Assessing Cortical Cerebral Microinfarcts on High Resolution MR Images
Summary
A high resolution ex vivo 7T MR imaging protocol is presented, to perform MR-guided histopathological validation of microvascular pathology in post-mortem human brain tissue. Furthermore, guidelines are provided for the assessment of cortical microinfarcts on in vivo 7T as well as 3T MR images.
Abstract
Cerebral microinfarcts are frequent findings in the post-mortem human brain, and are related to cognitive decline and dementia. Due to their small sizes it is challenging to study them on clinical MRI scans. It was recently demonstrated that cortical microinfarcts can be depicted with MRI scanners using high magnetic field strengths (7T). Based on this experience, a proportion of these lesions is also visible on lower resolution 3T MRI. These findings were corroborated with ex vivo imaging of post-mortem human brain tissue, accompanied by histopathological verification of possible cortical microinfarcts.
Here an ex vivo imaging protocol is presented, for the purpose of validating MR observed cerebral microvascular pathology with histological evaluation. Furthermore, guidelines are provided for the assessment of cortical microinfarcts on both in vivo 7T and 3T MR images. These guidelines provide researchers with a tool to rate cortical microinfarcts on in vivo images of larger patient samples, to further unravel their clinical relevance in cognitive decline and dementia, and establish these lesions as a novel biomarker of cerebral small vessel disease.
Introduction
The application of ultra-high field 7 Tesla (T) MRI in patient studies is rapidly progressing1. This paper introduces a representative application of 7T MRI in the context of cerebrovascular disease in the aging human brain. Cerebrovascular disease is a major cause of cognitive decline and dementia. This vascular contribution to dementia frequently involves the small vessels of the brain, such as arterioles, small veins, and capillaries. Hence, it is referred to as cerebral small vessel disease (SVD)2. Because the cerebral small vessels are too small to capture with conventional MRI, only the consequences of SVD – i.e., the resulting tissue injury – can be visualized. This includes white matter hyperintensities, cerebral microbleeds, and lacunar infarcts3.
Other important manifestations of SVD are cerebral microinfarcts (CMIs)4. Autopsy studies report high prevalence of CMIs in vascular dementia, and Alzheimer’s disease5. However, due to their small sizes (ranging from 50 µm to a few mm) they escape detection on conventional MRI4,5. 7T MRI provides high resolution images with improved signal-to-noise-ratio and contrast, which enables the detection of certain structures and lesions beyond the detection limit of conventional MRI. This technique was therefore applied to detect CMIs. To identify possible CMIs, many in vivo 7T MRI scans were previously screened for lesions with sizes <5 mm and imaging characteristics consistent with ischemic properties. Such lesions could reliably be identified in the cortex. These focal elongated lesions were hyperintense on 7T FLAIR (0.8 mm isotropic voxels), restricted to the cortex and seemed to extend from the cortical surface, hyperintense on T2 (0.7 mm isotropic voxels), and hypointense on T1 (1.0 mm isotropic voxels). It was confirmed that these lesions were cortical CMIs using an MR-guided histopathology approach in post-mortem human brain tissue6,7.
Here, the ex vivo MRI protocol is presented that was used in previous studies for the histopathological validation of cortical CMIs. Secondly, guidelines are provided for the assessment of cortical CMIs on in vivo 7T MRI. Finally, the assessment of cortical CMIs on 7T has been translated to more widely available 3T MRI, and guidelines are provided how to identify cortical CMIs on 3T MRI.
Protocol
The use of the autopsy samples and in vivo MR images for this protocol was in accordance with local regulations and approved by the local institutional review board of the University Medical Center Utrecht (UMCU).
1. MR-guided Histopathological Validation of Cortical Microinfarcts
- Ex vivo MRI
- When handling brain tissue, always wear gloves and suitable protective clothing.
