Atomic Force Microscopy Imaging and Force Spectroscopy of Supported Lipid Bilayers
Summary
We describe a protocol for preparation of supported lipid bilayers and its characterization using atomic force microscopy and force spectroscopy.
Abstract
Atomic force microscopy (AFM) is a versatile, high-resolution imaging technique that allows visualization of biological membranes. It has sufficient magnification to examine membrane substructures and even individual molecules. AFM can act as a force probe to measure interactions and mechanical properties of membranes. Supported lipid bilayers are conventionally used as membrane models in AFM studies. In this protocol, we demonstrate how to prepare supported bilayers and characterize their structure and mechanical properties using AFM. These include bilayer thickness and breakthrough force.
The information provided by AFM imaging and force spectroscopy help define mechanical and chemical properties of membranes. These properties play an important role in cellular processes such as maintaining cell hemostasis from environmental stress, bringing membrane proteins together, and stabilizing protein complexes.
Introduction
Atomic Force Microscopy (AFM) generates an image of a surface by scanning across an area of the sample using a cantilever with a very sharp tip1. The movement of the cantilever probes the surface topology of the sample. AFM has been widely applied to biological molecules — including proteins, DNA, and membranes, owing to its versatility in analyzing fixed samples in air or near-native state in liquid2-5.
Apart from its high-resolution imaging capability in the nanometer range, the AFM cantilever acts as a spring to probe interaction forces (adhesion and repulsion) and mechanical properties of the sample5,6. This is known as force spectroscopy. In this mode, the probe first approaches the sample and exerts a force on it, then is retracted until it loses contact with the sample (Figure 1A). The generated curves show force as a function of distance of the cantilever for both the approach and retraction. Several properties including the elastic modulus to measure the stiffness of a material, and adhesion forces can be derived.
Supported lipid bilayers are biological model membranes lying on top of a solid support — usually mica, borosilicate glass, fused silica, or oxidized silicon7. They are prepared using various techniques like vesicle deposition, Langmuir-Blodgett method and spin-coating8,9. AFM imaging has been used to follow the formation of these supported bilayers10, and probe different structures formed by membranes of different compositions11-15.
Performing force spectroscopy on supported bilayers results in a peak in the approach curve. This peak indicates the force needed to pierce the bilayer, and is called breakthrough force. The bilayer thickness can also be measured using the force curve6 . The typical breakthrough force of bilayers range between 1-50 nN6. These properties depend on lipid packing (liquid or gel phase) and structure (acyl chain length and degree of unsaturation) and altered by membrane-active agents16. The theory behind the rupture has been explained17 and other experimental parameters such as cantilever softness, tip radius and approach speed also affect the breakthrough force15,16,18. Force spectroscopy has been used to analyze properties of different lipid phases11,19, composition-dependent changes12,20, as well as effects of other biomolecules, like peptides, on the stability of the membrane21.
The flat orientation of supported bilayers is advantageous for combining AFM with other methods such as surface plasmon resonance22 and fluorescence microscopy11,19 to better characterize structure and properties of membranes.
This detailed video protocol is intended to prepare supported lipid bilayers using vesicle deposition and analyze them with AFM and force spectroscopy. While vesicles of various sizes may be used to prepare bilayers, this protocol focuses on small and large unilamellar vesicles. Supported bilayers that phase separate into liquid ordered (Lo) and liquid disordered (Ld) phases were characterized11,15. The membrane is composed of di-oleoyl-phosphatidylcholine (DOPC), sphingomyelin (SM), and cholesterol (Chol) at 2:2:1 ratio. This composition models the lipid rafts, which are proposed to behave as platforms important for protein trafficking and sorting, cell signaling and other cellular processes23,24.
Protocol
1. Preparation of Supported Lipid Bilayers (SLB)11,12,21
- Preparation of Lipid Mixture and Multilamellar Vesicle Suspensions
- Prepare the following buffers beforehand.
- Prepare PBS buffer at concentrations of 2.7 mM KCl, 1.5 mM KH2PO4, 8 mM Na2HPO4, and 137 mM NaCl, pH 7.2.
- Prepare SLB (supported lipid bilayer) buffer at concentrations of 150 mM NaCl, 10 mM HEPES, pH 7.4.
- Prepare a solution of 1 M CaCl2.
- Dissolve the following lipids in chloroform at a desired concentration: for example, di-oleoyl-phosphatidylcholine (DOPC) at 25 mg/ml, sphingomyelin (SM) at 25 mg/ml and cholesterol (Chol) at 10 mg/ml.
- Take 18.38 µl of DOPC, 17.10 µl of SM and 11.30 µl of Chol to make a solution with 1 mg of total lipids in a 2:2:1 DOPC:SM:Chol molar ratio. Vary the volumes accordingly based on the concentrations of the lipids prepared in 1.1.2. However, maintain the molar ratio at 2:2:1 DOPC:SM:Chol, as changing the molar ratio will change the phase characteristics of the membrane25.
- Mix the solution completely by vortexing. Add a fluorescent lipidic dye at 0.05 mol % if one wishes to visualize the bilayer by a fluorescence microscope.
