hind limb weight support. In contrast, the internal urinary infection may be caused by the inability of the injured mice to urinate independently. In order to avoid these problems it is absolutely necessary to inject the animals with the indicated antibiotic and to check the size of the bladder twice a day during the animal care procedures of the first week. The hydration status and the weight must be checked carefully during the first three weeks after lesioning.
Applying the described procedures we were able to obtain reproducible deficits of hind limb function that were evaluated through a specific behavioral test developed by Basso and colleagues (BMS) 18. Immediately after the injury the hind limb loss of function is complete, which is followed by a gradual recovery that is most important during the first 2-3 weeks. The behavioral recovery reached a higher level when lesioned mice were treated with adult PM-NPCs (Figure 1). We also serially sectioned spinal cords of cell treated mice at 4 weeks after injury, and detected PKH26 positive PM-NPCs by means of confocal microscopy at the edges of the lesion (Figure 3). We estimated that the total number of vital grafted PM-NPCs is greater than the grafted ESCs and adult NSCs as previously reported by our research group 20-21. At four weeks after lesioning and transplantation, most PM-NPCs have larger bodies and possess extended dendritic-like processes that are fully immunostained by the specific antibodies to MAP-2 (Figure 4).
The major advantage of this model of traumatic spinal cord injury is the standardization of the injury. A reproducible time-related curve of hind limb recovery of function is also achieved. Such reproducibility makes it possible to define proof-of-principle studies for investigated treatments including transplantation of cells for regenerative medicine studies with a reduced number of cases. In addition, several aspects of the pathophysiology of spinal cord injury can be analyzed in greater detail.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
The Authors acknowledge the economic support by FAIP (Federazione Associazioni Italiane Paraplegici), “Neurogel-en-Marche” Foundation (France), Fondazione “La Colonna”.
Materials
| Name of Material/ Equipment | Company | Catalog Number | Comments/Description |
| PKH26GL-1KT | Sigma | 091M0973 | |
| Infinite horizon (IH) Impactor device | Precision Systems and Instrumentation, LLC | Model 0400 Serial 0171 | |
| Gentamycin 10mg/ml | Euroclone | ECM0011B | 1mg/ml in sterile saline solution |
| Isoflurane-Vet 250ml | Merial | B142J12A | |
| Blefarolin POM OFT 10g | |||
| Slide Warmer | 2Biological Instruments | HB101-sm-402 | |
| Scalpel, size 10 | Lance Paragon | 26920 | |
| Small Graefe Forceps | 2Biological Instruments | 11023-14 | |
| Rongeur | Medicon Instruments | 07 60 07 | |
| Micro scissors | 2Biological Instruments | 15000-00 | |
| Absorbable sutures (4/0) | Safil Quick | C0046203 | |
| Hemostat | 2Biological Instruments | 13014-14 | |
| Reflex 7 wound clip applicator | 2Biological Instruments | 12031-07 | |
| 7mm Reflex wound clips | 2Biological Instruments | 12032-07 | |
| NGS | Euroclone | ECS0200D | |
| Triton X 100 | Merck Millipore | 1086431000 | |
| Anti Microtubule Assocoated Protein (MAP) 2 | Millipore | AB5622 | |
| Alexa Fluor 488 | Invitrogen | A11008 | |
| FluorSave Reagent | Calbiochem | 345789 | |
| Neural stem cells medium | DMEM-F12 medium (Euroclone) containing 2 mm l-glutamine (Euroclone), 0.6% glucose (Sigma-Aldrich), 9.6 gm/ml putrescine (Sigma-Aldrich), 6.3 ng/ml progesterone (Sigma-Aldrich), 5.2 ng/ml sodium selenite (Sigma-Aldrich), 0.025 mg/ml insulin (Sigma-Aldrich), 0.1 mg/ml transferrin (Sigma-Aldrich), and 2 μg/ml heparin (sodium salt, grade II; Sigma-Aldrich), bFGF (human recombinant, 10 ng/mL; Life Technologies) and EGF (human recombinant, 20 ng/mL; Life Technologies) | ||
| DMEM-F12 | Euroclone | ASM5002 | |
| l-glutamine | Euroclone | ECB3000D | |
| glucose | Sigma-Aldrich | G8270-100G | |
| putrescine | Sigma-Aldrich | P5780-25G | |
| progesterone | Sigma-Aldrich | P6149-1MG | |
| Sodium-selenite | Sigma-Aldrich | S9133-1MG | |
| transferrin | Sigma-Aldrich | T 5391 | |
| Insulin | Sigma-Aldrich | I1882 | |
| Heparin sodium-salt | Sigma-Aldrich | H0200000 | |
| bFGF | Life Technology | PHG0024 | |
| h-EGF | Life Technology | PHG6045 | |
| Syringe 0.33cc 29G | Terumo | MYJECTOR | |
| buprenorphine | Schering Plough SpA | TEMGESIC | |
| eye gel | Bausch & Lomb | LIPOSIC |
Riferimenti
- . Cord Injury Statistical Center: spinal cord injury facts and figures at glance Available from: https://www.nscisc.uab.edu/PublicDocuments/fact_figures_docs/Facts%202013.pdf (2013)
- Yip, P. K., Malaspina, A. Spinal cord trauma and the molecular point of no return. Molecular Neurodegeneration. 7, 6 (2012).
