Microscopy Based Methods for the Assessment of Epithelial Cell Migration During In Vitro Wound Healing
Summary
This manuscript describes how incision-like lesions made on cultured epithelial cell monolayers conveniently model wound healing in vitro, allowing for imaging by confocal or laser scanning microscopy, and which can provide high-quality quantitative and qualitative data for studying both cell behavior and the mechanisms involved in migration.
Abstract
Cell migration is a mandatory aspect for wound healing. Creating artificial wounds on research animal models often results in costly and complicated experimental procedures, while potentially lacking in precision. In vitro culture of epithelial cell lines provides a suitable platform for researching the cell migratory behavior in wound healing and the impact of treatments on these cells. The physiology of epithelial cells is often studied in non-confluent conditions; however, this approach may not resemble natural wound healing conditions. Disrupting the epithelium integrity by mechanical means generates a realistic model, but may impede the application of molecular techniques. Consequently, microscopy based techniques are optimal for studying epithelial cell migration in vitro. Here we detail two specific methods, the artificial wound scratch assay and the artificial migration front assay, that can obtain quantitative and qualitative data, respectively, on the migratory performance of epithelial cells.
Introduction
Cell migration is required for wound healing, as it is responsible for the final closure of the epithelial gap and restoration of the disrupted surface1. Performing artificial wounds in animal models allows for the replication of this complex process in near physiological conditions2. However, this approach often results in costly and complicated experimental procedures, that potentially lack precision for the study of distinct processes, due to the intricate nature of the wound-healing process.
In vitro culture of epithelial cell lines provides a helpful alternative to animal models for researching the role that these cells play in wound healing and the effects of treatment on cell migratory behavior. The physiology of epithelial cells is often studied by molecular techniques using non-confluent cultures3,4,5,6; however, the disruption of the epithelium integrity is usually achieved by fine mechanical incisions. In cell culture, this implies that negligible number of cells may be exposed to the wound gap, and they represent a too small sample for molecular biology techniques. However, these lesions can be studied at the microscopic scale, taking advantage of the innate migratory properties of some epithelial cell lines, such as the Mink Lung Epithelial cell (Mv1Lu) or the spontaneously immortalized human keratinocyte (HaCaT) cell lines.
Here we described a method for microscopy that is suitable to obtain quantitative data on the migration of epithelial cells in the context of wound healing3,4,7,8. Moreover, we present additional methods that are helpful to study qualitatively molecular and morphological changes occurring on epithelial monolayers during migration. Overall, these methods provide a framework to study both the dynamics and morphological changes involved with epithelial cell behavior and response to treatments during wound healing.
Protocol
1. Artificial Wound Scratch Assay for Quantitative Studies
- Cell monolayer preparation
- Working under sterile conditions, seed and grow Mv1Lu or HaCaT epithelial cells in culture flasks using serum-supplemented medium. Refresh medium once every 24-48 h. After cells reach 80% confluence, detach cells using an appropriate method, i.e., trypsinization9.
NOTE: Mv1Lu and HaCaT epithelial cell lines are cultured using EMEM and DEMEM culture medium, respectively, supplemented with 2 mM L-Glutamine, and incubated at 37 °C with a controlled atmosphere of 5% CO2. Every cell line must be assayed thoroughly to determine cell concentration and timing required to reach full confluency. Typically for Mv1Lu or HaCaT cells, 2-4 x 104 or 4-6 x 104 cells/well, respectively, are seeded in a 175 cm2 culture flask, and at 80% confluence yields up to 35 x 106 Mv1Lu or 1 x 107 HaCaT cells. - In either a 12-well or 24-well culture plate, seed in each well 2 mL of cells. Refresh medium once every 24-48 h. Ensure cell numbers are high enough to reach 100% confluence after 2 or 3 days.
- After the cells reach confluence, remove the serum-supplemented medium and wash twice with fresh serum-free medium. Keep cells in serum-free medium for 24 h before the scratching.
NOTE: Mv1Lu or HaCaT cells do not have special substrate requirements; however, for cell lines susceptible to detaching spontaneously in serum-free conditions, pre-coating of the culture plate surface using a poly-L-lysine solution should be considered10.
- Working under sterile conditions, seed and grow Mv1Lu or HaCaT epithelial cells in culture flasks using serum-supplemented medium. Refresh medium once every 24-48 h. After cells reach 80% confluence, detach cells using an appropriate method, i.e., trypsinization9.
- Monolayer scratching
- Working in a sterile environment, scratch the culture monolayer by firmly dragging the narrow end of a 20 µL or 200 µL sterile micropipette tip, depending on the desired scratch width. Keep the tip perpendicular to the culture surface to maximize gap homogeneity.