Note: The family of long-chain dialkylcarbocyanines, like DiD, DiI and DiO span a wide range of wavelengths and can be ideally used to label lipid membranes. Alternatively, fluorescently labeled lipids, like rhodamine-phosphatidylethanolamine or BODIPY-cholesterol can be used. In any case, the concentration of the dye should be optimized according to the microscopy setting, keeping in mind that high concentrations of the fluorophore bound to the lipid can change the lipid behavior19. - Dry off the chloroform using N2 gas (or any available inert gas such as Ar and He).
- Further dry the lipid mixture under vacuum for at least 1 hr.
Note: If storing this lipid mixture, purge air with inert gas (N2, Ar or He) and store at -20 °C. Aliquot excess lipids-in-chloroform into small brown vials. Dry them in a similar manner as the lipid mixture, displace air with inert gas (N2, Ar or He) and store at -20 °C. - Dissolve the dried lipids in PBS buffer to a concentration of 10 mg/ml.
- Vortex the mixture vigorously for about 20 sec to 1 min to get a cloudy suspension, containing multilamellar vesicles. Make sure that the there are no lipid films sticking to the sides of the vial.
- Distribute this suspension into 10 µl aliquots (1 mg of lipid makes 10 aliquots).
- Store at -20 °C until further use.
- Prepare the following buffers beforehand.
- Preparation of Unilamellar Vesicles
Note: Various sizes of unilamellar vesicles can be prepared using the sonication method or the extrusion method. Sonication uses sound energy to agitate the mixture and separate the layers of the multilamellar suspension. This is indicated by clearing up of the hazy suspension. Extrusion forces the multilamellar vesicles to pass through a membrane filter with a definite pore size.- Sonication Method
- Take the 10 µl of multilamellar lipid suspensions and add 150 µl of SLB buffer.
- Transfer the mixture into small (2 ml) capped glass vials and seal with Parafilm.
- Sonicate the mixture at room temperature in a bath sonicator for 10 min or until it becomes clear to yield small unilamellar vesicles (SUVs, below 50 nm in diameter).
- Extrusion Method
- Take the 10 µl of multilamellar lipid suspensions and add 150 µl of SLB buffer.
- Transfer the suspension into a cryogenic tube.
- Freeze the lipid suspension by submerging the cryogenic tube for a few seconds in liquid nitrogen.
- Thaw the lipid suspension by submerging the cryogenic tube in a water bath at room temperature or 37 °C for a few minutes. Repeat the freeze-thaw steps at least 10 times.
- In order to extrude the lipid suspension, use a polycarbonate membrane with 200 nm pore size (other pore sizes are available like, 100 nm or 400 nm) and a commercial extrusion device26.
Note: Currently, there are two extruder devices commercially available: a manual extruder is available for small volumes (200 µl to a few ml). In this case, the sample is extruded through the membrane by manually pushing the suspension back and forth between two syringes. Depending on the manufacturer, lipid aliquots would need to be combined to reach minimum volume of the set up. For bigger volumes (up to 50 ml) more sophisticated devices are used in which the suspension is forced through the polycarbonate membrane by using compressed gas. Set-up and extrusion conditions can change according to the model. Refer to the instructions from the manufacturer before use. This protocol refers to the manual extruder for small volumes. - Equilibrate the extruder in water by passing water through the extruder. Immerse the membrane in water.
- Place the membrane between the two pistons, assemble the device and equilibrate in buffer, by passing the buffer through the extruder, from one syringe to the other. This step allows additionally testing the leakiness of the system. If it leaks, disassemble and reassemble it again changing the membrane.
- Take the lipid suspension using one syringe of the extruder ('dirty side').
- Attach the syringe containing the lipid suspension on one chamber and the empty syringe on the opposing chamber.
- Push the syringe on one end. Pass the solution through the membrane and into the opposing syringe. This is the 1st pass.
- Pass it through the membrane for a total of 31 times such that the solution ends up on the opposing syringe ('clean side').
Note: Check the membrane after extruding. If it was broken, then extrusion process was not successful and needs to be repeated. - Empty the syringe into a small tube. Large unilamellar vesicles (LUVs) are stable for 1-2 days if kept at 4 °C.
- Sonication Method
- Preparation of Mica and Coverslips
- Wash coverslips first in water and then in ethanol. Air dry.
- Take mica discs and peel off the outer layer using adhesive tape.
- Attach the mica disc onto the coverslip. Transparent optical glues are optimal so that one can check the bilayers with a fluorescence microscope.
- Attach plastic cylinders (2.5 cm outer diameter, 2 cm inner diameter, and 0.5 cm height) using optical glue/grease. Make sure that everything is sealed and that there are no leaks. Use grease when wanting to re-use the cylinders as they can be easily detached from the coverslip and washed. The dimensions of the plastic cylinders depend on the size of the AFM cantilever holder and should be adjusted accordingly.
- Deposition of Unilamellar Vesicles on Solid Support
- Pipet the whole liposome solution directly onto the mica. Add more SLB buffer to reach a volume of 300 µl.
- Add 0.9 µl of 1 M calcium chloride solution to reach a final concentration of 3 mM Ca2+.
- Incubate at 37 °C for 2 min and then at 65 °C for at least 10 min. Always cover the bilayers with buffer. Contact with air and air bubbles will destroy it. During incubation, add more buffer if necessary, especially if incubating at higher temperatures.