- Based on the research question, select appropriate, preferably 10-mm thick, formalin-fixed brain slabs. The brain slabs for this paper were derived from the neuropathology department of the UMCU, and VU University Medical Centre (VUMC), based on known Alzheimer pathology.
- Formalin-fix whole brains for at least 3-4 weeks by immersion in 10% formalin, prior to cutting. Cut the brains into coronal slabs, containing both hemispheres.
- For post-mortem scanning, select for example three brain slabs per brain, taken from the frontal, temporo-parietal, and occipital regions of the brain. The current protocol is optimized for the use of three coronal brain slabs, containing both hemispheres, in one scan session.
- Take photographs of the brain slabs at both sides (dorsal and caudal), and take careful notes (or make sketches) of the orientation of the slabs in the container and in the scanner, for later co-localization of histology with MRI.
- Fill a purpose-built container (Figure 1) – in this case one that fits within the MR head coil – with 10% fresh formalin at RT. If MRI signal from the fluid is undesired, use a perfluoropolyether (PFPE) lubricant with a suitable density instead of formalin (such as Fomblin or Galden PFPE). Ensure to use a flow cabinet when handling formalin.
- When placing the brain slabs in the container, make sure to avoid air bubbles. Remove the majority of air bubbles by gently shaking the tissue, either by hand, or using a shaker or ultrasound bath.
- Make sure the slabs cannot move within the container and limit the amount of needed fluid, by using a smaller container to keep the slabs in place (Figure 1).
- Cover the container with plastic or parafilm, to prevent evaporation and to protect the MR (head) coil from potential contamination (Figure 2).
- Use a whole-body 7T MRI scanner with an appropriate coil. In this protocol a dual transmit and 32-channel receive head coil is used.
- Place the container in the head coil, wrapped in a towel or surgical underpad, to prevent potential spilling of fluid. Make sure the container cannot move, and that the slabs remain in horizontal position (Figure 2).
- Run a survey scan that can be used for the planning of the high resolution scans, correct B0 inhomogeneity by using an appropriate shimming tool, and calibrate the RF power to obtain the correct flip angles (the few slabs require less power compared to in vivo scanning of a whole head) according to manufacturer’s protocol.
- Plan the high resolution acquisitions on the survey scan, to ensure the brain slabs are fully included in the field-of-view. Scan the brain slabs O/N with the high resolution acquisitions shown in Table 1, which are optimized for ex vivo imaging. The acquisition protocol presented here includes a 3D FLAIR, T2, and T1 weighted image with an isotropic resolution of 0.4 mm, and a T2* weighted image with an isotropic resolution of 0.18 mm.
- Identify automatic software processes that may interrupt scanning, such as automated up-dates that run O/N, or warnings for peripheral nerve stimulation, and make sure the scanner procedures will not be interrupted by these.
- Monitor the scanner O/N for possible confirmation pop-ups that may interrupt scanning, by using for example a VPN connection.
- Return the following morning (after a total scan time of approximately 12 hr in the current protocol). Store the brain slabs in formalin, clean up.
- Save the images to an external hard disk.

Figure 1. Preparation of formalin-fixed brain slabs for post-mortem scanning at 7T MRI. A purpose-built Perspex container is filled with either 10% formalin or a perfluoropolyether (PFPE) lubricant if MRI signal from the fluid is undesired. Three 10-mm thick formalin-fixed coronal brain slabs are placed in the container. A smaller container is used to keep the slabs in place. Tape the second container to the first one, to prevent movement.
