Microdissection of Primary Renal Tissue Segments and Incorporation with Novel Scaffold-free Construct Technology
Summary
Tissue-engineered renal constructs provide a solution for the organ shortage and deleterious effects of dialysis. Here, we describe a protocol to micro dissect murine kidneys for isolation of cortico-medullary segments. These segments are implanted into scaffold-free cellular constructs, forming renal organoids.
Abstract
Kidney transplantation is now a mainstream therapy for end-stage renal disease. However, with approximately 96,000 people on the waiting list and only one-fourth of these patients achieving transplantation, there is a dire need for alternatives for those with failing organs. In order to decrease the harmful consequences of dialysis along with the overall healthcare costs it incurs, active investigation is ongoing in search of alternative solutions to organ transplantation. Implantable tissue-engineered renal cellular constructs are one such feasible approach to replacing lost renal functionality. Here, described for the first time, is the microdissection of murine kidneys for isolation of living corticomedullary renal segments. These segments are capable of rapid incorporation within scaffold-free endothelial-fibroblast constructs which may enable rapid connection with host vasculature once implanted. Adult mouse kidneys were procured from living donors, followed by stereoscope microdissection to obtain renal segments 200 – 300 µm in diameter. Multiple renal constructs were fabricated using primary renal segments harvested from only one kidney. This method demonstrates a procedure which could salvage functional renal tissue from organs that would otherwise be discarded.
Introduction
Chronic kidney disease (CKD) is one of the current major public health challenges worldwide1. The prevalence of CKD in the United States is over 14% of the total population, with over 600,000 Americans suffering from the most severe form, end-stage renal disease (ESRD)2. The current treatment options available for those with ESRD include dialysis and kidney transplantation. Although approximately 25,000 patients undergo renal transplantation each year, a significant number of patients are added annually leading to a large disparity between those awaiting a life-saving organ and those receiving transplantation3. In addition to its serious negative effects on longevity and quality of life, dialysis is associated with an astonishing financial burden. In 2014, Medicare paid claims totaled over $30 billion for ESRD patients2. With a limited organ supply and no apparent downtrend in patients requiring dialysis, research efforts aimed at identifying alternative solutions to dialysis and transplantation are ever important. Even a relatively short delay in the need for dialysis increases a patient's number of quality-adjusted life years and productivity substantially while postponing dialysis-related costs4,5,6.
Solutions for functional tissue loss, like that in ESRD, are currently being studied in tissue engineering and regenerative medicine laboratories, with widely varied approaches ranging from scaffold-based organoid fabrication to whole organ engineering using decellularized organ structures for cellular implantation7,8,9,10,11. Recapitulating complex renal structures from marginal or discarded kidneys has only partially been investigated. In fact, nearly 20% of kidneys procured for transplantation are discarded for various reasons12,13. The functional renal tissue from these putative grafts could be utilized and incorporated into one or many tissue-engineered constructs. Prior studies have demonstrated the feasibility of working with these discarded organs, utilizing kidneys for the extra-cellular matrix for tissue engineering purposes14,15. However, few have used primary nephronal tissue from healthy kidneys for tissue-engineering purposes16,17,18.
One method previously described by Kim et al. involves isolation of renal "segments" from healthy rat kidneys, which were then seeded on polyglycolic acid (PGA) scaffolds for construct fabrication16. However, little information is given regarding exact dissection methodology, and segments were obtained from a combination of fine mincing and filtration. We describe a modification of this protocol, which similarly produces discrete renal segments with intact nephronal architecture, but instead relies on microdissection techniques. Nephrectomies are performed on living adult mice, after which the kidneys are transferred to the dissection microscope where the renal capsule is removed, and the tissue is further dissected. Small-bore 30½ G needles are used as cutting instruments and also as guides aiding in dissection, as the needle diameter is equal to the target diameter of the renal segments. The isolated, in this case murine, renal segments maintain viability in culture and incorporate with scaffold-free endothelial-fibroblast cellular constructs19. These constructs have previously been used to engineer other organs, including a bio-artificial pancreas20.
Protocol
All animal surgical procedures described below were approved by the Institutional Animal Care and Use Committee (IACUC) at the Medical University of South Carolina prior to any animal surgeries or use of any animal tissues.
1. Murine Nephrectomy
- Don a surgical mask and bouffant cap to minimize the risk of contamination. Maintain sterility during set-up of the surgical area.