- Fehlings, M. G., Cadotte, D. W., Fehlings, L. N. A series of systematic reviews on the treatment of acute spinal cord injury: a foundation for best medical practice. J Neurotrauma. 28 (8), 1329-1333 (2011).
- Furlan, J. C., Noonan, V., Cadotte, D. W., Fehlings, M. G. Timing of decompressive surgery of spinal cord after traumatic spinal cord injury: an evidence-based examination of pre-clinical and clinical studies. J Neurotrauma. 28 (8), 1371-1399 (2011).
- Sekhon, L. H., Fehlings, M. G. Epidemiology, demographics, and pathophysiology of acute spinal cord injury. Spine. 26 (24), 2-12 (2001).
- Gorio, A., et al. Recombinant human erythropoietin counteracts secondary injury and markedly enhances neurological recovery from experimental spinal cord trauma. Proc Natl Acad Sci U S A. 99 (14), 9450-9455 (2002).
- Windle, W. F., Clemente, C. D., Chambers, W. W. Inhibition of formation of a glial barrier as a means of permitting a peripheral nerve to grow into the brain. J Comp Neurol. 96 (2), 359-369 (1952).
- Young, W. Spinal cord contusion models. Prog Brain Res. 137, 231-255 (2002).
- Stokes, B. T., Noyes, D. H., Behrmann, D. L. An electromechanical spinal injury device with dynamic sensitivity. J Neurotrauma. 9 (3), 187-195 (1992).
- Jakeman, L. B., et al. Traumatic spinal cord injury produced controlled contusion in mouse. J Neurotrauma. 17 (4), 299-319 (2000).
- Sahni, V., Kessler, J. A. Stem cell therapies for spinal cord injury. Nat Rev Neurol. 6, 363-372 (2010).
- Marfia, G., et al. Adult neural precursors isolated from post mortem brain yield mostly neurons: an erythropoietin-dependent process. Neurobiol Dis. 43 (1), 86-98 (2011).
- Gritti, A., et al. Multipotent neural stem cells reside into the rostral extension and olfactory bulb of adult rodents. J Neurosci. 22 (2), 437-445 (2002).
- Whetstone, W. D., Hsu, J. Y., Eisenberg, M., Werb, Z., Noble-Haeusslein, L. J. . J Neurosci Res. 74 (2), 227-239 (2003).
- Gonzalez-Lara, L. E., et al. The use of cellular magnetic resonance imaging to track the fate of iron-labeled multipotent stromal cells after direct transplantation in a mouse model of spinal cord injury. Mol Imaging Biol. 13 (4), 702-711 (2010).
- Ottobrini, L., et al. Magnetic resonance imaging of stem cell transplantation in injured mouse spinal cord. Cell R4. 2 (3), e963 (2014).
- Janowwski, M., et al. Neurotransplantation in mice: The concorde-like position ensures minimal cell leakage and widespread distribution of cells transplanted into the cistern magna. Neuroscience Letter. 430 (2), 169-174 (2008).
- Basso, D. M., et al. Basso Mouse Scale for locomotion detects differences in recovery after spinal cord injury in five common mouse strains. J Neurotrauma. 23 (5), 635-659 (2006).
- Hofstetter, C. P., et al. Allodynia limits the usefulness of intraspinal neural stem cell grafts; directed differentiation improves outcome. Nat Neurosci. 8 (3), 346-353 (2005).
- Bottai, D., Madaschi, L., Di Giulio, A. M., Gorio, A. Viability-dependent promoting action of adult neural precursors in spinal cord injury. Mol. Med. 14 (9-10), 634-644 (2008).
- Bottai, D., et al. Embryonic stem cells promote motor recovery and affect inflammatorycell infiltration in spinal cord injured mice. Exp Neurol. 223 (2), 452-463 (2010).