Note: Incubation temperatures vary according to the lipid mixtures and phases required. Incubation time of at least 10 min is needed to form supported bilayers, but it could be longer, depending on the temperature and composition. - Wash the bilayer with hot (65 °C) SLB buffer 15-20 times to remove calcium and unfused vesicles. Take extra care during the washing steps to keep the bilayer under buffer and gently pipet the washing solution to avoid destroying the bilayer.
- Cool supported bilayers to room temperature before AFM measurements. This is needed to form the different phases of the membrane as well as minimize thermal drift in the instrument.
2. Atomic Force Microscopy Measurements11,12
- Imaging
Note: Perform atomic force microscopy imaging in contact mode or tapping mode. Image supported bilayers in solution; do not allow solution to evaporate completely as this will destroy the bilayers. As one would be working with buffer, please observe safety precautions in making sure that any AFM part is protected from liquid spills.- Select a soft cantilever (below 0.2 N/m spring constant is advised) and mount it to the AFM. The choice of cantilever stiffness is more critical for force spectroscopy (and this will be discussed later).
- Mount the sample on the AFM stage and make sure that all vibration-isolation modules are turned on. Wait for at least 15 min to equilibrate and reduce thermal drift of the system. Depending on the AFM set-up, tuning and imaging specifications may vary. Consult the manufacturer’s handbook.
- Approach the sample with the AFM tip until contact.
- Since lipid bilayers are usually 4 nm in height, use the Z-range of the instrument that will give the highest z-resolution (for example 1.5 µm).
- Select a region to image. To get an overview of the lipid bilayer, a 50 µm x 50 µm or 25 µm x 25 µm region will be a good starting point to image. If smaller features appear, one can image in smaller areas (10 µm x 10 µm, or 2 µm x 2 µm). The choice of the imaging area also depends on the working range of the piezoelectric scanner. Please consult the instrument manual for this.
- As bilayers are very fragile, adjust the set point of the AFM tip during imaging to keep the force at minimum and correct for thermal drift, while maintaining contact with the sample. In contact mode, minimize the set point. In tapping mode, maximize the set point.
- Process images by fitting each scan line to polynomial leveling functions. Perform line removal for scans that lose contact with the membrane using the proprietary software of the AFM manufacturing company.
- Force Spectroscopy
- Calibrate the cantilever following the instructions according to manufacturer’s protocol. Calibration allows the conversion of the electrical signal of the photodiodes to force (Figure 1B).
- Calibrate the tip’s sensitivity to measure deflection as z-piezo movement (in units of length) instead of electrical signal in volts.
Note: AFM detects changes in the deflection of the cantilever using a laser reflected at the back of the cantilever. The laser reaches a photodiode that can spatially detect the position of the laser spot. A change in the deflection of the cantilever as it bends upon contact with the sample is detected by this photodiode as “vertical deflection”, and this is in the units of voltage. Since the cantilever is moved by the z-piezo, the vertical deflection is related to the movement in the z-piezo. The ratio between vertical deflection and z-movement is called sensitivity and converts voltage to distance. - Calculate the cantilever spring constant using the thermal noise frequency method, usually incorporated in the AFM software. In this method, plot the amplitude of vibration from noise with respect to frequency of oscillation, creating a power spectral density plot. This plot will show a peak around the resonance frequency. The frequency where the amplitude is the greatest is the resonance frequency. Fit a Lorentzian function around the peak to automatically calculate the spring constant by the software. Some useful resources for the theory of the thermal noise method are given in references27-30.
- Calibrate the tip’s sensitivity to measure deflection as z-piezo movement (in units of length) instead of electrical signal in volts.
- After imaging an area of the bilayer, select a 5 µm x 5 µm region in the bilayer to perform force spectroscopy.
- Set up the AFM so that it takes force curves in a 16 x 16 grid of that region. This results in 256 force curves per region. The space between two neighboring points should be enough to avoid that the membrane area being probed by one point would coincide with the next point. This is dependent on the tip radius. A 100 nm distance between two points would be ideal. Divide the 5 µm x 5 µm region into 16 x 16 grid. Depending on the size of the phase, one can select a smaller or bigger region, and then adjust the grid size accordingly.
- Set z-piezo displacement to 400 nm. This determines the maximum range of movement of the z-piezo. It should be big enough to make sure that the cantilever has fully retracted away from the sample in between measurements.
- Set approach speed to 800 nm/sec and retract speed to 200 nm/sec. Use an approach speed between 400 to 6,000 nm/sec depending on the bilayer composition and lipid phase to be studied6,16.
- After setting up all the parameters, acquire force curves, by pressing the “run” button of the AFM software.
- Save force curves as force vs. height curves (a measure of z-piezo movement). This does not take into account the bending of the cantilever when in contact with the sample. During the data processing step, the AFM software allows correction for cantilever bending to get force vs. tip-sample separation curves (Figure 1C), which will give proper values for bilayer thickness. The correction values are usually incorporated in the calibration, which is saved with each force curve. Calculate tip-sample separation by subtracting the vertical deflection from the piezo movement at a given point.
Note: The peak in the approach force curve (the force value goes down even if the cantilever movement is still on the approach cycle) is the breakthrough force of the membrane, while the difference between the position of this peak and the position of the point where the force begins to rise up again provides the thickness of the membrane. - Acquire at least 500 force curves for each condition to get good statistics. Plot a histogram of the values using programs like Origin or MatLab, and fit the histograms to a Gaussian distribution to get the peak and width of the distribution.