- Place non-fenestrated surgical drapes on the operating table.
- Open the pack of autoclaved instruments onto the sterile drapes. The instruments required for nephrectomy include 3 small hemostats, fine forceps with teeth, fine forceps without teeth, and straight iris scissors.
- Place another non-fenestrated surgical drape in the center of the operating table.
- Prepare 5 mL of Dulbecco's Phosphate Buffered Saline (DPBS) with calcium and magnesium, supplemented with 1% penicillin/streptomycin in a 15-mL sterile conical tube. Place this solution on ice next to the operating table.
- Obtain an 8 – 12 week old C57BL/6 mouse (male or female) from the animal housing complex.
- Place the mouse inside an anesthesia induction chamber and induce with 3.5% inhaled isoflurane. Place a nose cone on the mouse, using 2% isoflurane for maintenance anesthesia.
- Monitor for adequate depth of anesthesia periodically throughout the surgical procedure by assessing for pedal reflex (firm toe pinch).
- Remove all of the hair from the ventral torso using hair removal cream. Rinse this area with distilled water to ensure all hair has been removed from the abdomen.
- Transfer the mouse to the sterile operating table in supine position under the top non-fenestrated drape. Cut out a 2 x 2 cm2 fenestration in the drape just overlying the mouse abdomen.
- Apply povidone-iodine followed by 70% ethanol to the abdomen using sterile gauze or cotton balls.
- Using fine forceps with teeth and iris scissors, make a 3 – 4 cm vertical abdominal skin incision parallel to the midline, 0.5 cm to the left of midline
Note: If attempting to remove the right kidney, this will be an incision 0.5 cm to the right of midline.- Grasp the peritoneum with fine forceps without teeth and incise with iris scissors to enter the abdominal cavity. Apply hemostats to the superior and inferior lateral edges of skin and peritoneum to place tension laterally and enhance exposure.
- Shift the intestinal contents delicately to the right side of the abdomen to expose the left kidney. Gently place traction on the kidney to elevate it out of the abdomen.
- Place a hemostat across the hilum of the left kidney. Use iris scissors to transect the ureter and renal vessels.
- Place the left kidney in the 1% Penicillin/Streptomycin DPBS Solution on ice.
- Transfer the tube containing the kidney to the sterile tissue culture hood. Transfer the kidney from the 15-mL conical tube to a 60-mm petri dish (Figure 1). Rinse twice using 5 mL DPBS with calcium and magnesium. Keep the kidney suspended in 5 mL DPBS in the 60-mm dish.
- Prepare an additional 60 mm petri dish with 5 mL DPBS to be used during the microdissection protocol.
- Euthanize the mouse by thoracotomy and exsanguination following the procurement of the kidney, according to an approved IACUC animal protocol.
2. Murine Kidney Microdissection for Renal Segment Isolation
- Continue using sterile technique for this portion of the protocol, including sterile gloves, surgical mask, and bouffant to minimize contamination risk.
- Place sterile drapes on the stereo microscope platform as well as to the areas just left and right of the microscope to have a sterile field for instruments. Place plastic adhesive sheets on microscope focus knobs and spray with 70% ethanol. Turn on the dual gooseneck illuminator to light the stage.
- Open sterilized instruments (fine forceps without teeth, scalpel handle, #15 scalpel blade, two straight hemostats, two 30½ gauge hollow-bore needles) onto sterile drapes. Place the #15 blade onto the scalpel handle now and attach one hemostat to each needle adaptor/hub to create needle dissection instruments for later use, as shown in Figure 1.
- Move the 60-mm petri dishes, one containing the kidney in DPBS and the other only DPBS, from the tissue culture hood to the microscope area.
- Place the 60-mm petri dish under the objective and remove the lid to expose the kidney. Leave the DPBS-containing dish to the side on the sterile field for later use.
- Move the dish so that only the anterior or posterior surface of the kidney is in view (i.e., the large flat face of the kidney, with the other face touching the bottom of the dish). Zoom to 3.2 – 4X for this portion of the procedure. Using the non-dominant hand, use fine forceps without teeth to pierce and pin the kidney against the dish near the inferior and superior poles of the kidney.
