Invasive Hemodynamic Characterization of the Portal-hypertensive Syndrome in Cirrhotic Rats
Summary
Here we describe a detailed protocol for invasive measurements of hemodynamic parameters including portal pressure, splanchnic blood flow, and systemic hemodynamics in order to characterize the portal hypertensive syndrome in rats.
Abstract
This is a detailed protocol describing invasive hemodynamic measurements in cirrhotic rats for the characterization of portal hypertensive syndrome. Portal hypertension (PHT) due to cirrhosis is responsible for the most severe complications in patients with liver disease. The full picture of the portal hypertensive syndrome is characterized by increased portal pressure (PP) due to the increased intrahepatic vascular resistance (IHVR), hyperdynamic circulation, and increased splanchnic blood flow. Progressive splanchnic arterial vasodilation and increased cardiac output with elevated heart rate (HR) but low arterial pressure characterizes the portal hypertensive syndrome.
Novel therapies are currently being developed that aim to decrease PP by either targeting IHVR or increased splanchnic blood flow — but side effects on systemic hemodynamics may occur. Thus, a detailed characterization of portal venous, splanchnic, and systemic hemodynamic parameters, including measurement of PP, portal venous blood flow (PVBF), mesenteric arterial blood flow, mean arterial pressure (MAP), and HR is needed for preclinical evaluation of the efficacy of novel treatments for PHT. Our video article provides the reader with a structured protocol for performing invasive hemodynamic measurements in cirrhotic rats. In particular, we describe the catheterization of the femoral artery and the portal vein via an ileocolic vein and the measurement of portal venous and splanchnic blood flow via perivascular Doppler-ultrasound flow probes. Representative results of different rat models of PHT are shown.
Introduction
PHT is defined as pathologically increased blood pressure in the portal venous system that can cause severe complications in patients with cirrhosis such as variceal bleeding and ascites1. While pre-hepatic (e.g., portal vein thrombosis) and post-hepatic (e.g., Budd-Chiari Syndrome) PHT are rare, intrahepatic PHT due to liver cirrhosis represents the most common cause of PHT2.
In liver cirrhosis, PP is primarily increased as a consequence of elevated IHVR3. In advanced stages, PHT is aggravated by the increased PVBF due to increased cardiac output and decreased systemic and splanchnic vascular resistance — defining the portal hypertensive syndrome4. Ohm's law (ΔP = Q * R) implies that the IHVR and blood flow are proportional to PP5. In patients, direct measurement of PP is risky and not routinely performed; instead, the hepatic venous pressure gradient (HVPG) is used as an indirect measure of PP6,7. The HVPG is calculated by subtracting the free hepatic venous pressure (FHVP) from the wedged hepatic venous pressure (WHVP), which are measured using a balloon catheter placed in a hepatic vein8. The physiological HVPG ranges between 1–5 mmHg, while an HVPG ≥10 mmHg defines clinically significant portal hypertension (CSPH) and indicates increased risk for PHT-related complications, such as variceal bleeding, ascites, and hepatic encephalopathy9. Although PP (i.e., HVPG) is the most relevant parameter for PHT severity, information about other components of PHT, including the severity of hyperdynamic circulation (HR, MAP), splanchnic/mesenteric arterial blood flow, and IHVR, are critical to obtain a comprehensive understanding of the distinct underlying mechanism of PHT.
Thus, in contrast to indirect measurements of PP in humans, the introduced methodology for rats offers the advantage of a direct measurement of PP and allows the recording of additional hemodynamic parameters characterizing the portal hypertensive syndrome. In addition, the direct measurement of PP is an excellent integrative readout of the amount of liver fibrosis (a major determinant of IHVR) and overcomes certain limitations of fibrosis quantification related to liver tissue sampling errors.
The most commonly used rodent models of cirrhotic PHT include surgical bile duct ligation (BDL), toxin-induced liver injury (i.e., by carbon tetrachloride, thioacetamide, or dimethylnitrosamine administration), and diet-induced metabolic liver disease models. Prehepatic (non-cirrhotic) PHT can be induced by partial portal vein ligation (PPVL)10.
Small rodents are well suited for the presented method, including mice, hamsters, rats, or rabbits, and are associated with relatively low maintenance costs. Despite that all the hemodynamic assessments are feasible to perform in mice, better accuracy and reproducibility of results are seen with rats or larger rodents due to the obvious advantage of animal size. In addition, specific micro-instruments and devices are needed to obtain similar hemodynamic parameters in mice. Finally, rats are more robust with lower associated morbidity and mortality and thus, the drop-out rates are likely lower in rats than in mice.
The presented methodology is well-suited for evaluating specific treatments of liver disease (i.e., anti-fibrotic or anti-inflammatory drugs) or novel pharmacological approaches that influence vascular tone and/or endothelial biology; and thus, likely effect hemodynamic parameters in PHT.
Protocol
All methods described here have been approved by the ethics committee of the Medical University of Vienna and the Austrian Ministry of Science, Research and Economy (BMWFW). Procedures must be performed in aseptic conditions in an operation room or similar clean working area since the hemodynamic measurements represent surgical interventions. Generally, working in sterile conditions is recommended. When using an inhalation anesthesia, consider adequate ventilation of the surgery room for work safety. A time period of 40–50 min/animal has to be considered in the case all hemodynamic readouts presented in this protocol.
1. Pre-surgical Preparations
- Turn on and calibrate the electronic multichannel recorder including the pressure transducers according to the manufacturer's instruction.
- Connect the ultrasonic flow probes (1 mm and 2 mm) to the bridge amplifier.
- Prepare a reservoir of sterile physiological saline solution, heated to body temperature, 37 °C, for moistening tissues or gauze compresses.
- Record the animal body weight to provide weight-adjusted anesthesia and normalization of hemodynamic parameters according to the body weight.
- Prepare all equipment for an inhalation anesthesia.