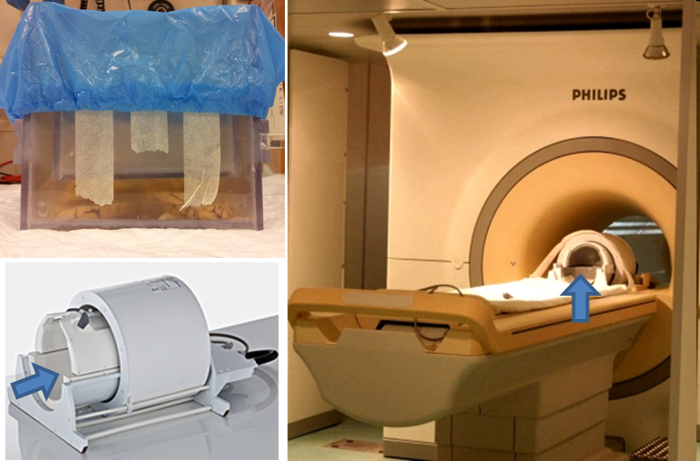
Figure 2. Placement of purpose-built container in 7T head coil. Cover the container with plastic or parafilm to prevent evaporation of the formalin. Place the container, enclosed in a towel or surgical underpad, in the head coil of a 7T MR scanner. Make sure the container cannot move, and that the slabs remain in horizontal position.
- Histopathology
- Identify possible cortical CMIs – or other lesions of interest – on the acquired images. These lesions are the targets for histological analysis. Watch out for artifacts, such as post-mortem tissue damage (which sometimes appear at the surface of brain slabs due to cuts) or long-term formalin storage artifacts (e.g., coarse MRI hypointensities representing neuropil changes8).
Note: Different histopathological subtypes of cortical CMIs have different MR characteristics. For further details on CMI subtypes, the reader is referred to a recent ex vivo study7. - After identifying possible cortical CMIs on the MR images, sample the region of interest for histopathological validation. Make sure to cut out a region, containing anatomical landmarks, for later matching of the MRI with histopathology. Perform standard histopathology, as followed (but other approaches might also apply).
- Cut out a region of approximately 30 x 20 x 5 mm3 containing a possible cortical CMI.
- To obtain accurate sampling, estimate the lesion location by the slice thickness of the MR images, and tissue architecture. Manually cut the tissue slightly above the estimated lesion location to limit the amount of serial sectioning (after paraffin embedding) that is needed for targeting the lesion.
- Make sure the sampled tissue fits a tissue cassette. Place the surface to be cut face-down in the cassette.
- Keep all tissue cassettes in 10% formalin, until processing of the tissue.
- Process the tissue for paraffin embedding. This usually involves an automated procedure of dehydrating the tissue, through a series of graded alcohol (e.g., 70% to 95% to 100%) baths, and clearing of the tissue in xylene.
- Embed the tissue in paraffin wax blocks. Ensure the surface to be cut faces up after embedding.
- Cut 4-6 µm serial sections with a microtome, until the targeted lesion is retrieved.
- Float the sections on the surface of a 37 °C water bath. Mount the sections on glass slides. Place the slides on a warming block to bond the tissue to the glass. Store slides O/N at RT.
- Perform an appropriate staining (e.g., H&E staining) on the first sections, keep adjacent blank sections for further use (e.g., immunohistochemistry).
- Coverslip the H&E stained sections, using a drop of mounting medium of choice. Gently lower the slip, avoiding air bubbles.
- Study the sections using a light microscope, at an appropriate magnification. Compare sections to the previously obtained MR images.
- Identify possible cortical CMIs – or other lesions of interest – on the acquired images. These lesions are the targets for histological analysis. Watch out for artifacts, such as post-mortem tissue damage (which sometimes appear at the surface of brain slabs due to cuts) or long-term formalin storage artifacts (e.g., coarse MRI hypointensities representing neuropil changes8).
2. Assessing Cortical Microinfarcts on In Vivo 7T MRI
- Perform 7T MRI in the patient population of your interest, using the in vivo MRI protocol (which includes at least a 3D FLAIR) as described in6.
- Assess cortical CMIs on the in vivo 7T MR images as detailed in the steps below, using the following 7T rating criteria for CMIs: cortical CMIs are hyperintense on FLAIR (with or without a hypointense center), hyperintense on T2, hypointense on T1, detectable on at least two views of the brain (e.g., sagittal and transversal), restricted to the cortex, distinct from perivascular spaces, with a greatest dimension ≤4 mm6,7.