- Keep the non-dominant hand in place to pin the kidney. With the dominant hand, use the #15 blade to remove the translucent fibrous capsular layer from the exposed surface. Shave the most superficial 0.5 mm from the exposed surface of the kidney to remove the remainder of the capsule.
- Use the #15 blade to dissect an inverted pyramid of tissue from the decapsulated area, approximately 2 mm3 in size. Retrieve the 60-mm dish containing only DPBS and place the pyramid of tissue into the clean dish. Discard the remainder of the kidney.
- Decrease magnification to 1.5 – 2.0X for the rest of the dissection. Using two hemostat-needle instruments as cutting instruments, slice the tissue fragment into progressively smaller pieces until the tissue segments are less than or equal to the diameter of the needle tips. A renal tissue 2 mm3 in size will produce more than 50 segments once completely dissected.
- Move the 60-mm dish with renal segments to the tissue culture hood. Remove DPBS with 1000 µL pipette. Add 5 mL DMEM, supplemented with 10% Fetal Bovine Serum (FBS) and 1% penicillin/streptomycin.
- Culture for 3 days at 37 °C in an incubator or implant into cellular constructs immediately.
3. Renal Segment Cellular Construct Fabrication
- Culture normal human dermal fibroblasts (NHDF) and human adipose microvascular endothelial cells (HAMEC) for several weeks to obtain 7.2 x 105 fibroblasts and 1.8 x 105 endothelial cells per construct desired. In the tissue culture hood, seed the appropriate ratio of fibroblasts to endothelial cells (4:1) into rod-shaped wells in agarose molds to form scaffold-free pre-vascular endothelial-fibroblast constructs (SPEC) as previously described by Czajka et al19.
- Obtain a sterilized hemostat, 30½ G needle, and a sterile spatula with flat end.
- Remove the majority of the media from the 60-mm plate containing renal segments, being careful not to aspirate and remove the renal segments.
- Equip surgical Loupes to visualize implantation of the segments.
- Scrape the end of the spatula gently along the bottom of the 60-mm dish to obtain 10 – 15 segments, spread out evenly along the very tip of the spatula. Place the tip of the spatula as close as possible to the lip of the well where the SPEC was seeded.
- Use a hemostat-30½ G needle instrument in the other hand to move each individual segment from the spatula tip to the edge of the well. Gently move each segment down into the well using the needle tip until slightly submerged in the previously seeded cellular suspension.
- Culture renal segment-SPEC in 2:1:1 ratio of FGM-2/EGM-2/DMEM medium (2.5 mL volume total). Change culture media every 24 h for 3 days. Remove the constructs from the molds at 72 h and fix or flash-freeze for processing.
Representative Results
The protocol described produces approximately 50 renal segments per pyramidal 2 mm3 section of renal tissue. The renal segments that have been processed and imaged have tubular and glomerular components in differing proportions (see Figure 2). The intact segments were subjected to an assay in order to determine the viability of different segments once every 24 h for three days. Green-fluorescent calcein-AM is present with intracellular esterase activity, indicative of living cells. Red-fluorescent ethidium homodimer-1 is seen with loss of the integrity of the plasma membrane. Though these segments are intact, three-dimensional structures, significant assay penetration is appreciable with confocal microscopy (see Figure 3).
Segments were further characterized for size uniformity using the same cell viability assay. Using glass slides with depressions, segments were placed under cover slips without mechanical force placed on any dimension of the segments. Free floating, the segments were imaged 30 minutes after placement in the cell viability/toxicity assay. A z-projection with 10-µm step size was taken through the entire segment. The largest 'X' and 'Y' dimensions were obtained and recorded. The 'Z' dimension was obtained by calculating the distance from the first to the last visible fluorophore. Given that the segments are roughly cubic, a simple cubic volume measurement was obtained and plotted to compare 10 randomly selected segments from one microdissection experiment. Target volume of (300 µm)3 = 0.027 mm3 is shown with a red marker on the histogram (Figure 4A). Representative 'X' and 'Y' measurements are shown in Figure 4B. Although there are outliers, there are many segments close to the target volume. Repeat experimentation has shown consistent results (n = 20). The raw measurements (not shown here) show that in the vast majority of segments, there is at least one dimension measuring 200 – 300 µm. This has important implications for diffusion limitations.