NOTE: If inhalation anesthesia and/or necessary equipment is not on site or feasible, then injection anesthesia using ketamine and xylazine can be used (80–100 mg/kg ketamine with 5–10 mg/kg xylazine in physiological saline solution, intraperitoneal (i.p.)). A re-dosing of ketamine (reduced dose of 20–30 mg/kg, intramuscular (i.m.)) after 30–45 min is necessary to induce continuous surgical-plane anesthesia. - Put the animal under short-term isoflurane anesthesia using an induction box for inhalation anesthesia (5 min, 5% v/v isoflurane, 3–4 L O2-flow). Carefully tilt the box and check the depth of anesthesia by the immobility status of the animal.
- Intubate the rat with a suitable self-made endotracheal tube.
NOTE: For personnel who are new to the intubation technique, the analgesia/anesthesia (step 1.10) can be given right after short term inhalation anesthesia to allow extra time for intubation. Another option is an animal-specific facemask for inhalation anesthesia.- Use a self-made endotracheal tube from a modified peripheral venous catheter (14 G). Cut off the handling wings and affix an adhesive tape loop for posterior fixation to the rat's cheek to prevent dislocation of the endotracheal tube (Figure 1A).
- Use a self-made guide wire device from a modified arterial cannula as a guide wire holder and a suitable blunt-tipped wire (Figure 1B).
- Use a suitable intubation desk for proper positioning of the animal. Place the animal in the supine position with the head in the angled position.
NOTE: If no intubation desk is available, it is possible to place the animal in supine position with the neck carefully stretched over the table's edge. However, this procedure is not recommended due to increased risk of injuries. - Fix a suture behind the incisors on one side of the rat, and gently stretch the animal's neck by tying the suture down on the other side (Figure 1C).
- Illuminate the pre-tracheal ventral collar area by a focused light beam. Especially in albino animals, ensure that the vocal cords are illuminated through the skin to allow for improved visualization and rapid intubation.
NOTE: Use an animal-specific laryngoscope for intubation of pigmented animals. - Grab the tongue and gently pull it by using two fingers.
- Use a cotton bud treated by lidocaine (pump spray) to carefully anesthetize the laryngeal area.
- Intubate the animal by inserting the endotracheal tube between the vocal folds and into the trachea, using the support of the guide wire device (Figure 1D).
- Remove the guide wire device.
- Connect the tube to the ventilator.
- Start the ventilator using the appropriate settings for the animal (1 L/min O2-Flow, Auto Flow = 90/min; inspiratory pressure: 18 mmHg; PEEP: 3 mmHg, I/E = 1:2) and check for proper intubation.
NOTE: If inflation of the stomach is noted, remove the tube and try again. Additionally, compare the respiratory activity to the ventilator rhythm or place two fingers on the abdomen wall right over the stomach to assess potential inflation of the stomach.
- Start isoflurane anesthesia at 0.5–1.0% v/v and 1 L/min O2-Flow immediately after successful intubation (Figure 2A).
- Fix the endotracheal tube by a transbuccal suture through the cheek and the affixed adhesive tape loop of the tube.
- Administer additional anesthesia and analgesia by two 1 mL syringes, e.g., ketamine (100 mg/kg) i.p. (23 G cannula) [or i.m. by distributing the injected volume (dose) in bilateral injections in the caudal thigh muscle (30 G cannula)] and piritramide (2 mg/kg) by subcutaneous (s.c.) injection (23 G cannula). Note the maximum volume of i.m. injections per injection site (Figure 2B).
- Apply eye ointment. Clip the body hair at the abdominal region and both inner thighs. Disinfect the skin.
NOTE: Both inner thighs should be shaved to allow the use the contralateral femoral artery for HR and MAP measurements in case the catheterization of the femoral artery failed on one side. Shaving at a later time point may cause hair to contaminate the surgical field. - Fix the animal in the supine position on a heating pad (38 °C) with adhesive tape (Figure 2C).
- Monitor the body temperature of the animal continuously, e.g., by using a rectal temperature probe (Figure 2D).
- Assess the anesthesia depth by lid closure reflex and toe-pinch-test prior to any intervention or surgery.
2. Measurement of HR and MAP
- Incise the skin on the inner thigh (select one side) above the presumed location of the femoral artery by lifting the skin by tissue forceps and removing a skin area of about 2 cm length by Mayo scissor (Figure 3A).
- Expose and bluntly dissect the artery-vein-nerve complex that includes the femoral artery from connective tissue (adventitia) by repeatedly opening a hemostat along the complex.
- Dissect the artery-vein-nerve complex from the surrounding tissue along ~1–1.5 cm (Figure 3B).
- If necessary, remove subcutaneous/perivascular fat for a better view and dissection.
NOTE: Be careful when removing subcutaneous/perivascular fat by cuticle scissor, since injured blood vessels may cause bleedings. If bleeding is noted, apply pressure on the bleeding area using a small gauze or stop the bleeding by a hemostat. - Separate the femoral artery from the femoral vein and nerve with two high precision 45° angle broad point forceps (consider that the femoral vein is the most medial structure and the femoral artery is located more lateral) (Figure 3C-F).
- Place a ligature on the femoral artery as distal as possible and use a curved clamp on the suture to apply gentle longitudinal traction to the femoral artery.
- Place a second pre-knotted (but not closed knot) suture on the femoral artery as proximal as possible (Figure 3G).
- Prepare a suitable catheter (PE-50 for rat femoral artery) with a slanted (~45°) cut-off tip flushed with heparinized sterile physiological saline solution (5 mL syringe and 23 G blunt cannula). Ensure no air bubbles are inside the catheter as they hamper the arterial pressure and HR readout (Figure 3H).
- Compress the femoral artery at the proximal end of the exposed section using a micro clamp to temporarily stop the arterial blood flow (Figure 3I).