- Use an interface with three image viewers, to simultaneously view FLAIR, T1, and T2 images, e.g., MeVisLab (Figure 3). This platform allows to incorporate multiple viewers and to place markers on possible lesion locations.
- First assess one hemisphere on FLAIR in sagittal view. Screen the whole cortex for hyperintense lesions. Any hyperintens lesion ≤4 mm is a possible CMI. Place markers by clicking on each possible CMI.
- Repeat for the other hemisphere.
- Verify all marked locations on T1 and T2. Discard a location if it is not hypointense on T1 or hyperintense on T2.
- Assess transversal view, on FLAIR, T1, and T2. Discard a location if it is not visible. Check the coronal view in case of doubt.
- Watch out for MRI artifacts and anatomical variations (especially sulcal edges).
- Save markers.
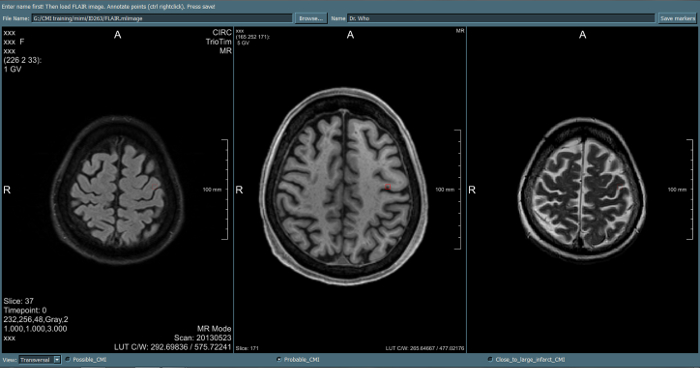
Figure 3. Example image viewing platform for the assessment of cortical microinfarcts. An interface is used, integrated in MeVisLab. This program allows to incorporate multiple viewers simultaneously, to switch easily between sagittal / transversal / coronal orientation, and to place and save markers on possible lesion locations. (Different markers can be chosen for different types of lesions).
3. Assessing Cortical Microinfarcts on In Vivo 3T MRI
- Acquire 3T MR images of the patient population of your interest. Existing data can also be used as long as the MR imaging protocol contained at least a 3D T1, and a FLAIR and T2.
- Assess cortical CMIs on the in vivo 3T MR images as detailed in the steps below, using the following 3T rating criteria for CMIs: cortical CMIs are hypointense on T1 (isointense with CSF), detectable on at least two views of the brain (e.g. sagittal and transversal), restricted to the cortex, distinct from perivascular spaces, with a greatest dimension ≤4 mm.
- Explore the location of a hypointense cortical lesion found on T1 on FLAIR and T2 weighted images. Rate the lesion as a probable cortical CMI if the location is hyperintense or isointense (with the grey matter) on FLAIR and T2. Discard the lesion if at the same location a hypointense signal is found on T2, indicating the T1 hypointense lesion is either due to a hemorrhagic lesion, a vessel, or an artifact. In case of doubt, check the location on a T2* weighted image9.
- Use the same interface as described above.
- First assess one hemisphere on T1 in sagittal view. Screen the whole cortex for focal hypointense lesions. Any hypointense lesion ≤4 mm is a possible CMI. Place markers by clicking on each possible CMI.
- Repeat for the other hemisphere.
- Assess transversal T1, and simultaneously verify all marked locations on transversal FLAIR and T2. Regard the location as a probable CMI if it is hyperintense or isointense on FLAIR and T2. Discard a location if it seems to be an artifact or anatomical variation. Discard a location if it is hypointense on T2.