- Place the culture plates over a dark surface to clearly monitor the cell monolayer. If needed, perform two perpendicular scratches (cross-shaped) on each well to study up to 4 migration areas.
- Wash detached cells by gently removing culture medium and gently adding fresh serum-free medium. Repeat two times, or until most unattached cells have been removed.
- Working in a sterile environment, scratch the culture monolayer by firmly dragging the narrow end of a 20 µL or 200 µL sterile micropipette tip, depending on the desired scratch width. Keep the tip perpendicular to the culture surface to maximize gap homogeneity.
- Experimental procedure and data collection
- Take up to 4 pre-treatment reference images centered on the scratch gap from each well using an inverted phase-contrast microscope incorporating a CCD camera. Select interest areas by aligning the edge of microscope field to the adjacent intersection of scratches.
NOTE: Typically, images are acquired using a 10x magnification and a 1,280 x 1,024 pixel image size. Selecting interest areas as described above helps for easy localization and validation of up to 4 measurements per well. - Once all the images are acquired, designate treatment wells; exchange the medium in the wells with fresh medium that contains selected treatments.
NOTE: Alternatively, for chemicals or compounds that do not disturb culture medium homogeneity, treatments can be inoculated directly into the culture medium. - Incubate the scratched plate (37 °C with a controlled atmosphere containing 5% CO2) until the conditions under observation reach 90% scratch gap closure. Avoid full closure.
NOTE: Total gap closure nullifies post-treatment picture collection as reference areas are not detectable and the time reference for gap closing cannot be established. In the case of Mv1Lu or HaCaT cells, 16-19 h are required to reach 90% confluence. - Stop cell migration by fixing the cells; gently replace the experimental culture medium with 1 mL of 4% formalin in PBS. Incubate for 15 min at RT.
- Wash the cells to remove excess formalin; gently replace the solution in the wells with fresh PBS. Repeat at least two times.
NOTE: At this stage, after sealing, plates can be stored at 4 °C indefinitely. - Take post-treatment reference images of each well from the original reference areas captured after scratching. Use the same equipment (inverted optical phase-contrast microscope incorporating a CCD camera) and matching picture settings (magnification and digital resolution/density).
NOTE: For cells that migrate individually and fill the gap independently of the formation of a coherent front (e.g., MDA-MB-231 breast cancer human cells), the time-course images may allow the calculation of individual cell migration speeds.
- Take up to 4 pre-treatment reference images centered on the scratch gap from each well using an inverted phase-contrast microscope incorporating a CCD camera. Select interest areas by aligning the edge of microscope field to the adjacent intersection of scratches.
- Image analysis and quantification of migration
- Using image processing software (e.g., ImageJ), define scratch gap limits and determine the gap surface for up to four measurements in pre-treatment pictures. Record the data as "pre-treatment gap surface" (PREGAP). Repeat the same procedure for post-treatment pictures. Record the data as "post-treatment gap surface" (POSTGAP).
- Open the recorded image using ImageJ. In the "image" menu, set image type to 8 bit. In the "process" menu, go to "filters" submenu and apply "variance" filtering.
- In the "image" menu, enter "adjust" submenu and set threshold to black and white (B&W), make sure "Dark background" is not selected. In the "process" menu, go to "binary" submenu, and select "fill holes".
- In the "analyze" menu, select "set measurements" and activate "area". Then draw the measurable area following the migrating edge contour thus delimiting the gap.
- In the "analyze" menu, select "analyze particles" and record "total area" values for the drawn area surface.
- Introduce PREGAP and POSTGAP total area values in a spreadsheet to quantify absolute migration for each set of individual samples as the difference of gap surface measurements: PREGAP − POSTGAP [Arbitrary units]. Additionally, normalize absolute migration data of each condition to control samples: (SAMPLE / CONTROL) * 100 [%].
NOTE: For cells that migrate individually and fill the gap independently of the formation of a coherent front, (e.g., MDA-MB-231 breast cancer human cells), the absolute migration can be calculated by counting cells invading a central strip either in the control or the treated samples. - Plot quantification outputs (see Figure 1). Perform statistical analysis if necessary. Consider Student's t-test when comparing two conditions or Analysis of Variance test for greater number of conditions.
- Using image processing software (e.g., ImageJ), define scratch gap limits and determine the gap surface for up to four measurements in pre-treatment pictures. Record the data as "pre-treatment gap surface" (PREGAP). Repeat the same procedure for post-treatment pictures. Record the data as "post-treatment gap surface" (POSTGAP).
2. Artificial Migration Front Assay for Topographical Studies
- Cell monolayer preparation
- Working in sterile conditions, place one layer of sterilized round coverslips in empty culture plates until the plate's surface is completely covered.