- Perforate the arterial wall at a distal location of the dissected section by using a bent cannula (23 G) while placing a supporting micro metal spatula underneath the femoral artery (Figure 3J).
- Carefully catheterize the femoral artery through the perforation with the slanted tip of the catheter facing upwards. Advance the catheter until the micro clamp is approximated (Figure 3K).
NOTE: In case of unsuccessful catheterization of the femoral artery at first try, a more proximal second attempt can be performed (begin at step 2.8). If the artery ruptures or severe bleeding occurs, ligate or clamp the artery as proximal as possible to prevent further blood loss. If blood loss is minimal, attempt catheterizing the contralateral femoral artery (begin at step 2.1). - Open the micro clamp and check for pulsatile arterial blood flow into the catheter (Figure 3L).
- Prevent further influx of blood into the catheter by blocking the outflow of the catheter.
- Fix the catheter in its intraluminal location by closing the prepared proximal ligature around the femoral artery and the introduced catheter (Figure 3M).
- Repeatedly rinse and aspirate the catheter in the femoral artery to assess proper intravascular placement. Arterial pulsation in the column of aspirated blood should be easily noticeable.
- Fix the catheter position along the vessel using the ends of the distal ligature to prevent dislocation and ensure a longitudinal position of the catheter (Figure 3N).
- Additionally, tape the catheter close to the animal on the surgical table to secure it and prevent accidental dislocation.
- Connect the catheter filled with physiological saline solution to the pressure transducer while avoiding the formation of air bubbles.
- Start recording the HR and MAP using the digital interface (Figure 3O).
- Cover the exposed area on the inner thigh with a small moistened gauze compress (Figure 3P).
- Calculate the hyperdynamic (HD) index: HD = MAP/HR.
NOTE: In advanced PHT, the HD index is elevated as compared to non-portal hypertensive animals. However, an increase in HD index during surgery might also indicate bleeding, hypovolemia, or pain. If recorded MAP values are very low but the signal is good and pulsatile flow is detected in the catheter, check for the level of anesthesia and potentially reduce the level of anesthesia. Do not completely stop isoflurane anesthesia, since this may lead to insufficient anesthesia depth according to animal welfare and good scientific practice.
3. Superior Mesenteric Artery Blood Flow (SMABF)
- Perform a median laparotomy (Figure 4A-C)
- Lift the skin layer with tissue forceps 5–6 cm below the xiphoid and remove a thin strip of skin using a Mayo scissor above the linea alba until the xiphoid is reached.
- At the middle the skin incision, lift the muscular layer by tissue forceps along the linea alba to create distance between the abdominal wall and splanchnic organs.
- Open the peritoneal cavity by incising the abdominal wall with a scalpel at the linea alba. Extend the opening while lifting the abdominal wall by tissue forceps with a Metzenbaum scissor along the linea alba over the same distance as the skin layer.
- Carefully excavate the intestine using wet cotton buds by starting with the coecum and place it on a large gauze compress soaked in sterile physiological saline solution next to incision (Figure 4D-F).
- Wrap the intestine into the gauze compress and make sure it is moistened with sterile physiological saline solution (Figure 4G).
- Locate and expose the superior mesenteric artery.
- Dissect the superior mesenteric artery with two self-made blunt 'Schwabl'-Hooks: Lift the artery with the first hook and try to place the second one through the same tissue tunnel. Expose the superior mesenteric artery along a 5 mm distance to ensure the flow probe (1 mm) can be placed around it (Figure 4H-K).
NOTE: If preferred, a precision 45° angle broad point forceps can be used to lift the artery as well. 'Schwabl'-Hooks are prepared from 30 G cannulas with bluntly broken tips bent to a hook shape. If collateral arteries are extensive, the 'Schwabl'-Hooks may be safer as bleedings from collaterals while dissecting the superior mesenteric artery can be avoided. If bleeding occurs while preparing the superior mesenteric artery, place a small gauze compress on the bleeding site for 1–2 min with gentle pressure. Small bleeding will usually stop quickly; always keep in mind to moistening tissue periodically (step 1.12). If the superior mesenteric artery itself is harmed, the hemodynamic assessments must be terminated. - Apply ultrasound gel to the ultrasonic flow probe's sensor and attach it to the splanchnic mesenteric artery. Align it to the natural route of the superior mesenteric artery (Figure 4L-M).
- Close the flow probe (1 mm) and if needed gently apply additional ultrasound gel on the Doppler sensor to improve signal quality. Do so by using a syringe (20 mL) filled with ultrasound gel and a blunt-tipped cannula (18 G) (Figure 4N-O).
NOTE: If the flow probe is not well aligned along the natural course of the vessel, tension may cause vascular constriction and thus, turbulent flow, which reduces the accuracy of the flow measurements. Try to re-align the direction of the flow probe along the natural route of the vessel and then sufficiently fix the flow probe in an appropriate position. - Measure the SMABF and assess the accordance of the pulsatile flow signal to the systolic peaks of the femoral artery recording.
NOTE: If recorded MAP values are very low but the signal is good and pulsatile flow is detected in the catheter, check for the level of anesthesia and potentially reduce the level of anesthesia. Do not completely stop isoflurane anesthesia, since this may lead to insufficient depth of anesthesia according to animal welfare and good scientific practice. - Find a stable position of the flow probe (1 mm) and fix the cable of the flow probe. Start recording the SMABF without further manipulation of the flow probe (1 mm) (Figure 4P).
4. PVBF
- Locate and expose the portal vein at the dorsal face of the mesentery that is close to the liver hilum (Figure 5A).
- Gently dissect the portal vein from the surrounding tissue using a high precision 45° angle broad point forceps: Isolate the portal vein by gently and repeatedly pushing the forceps under the portal vein to create a tissue tunnel (Figure 5B).