- Watch out for artifacts that look like CMIs on T1 weighted images, especially ringing artifacts at the ‘edges’ of the brain which will appear at several adjacent gyri, watch out for edges of sulci, watch out for large vessels in the temporal lobes (at the poles). Finally, it is recommended to discard possible cortical CMIs in tissue in close proximity to a larger cortical infarct.
- Save markers.
Representative Results
An impression of the high resolution and high image quality of an ex vivo sequence acquired at 7T is provided here (Figure 4). This is a 3D T2* weighted ex vivo scan, with an isotropic resolution of 0.18 mm. Tissue was derived from an 84-year old demented female with pathologically proven Alzheimer’s disease and severe cerebral amyloid angiopathy (CAA). The detail of the image allows the identification of cortical microvascular pathology. T2* is susceptible for iron, as well as air. This tissue contains a high burden of microvascular pathology within the cortex. The hypointensities in the sulci of these slabs are the results of air bubbles, which can interfere with rating cortical microvascular pathology. For the identification of cortical CMIs, a T2 weighted sequence is required.
Figure 5 represents a cortical CMI identified on ex vivo images at 7T. This cortical CMI was found in the post-mortem brain tissue of an 86-year old female with moderate Alzheimer pathology (Braak & Braak stage IV). The corresponding H&E section verified that this lesion is a chronic gliotic CMI with cavitation7.
Figure 6 is a representative probable cortical microinfarct, detected on in vivo 7T MRI.
Figure 7 is a representative probable cortical microinfarct, detected on in vivo 3T MRI.
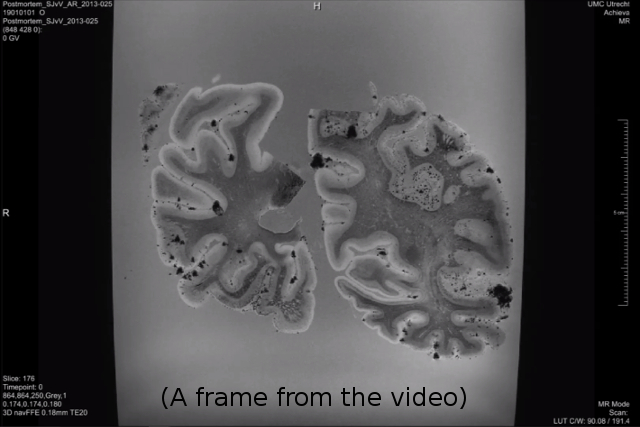
Figure 4. Representative post-mortem images acquired at 7T. Please click here to view this video.
This is a 3D movie of a 0.18 mm isotropic T2* weighted image of a case with severe amyloid angiopathy. These brain slabs have been generously provided by Dr. Annemieke Rozemuller, VUMC, Amsterdam.
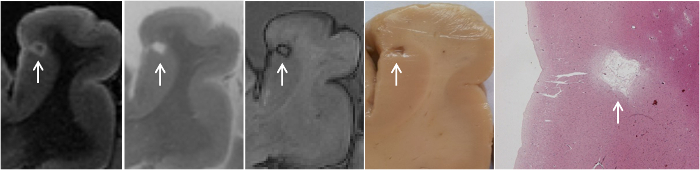
Figure 5. MR-guided histopathology of cortical microinfarct.
Depicted are a FLAIR, T2, T1, wet tissue, and H&E staining, showing a cortical chronic gliotic microinfarct with cavitation. This figure has been modified from7. Please click here to view a larger version of this figure.
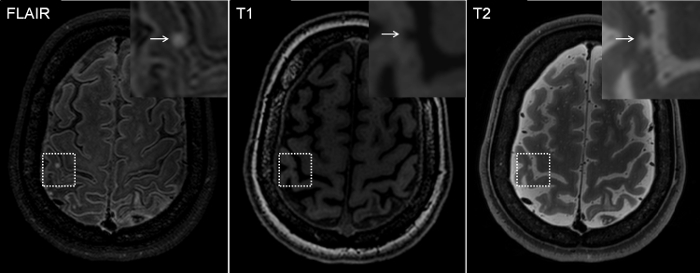
Figure 6. Representative probable cortical microinfarct on 7T MRI.