NOTE: Up to 12 and 33 coverslips fit on 5 cm and 10 cm diameter plates, respectively. - On the culture plate, gently seed an adequate volume of cells at a concentration that allows 100% confluence after 2 or 3 days. Make sure no coverslip overlaps neighboring coverslips and prevent coverslips from floating by applying gentle pressure with a sterile micropipette tip. Refresh the medium every 24-48 h.
NOTE: Every cell line must be assayed thoroughly to determine cell concentration and timing required to reach full confluency. Typically for Mv1Lu or HaCaT cells, 2-3 x 106 or 2.5-4 x 106 cells/10 cm-diameter plate, respectively, are seeded. For smaller plates, cell numbers must be scaled down. - After the cells reach confluence, remove the serum-supplemented medium and replace with fresh serum-free medium. Keep the cells in serum-free medium for 24 h before the artificial wound is performed.
NOTE: Mv1Lu or HaCaT cells do not have special substrate requirements; however, for cell lines susceptible of detaching spontaneously in serum-free conditions, pre-coating of the culture surface using a poly-L-lysine solution should be considered10.
- Working in sterile conditions, place one layer of sterilized round coverslips in empty culture plates until the plate's surface is completely covered.
- Monolayer wounding
- Using sterile tweezers, gently move a coverslip to a clean 10 cm plate containing fresh serum-free medium. Avoid damaging the monolayer by holding the coverslip at the periphery with tweezers. Avoid excessive pressure that could result in coverslip breakage.
- Create artificial wounds by dragging a sterilized razor blade in a transverse line over the center of the coverslips. Drag 3-4 mm back-and-forth, in order to completely remove the central monolayer strip. Make sure no cell debris remains attached to the wounded edges.
NOTE: On a 12-mm diameter round coverslip, the incision is made at the mid of the coverslip. Make sure to leave a streak of cells opposite the main front large enough (at least 2 mm wide) to avoid tilting of the coverslip in the following steps. - Transfer wounded coverslips with cells facing up, to a clean 6-well plate containing at least 2 mL fresh serum-free medium. Fit up to 4 wounded coverslips/well.
- Gently wash two times using fresh serum-free medium to remove detached cells.
- Experimental procedure and sampling
- Gently exchange the medium in the wells with fresh medium that contains selected treatments.
NOTE: Alternatively, for chemicals or compounds that do not disturb culture medium homogeneity, treatments can be inoculated directly into the culture medium. Proceed with caution to avoid any potential cell detachment. - Keep the plate inside a cell incubator (37 °C, 5% CO2) for the desired experimental timing.
- Stop cell migration by fixing the cells; gently replace the experimental culture medium with 1 mL of 4% formalin in PBS. Incubate for 15 min at RT.
NOTE: For time course experiments, remove the samples from the culture plate and fix them in a separate plate to avoid formalin fumes affecting the other, ongoing experimental conditions. - Wash the cells to remove excess formalin; gently replace the solution in the wells with fresh PBS. Repeat at least two times.
NOTE: At this stage, after sealing, plates can be stored at 4 °C indefinitely.
- Gently exchange the medium in the wells with fresh medium that contains selected treatments.
- Immunofluorescence (IF) and topological analysis
- Perform IF staining and imaging on individual coverslips as described elsewhere3,4,11.
- Permeabilize for 10 min by immersing coverslips in a 0.3% Triton X-100 solution in PBS.
- Block for 30 min using the following blocking solution: 0.3% Bovine Serum Albumin; 10% Fetal Bovine Serum; 5% skim milk; 0.3% Triton X-100 solution in PBS.
NOTE: Blocking solution without milk can be prepared in advance and stored at -20 °C. - Incubate for 1 h at room temperature inside a moist chamber, with primary antibody diluted in milk-free blocking solution. Place the coverslips upside-down in the 15 µL antibody solution to ensure proper distribution.
NOTE: The antibody working dilution is variable and will depend on the antibody in use. For example, the Rabbit anti-c-Jun antibody requires a 1/100 dilution. - Wash three times by dipping the coverslips in a in 0.1% Triton X-100 solution in PBS.
- Incubate the coverslips in secondary antibody diluted in milk-free blocking buffer for 30 min.
NOTE: The antibody working dilution is variable and will depend on the antibody in use. For example, the Goat-anti-Rabbit (488) requires a 1/400 dilution for optimal resolution. Other labeling reagents such as phalloidin and Hoechst-33258 can be added to the incubation buffer at this stage. - Wash three times by dipping the coverslips in 0.1% Triton X-100 solution in PBS.
- Mount the coverslips upside-down in a 10 µL mounting media drop (e.g., Vectashield) placed on a microscope slide. Leave the slides in the dark at room temperature overnight for the mounting medium hardening. Afterwards, store at 4 °C in the dark indefinitely.