NOTE: If a bleeding occurs from the periportal tissue while preparing the portal vein, apply gentle pressure on the bleeding site for 1-2 min using a cotton bud; this often stops the bleeding. - Enlarge the tunnel by opening the high precision 45° angle broad point forceps slowly and expose the portal vein along a distance of 5-6 mm to allow the placement of the perivascular flow probe (2 mm) (Figure 5C, D).
- Apply ultrasound gel to the ultrasonic flow probe's sensor and attach it to the portal vein aligned with its natural route (Figure 5E).
- Close the flow probe (2 mm) and apply additional ultrasound gel if needed, as previously described (step 3.7) (Figure 5F).
- Make sure the flow probe is placed in a non-constrictive way around the portal vein (Figure 5G).
NOTE: If the flow probe is not well aligned along the natural course of the vessel, tension may cause vascular constriction and thus, turbulent flow, which reduces the accuracy of the flow measurements. Try to re-align the direction of the flow probe along the natural route of the vessel and then sufficiently fix the flow probe in an appropriate position. - Find a stable position of the flow probe (2 mm) and fix the cable of the flow probe. Then, start recording the PVBF (Figure 5H).
NOTE: If recorded MAP values are very low but the signal is good and pulsatile flow is detected in the catheter, check for the level of anesthesia and potentially reduce the level of anesthesia. Do not completely stop isoflurane anesthesia, since this may lead to insufficient depth of anesthesia according to animal welfare and good scientific practice.
5. PP
- Prepare a catheter (PE-50 for rat mesenteric veins) with a slanted (about 45°) cut-off tip that is flushed with sterile physiological saline solution (5 mL syringe and blunt 23 G cannula). Pay attention that no air bubbles are inside the catheter as they obstruct the PP readout (Figure 6A).
- Handle the intestine with wet gloves and place it scattered across fingers (Figure 6B).
- Optimize the view of the mesenteric vascular bed close to the small intestine (Figure 6C).
- Identify the main venous mesenteric vessels leading to the portal vein (vena ileocolica – vena mesenterica superior – vena portae).
- Find a suitable junction of the ileocolonic vein that is accessible for catheterization.
- First stick the catheter into the mesenteric tissue by perforating the visceral peritoneum of the mesentery close to the vascular junction chosen for catheterization.
- Carefully advance the slanted tip of the catheter closer to a junction of the ileocolic vein until a slight impression of the vessel junction is seen (Figure 6D).
- Finally, catheterize the venous system in line with the joining vessel route by perforating the vascular wall at the crossing angle of the vessels. (Figure 6E).
NOTE: If bleeding occurs at the catheterization of the ileocolic vein, using a thumb press with a small gauze compress at the bleeding area. This pressure should be maintained for 1–2 min to stop the bleeding. Afterwards, try to insert the catheter at a more proximal branch of the ileocolic vein. - Advance the catheter carefully further along the main venous vessel route to the portal vein without perforating the vein (Figure 6F).
NOTE: Keep the catheter at a sufficient distance to the flow probe placed around the main branch of the portal vein to avoid artifacts in the flow signal and to prevent perforation of the portal vein. - Connect the catheter filled with physiological saline solution to the pressure transducer while avoiding the formation of air bubbles.
- Start recording the PP.
- Record all hemodynamic parameters simultaneously under stable conditions for several min (Figure 6G, H). Optionally, the portal venous catheter can be fixed in place using tissue glue and the intestines can be re-located into the abdominal cavity.
NOTE: If recorded MAP values are very low but the signal is good and pulsatile flow is detected in the catheter, check for the level of anesthesia and potentially reduce the level of anesthesia. Do not completely stop isoflurane anesthesia, since this may lead to insufficient depth of anesthesia according to animal welfare and good scientific practice.
6. IHVR
- After sacrificing the animal, measure the liver weight. Calculate the IHVR: IHVR = PVBF/PP. Normalize this PVBF value to the weight of the liver.
Representative Results
Depending on the animal model and the severity of liver disease, the degree of PHT and severity of the portal hypertensive syndrome is different (Figure 7).
The BDL model causes biliary cirrhosis due to cholestasis. Accordingly, PP increases over time and a hyperdynamic circulation develops, as seen by an increase of HR and decrease of MAP. In cirrhotic animals, SMABF, PVBF and IHVR also increase concordantly to the hepatic and hemodynamic alterations (Figure 7A–F).
In contrast, PPVL causes prehepatic, non-cirrhotic PHT, which is characterized by an immediate increase in PP and corresponding changes in systemic hemodynamics (Figure 7G-I). However, during the time-course portosystemic collaterals develop which may lower PP.
The hemodynamic values of sham-operated animals remain at physiological levels and do not significantly change over time. The portal pressure in healthy SO animals is at maximum 5 to 6 mmHg (Figure 7J-L).

Figure 1: Self-made intubation devices: (A) Endotracheal tube. (B) Guide wire device (C) Intubation desk. (D) Tube attached to guide wire device. Please click here to view a larger version of this figure.
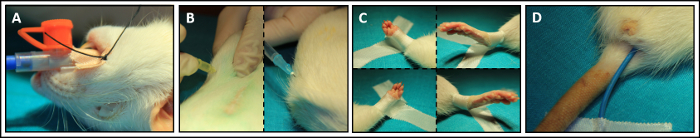
Figure 2: Pre-surgical Preparations: (A) Intubation of animal. (B) Intramuscular and subcutaneous injection for anesthesia. (C) Fixation of animal on heating mat. (D) Placing and fixing rectal temperature probe. Please click here to view a larger version of this figure.