The renal segments are embedded in scaffold-free endothelial-fibroblast constructs and cultured for 3 days. The renal segments incorporate with the cellular constructs to form intact structures by day 3 (see Figure 5A). The constructs maintain their pre-vascular endothelial network, shown by labeling with von Willebrand Factor (Figure 5E). However, it does not appear that the network invades the renal cellular material. Renal epithelial cells are labeled with Cytokeratin-18 (Figure 5B, green). These constructs were incubated with FITC-labeled albumin (Figure 5C, gray) in order to test in vitro renal functionality. While there is residual albumin found away from the segments of embedded renal tissue, there are "hot spots" in the area of the renal tubular epithelial cells, those known to take up albumin that traverses intra-luminally. This is thought to represent albumin reuptake in the renal segment cellular construct.
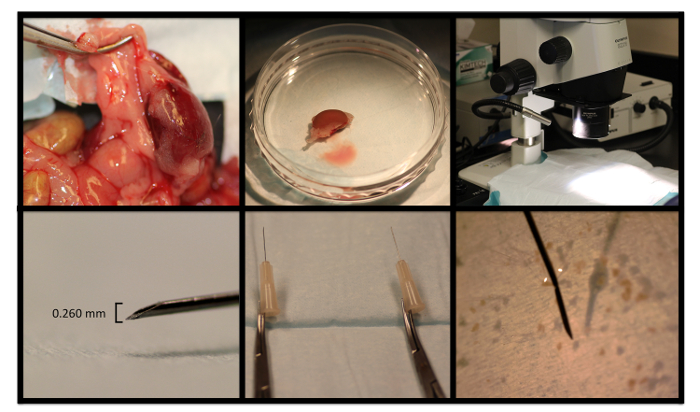
Figure 1: Representative Photographs from Nephrectomy and Renal Segment Microdissection. A murine kidney is removed, rinsed, and placed in a 60-mm dish, and moved to the stereoscope microscope stage. The diameter of the needle tip is used to guide dissection. Hemostats are attached to needle tips for the dissection instruments. Please click here to view a larger version of this figure.
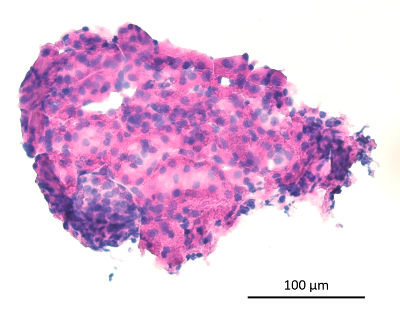
Figure 2: Microdissected Renal Segment. The microdissected renal segments contain varying proportions of glomeruli (lower left corner, basophilic structure) and tubules (remainder of the image) in their native arrangement. 10X image, scale bar as indicated. Please click here to view a larger version of this figure.
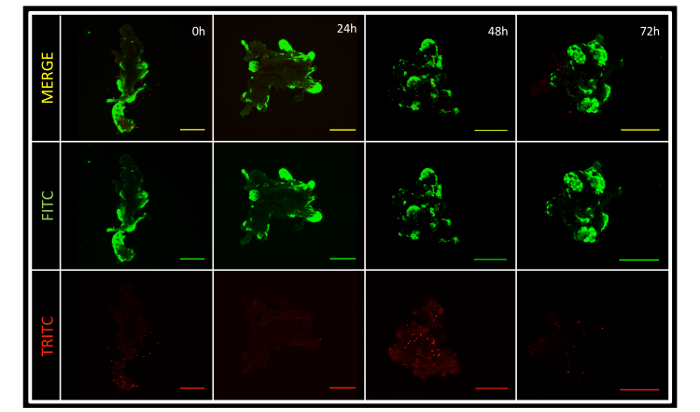
Figure 3: Renal Segment Viability. The microdissected renal segments contain portions of live and dead tissue. Predominantly, the segments are living, and remain so in the culture at 72 h. Note that different segments were used for the different time points, as the viability assay itself is toxic to cells. 10X images, Scale bars = 200 µm. Please click here to view a larger version of this figure.