- Calibrate the cantilever following the instructions according to manufacturer’s protocol. Calibration allows the conversion of the electrical signal of the photodiodes to force (Figure 1B).
Representative Results
Supported lipid bilayers composed of DOPC:SM:Chol (2:2:1) were imaged in AFM (Figure 2 A-C). Because of the lipid composition, SM/Chol-rich Lo and DOPC-rich Ld phases were observed. The height profile from the AFM imaging can provide important information on the membrane structure. By looking at the height profile, the bilayer thickness can be measured in presence of defects in the membrane (Figure 2B), or the difference in height between the Lo/Ld phases can be provided. In addition, AFM allows to specifically select regions representing these phases and analyze their mechanical properties using force spectroscopy (Figure 2 D–F). The force curves derived from the force spectroscopy mode are used to measure the breakthrough force by finding the force value at the peak of the force curve (Figure 2D and E), and the membrane thickness by subtracting the distance value from the peak of the force curve to the distance value when the force curve begins to rise again (Figure 2D and F).
The sensitivity of AFM measurements is dependent on piezoelectric material and electronic feedback loops. As such, before doing experiments, is important to get familiar with the AFM set up. There are two common instrument set up for the piezoelectric materials: (1) tube scanner in which the sample is placed on top of the piezoelectric material and the sample is scanned on an immobile tip; or (2) flexure-stage where the tip is mounted on the piezoelectric material instead of the sample. In the latter case, the piezoelectric material and other electronics are protected from spills. A lot of AFM models for biological applications have this configuration.
During imaging, the AFM tip may lose contact with the sample and lead to erroneous measurements. Depending on how many scans are lost, the image can be corrected by removing the scan lines and using a mean intensity filter to derive the new values from surrounding area (Figure 3). However in most cases, this does not yield a good image and it is recommended to simply discard the image. There are other artifacts that need to be monitored. These arise from blunt/damaged/dirty tips as well as the diffraction of the laser spot behind the cantilever31. In these cases, images should be discarded and the AFM tip should be replaced.
During force spectroscopy measurements, several challenges may arise: (1) absence of peaks (Figure 4A), (2) presence of shoulders instead of well-defined peaks (Figure 4B), (3) a small rise and subsequent plateauing at very low forces (Figure 4C–D). The absence of a peak can be attributed to measuring in a region without a bilayer or to acquisition conditions not being optimized. It is recommended that an AFM image is acquired before doing force spectroscopy to make sure that the bilayer is indeed being measured. Acquisition conditions as well as tip/cantilever specifications affect the probability of a breakthrough event17. A sharper tip can easily puncture a bilayer, as such, the breakthrough force may appear low or not appear at all because it is below the noise levels. . Increasing the approach speed also increases the breakthrough force16. As the bilayer is pressed by the tip, the lipid molecules also react to dissipate this force. At a lower approach speed, the lipid bilayer has enough time to equilibrate before the next step in force, and this increases the probability that a breakthrough event can happen. At a higher approach speed, the force ramps faster than the reaction of the lipids and, as such, the probability of a breakthrough event is lower at the same force. It is possible that by using high approach speed, the set point is reached before the bilayer breaks resulting in no observable peaks. There are no reports to explain the appearance of shoulders on the curve but we postulate that it could be due to material compression before breakthrough. Furthermore, a plateau or wide peaks at very low forces may indicate the presence of dirt or loose lipid material in the tip. Tips may accumulate dirt and lipidic material in the span of several measurements. Dirt and material attached to the tip makes it blunt, thus increasing the breakthrough force.
It is recommended that force curves are acquired in different areas if these challenges arise. A second recommendation is to change tips (this will require recalibration). Lastly, the approach speed may need to be adjusted or the use of a tip with different specifications (tip radius and stiffness) may be considered. The breakthrough force varies between 0-50 nN6, as such, the spring constant of the cantilever can be in the range of 0.05-0.7 N/m. Most commercial cantilevers have a nominal tip radius around 20-40 nm. Instead of using conventional tips, tipless cantilevers could also be bought and spheres of known radius (which are usually monodisperse in size distribution) could be attached to them. To get reproducible data, always use the same acquisition conditions and cantilever specifications for the same set of experiments.
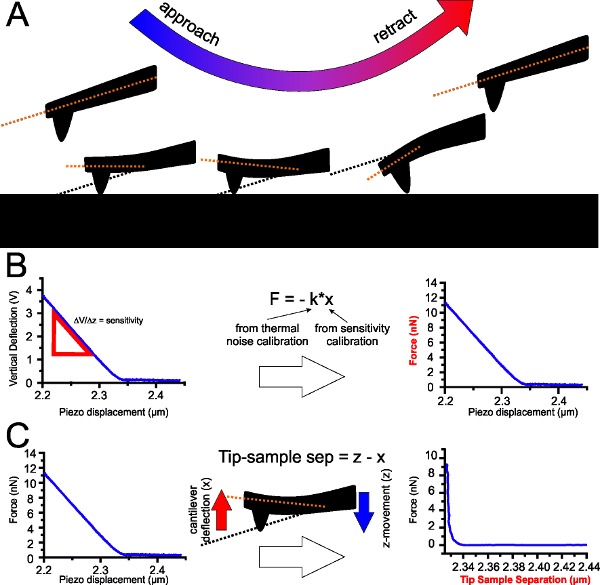
Figure 1: Principles of Force Spectroscopy. (A) Sequence of approach-retraction of the cantilever from the sample. The red line shows the cantilever deflection from its equilibrium position (black line). (B) Cantilever calibration converts electrical signal (in Volts) to force (in Newtons). By acquisition of the cantilever sensitivity and spring constant the vertical deflection (in volts) is converted into distance and the distance into force using Hooke’s law. (C) Upon contact in force spectroscopy measurements, z-piezo scan movement does not take into account the cantilever bending. A correction for this can be done by subtracting the vertical deflection, x (in distance units) from the z-piezo movement, z (also in distance units). In this way, the actual tip-sample separation, instead of simply z-piezo movement, can be reported, which is a more accurate measure of distances and is important in order to measure the thickness of the sample.