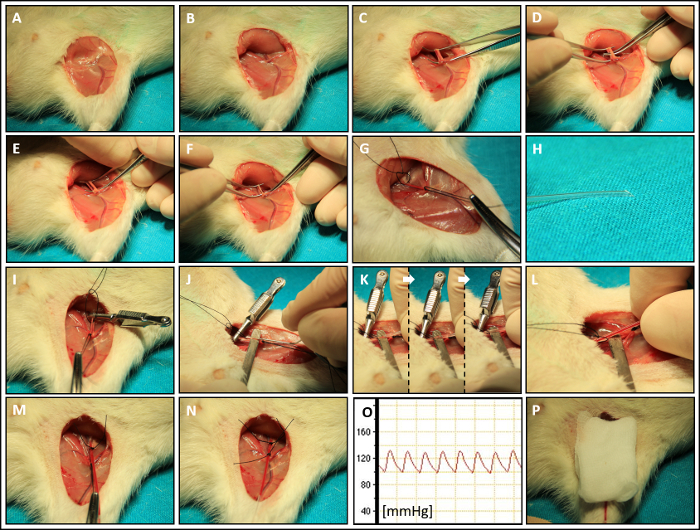
Figure 3: Heart Rate (HR) and Mean Arterial Pressure (MAP): (A) Skin incision. (B) Preparation of the femoral vascular and nerve structures. (C–F) Dissection of the femoral artery. (G) Distal suture and fixation – proximal pre-knot proximal suture. (H) Preparation of the femoral catheter. (I) Placement of the vascular micro clamp. (J) Perforation of the femoral artery with a bend needle. (K) Catheterization of the femoral artery. (L) Opening of the micro clamp for assessment of pulse. (M) Proximal fixation of catheter. (N) Distal fixation of catheter. (O) Measurement of MAP and HR. (P) Covering the surgical field with soaked small gauze compress. Please click here to view a larger version of this figure.
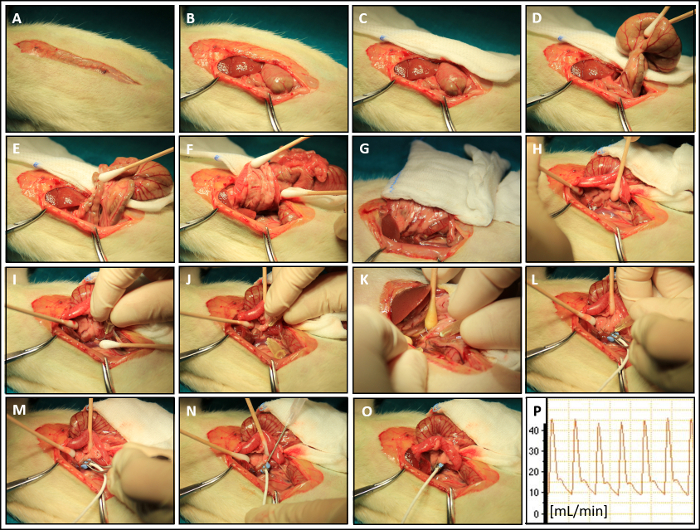
Figure 4: Superior Mesenteric Arterial Blood Flow (SMABF): (A–C) Median laparotomy. (D) Excavation of coecum. (E–F) Excavation of intestine. (G) Wrapping of the intestines in soaked gauze compress. (H–K) Preparation of the splanchnic mesenteric artery with blunt cannula hooks. (L, M) Attachment of the flow probe. (N) Application of ultrasound gel on the flow probe sensor. (O) Correct 'non-constrictive' placement of the of the flow probe. (P) Measurement of SMABF. Please click here to view a larger version of this figure.
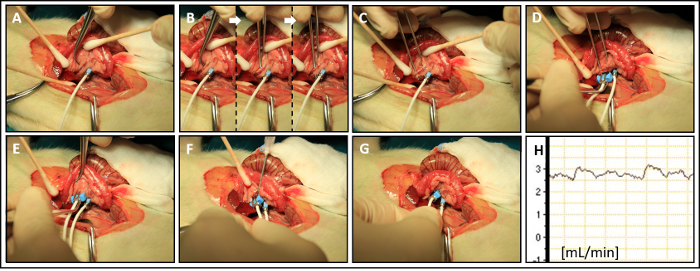
Figure 5: Portal Venous Blood Flow (PVBF): (A) Optimized dorsal view on portal vein (B) Dissection of the portal vein from mesenteric fat tissue. (C) Creation of a tissue tunnel for the portal vein flow probe. (D, E) Attachment of the flow probe to portal vein. (F) Application of ultrasound gel on the flow probe sensor. (G) Correct 'non-constrictive' placement of the flow probe. (H) Measurement of PVBF. Please click here to view a larger version of this figure.
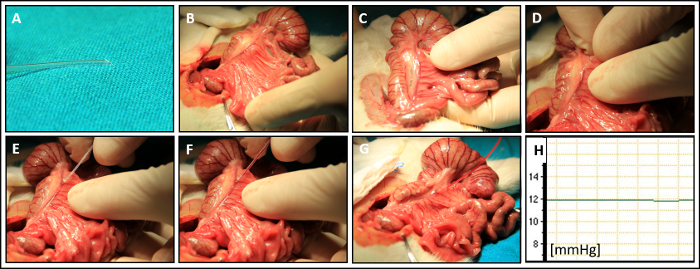
Figure 6: Portal Pressure (PP): (A) Preparation of catheter. (B) Preparation of the intestines. (C) Optimized view on the main mesenteric venous vasculature. (D) Perforation of the visceral peritoneum and advancement of the catheter closer to suitable vascular branch. (E) Catheterization of the ileocolic vein at junction angle between the main branch and a side-branch. (F) Advancement of the catheter tip into the portal vein closer to liver hilum. (G, H) Measurement of PP. Please click here to view a larger version of this figure.