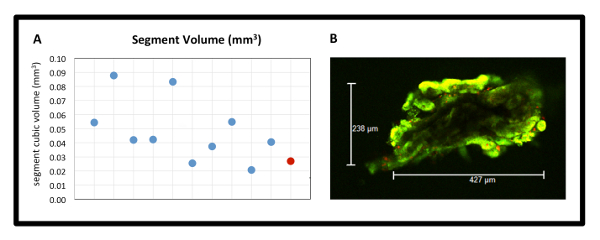
Figure 4: Renal Segment Volume Uniformity. (A) Segment cubic volume in mm3 from one dissection experiment (n = 10). Individual segments are represented by blue points, in comparison to target red point (target volume 0.027 mm3). (B) Representative X and Y dimension measurements from the live-dead assay, 10X image. Please click here to view a larger version of this figure.
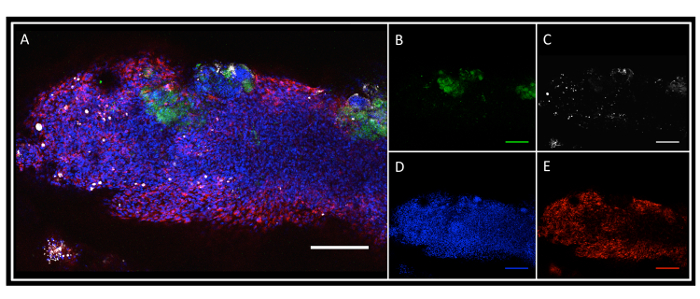
Figure 5: Tissue-Engineered Scaffold-Free Renal Segment Construct. (A) Merged image showing incorporation of renal segments into the SPEC. (B) Cytokeratin-18-positive renal tubular epithelial cells (green). (C) FITC-labeled albumin. (D) Hoescht stain for nuclei. (E) von-Willebrand factor (vWF) positivity highlighting the prevascular network. 10X images, Scale Bar = 200 µm. Please click here to view a larger version of this figure.
Discussion
Methods used to engineer living renal tissue constructs vary widely with regard to both the type of cells and biomaterials utilized, and in many cases, are outdated or not well-characterized in the literature7. While many are using stem cell approaches or recapitulating individual components of the renal architecture in isolation, the prospect of artificially recreating an entire organ with over 26 different differentiated cell types from cellular suspensions is overwhelming to consider21. This is likely why others have moved to approaches using segments of renal tissue in tissue engineering applications. As described above, previous work has been done in isolation of renal segments for seeding on PGA scaffolds16. However, the methodology used to describe the isolation of segments was described in little detail and the segments were not cultured for any period of time to assess for viability. The method described here, along with the use of the cellular viability assay, shows that at the minimum, portions of the renal segments are maintaining viability to the 72-h mark, which has not been described before. The method also has the advantage of producing segments with fairly predictable dimensions. Additionally, these methods provide a clinically translatable approach to augmenting failing renal function, primarily demonstrating a feasible method for isolation of segments of primary renal tissue from organs that would otherwise be discarded.
In developing the method, the greatest challenges were, first, identifying a sterile object of the appropriate size, and second, maintaining sterility as the procedure involved many steps outside of the cell culture hood. 30½ G needle tips proved to be useful measurement as well as dissection devices. Sterility was a challenge early in experimentation, mostly related to surgical set-up and controlling murine fur.
Limitations of the procedure are related to the three-dimensional nature of the renal tissue segments. In one view under the microscope, the dimensions of the segment may appear to be roughly the size of the needle diameter, 0.260 mm. However, the Z-dimension is not visible on a stereoscope. The microdissection is done by hand, which itself is another limitation. In the future, robotic standardization would be ideal. If the 2 mm3 pyramidal portion of renal tissue was maintained in the correct orientation during dissection, dissecting out intact nephrons would not be unfathomable. Finally, comparing these constructs to the bio-artificial pancreas, in which islet cells were embedded in the SPEC, one must acknowledge key differences20. The kidney is functionally and architecturally unique from the pancreas and arguably more complex. Additionally, islet cells are only one subtype of functional cell within the pancreas, as opposed to the conglomerate of functional cells isolated with renal segments in this method. With architecture and specifically directionality playing such a large role in the function of the kidney and particularly filtration, the lack of uniform orientation of the segments within the construct is an additional weakness of this methodology.