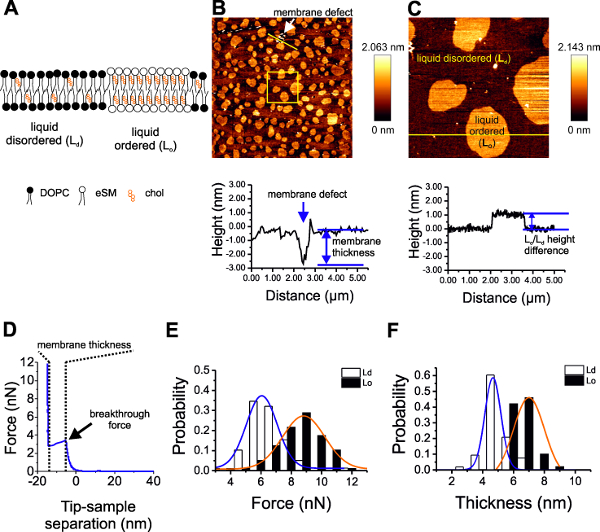
Figure 2: AFM of SLBs. (A) Scheme of SLBs composed of DOPC:SM:Chol (2:2:1) separate into liquid disordered (Ld) and liquid ordered (Lo) phases. (B) A 50 µm x 50 µm AFM image of the SLBs. The line profile below corresponds to the yellow line in the image. Membrane defects (blue arrow) show the thickness of the bilayer to be approx. 4 nm. (C) corresponds to the yellow square in B (10 µm x 10 µm). Lo and Ld phases are labeled. The line profile below corresponds to the yellow line in the image. The Lo phase appears 1-2 nm higher than the Ld phase. (D) A sample force curve showing how the breakthrough force and membrane thickness are derived. (E) Breakthrough force and (F) membrane thickness distribution for Ld and Lo phases with Gaussian fitting for quantification.
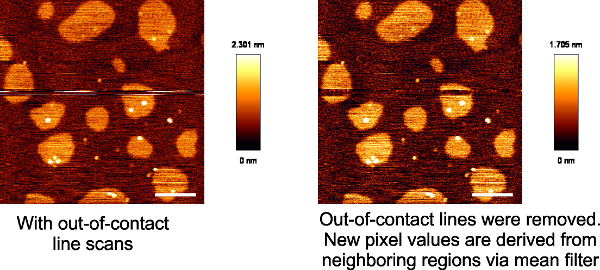
Figure 3: Loss of contact during imaging leads to scan lines that do not correspond to the rest of the image. A supported bilayer composed of DOPC:SM:Chol (2:2:1) with loss of contact in some line scans. Scale bar 2 µm.
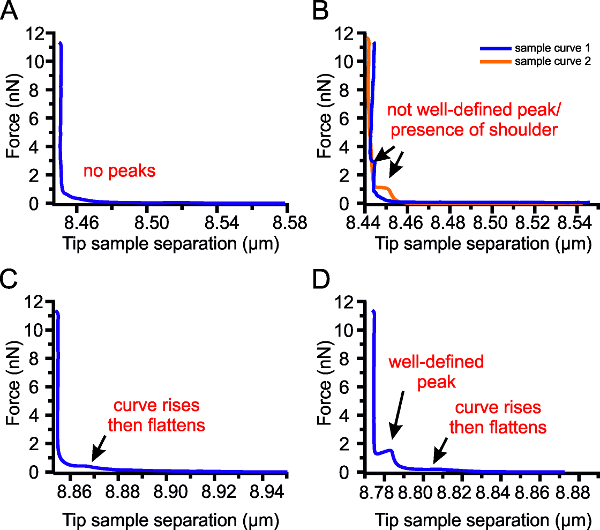
Figure 4: Challenges in Force Spectroscopy. (A) Some force curves will have no peaks. (B) The curve may have shoulders instead of well-defined peaks. (C) After a small increase a plateau may occur in the absence of the peak. (D) Appearance of a plateau together with a well-defined peak.
| Membrane region | Breakthrough Force (nN) | Membrane Thickness (nm) |
| Ld phase | 6 ± 1 | 4.7 ± 0.5 |
| Lo phase | 8 ± 1 | 6 ± 1 |
Table 1: Membrane properties of DOPC:SM:Chol (2:2:1) SLBs. The Ld phase has a breakthrough force of 6 ± 1 nN and a thickness of 4.7 ± 0.5 nm. The Lo has 8 ± 1 nN and a thickness of 6 ± 1 nm. The difference in height between the two phases is 2 ± 1.4 nm.