- Obtain either epifluorescence or confocal microscopy images of structural changes occurring at the migrating front edge.
NOTE: Topological studies can be performed by tiling pictures from the migrating front to the inner monolayer. Semi-quantitative studies are possible by software based analysis on local fluorescence intensities.
- Perform IF staining and imaging on individual coverslips as described elsewhere3,4,11.
Representative Results
Artificial Wound Scratch Assay for Quantitative Studies: Assessing Epidermal Growth Factor (EGF) Promotion of Migration:
EGF is a well-known inducer of epithelial cells proliferation and migration, and thus a positive control for quantifying migration promotion. Mv1Lu and HaCaT cell monolayers were used in the wound scratch assays and pre-treatment pictures were obtained. After inoculation with 10 ng/mL EGF, cells were incubated for 19 h before fixation and post-treatment pictures. Proliferation was kept to a minimum by maintaining serum starvation conditions. Both pre- and post-treatment sets of pictures were analyzed using ImageJ and the central gap areas were determined. The experiment was run in duplicate, registering four measurements for each well. Quantification of the absolute migration was calculated as the difference of surfaces: (PREGAP − POSTGAP). There was a greater migratory potential in the stimulated Mv1Lu cells (Figure 1A) compared to the stimulated HaCaT cells (Figure 1B). The differences are explained by the cell sources, being mucosa epithelial cells and skin keratinocytes, respectively.
Artificial Migration Front Assay for Topographical Studies: Variations in Cytoskeletal Conformation and Spatial Expression of C-Jun:
Cell migration involves continuous and deep changes of the cytoskeleton structure in order to sustain cell displacement. HaCaT cell monolayers were used in the migration front assays under control and stimulation conditions. Treatment with EGF potentiates the cytoskeletal dynamics, which are observed by IF and laser scanning microscopy (LSM; Figure 2A). EGF treatment also results in robust Mitogen-Activated Protein (MAP) kinases signaling and c-JUN overexpression. Tiling LSM images allows for the configuration of spatial patterns. Increased expression of c-JUN at cells adjacent to the migrating front can be easily revealed by software image analysis; here, we implemented a rainbow scale for the green channel, which correspond to c-JUN labeling (Figure 2B).
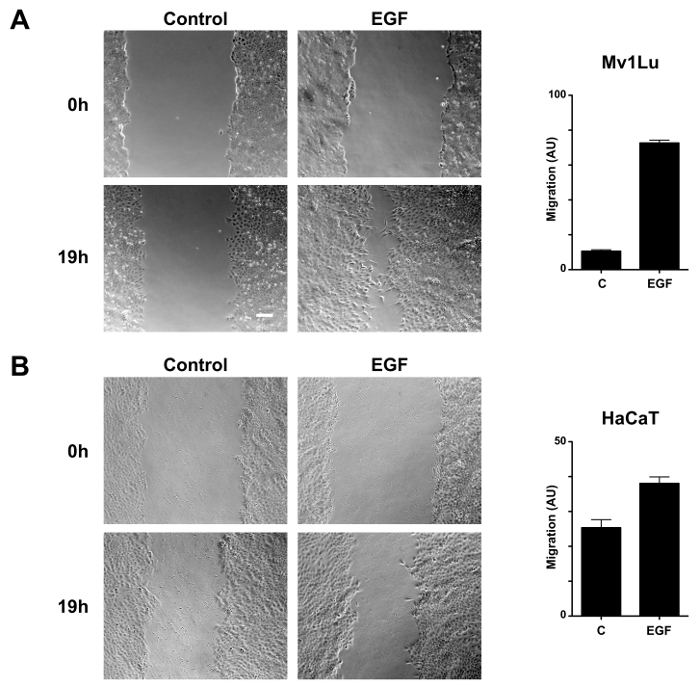
Figure 1. Promotion of migration by EGF in Mv1Lu and HaCaT cells. Phase-contrast microscopy images of the wounded area of Mv1Lu (A) and HaCaT (B) cells were taken under control conditions and compared to images of the cells treated with 10 ng/mL EGF for 19 h in serum-free conditions. Representative micrographs show the extent of scratch gap and closure obtained after wounding with a 20 µL micropipette tip. Magnification = 10x; Scale bar = 100 µm. Cell migration was quantified and represented as variation of the area differences between treatments and control samples for each assay (arbitrary units; AU). The plot is representative of eight independent measurements for each condition. Please click here to view a larger version of this figure.