Ongoing research is aimed at assessment of renal segment and renal construct functionality, to clarify the extent to which the albumin uptake is occurring and evaluate other renal functions. Future animal experiments would involve the implantation of the renal constructs in the bladder wall. Embedding the construct in this highly vascular bodily structure would allow rapid anastomosis as well as obviate the need for a collection system, with filtrate excreting directly into the bladder. Considering that in vitro functionality of renal tissues is extremely limited, in vivo testing is vital. The only in vitro tests currently reported in the literature are centered around albumin uptake and erythropoietin expression17,22. These living segments of intact renal tissue in its native architecture embedded in constructs with primitive endothelial networks, which do not rely on any foreign biomaterials, show promise in moving towards functional, biocompatible renal tissue replacement.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
NIH Institutional Postdoctoral Training Grant, NIH-HL-007260
Materials
| Non-fenestrated Sterile Field | Busse Hospital Disposables | 696 | |
| Fenestrated Sterile Field | Busse Hospital Disposables | 697 | |
| Halsted Mosquito Forceps 5 Curved | Miltex | Mil-7-4 | "Hemostat" in manuscript |
| Extra Fine Graefe Forceps, Curved with teeth | Fine Science Tools | 11155-10 | Fine forceps with teeth |
| Extra Fine Graefe Forceps, Serrated (without teeth) | Fine Science Tools | 11152-10 | Fine forceps without teeth |
| Fine Scissors – Tungsten Carbide | Fine Science Tools | 14568-09 | Iris Scissors |
| Betadine Surgical Scrub with Pump, Povidone-iodine 7.5% | Purdue Products L.P. | 67618-151-17 | |
| Sterile Cotton Gauze Pad (4" x 4") | Fisher Healthcare | 22-415-469 | |
| Dulbecco's Phosphate Buffered Solution | Corning | 21-030-CV | |
| Penicillin/Streptomycin Solution, 100X | Corning | 30-002-Cl | |
| Isoflurane, USP | Manufacturer: Piramal, Distributor: McKesson | 2254845 | |
| Nair Hair Remover | Nair | 22600-23307 | Hair Removal Cream in text |
| 200 Proof Ethanol | Decon Laboratories | 2705 | Diluted to 70% Ethanol Solution |
| BioLite 60mm Tissue Culture Dish | Themo-Scientific | 130181 | |
| Press'n Seal | Glad | 12587-70441 | Applied to Stereoscope |
| SZX16 Stereo Microscope | Olympus | SZX16 | |
| Fiber Optic Illuminator | Cole Parmer | 41720-20 | |
| Self-Supporting Dual-Light Pipe, 23" L Gooseneck | Cole Parmer | EW-41720-60 | |
| Scalpel Handle #3 | Miltex | Mil-4-7 | |
| Sterile Rib-Back Carbon Steel Blade, Blade Size 15 | Bard-Parker | 371115 | |
| 31 1/2 Gauge Needle | ThermoFisher Scientific | 14-826F | Becton Dickinson 305106 |
| Dulbecco's Modified Eagle's Medium | Corning | 10-017-CV | |
| Fetal Select 100% Bovine Serum | Atlas Biologicals | FS-0500-AD | |
| Normal Human Dermal Fibroblasts | Lonza | CC-2511 | |
| Human Adipose Microvascular Endothelial Cells | Sciencell Research Laboratories | 7200 | |
| Surgical Loupes (2.5x) | Orascoptic | (N/A) Custom Order | |
| FGM-2 (Fibroblast Basal Medium with FGM-2 SingleQuots Added) | Lonza | CC-3131, CC-4126 | |
| EGM-2 (Endothelial Basal Medium with EGM-2 SingleQuots Added) | Lonza | CC-3156, CC-4176 | |
| Live/Dead Viability/Cytotoxicity Kit for Mammalian Cells | ThermoFisher Scientific | L3224 | |
| Anti-Cytokeratin-18 Antibody | Abcam | ab668 | |
| Goat anti-Mouse IgG, Alexa Fluor 633 | ThermoFisher Scientific | A-21052 | |
| Goat anti-Rabbit IgG, Alexa Fluor 546 | ThermoFisher Scientific | A-11010 | |
| Anti-Von Willebrand Factor Antibody | Abcam | ab6994 | |
| Albumin, Fluorescein isothiocyanate Conjugate | Sigma Aldrich | A9771-50MG | |
| Hoescht 33342 | BD Pharmingen | 561908 | |
| Background Buster | Innovex Biosciences | NB306 |
Riferimenti
- Jha, V., et al. Chronic kidney disease: global dimension and perspectives. Lancet. 382 (9888), 260-272 (2013).