Discussion
SLBs composed of DOPC:SM:Chol (2:2:1) were formed on mica after vesicle adsorption and rupture induced by calcium chloride. This lipid composition separated into Ld and Lo phases. The Lo phase is enriched in sphingomyelin and cholesterol and is less fluid/more viscous (Figure 1A) than the Ld phase11. The separation of Lo from Ld phase manifests as circular structures elevated above the surrounding (Figure 1B, C). The platforms are the Lo phases surrounded by the Ld phases. Membrane defects (or holes in the membrane) are also seen (Figure 1B, blue arrow) and a height profile across this defect shows the thickness of the bilayer. The Ld/Lo height difference can also be examined by looking at an AFM height profile (Figure 1C).
The preparation of lipid mixture is very important as the composition determines the bilayer properties. During the preparation of the supported bilayer, it is important to work at the indicated temperatures for the proper formation of lipid phases (step 1.4.5). During imaging, keeping a low force is very important so as not to destroy the bilayer nor to accumulate dirt on the tip.
After AFM imaging, regions of the SLB are selected and probed using force spectroscopy. Force spectroscopy allows us to probe other properties of the membrane, specifically the breakthrough force (Figure 1D). The Ld and Lo phases show different breakthrough force and thickness (Figure 1E, F). The properties are summarized in Table 1. We note that compared to Chiantia et al.11, similar values for breakthrough force in the Ld phase were obtained but a lower value for the Lo phase. This could be due to lower amounts of cholesterol used in our membranes (we used DOPC:SM:Chol ratio 2:2:1, while they used of 1.5:1.5:1). This also demonstrates the importance of mixing the right lipid ratio — a critical factor in reproducibility of results. Furthermore, using different lipids (chain length, and degree of unsaturation) also changes the bilayer properties6,15,18.
While plotting the distribution of breakthrough forces is useful and can already show differences in the bilayer properties, further analysis of the breakthrough force distribution can be used to deduce other mechanical properties including line tension and spreading pressure11,17,21. Briefly, the probability, P(F) of measuring a certain breakthrough force, F, is described by the continuum nucleation model11,15,17:
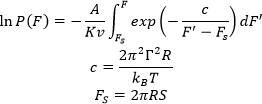
where A is the resonance frequency of the cantilever, K is the spring constant of the cantilever, v is the speed of the approach, Γ is the line tension (the edge energy required to keep a hole in the membrane open), R is the radius of the AFM tip, kB is the Boltzmann constant, T is temperature, and S is the spreading pressure or the energy associated to close the hole (opposite of membrane tension — the energy needed to be exerted by the membrane to open a hole32). This equation can be integrated and dP/dF can be calculated as:
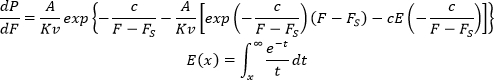
A, K, R are properties of the AFM cantilever/tip that can be acquired through cantilever calibration. T and v are experimental parameters that should be kept constant throughout the experiments. dP/dF is then fitted to the histogram to acquire the line tension and the spreading pressure. As several parameters affect P(F), experimentalists are advised to use similar tips and acquisition conditions to have comparable values for line tension and spreading pressure. Contamination of the AFM tip (including dirt and unfused vesicles on the tip acquired during imaging) leads to non-reproducible results. Furthermore, the choice of support affects the mechanical properties of the membrane20.
Another technique that can characterize mechanical properties of lipid bilayer is the micropipette aspiration of giant unilamellar vesicles (GUVs)33. In this technique, membrane tension is calculated from the deformation of GUVs during aspiration. Furthermore, by increasing the suction, one can calculate the tension at which the GUV ruptures (or lysis tension)34, which could be related to the line tension in the continuum nucleation model. One drawback of this techniques is the complexity of calculating membrane tension to phase separating membranes, as the different phases have different mechanical properties35. This is in contrast to AFM force spectroscopy, where individual phases can be characterized separately. Furthermore, whereas many GUVs are needed to produce statistically relevant results, the AFM can produce one distribution per SLB. However, membrane remodeling effects (for example, tubulation and invaginations) are more easily visualized with GUVs, making them better systems for characterizing these shape-related effects together with mechanical properties.
In further applications to study the effects of membrane proteins on bilayer properties, purified proteins may be incorporated in these vesicles to make proteoliposomes. One technique for doing this uses detergents to help the proteins to stabilize and insert into the membrane. Subsequent purification via dialysis or size-exclusion chromatography removes free proteins and detergent in solution36. Another approach requires the tagging of the purified proteins (for example, Histidine or Streptavidin tags) and incorporating affinity partners for this tag in the liposomes (with nickel-nitriloacetic acid (Ni-NTA) or biotinylated lipids).
In summary, we described a method to analyze lipid bilayers. Due to its high-resolution, AFM can reveal sub-micrometer structures in the membrane, which include lipid phases among others. Force spectroscopy, here used for characterizing lipid membranes, can also be used on membrane proteins and other biomolecules on the membrane.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Max Planck Society, the German Cancer Research Center, the University of Tübingen, and the Bundesministerium für Bildung und Forschung (grant no. 0312040).
We thank Eduard Hermann for helping us automate the analysis of the force curve data and Dr. Jakob Suckale for careful reading of this manuscript.