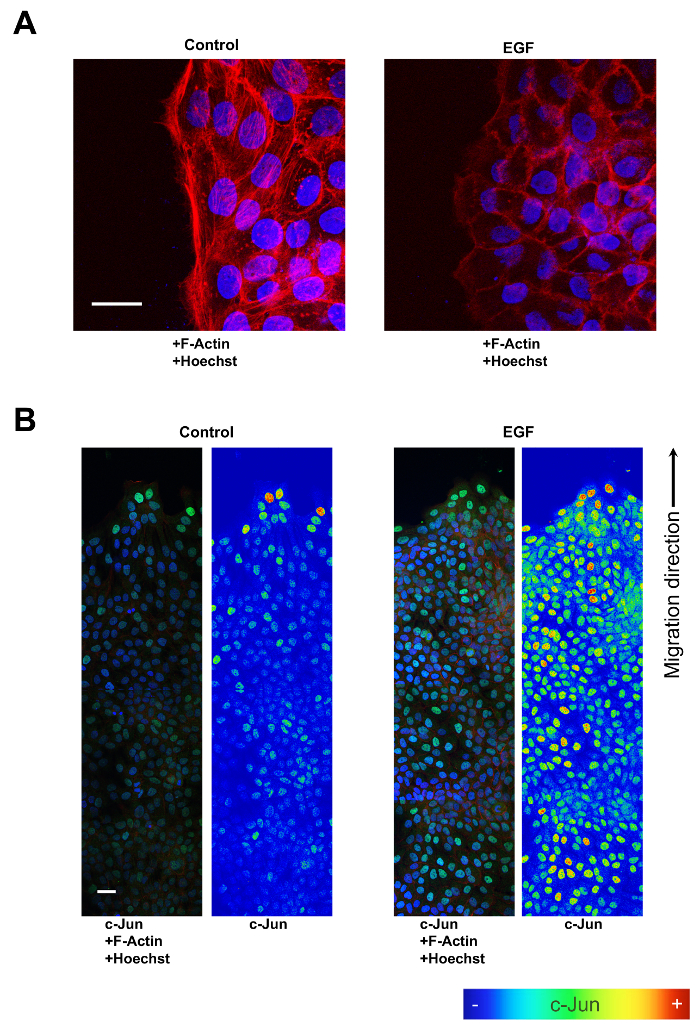
Figure 2. Actin filaments and c-JUN expression changes promoted by EGF in migrating HaCaT cells. (A) Overnight 10 ng/mL EGF treatment promotes profound changes in the conformation of actin filaments in migrating HaCaT cells. Cells at the migration front of control conditions portray a tightly structured actin filaments cytoskeleton, while cells treated with EGF show poor actin filaments organization at the migration front. Magnification = 63x; Scale bar = 20 µm. F-Actin is color-coded red, and Hoechst is represented by a blue color. (B) When immuno-fluorescence is used in combination with LSM imaging to study c-JUN expression after 10 ng/mL EGF treatment on migrating HaCaT cells, tiling compositions reveal spatial expression patterns. Images of c-Jun fluorescence (green) were converted into pseudo-color to show the intensity of c-Jun staining. The color rainbow scale represents fluorescence intensity for c-Jun, corresponding to colormaps on the right panels for EGF and Control. Co-staining with phalloidin (red) and Hoechst-33258 (blue) was used to show cell structure and nuclei, respectively. Images were taken by a confocal microscope. Note the increasing expression levels of nuclear c-Jun in areas nearing the migrating front. Scale bar = 40 µm. Please click here to view a larger version of this figure.
Discussion
Upon skin or mucous membrane disruption, barrier function is restored by the actions of numerous cell types, including fibroblasts or epithelial and immune cells. Conjointly, these cells undergo a complex process involving apoptosis, proliferation, differentiation, and importantly, fibroblast and epithelial cell migration, which is the ultimate mechanism responsible for restoration of the disrupted tissue and the closure of the superficial epithelial gap1,12 Thus, studying cell migration helps precisely describe the physiological behavior of epithelial cells, while also determining the modulatory potential of treatments for wound healing.
Performing artificial wounds on different research animal models allows for the replication of this complex process in near physiological conditions2, and thus the assessment of some pathological conditions or the effects of pharmaceutical treatments13. However, this approach often results in costly and complicated logistics, as well as burdensome experimental procedures that may generate data in a delayed fashion13. On the contrary, in vitro cell culture provides a more convenient framework for researching the behavior of cells involved in the wound healing process. While primary cell culture of epithelial cells constitutes a viable option, it can be difficult to obtain and handle the cells, which can complicate data collection. For instance, in addition to the typical low-passage restriction of primary cultures, primary human keratinocytes need a fibroblast feeding layer for growth in vitro3. On the contrary, well-established epithelial cell culture lines, like Mv1Lu or HaCaT, constitute an obstacle-free research model that features behavior and characteristics like those in primary culture cells3,4,14,15. Precisely, for the study of wound healing, these two cell lines retain critical traits in their ability to migrate and stop proliferation due to cell-to-cell contact inhibition upon confluence16,17.