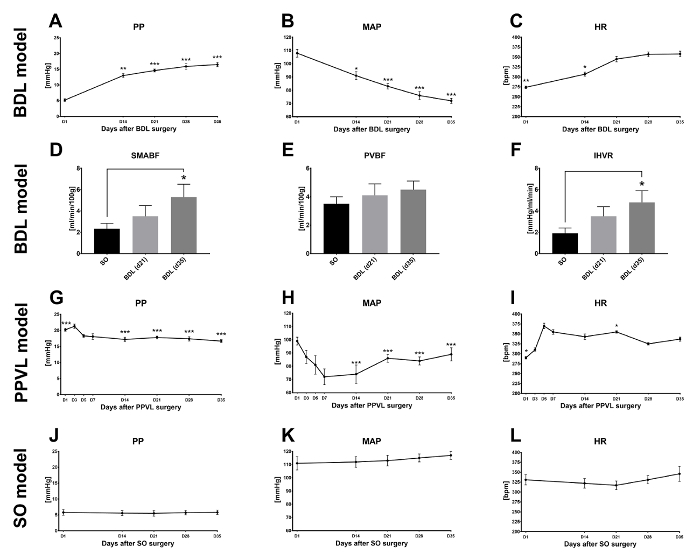
Figure 7: Representative Results: Time course of (A) PP, (B) MAP, and (C) HR in BDL rats. Accordingly, changes in (D) SMABF, (E) PVBF, and (F) IHVR are observed. In the PPVL, hemodynamic changes of (G) PP, (H) MAP, and (I) HR are most pronounced in the early days after surgery. In healthy sham-operated (SO) animals, (J) PP, (K) MAP and (L) HR remain within physiological values and do not change over time. Please click here to view a larger version of this figure.
Discussion
PP is the main outcome parameter for evaluation of the portal hypertensive syndrome and reflects the severity of underlying cirrhosis. Both matrix deposition (i.e., fibrosis) and sinusoidal vasoconstriction (due to increased hepatic expression of vasoconstrictors and decreased responsiveness to vasodilators) cause increased IHVR. The importance of PP and its impact on chronic liver disease has been shown in multiple preclinical11,12,13,14 and clinical studies15,16,17,18. Hence, in cirrhotic patients, PP is a hard outcome parameter, and its reduction is recommended by treatment guidelines19,20 and a main research goal of current hepatology. Comprehensive animal models are needed to characterize and translate16,21 novel treatment options of PHT22. This protocol presents the methodology needed for a detailed hemodynamic characterization, including assessment of portal pressure, the hyperdynamic circulation, splanchnic vasodilation and intrahepatic resistance. To achieve a representative and full hemodynamic data set from rodent models, experience and training of the performing operator is of utmost importance.
Prevention and control of severe bleedings are especially key skills. Blunt and precise preparations of the vascular sections of interest is critical in order to avoid cannulation failures and severe bleedings. Significant blood loss has an impact on hemodynamics and precludes accurate measurements of PP or may even result in death of the laboratory animal. Document bleedings that have occurred during the measurements in the protocols and characterize the severity and location of bleeding.
Of note, using perivascular ultrasound flow probes to assess blood flow generates only an approximation and might be subjected to reading errors, due to different vessel sizes and incorrect probe alignment. Another technique to measure blood flow, and especially blood-flow distribution (including calculation of portosystemic shunting) is the colored microsphere technique23. However, whole organs must be harvested, dissolved, and analyzed, and this omits the possibility to perform histological or expression analysis. Hence, the ultrasound technique supports the principles of the 'three Rs' in animal research (reduce, refine, and replace) by Russell and Burch24. In addition, flow probes are suitable to monitor splanchnic blood flow in real-time and parallel to other hemodynamic parameters, while the colored microsphere technique requires integrating organ (mesenteric blood) flow over time. Moreover, colored microspheres, which usually have a diameter of 15 µm, require a normal distribution of micro-vessels with a diameter < 15 µm in the respective organs to avoid becoming trapped and immobile, which might be not the case in cirrhotic livers.
The main limitation of this method is the need for a state of unconsciousness and anesthesia during the hemodynamic characterization of the PHT syndrome in animals. The most common and widely used injection anesthesia ketamine/xylazine often requires redosing after 30–45 min to obtain a necessary depth of anesthesia25,26; this adds time pressure especially if troubleshooting is needed. Using inhalation anesthesia involves many advantages, but special equipment is required, and safety regulations related to volatile anesthetics must be followed. The depth of anesthesia can be rapidly adapted without interfering with the surgery procedures by adjusting the anesthesia concentration. The endotracheal tube secures airways especially after activation of salvation by ketamine and the ventilation ensures sufficient oxygenation and ventilation of the animal to lower the risk of anesthesia-induced death27. While ketamine/xylazine is still widely used, low-dose isoflurane anesthesia causes no significant changes of hemodynamic or cardiovascular parameters in rats28,29.
Local experience and regulations provide stateoftheart recommendations and best practices of anesthesia, and the researchers must continuously reconsider the type of anesthesia used to perform these hemodynamic assessments30. Future experiments might use telemetry with implanted wireless pressure transducers that will overcome the current limitations related to general anesthesia and allow hemodynamic characterization of conscious animals.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We thank the veterinarians, nurses, and animal keepers at the Center of Biomedical Research for their continuous support during our research projects. The authors acknowledge the important input of all reviewers of this protocol. Some of the research was funded by the "Young Science Award" of the Austrian Society of Gastroenterology and Hepatology (ÖGGH) to PS and the "Skoda Award" of the Austrian Society of Internal Medicine to TR.