A cortical microinfarct on 7T is hyperintense on FLAIR and T2, and hypointense on T1. This case is a 45-year old female who suffered from a lobar intracerebral hemorrhage. MR images are a courtesy of Dr. Karin Klijn, UMCU, Utrecht. Please click here to view a larger version of this figure.
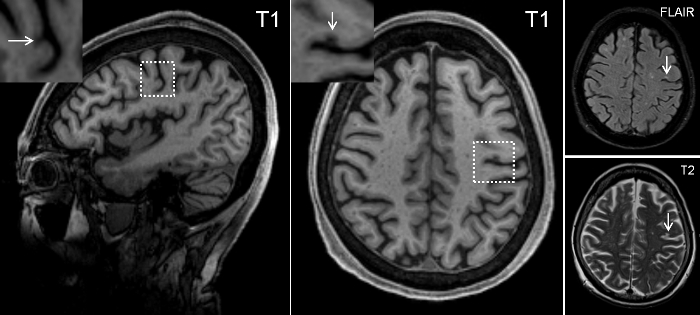
Figure 7. Representative probable cortical microinfarct on 3T MRI.
A cortical microinfarct on 3T can best be identified as a hypointense lesion on a 3D T1. The corresponding location on FLAIR and T2 should be hyperintense (in this case) or isointense. This case is a 76-year old female with a clinical diagnosis of Alzheimer’s disease. MR images are a courtesy of Dr. Christopher Chen, NUS, Singapore. Please click here to view a larger version of this figure.
| TI | TR / TE | Flip / refocusing angle | Acquired resolution | Matrix size | Slices | Averages | Scan duration | |
| (ms) | (ms) | (°) | (µm3) | (h:min:sec) | ||||
| T2 | – | 3,500 / 164 | 90 / 40 | 400x400x400 | 500×280 | 100 | 4 | 1:52:03 |
| FLAIR | 1,600# | 8,000 / 164 | 90 / 40 | 400x400x400 | 500×280 | 100 | 4 | 4:16:08 |
| T1 | 280 | 7.7 / 3.5 | 6 / – | 400x400x400 | 348×348 | 80 | 3 | 0:55:38 |
| T2* | – | 75 / 20 | 25 / – | 180x180x180 | 832×834 | 278 | 1 | 4:59:31 |
| No sensitivity encoding (SENSE) acceleration was applied. # The TI was determined based on 10% formalin. | ||||||||
Table 1. Post-mortem scan parameters.
Discussion
CMIs have attracted increasing attention over the last few years. A growing body of evidence derived from autopsy studies has identified CMIs as important contributors to age-related cognitive decline and dementia4,5. CMIs are now detectable on 7T and also 3T MRI. Optimization and standardization of assessment protocols for these lesions will support rapid implementation of robust and valid CMI detection in cohort studies throughout the world. This will enable a widespread evaluation of the clinical relevance of CMIs in the context of aging, cerebrovascular disease, and dementia in future clinical studies, both cross-sectional as well as longitudinal9,10.
The described method in this work is actually a ´meta-method’ in the sense that it can be regarded as a procedure to develop (and successively improve) methods for in vivo detection of CMIs, which could thus far only be assessed post-mortem by a neuropathologist. The most critical step in developing improved in vivo CMI detection methods by new MRI protocols and image processing techniques is to validate it with histology, which is currently the gold standard. An important limitation of the in vivo detection of CMIs compared to histology is resolution. However, despite the fact that in vivo MRI will not be able to detect the smaller CMIs, it does provide whole-brain coverage, which might prove to be as effective as looking for microscopic CMIs on just a few histological sections.