Scratch assays are a reliable and versatile method for quantifying the migratory capabilities of different cell types, especially epithelial and cancer cells. For the case of cancer cells, the scratch method has been proposed for assessment of invasive potential. However, this approach falls short as an indicator for invasiveness when compared to more specific methods relying on the expression of metalloproteases needed for penetrating the extracellular matrix18,19. On the contrary, scratch assays are reliable for assessing the migratory performance of epithelial cells and the effect of different treatments on this trait. In contrast to cancer cells lines, which frequently show poor cohesion and the ability to grow on several layers, Mv1Lu, HaCaT, and other epithelial cell lines form tightly associated "growth islands", which expand their margins in a process depending on both proliferation, but more importantly on cell migration. Consequently, sparse or sub-confluent cell density conditions are conventionally used for studying the effects of different treatments on the migratory physiology; especially when subsequent molecular biology techniques are applied, as this cell environment minimizes contact inhibition while maximizing the ratio of actively migrating cells. However, for epithelial cells, non-confluent conditions pose the risk of excluding important pre-existing contact inhibition influences, for instance in the form of epigenetic regulation. Thus, to study wound healing, confluent conditions confer increased fidelity of the model.
To obtain precise estimates, concomitant factors affecting the conformation of the epithelial cell layer or contributing to migration should be addressed. While confluence is desired, it is important to avoid excessive cell density or multilayered cultures, since these conditions may affect negatively. Therefore, proliferation is commonly controlled by starvation in serum-free media or by the addition of chemicals like Mitomycin C, which irreversibly arrest cell proliferation by DNA crosslinking3,20,21. The use of one or the other relies on the behavior of specific cell lines; Mitomycin C is especially indicated when reduced serum supplementation is required to avoid massive cell detachment. That is the case for HEK-293T cells, where pre-coating the culture surface using poly-L-lysine is a good option to avoid Mytomycin C treatment. The release of inflammatory factors that could alter migration also needs to be addressed. Different strategies have been used, mainly based on silicone inserts to mimic the disruption of the epithelial surface achieved with the scratch assay22. While inserts generate pre-existing gaps that allow cell migration after removing the silicone piece, the scratch method relies directly on removing some of the epithelial monolayer to generate the gap. Preference on how to generate the gap relies on the ability of inserts to protect the culture surface coating (i.e., culture plate or poly-L-lysine) from damage during scratching, while also minimizing the release of factors from damaged cells that may compromise the cell culture behavior23. Although these characteristics might be useful for some experimental settings, i.e., assessing the ability of cancer cells to migrate, in our experience we found significant drawbacks for using inserts in wound healing studies. Mv1Lu cells are capable of attaching to the silicone insert outer surface, which results in unintentionally tearing of the cell monolayer upon removal of the silicone pieces. Also, although HaCaT cells do not attach to silicone, we found a diminished ability for these cells to migrate into insert-generated gaps. To that extent, inflammation and migration are tightly associated responses, and these appear to be critical together in the context of wound healing in HaCaT cells. Moreover, since applying a plastic tip for scratching the culture surface has no apparent consequence on the ability of cells to repopulate the gap, in the case of HaCaT cells, such behavior suggests that extracellular matrix remnants in the gap may guide migration in a similar fashion as dermis in a natural wound. However, the main source of variation remains the scratching procedure itself, as the use of a plastic tip to make the wound does not always render the same size gap. We have observed variations up to 20% among different samples, due to inconsistency of pressure and position of the tip at the time of scratch. These variations can be addressed by wounding the monolayer with a more rigid object; however, any scratch on the plastic surface would jeopardize the free movement of the cells. So, variations in the initial scratch must be taken into account to calculate migration; typically, by increasing the number of measurement per condition.
In addition to quantification, applying wounding procedures together with classic IF techniques can qualitatively assess the molecular processes of cell migration. The capability of epithelial cells to grow on coverslips is well-know; in the past, variations of the scratch assays involved cells migrating on coverslips exposed to different conditions followed by fixing and staining for comparison within the wound-scratch experimental setup24. Here, we found that creating wide gaps in monolayers growing on coverslips allows for extending the experimental time courses3,4,7. Moreover, in these experiments, IF increases the detection capabilities of the procedure to potentially study any precise cellular and subcellular mechanisms involved, and it is only limited by the availability of antibodies suitable for this technique and the cells of interest. Samples on the coverslips used for IF are mounted on microscope slides, which allow for extended preservation. This aids the application of experimental setups that require long study periods, like the "tiling" approaches showcased here and elsewhere3,4,7. While the "tiling" strategy integrates spatial patterns with temporal data for reconstructing the mechanisms involved in migration, the implementation of software analysis tools can provide semi-quantitative assessments based on local fluorescence intensity, and further enrich the quality of the information obtained. In our experience, tiling also offers the possibility of studying cell-positional information for the expression of proteins in this type of assay. This positional information has been shown to be crucial to understand and better translate the effects of different agents on cell migration, especially in setups where the position of epithelial cells is very important to determine its migratory behavior3,4,14,15.