Materials
| 1,2-dioleoyl-sn-glycero-3-phosphocholine | Avanti Polar Lipids, Inc. | 850375P | Comes as lyophilized powder in sealed vials. Dissolve all powder in chloroform upon opening. Store extra as dried lipid films, under inert atmosphere, at -20°C. For more information on storage and handling visit http://www.avantilipids.com/index.php?option=com_content&view=article&id=1679&Itemid=398 |
| Sphingomyelin (Brain, Porcine) | Avanti Polar Lipids, Inc. | 860062P | Comes as lyophilized powder in large sealed plastic containers. Dissolve a spatula point of powder powder in chloroform upon opening. Store extra as dried lipid films, under inert atmosphere, at -20°C. For more information on storage and handling visit http://www.avantilipids.com/index.php?option=com_content&view=article&id=1679&Itemid=398 |
| Cholesterol | Avanti Polar Lipids, Inc. | 700000P | Comes as lyophilized powder in large sealed plastic containers. Dissolve a spatula point of powder powder in chloroform upon opening. Store extra as dried lipid films, under inert atmosphere, at -20°C. For more information on storage and handling visit http://www.avantilipids.com/index.php?option=com_content&view=article&id=1679&Itemid=398 |
| Sodium chloride (NaCl), 99.8% | Carl Roth GmbH + Co. KG | 9265.1 | |
| Potassium chloride (KCl), 88% | Sigma | P9541 | |
| Sodium hydrogenphosphate (Na2HPO4), >99% | AppliChem GmbH | A1046 | |
| Potassium dihydrogenphosphate (KH2PO4), 99% | Carl Roth GmbH + Co. KG | 3904.1 | |
| Calcium chloride dihydrate (CaCl2), molecular biology grade | AppliChem GmbH | A4689 | |
| HEPES, molecular biology grade | AppliChem GmbH | A3724 | |
| Glass coverslip, 24×60 mm, 1mm thickness | Duran Group | 2355036 | |
| Mica blocks | NSC Mica Exports Ltd. | These are mica pieces at least 1 sq. Inches in area and thickness randing from 0.006 inches to 0.016 inches. They are cut to a specific size by the company for shipping. Small mica discs can be punched from the mica blocks using the punch and die set. Always handle mica with gloves or tweezers. | |
| Punch and Die Set | Precision Brand Products, Inc | 40105 | |
| Optical Adhesive | Norland Products, Inc. | NOA 88 | Liquid adhesive that hardens when cured under long wavelength UV light. |
| Laboratory Equipment Grease | Borer Chemie AG | Glisseal N | |
| Liposome Extruder | Avestin | LiposoFast-Basic | As an alternative one can also look at offers from Northern Lipids, Inc. |
| Adhesive Tape | 3M | Scotch(R) Magic (TM) Tape 810 (1-inch) | |
| Bath Sonicator | Bandelin Sonorex Digitec | DT-31 | No heating, Frequency: 35 kHz, Ultrasonic Peak Output: 160 W, HF Power: 40 W. http://www.bandelin.com/datenblaetter/dt/DT_31_H_1798d_DE_GB_FR_BANDELIN.pdf |
| Silicon Nitride AFM Cantilever | Bruker AFM Probes | DNP-10 | Each cantilever has four tips and their nominal tip radius is 20 nm (with possible maximum at 60 nm). Based on the specifications, we use tip D with resonance frequency of 18 kHz, and nominal spring constant of 0.06 N/m. |
| AFM | JPK | JPK Nanowizard II mounted on Zeiss Axiovert 200 |
Riferimenti
- Binnig, G., Quate, C. F., Gerber, C. Atomic Force Microscope. Phys. Rev. Lett. 56, 930-933 (1986).
- Hansma, P. K., Elings, V. B., Marti, O., Bracker, C. E. Scanning Tunneling Microscopy and Atomic Force Microscopy: Application to Biology and Technology. Science. 242, 209-216 (1988).
- Gaczynska, M., Osmulski, P. A. AFM of biological complexes: What can we learn. Curr, Opin. Colloid In. 13, 351-367 (2008).
- Goksu, E. I., Vanegas, J. M., Blanchette, C. D., Lin, W. -. C., Longo, M. L. AFM for structure and dynamics of biomembranes. BBA-Biomembranes. 1788, 254-266 (2009).
- Muller, D. J. AFM: A Nanotool in Membrane Biology. Biochemistry-US. 47, 7986-7998 (2008).
- Redondo-Morata, L., Giannotti, M. I., Sanz, F., Baró, A. M., Reifenberger, R. G., Sanz, F. . Atomic Force Microscopy in Liquid: Biological Applications. , (2012).
- Castellana, E. T., Cremer, P. S. Solid supported lipid bilayers: From biophysical studies to sensor design. Surf. Sci. Rep. 61, 429-444 (2006).
- Frederix, P. L. T. M., Bosshart, P. D., Engel, A. Atomic Force Microscopy of Biological Membranes. Biophys. J. 96, 329-338 (2009).
- Mennicke, U., Salditt, T. Preparation of Solid-Supported Lipid Bilayers by Spin-Coating. Langmuir. 18, 8172-8177 (2002).
- Raviakine, I., Brisson, A. R. Formation of Supported Phospholipid Bilayers from Unilamellar Vesicles Investigated by Atomic Force Microscopy. Langmuir. 16, 1806-1815 (2000).