Materials
| Instruments | |||
| LabChart 7 Pro software | ADInstruments, Colorado Springs, CO, USA | - | Software |
| ML870 PowerLab 8/30 | ADInstruments, Colorado Springs, CO, USA | - | Electronic multichannel recorder |
| MLT0380/D | ADInstruments, Colorado Springs, CO, USA | - | Pressure transducer (x2: for Portal Pressure and Arterial Pressure) |
| ML112 Quad Bridge Amplifier | ADInstruments, Colorado Springs, CO, USA | - | Bridge amplifier |
| TS420 | Transonic Systems Inc., Ithaca, NY, USA | - | Flowmeter module |
| Biological Research Apparatus 7025 | UGO BASILE S.R.L., Comerio, Italy | - | Ventilator |
| Vapor 2000 | Dräger Medical AG & Co. KG, Lübeck, Germany | - | Isofluran Vaporizer |
| Perivascular probes (rat) for Transonic systems (Superior Mesenteric Artery) | Transonic Systems Inc., Ithaca, NY, USA | #MA1PRB | Ultrasonic flow probe (1mm) |
| Perivascular probes (rat) for Transonic systems (Portal Vein) | Transonic Systems Inc., Ithaca, NY, USA | #MA2PSB | Ultrasonic flow probe (2mm) |
| 1st for intubation & 2nd for clean skin incisions | - | - | Mayo scissor [x2] |
| Metzenbaum scissor | - | - | - |
| Cuticle scissor | - | - | - |
| e.g. Adson Brown tissue forceps | - | - | Tissue Forceps |
| High precision 45° angle broad point forceps [x2] | - | - | - |
| Hemostat [x4] | - | - | - |
| e.g. Mikulicz peritoneal clamp | - | - | Curved clamp |
| e.g. Dieffenbach clamp | - | - | Micro clamp |
| e.g. micro spatula with flat ends, width 4 mm, | - | - | Micro metal spatula |
| for transbuccal suture at intubation | - | - | Needle holder |
| Scalpel grip | - | - | - |
| selfmade | - | - | Intubation desk |
| blut, flexible and with a suitable diameter for arterial cannula and venflow | - | - | Blunt steel wire |
| modified arterial line 20G with Flowstich | Becton Dickinson, Farady Road, Swindon, UK | #682245 | Arterial line |
| Heating pad | - | - | - |
| Rectal temerature probe | - | - | - |
| Saline heater | - | - | - |
| Laryngoscope (specific for animal size, e.g. rat) | - | - | - |
| Inductionbox for inhalation anesthesia | - | - | - |
| Scale (able to measure mg) | - | - | - |
| Hair clipper | - | - | - |
| Name | Company | Catalog Number | Comments |
| Consumables | |||
| e.g. modified BD Venflon Pro Safety 14GA | Becton Dickinson Infusion Therapy, AB, SE251 06 Helsingborg, Sweden | #393230 | Peripheral venous catheter (14G) |
| Fine-Bore Polyethylene Tubing, ID 0.58mm, OD 0.96mm, Portex, | Smiths Medical International Ltd., Kent, UK | #800/100/200 | Catheter tube (PE-50) |
| e.g. Omnifix-F Solo | B. Braun Melsungen AG, Melsungen, Germany | #9161406V | Syringe 1mL |
| e.g. Injekt Solo | B. Braun Melsungen AG, Melsungen, Germany | #4606051V | Syringe 5mL |
| e.g. Injekt Solo | B. Braun Melsungen AG, Melsungen, Germany | #4606205V | Syringe 20mL |
| e.g. BD Microlance 3, 18G – 1 1/2" | Becton Dickinson S.A., Fraga, Spain | #304622 | Cannula (18G) |
| e.g. BD Microlance 3, 23G – 1" | Becton Dickinson S.A., Fraga, Spain | #300800 | Cannula (23G) |
| e.g. BD Microlance 3, 30G – 1/2" | Becton Dickinson S.A., Fraga, Spain | #304000 | Cannula (30G) |
| e.g. Leukoplast S | BSN medical GmbH, Hamburg, Germany | #47619-00 | Adhesive tape |
| e.g. Gazin RK Mullkompressen (18x8cm) | Lohmann & Rauscher, Vienna, Austria | #10972 | Gauze compress (small) |
| e.g. Gazin RK Mullkompressen (5x5cm) | Lohmann & Rauscher, Vienna, Austria | #10961 | Gauze compress (big) |
| Silk Braided black, USP 4/0, EP 1.5 | SMI AG, St. Vith, Belgium | #2021-04 | Suture (Silk 4/0, EP 1.5) |
| e.g. Mersilk, 2-0 (3 Ph. Eur.), PS-1 Prime | Johnson & Johnson Medical GmbH – Ethicon Deutschland, Germany | #EH7552 | Transbuccal suture |
| e.g. Cottonbuds (2.2mm, 15cm) | Paul Hartmann AG, Heidenheim, Germany | #967936 | Cotton buds |
| e.g. Vue Ultrasoundgel | Optimum Medical Limited, UK | #1157 | Ultrasound gel |
| e.g. Glubran 2 | Gem srl, Viareggio, Italy | #G-NB2-50 | Tissue glue |
| e.g. Surgical scalpell knife Nr. 10 – carbon steel | Swann-Morton, England, B.S. | #202 | Scalpel Knife |
| Heparin, 5000 i.E./mL (Natriumheparin) | Medicamentum Pharma GmbH, Allerheiligen im Mürztal, Austria | - | Heparin |
| Florane | Aesica Queenborough Ltd., Queenborough, UK | - | Isoflurane |
| OeloVital (5g) | Fresenius Kabi Austira Gmbh, Graz, Austria | - | Eye gel |
| Ketasol | aniMedica GmbH, Senden-Bösensell, Germany | - | Ketamine |
| Rompun | Bayer Austria Ges.m.b.H., Vienna, Austria | - | Xylazine |
| Xylocain 10% Pumpspray | AstraZeneca Österreich GmbH, Vienna, Austria | - | Lidocaine pump spray |
| Dipidolor | Jansen-Cilag Pharma GmbH, Vienna, Austria | - | Piritramide |
| NaCl 0.9% Fresenius, 1L | Fresenius Kabi Austira GmbH, Graz, Austria | #13LIP132 | Physiological saline solution |
Riferimenti
- Ripoll, C., et al. Hepatic venous pressure gradient predicts clinical decompensation in patients with compensated cirrhosis. Gastroenterology. 133 (2), 481-488 (2007).