Both procedures described demonstrate great convenience by uncomplicated implementation, as well as producing quality data. Thus, these approaches that are based on microscopy studies, offer a unique and powerful framework to study both the dynamics and morphological changes involved in epithelial cell behavior and response to treatments during wound healing.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We want to give thanks to older members of the lab that help to improve and refine these techniques to its actual state: Dr. Celia Martinez-Mora; Dr. Anna Mrowiec, Dr. Catalina Ruiz-Cañada and Dr. Antonia Alcaraz-García. We are indebted to the Hospital Clínico Universitario Virgen de la Arrixaca for strongly supporting the development of these techniques. Also the Instituto de Salud Carlos III, Fondo de Investigaciones Sanitarias. Plan Estatal I+D+I and Instituto de Salud Carlos III-Subdirección General de Evaluación y Fomento de la Investigación (Grant no.: PI13/00794); www.isciii.es. Fondos FEDER “Una manera de hacer Europa”. We also thank Universidad de Murcia, IMIB-Arrixaca and FFIS for administrative support and assistance. Finally, we want to give a special thanks to Dr. Isabel Martínez-Argudo and the Facultad de Ciencias Ambientales y Bioquímica, Campus Tecnológico de la Fábrica de Armas, Universidad Castilla la Mancha, Toledo for their kind support in willingly ceding the Biomedicine and Biotechnology Laboratory to make possible the filmed part of this paper.
Materials
| Dulbecco’s Modified Eagle Medium (DMEM) | Biowest, Nuaillé, France | L0102-500 | Optional 10 % FBS supplement |
| Eagles’s Minimum Essential Medium (EMEM) | Lonza | BE12 -662F | Optional 10 % FBS supplement |
| L-Glutamine | Lonza | BE17-605E | Use at 2 mM |
| Fetal Bovine Serum (FBS) | Thermo Fisher Scientific, Waltham, MA USA | DE17603A | |
| Trypsin-EDTA | Sigma-Aldrich, St Louis, MO, USA | T4049 | Dilute as appropriate |
| Poly-L-Lysine | Sigma-Aldrich, St Louis, MO, USA | P9155 | |
| Dulbecco's Phosphate Buffered Saline (DPBS) (10x) | Gibco by Life Technologies | 14200-067 | Dilute to 1x |
| 24-well culture plates | BD FALCON//SARSTED | 734-0020 | |
| 6-well culture plates | SARSTEDT | 83-3920 | |
| Epidermal Growth Factor (EGF) | Sigma-Aldrich, St Louis, MO, USA | E9644 | Used 10 ng/mL |
| Round cover glass | MENZEL-GLÄSER | MENZCB00120RA020 | SHORT DEPTH OF FIELD |
| Reinforced razor blade no. 743 | Martor (through VWR) | MARO743.50 | |
| 200 µl sterile aerosol pipet tips | VWR | 732-0541 | |
| 20 µl sterile aerosol pipet tips | VWR | 732-0528 | |
| Digital camera coupled phase contrast microscope | Motic Spain | Moticam camera 2300 3.0 M Pixel USB 2.0; Motic Optic AE31 | |
| Confocal microscope | ZEISS Microimaging, Germany | LSM 510 META | |
| 10 cm Culture dish | BD FALCON | 353003 | |
| Rabbit polyclonal anti c-Jun antibody | Santa Cruz Biotechnology | sc-1694 | Used 1:100 |
| Anti-rabbit IgG (polyclonal goat ) AF 488 | Invitrogen | A11008 | Used 1:400 |
| Hoechst-33258 | Sigma-Aldrich, St Louis, MO, USA | 14530 | Used 1:1000 |
| Alexa Fluor 594 phalloidin (in methanol) (red) | Invitrogen | A12381 | Used 1:100 |
| Bovine Serum Albumin | Santa Cruz Biotechnology | SC-2323 | |
| Triton X-100 | Sigma-Aldrich, St Louis, MO, USA | T9284 | |
| Skim milk | BD DIFCO | 232100 | |
| ImageJ | National Institutes of Health, USA | Release 1.50i | |
| Zen LSM 510 image processing software | ZEISS Microimaging, Germany | Release 5.0 SP 1.1 |
Riferimenti
- Singer, A. J., Clark, R. A. Cutaneous wound healing. N Engl J Med. 341 (10), 738-746 (1999).