An important step to establish the in vivo guidance for CMI rating was the histopathological validation of CMIs, guided by ex vivo high resolution MRI of post-mortem human brain tissue. The ex vivo scan protocol presented here is optimized for the validation of cortical microvascular pathology, but can be applied in a broader research context, to support in vivo rating of other novel brain imaging markers. Scanning post-mortem human brain tissue has its challenges, which should be acknowledged here. Prolonged storage of formalin-fixed tissue can cause artifacts8. Other challenges are MRI artifacts caused by air bubbles, because air bubbles can interfere with the MR signal, especially at fluid-tissue boundaries. Therefore, the removal of the air bubbles is an important step. Air easily accumulates in empty blood vessels, between gyri, and between slabs. To overcome the latter, one would ideally scan a whole block of un-cut tissue. However, part of standardized autopsy procedures is cutting the formalin-fixed tissue in 10-mm thick slabs. Scanning post-mortem tissue requires extra attention to ensure sufficient B0 shimming and correct RF power optimization (step 1.1.10). This may need specific attention from a local MRI physicist. These steps are dependent on the vendor of the 7T MRI scanner and may be optimized according to the desires of the individual research group. During scanning, slabs are either submerged in 10% formalin or in a PFPE lubricant, which is a proton-free fluid without MR signal. The advantage of using a proton-free fluid is that it minimizes the required field-of-view, it enables better B0 shimming, and it doesn’t penetrate the tissue as it is highly hydrophobic. Disadvantages are that it is expensive, and can be impractical in use (being an oily substance). Formalin is much easier in use, is cheap, and doesn’t interfere with the tissue when the tissue is already formalin-fixed. The disadvantage of formalin is that it may cause RF inhomogeneity at 7T, when scanning large volumes (e.g., whole brains), and that the substance is toxic. Another frequently applied embedding substance for ex vivo MRI is agar gel. Agar is ideal for scanning single slabs or individual pathological specimens, and the major advantage is the reduction of potential movement. Also, it allows the placement of fiducials to use as artificial landmarks11.
In the current in vivo imaging example of cerebral SVD at 7T, the following sequences were used: a 3D FLAIR (0.8 mm isotropic voxels), 3D T2 (0.7 mm isotropic voxels), 3D T1 (1.0 mm isotropic voxels), and T2* (0.5×0.5×0.7 mm3 voxels). The applied 7T FLAIR is heavily T2 weighted, and therefore highly suitable for visualizing minute ischemic lesions12. The T2* sequence has been included for the detection of cerebral microbleeds13, but may also be used to check CMI locations in the absence of a T2. It should be noted that the current 7T FLAIR sequence does not allow a reliable assessment of the temporal lobes, due to a low signal-to-noise ratio in these areas. Other research groups inevitably want to use their own FLAIR and T2 weighted protocols, but this may lead to different sensitivity regarding CMI detection.
The translation of the 7T rating criteria to the assessment of cortical CMIs on in vivo 3T MR images is important to allow the investigation of CMIs in larger patient samples. However, there are a few challenges to take into account. First, ensure to include at least one 3D sequence in the 3T MRI scan protocol, which might not be the standard procedure in most clinically applied protocols. Secondly, a FLAIR sequence at 3T is usually less heavily T2 weighted than on 7T. That is the reason it is recommended to assess cortical CMIs on 3D T1 weighted images, with confirmation on FLAIR and T2 (if available), and in case of doubt T2*. For the purpose of the current CMI rating guidelines described in this protocol, a high resolution 3D T1 (1.0 mm isotropic voxels), 2D FLAIR (1.0×1.0x3.0 mm3 voxels), and 2D T2 (1.0×1.0x3.0 mm3 voxels)9 were used. These images were acquired on a 3T MRI system, with a 32-channel receive head coil.