- 2016 USRDS Annual Data Report: Epidemiology of Kidney Disease in the United States. U.S.R.D Available from: https://www.usrds.org/2016/download/v2_ESRD_16.pdf (2016)
- Hart, A., et al. OPTN/SRTR 2015 Annual Data Report: Kidney. American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons. 17, 21-116 (2017).
- de Vries, E. F., Rabelink, T. J., van den Hout, W. B. Modelling the Cost-Effectiveness of Delaying End-Stage Renal Disease. Nephron. 133 (2), 89-97 (2016).
- Lefebvre, P., Duh, M. S., Mody, S. H., Bookhart, B., Piech, C. T. The economic impact of epoetin alfa therapy on delaying time to dialysis in elderly patients with chronic kidney disease. Disease management : DM. 10 (1), 37-45 (2007).
- Mennini, F. S., Russo, S., Marcellusi, A., Quintaliani, G., Fouque, D. Economic effects of treatment of chronic kidney disease with low-protein diet. Journal of renal nutrition : the official journal of the Council on Renal Nutrition of the National Kidney Foundation. 24 (5), 313-321 (2014).
- Moon, K. H., Ko, I. K., Yoo, J. J., Atala, A. Kidney diseases and tissue engineering. Methods. 99, 112-119 (2016).
- Wobma, H., Vunjak-Novakovic, G. Tissue Engineering and Regenerative Medicine 2015: A Year in Review. Tissue engineering. Part B, Reviews. 22 (2), 101-113 (2016).
- Langer, R., Vacanti, J. Advances in tissue engineering. Journal of pediatric surgery. 51 (1), 8-12 (2016).
- Jakab, K., et al. Tissue engineering by self-assembly and bio-printing of living cells. Biofabrication. 2 (2), 022001 (2010).
- Fisher, M. B., Mauck, R. L. Tissue engineering and regenerative medicine: recent innovations and the transition to translation. Tissue engineering. Part B, Reviews. 19 (1), 1-13 (2013).
- Stewart, D. E., Garcia, V. C., Rosendale, J. D., Klassen, D. K., Carrico, B. J. Diagnosing the Decades-Long Rise in the Deceased Donor Kidney Discard Rate in the United States. Transplantation. 101 (3), 575-587 (2017).
- Mohan, S., et al. The weekend effect alters the procurement and discard rates of deceased donor kidneys in the United States. Kidney international. 90 (1), 157-163 (2016).
- Orlando, G., et al. Discarded human kidneys as a source of ECM scaffold for kidney regeneration technologies. Biomaterials. 34 (24), 5915-5925 (2013).
- Katari, R., et al. Renal bioengineering with scaffolds generated from human kidneys. Nephron. Experimental nephrology. 126 (2), 119 (2014).
- Kim, S. S., Park, H. J., Han, J., Choi, C. Y., Kim, B. S. Renal tissue reconstitution by the implantation of renal segments on biodegradable polymer scaffolds. Biotechnology letters. 25 (18), 1505-1508 (2003).
- Guimaraes-Souza, N. K., Yamaleyeva, L. M., AbouShwareb, T., Atala, A., Yoo, J. J. In vitro reconstitution of human kidney structures for renal cell therapy. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association – European Renal Association. 27 (8), 3082-3090 (2012).
- Kelley, R., et al. Tubular cell-enriched subpopulation of primary renal cells improves survival and augments kidney function in rodent model of chronic kidney disease. American journal of physiology. Renal physiology. 299 (5), 1026-1039 (2010).
- Czajka, C. A., Drake, C. J. Self-assembly of prevascular tissues from endothelial and fibroblast cells under scaffold-free, nonadherent conditions. Tissue engineering. Part A. 21 (1-2), 277-287 (2015).
- Rhett, J. M., Wang, H., Bainbridge, H., Song, L., Yost, M. J. Connexin-Based Therapeutics and Tissue Engineering Approaches to the Amelioration of Chronic Pancreatitis and Type I Diabetes: Construction and Characterization of a Novel Prevascularized Bioartificial Pancreas. Journal of diabetes research. 2016, 7262680 (2016).
- Al-Awqati, Q., Oliver, J. A. Stem cells in the kidney. Kidney international. 61 (2), 387-395 (2002).
- Aboushwareb, T., et al. Erythropoietin producing cells for potential cell therapy. World journal of urology. 26 (4), 295-300 (2008).