- Bosch, J., Groszmann, R. J., Shah, V. H. Evolution in the understanding of the pathophysiological basis of portal hypertension: How changes in paradigm are leading to successful new treatments. J Hepatol. 62, S121-S130 (2015).
- Blachier, M., Leleu, H., Peck-Radosavljevic, M., Valla, D. C., Roudot-Thoraval, F. The burden of liver disease in Europe: a review of available epidemiological data. J Hepatol. 58 (3), 593-608 (2013).
- Colle, I., Geerts, A. M., Van Steenkiste, C., Van Vlierberghe, H. Hemodynamic Changes in Splanchnic Blood Vessels in Portal Hypertension. Advances in Integrative Anatomy and Evolutionary Biology. 291 (6), 699-713 (2008).
- Laleman, W., Van Landeghem, L., Wilmer, A., Fevery, J., Nevens, F. Portal hypertension: from pathophysiology to clinical practice. Liver International. 25 (6), 1079-1090 (2005).
- Franchis, R. d. . Updating Consensus in Portal Hypertension: Report of the Baveno III Consensus Workshop on definitions, methodology and therapeutic strategies in portal hypertension. Journal of Hepatology. 33 (5), 846-852 (2000).
- Zardi, E. M., Di Matteo, F. M., Pacella, C. M., Sanyal, A. J. Invasive and non-invasive techniques for detecting portal hypertension and predicting variceral bleeding in cirrhosis: a review. Annals of medicine. 46 (1), 8-17 (2014).
- Kumar, A., Sharma, P., Sarin, S. K. Hepatic venous pressure gradient measurement: time to learn. Indian J Gastroenterol. 27 (2), 74-80 (2008).
- Tsochatzis, E. A., Bosch, J., Burroughs, A. K. Liver cirrhosis. Lancet. 383 (9930), 1749-1761 (2014).
- Abraldes, J. G., Pasarín, M., García-Pagán, J. C. Animal models of portal hypertension. World Journal of Gastroenterology : WJG. 12 (41), 6577-6584 (2006).
- Reiberger, T., et al. Sorafenib attenuates the portal hypertensive syndrome in partial portal vein ligated rats. Journal of Hepatology. 51 (5), 865-873 (2009).
- Schwabl, P., et al. Pioglitazone decreases portosystemic shunting by modulating inflammation and angiogenesis in cirrhotic and non-cirrhotic portal hypertensive rats. Journal of Hepatology. 60 (6), 1135-1142 (2014).
- Reiberger, T., et al. Nebivolol treatment increases splanchnic blood flow and portal pressure in cirrhotic rats via modulation of nitric oxide signalling. Liver International. 33 (4), 561-568 (2013).
- Schwabl, P., et al. The FXR agonist PX20606 ameliorates portal hypertension by targeting vascular remodelling and sinusoidal dysfunction. Journal of Hepatology. 66 (4), 724-733 (2017).
- Mandorfer, M., et al. Sustained virologic response to interferon-free therapies ameliorates HCV-induced portal hypertension. J Hepatol. 65 (4), 692-699 (2016).
- Schwabl, P., et al. Interferon-free regimens improve portal hypertension and histological necroinflammation in HIV/HCV patients with advanced liver disease. Aliment Pharmacol Ther. 45 (1), 139-149 (2017).
- Reiberger, T., Mandorfer, M. Beta adrenergic blockade and decompensated cirrhosis. Journal of Hepatology. 66 (4), 849-859 (2017).
- Reiberger, T., et al. Carvedilol for primary prophylaxis of variceal bleeding in cirrhotic patients with haemodynamic non-response to propranolol. Gut. 62 (11), 1634-1641 (2013).
- Reiberger, T., et al. Austrian consensus guidelines on the management and treatment of portal hypertension (Billroth III). Wiener klinische Wochenschrift. 129 (3), 135-158 (2017).
- de Franchis, R. Expanding consensus in portal hypertension. Journal of Hepatology. 63 (3), 743-752 (2015).
- Pinter, M., et al. The effects of sorafenib on the portal hypertensive syndrome in patients with liver cirrhosis and hepatocellular carcinoma – a pilot study. Alimentary Pharmacology & Therapeutics. 35 (1), 83-91 (2012).
- Schwabl, P., Laleman, W. Novel treatment options for portal hypertension. Gastroenterol Rep (Oxf). 5 (2), 90-103 (2017).
- Klein, S., Schierwagen, R., Uschner, F., Trebicka, J. . Mouse and Rat Models of Induction of Hepatic Fibrosis and Assessment of Portal Hypertension. , (2017).
- Russell, W. M. S., Burch, R. L. . The Principles of Humane Experimental Technique. , (1959).
- Langhans, W., Myrtha, A., Riediger, T., Lutz, T. A. . Routine animal use procedures. , (2016).
- Animal Care and Use Program. . Rat and Mouse anesthesia and analgesia: Formulary and General Drug Information. , (2016).
- Davis, J. A. . Current Protocols in Neuroscience. , (2001).
- Albrecht, M., Henke, J., Tacke, S., Markert, M., Guth, B. Effects of isoflurane, ketamine-xylazine and a combination of medetomidine, midazolam and fentanyl on physiological variables continuously measured by telemetry in Wistar rats. BMC Veterinary Research. 10 (1), 198 (2014).
- Redfors, B., Shao, Y., Omerovic, E. Influence of anesthetic agent, depth of anesthesia and body temperature on cardiovascular functional parameters in the rat. Laboratory Animals. 48 (1), 6-14 (2014).
- Becker, K., et al. . Statement on anesthesia methodologies: Recommondations on anaesthesia methodologies for animal experimentation in rodents and rabbits. , (2016).