A few limitations of in vivo CMI rating are in place. Assessing CMIs visually on in vivo MR images remains challenging and is clearly rater dependent. It requires training, but even with the proper experience these small lesions easily escape detection by the human eye. Furthermore, rating CMIs is rather labor intensive and time-consuming, especially when applied to larger samples. Therefore, it is of importance to develop (semi-)automatic detection methods for the identification of CMIs that aid the visual rating14. It should be acknowledged that 3T MRI only detects the larger CMIs. The same applies for 7T, albeit to a lesser extent. Nevertheless, the CMIs that are detected on MRI do have important and specific clinical correlates9.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
The research leading to these results has received funding from the European Research Council under the European Union’s Seventh Framework Programme [FP7/2007-2013] / ERC grant agreement [337333]. The research of SvV and GJB is supported by a VIDI grant [91711384] from ZonMw, the Netherlands Organization for Health Research and Development.
Materials
| Fomblin / Galden PFPE | Solvay Solexis, Bollate, Italy | ||
| 7T MR system | Philips Healthcare, Cleveland, OH, USA | ||
| 32-channel receive head coil | Nova Medical, Wilmington, MA, USA | ||
| MeVisLab | MeVis Medical Solutions AG, Bremen, Germany |
Riferimenti
- Vander Kolk, A. G., Hendrikse, J., Zwanenburg, J. J., Visser, F., Luijten, P. R. Clinical applications of 7 T MRI in the brain. Eur J Radiol. 82 (5), 708-718 (2013).
- Pantoni, L. Cerebral small vessel disease: from pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol. 9, 689-701 (2010).
- Gouw, A. A., et al. Heterogeneity of small vessel disease: a systematic review of MRI and histopathology correlations. J Neurol Neurosurg Psychiatry. 82 (2), 126-135 (2011).
- Smith, E. E., Schneider, J. A., Wardlaw, J. M., Greenberg, S. M. Cerebral microinfarcts: the invisible lesions. Lancet Neurol. 11, 272-282 (2012).
- Brundel, M., de Bresser, J., van Dillen, J. J., Kappelle, L. J., Biessels, G. J. Cerebral microinfarcts: a systematic review of neuropathological studies. J. Cereb. Blood Flow Metab. 32 (3), 425-436 (2012).
- Van Veluw, S. J., et al. In vivo detection of cerebral cortical microinfarcts with high-resolution 7T MRI. J Cereb Blood Flow Metab. 33 (3), 322-329 (2013).
- Van Veluw, S. J., et al. The spectrum of MR detectable cortical microinfarcts; a classification study with 7 tesla post-mortem MRI and histopathology. J Cereb Blood Floow Metab. Jan. 21, 10-1038 (2015).
- Van Duijn, S., et al. MRI artifacts in human brain tissue after prolonged formalin storage. Magn Reson Med. 65 (6), 1750-1758 (2011).
- Van Veluw, S. J., et al. Cortical microinfarcts on 3T MRI: clinical correlates in memory-clinic patients. Alzheimers Dement. 5, 10-1016 (2015).
- Van Dalen, J. W., et al. Cortical microinfarcts detected in vivo on 3 Tesla MRI: clinical and radiological correlates. Stroke. 46 (1), 255-257 (2015).
- Vander Kolk, A. G., et al. Imaging the intracranial atherosclerotic vessel wall using 7T MRI: initial comparison with histopathology. AJNR Am J Neuroradiol. 36 (4), 694-701 (2015).
- Visser, F., Zwanenburg, J. J., Hoogduin, J. M., Luijten, P. R. High-resolution magnetization-prepared 3D-FLAIR imaging at 7.0 Tesla. Magn Reson Med. 64 (1), 194-202 (2010).
- Brundel, M., et al. High prevalence of cerebral microbleeds at 7Tesla MRI in patients with early Alzheimer’s disease. J Alzheimers Dis. 31 (2), 259-263 (2012).
- Kuijf, H. J., et al. Detecting cortical cerebral microinfarcts in 7.0 T MR images. IEEE International Symposium on Biomedical Imaging. , 982-985 (2013).