- Chiantia, S., Ries, J., Kahya, N., Schwille, P. Combined AFM and Two-Focus SFCS Study of Raft-Exhibiting Model Membranes. . ChemPhysChem. 7, 2409-2418 (2006).
- Unsay, J., Cosentino, K., Subburaj, Y., Garcia-Saez, A. Cardiolipin effects on membrane structure and dynamics. Langmuir. 29, 15878-15887 (2013).
- Domènech, &. #. 2. 1. 0. ;., Sanz, F., Montero, M. T., Hernández-Borrell, J. Thermodynamic and structural study of the main phospholipid components comprising the mitochondrial inner membrane. BBA-Biomembranes. 1758, 213-221 (2006).
- Domènech, &. #. 2. 1. 0. ;., Morros, A., Cabañas, M. E., Teresa Montero, M., Hernández-Borrell, J. Supported planar bilayers from hexagonal phases. BBA-Biomembranes. 1768, 100-106 (2007).
- Garcia-Saez, A. J., Chiantia, S., Schwille, P. Effect of line tension on the lateral organization of lipid membranes. J Biol Chem. 282, 33537-33544 (2007).
- Alessandrini, A., Seeger, H. M., Caramaschi, T., Facci, P. Dynamic Force Spectroscopy on Supported Lipid Bilayers: Effect of Temperature and Sample Preparation. Biophys. J. 103, 38-47 (2012).
- Butt, H. -. J., Franz, V. Rupture of molecular thin films observed in atomic force microscopy I. Theory. Physical Review E. 66, 031601 (2002).
- Garcia-Manyes, S., Oncins, G., Sanz, F. Effect of Temperature on the Nanomechanics of Lipid Bilayers Studied by Force Spectroscopy. Biophys. J. 89, 4261-4274 (2005).
- Chiantia, S., Kahya, N., Schwille, P. Raft domain reorganization driven by short- and long-chain ceramide: a combined AFM and FCS study. Langmuir. 23, 7659-7665 (2007).
- Canale, C., Jacono, M., Diaspro, A., Dante, S. Force spectroscopy as a tool to investigate the properties of supported lipid membranes. Microsc. Res. Techniq. 73, 965-972 (2010).
- García-Sáez, A. J., Chiantia, S., Salgado, J., Schwille, P. Pore Formation by a Bax-Derived Peptide: Effect on the Line Tension of the Membrane Probed by AFM. Biophys. J. 93, 103-112 (2007).
- Moreno Flores, S., Toca-Herrera, J. L. The new future of scanning probe microscopy: Combining atomic force microscopy with other surface-sensitive techniques, optical microscopy and fluorescence techniques. Nanoscale. 1, 40-49 (2009).
- Simons, K., Vaz, W. L. C. Model Systems, Lipid Rafts, and Cell Membranes1. Annu. Rev. Bioph. Biom. 33, 269-295 (2004).
- Pike, L. J. Rafts defined: a report on the Keystone symposium on lipid rafts and cell function. The Journal of Lipid Research. 47, 1597-1598 (2006).
- Kahya, N. Probing Lipid Mobility of Raft-exhibiting Model Membranes by Fluorescence Correlation Spectroscopy. J. Biol. Chem. 278, 28109-28115 (2003).
- Akbarzadeh, A., et al. Liposome: classification, preparation and applications. Nanoscale Research Letters. 8, 102 (2013).
- Butt, H. -. J., Jaschke, M. Calculation of thermal noise in atomic force microscopy. Nanotechnology. 6, 1-7 (1995).
- Chon, J. W. M., Mulvaney, P., Sader, J. E. Experimental validation of theoretical models for the frequency response of atomic force microscope cantilever beams immersed in fluids. Journal of Applied Physics. 87, 3973 (2000).
- Sader, J. E. Frequency response of cantilever beams immersed in viscous fluids with applications to the atomic force microscope. Journal of Applied Physics. 84, 64 (1998).
- Sader, J. E., Pacifico, J., Green, C. P., Mulvaney, P. General scaling law for stiffness measurement of small bodies with applications to the atomic force microscope. Journal of Applied Physics. 97, 12490310 (2005).
- Canale, C., Torre, B., Ricci, D., Braga, P. C. Recognizing and avoiding artifacts in atomic force microscopy imaging. Methods Mol Biol. 736, 31-43 (2011).
- Lee, M. -. T., Chen, F. -. Y., Huang, H. W. Energetics of Pore Formation Induced by Membrane Active Peptides. Biochemistry-US. 43, 3590-3599 (2004).
- Henriksen, J. R., Ipsen, J. H. Measurement of membrane elasticity by micro-pipette aspiration. The European physical journal. E, Soft matter. 14, 149-167 (2004).
- Nichols-Smith, S., Teh, S. -. Y., Kuhl, T. L. Thermodynamic and mechanical properties of model mitochondrial membranes. BBA-Biomembranes. 1663, 82-88 (2004).
- Tian, A., Johnson, C., Wang, W., Baumgart, T. Line Tension at Fluid Membrane Domain Boundaries Measured by Micropipette Aspiration. Phys. Rev. Lett. 98, (2007).
- Rigaud, J. -. L. Membrane proteins: functional and structural studies using reconstituted proteoliposomes and 2-D crystals. Brazilian Journal of Medical and Biological Research. 35, 753-766 (2002).