- Davidson, J. M. Animal models for wound repair. Arch Dermatol Res. 290, S1-S11 (1998).
- Alcaraz, A., et al. Amniotic Membrane Modifies the Genetic Program Induced by TGFss, Stimulating Keratinocyte Proliferation and Migration in Chronic Wounds. PLoS One. 10 (8), e0135324 (2015).
- Ruiz-Canada, C., et al. Amniotic membrane stimulates cell migration by modulating Transforming Growth Factor-beta signaling. J Tissue Eng Regen Med. , (2017).
- Schmierer, B., Hill, C. S. TGFbeta-SMAD signal transduction: molecular specificity and functional flexibility. Nat Rev Mol Cell Biol. 8 (12), 970-982 (2007).
- Hu, Y. L., et al. FAK and paxillin dynamics at focal adhesions in the protrusions of migrating cells. Sci Rep. 4, 6024 (2014).
- Martinez-Mora, C., et al. Fibroin and sericin from Bombyx mori silk stimulate cell migration through upregulation and phosphorylation of c-Jun. PloS one. 7 (7), e42271 (2012).
- Bernabe-Garcia, A., et al. Oleanolic acid induces migration in Mv1Lu and MDA-MB-231 epithelial cells involving EGF receptor and MAP kinases activation. PLoS One. 12 (2), e0172574 (2017).
- Huang, H. L., et al. Trypsin-induced proteome alteration during cell subculture in mammalian cells. J Biomed Sci. 17, 36 (2010).
- Liberio, M. S., Sadowski, M. C., Soekmadji, C., Davis, R. A., Nelson, C. C. Differential effects of tissue culture coating substrates on prostate cancer cell adherence, morphology and behavior. PLoS One. 9 (11), e112122 (2014).
- Pierreux, C. E., Nicolas, F. J., Hill, C. S. Transforming growth factor beta-independent shuttling of Smad4 between the cytoplasm and nucleus. Mol Cell Biol. 20 (23), 9041-9054 (2000).
- Barrientos, S., Stojadinovic, O., Golinko, M. S., Brem, H., Tomic-Canic, M. Growth factors and cytokines in wound healing. Wound repair and regeneration: official publication of the Wound Healing Society [and] the European Tissue Repair Society. 16 (5), 585-601 (2008).
- Ansell, D. M., Holden, K. A., Hardman, M. J. Animal models of wound repair: Are they cutting it?. Exp Dermatol. 21 (8), 581-585 (2012).
- Gurtner, G. C., Werner, S., Barrandon, Y., Longaker, M. T. Wound repair and regeneration. Nature. 453 (7193), 314-321 (2008).
- Insausti, C. L., et al. Amniotic membrane induces epithelialization in massive posttraumatic wounds. Wound Repair Regen. 18 (4), 368-377 (2010).
- Wu, F., et al. Cell cycle arrest in G0/G1 phase by contact inhibition and TGF-beta 1 in mink Mv1Lu lung epithelial cells. Am J Physiol. 270 (5 Pt 1), L879-L888 (1996).
- Boukamp, P., et al. Normal keratinization in a spontaneously immortalized aneuploid human keratinocyte cell line. J Cell Biol. 106 (3), 761-771 (1988).
- Marshall, J., Wells, C. M., Parsons, M. . Cell Migration: Developmental Methods and Protocols. , 97-110 (2011).
- Nyegaard, S., Christensen, B., Rasmussen, J. T. An optimized method for accurate quantification of cell migration using human small intestine cells. Metabolic Engineering Communications. 3, 76-83 (2016).
- Szybalski, W., Iyer, V. N. Crosslinking of DNA by Enzymatically or Chemically Activated Mitomycins and Porfiromycins, Bifunctionally “Alkylating” Antibiotics. Fed Proc. 23, 946-957 (1964).
- Tomasz, M. Mitomycin C: small, fast and deadly (but very selective). Chem Biol. 2 (9), 575-579 (1995).
- Sherry, D. M., Parks, E. E., Bullen, E. C., Updike, D. L., Howard, E. W. A simple method for using silicone elastomer masks for quantitative analysis of cell migration without cellular damage or substrate disruption. Cell Adh Migr. 7 (6), 469-475 (2013).
- Dennis, E. A., Rhee, S. G., Billah, M. M., Hannun, Y. A. Role of phospholipase in generating lipid second messengers in signal transduction. FASEB J. 5 (7), 2068-2077 (1991).
- Lampugnani, M. G. Cell migration into a wounded area in vitro. Methods Mol Biol. 96, 177-182 (1999).

