A Converging Strategy for the Generation of a Virtually Sequenced cDNA Library from Unreferenced Pacific Oysters
Summary
We describe a strategy for how to use RNA samples from unreferenced Pacific oyster specimens, and evaluate the genetic material by comparison with publicly available genome data to generate a virtually sequenced cDNA library.
Abstract
The access to biological material of reference species, which were used previously in key experiments such as in the development of novel cell lines or genome sequencing projects, are often difficult to provide for further studies or third parties due to the consumptive nature of the samples. Although now widely distributed over the Pacific coasts of Asia, Australia and North America, individual Pacific oyster specimens are genetically quite diverse and are therefore not directly suitable as the starting material for gene libraries. In this article, we demonstrate the use of unreferenced Pacific oyster specimens obtained from regional seafood markets to generate cDNA libraries. These libraries were then compared to the publicly available oyster genome, and the closest related library was selected using the mitochondrial reference genes Cytochrome C Oxidase subunit I (COX1) and NADH Dehydrogenase (ND). The suitability of the generated cDNA library is also demonstrated by cloning and expression of two genes encoding the enzymes UDP-glucuronic acid dehydrogenase (UGD) and UDP-xylose synthase (UXS), which are responsible for the biosynthesis of UDP-xylose from UDP-glucose.
Introduction
The acquisition of living referenced biological material may be challenging due to long delivery times, entrepreneurial reasoning, or country-specific customs regulations. As an alternative, the required biological material may be also collected from phenotypically identical specimens. However, these samples may vary significantly at the level of genotype, and therefore comparisons with digitally-stored reference genomes of the same species are often rendered difficult or even futile due to incompatibility of the newly-sourced material with existing DNA amplification methods. Sequencing highly conserved genes of individual samples is a widely used and powerful tool for identifying species1, such as conserved mitochondrial genes that are frequently used as reference genes for the quality assessment of cDNA libraries2,3,4,5,6. The underlying rationale for the herein presented method is that high conservation of mitochondrial gene sequences in individual anonymous oyster samples compared to the corresponding sequences of the reference genome indicates that other genes may also show a low level of divergence, given the generally faster rate of mitochondrial DNA evolution relative to nuclear DNA7, allowing the amplification and isolation of a wide range of scientifically and industrially relevant genes by simply using publicly available sequencing data as a reference.
The overall goal of the herein described method is to present an optimized workflow for generating a virtually sequenced oyster cDNA library which can be used as template DNA for the cloning of oyster genes. In virtual sequencing, de novo genome sequencing is circumvented; instead, a known, digitally-stored reference sequence is used directly to utilize or design primers for production of cDNAs that will eventually comprise a library (or be added to a pre-existing one). The aim is to produce a convergent cDNA library, meaning that similarities between the generated cDNA sequences and the reference sequence can be ranked from low to high divergence. A key advantage of using Cytochrome C Oxidase subunit 1 (COX1) and NADH Dehydrogenase (ND) as reference genes is that even highly geographically disjunct oyster specimens can be profiled due to the high conservation of these mitochondrial genes. Having proven the approach with these well-established markers, we then demonstrate its application to two enzyme candidates which are involved in sugar nucleotide biosynthesis and may be of industrial relevance8,9,10. The biotechnological potential of the Pacific oyster is still unexplored. Thus, we believe that this converging method for preparing a virtually sequenced cDNA library will also be appropriate for non-specialist researchers who want to generate cDNA from this relevant biological material.
Protocol
NOTE: A schematic overview is shown in Figure 1.
1. Sample collection
- Obtain oyster specimens. Keep oysters on ice during the postharvest period, the transport and prior to laboratory use and process within 4-7 days after purchase.
NOTE: For this protocol, oysters were purchased from Zhong Cai Wholesale Market in Nanjing (originating from Ningde, Fujian, China and Lianyungang, Jiangsu, China), Haijie Aquatic Product Company in Qingdao (originating from Qingdao, Shandong, China), Jucheng Aquatic Product Company in Yantai (originating from Yantai, Shandong, China) and Jinxiu Aquatic Product Company in Qingdao (originating from Qingdao, Shandong, China)).
2. RNA isolation by guanidinium thiocyanate-phenol extraction
- Preparation of oyster tissue sample
- Cut out approximately 100 mg of homogeneous soft tissue from the approximate geometric center of each oyster specimen with a sterilized scalpel, and transfer the samples into liquid nitrogen.
- Euthanize the remaining part of the oysters by freezing at -80 °C for 1 h and discard as biological waste.
- Grind the flash-frozen oyster tissue into a fine powder in a mortar (200 mL) filled with 50 mL of liquid nitrogen.
- Weigh out 75 mg of each specimen’s frozen tissue into a sterile 1.5 mL centrifuge tube and mix with 1 mL of the guanidinium thiocyanate-phenol extraction reagent. Centrifuge the sample at 14,000 x g at 4 °C for 15 min.
- Transfer the supernatant to a new 1.5 mL centrifuge tube, add 200 µL of chloroform and mix thoroughly by using a vortex mixer for 10-15 s until the mixture turns milky-white.
- Centrifuge at 14,000 x g at 4 °C for 15 min, and transfer the upper aqueous layer carefully with a 200 µL pipette without disturbing the interphase into a new 1.5 mL centrifuge tube.
- Add 500 µL of isopropyl alcohol and mix the samples gently by inversion, then leave the samples for 20 min on ice. Centrifuge at 14,000 x g at 4 °C for 8 min and remove the supernatant.
- Resuspend each of the pellets in 1 mL of 75% EtOH, and centrifuge at 14,000 x g at 4 °C for 5 min. Remove all of the supernatant.
- Repeat step 2.5 once. Dry the pellets for 6 min at room temperature. Do not dry for longer; otherwise, it may be difficult to dissolve the RNA pellet in the next step.
- Dissolve the dried RNA pellet in 25 µL of DEPC (diethylpyrocarbonate)-treated water and keep the tube on ice. Use RNA samples within 24 h.
3. cDNA library generation by reverse transcription
- For each RNA sample, prepare a reaction mixture using the a commercial reverse transcription system using a 10 µL pipette: Add 4 µL of MgCl2 solution, 2 µL of 10x Reaction Buffer, 2 µL of dNTP solution, 0.5 µL of RNase inhibitor, 0.7 µL of AMV Reverse Transcriptase, 0.5 µL of Oligo(dT) 15 primer, 1 µL of the extracted RNA sample and 9.3 µL of H2O into a 300 µL PCR tube.
- Incubate mixture in a PCR Thermocycler for 60 min at 42 °C, and then increase the temperature to 95 °C for 5 min.
- Store the generated cDNA library for up to 12 months at -20 °C.
4. Mitochondrial gene amplification and purification
- Prepare PCR mixture using a 10 µL pipette. Add 0.25 µL (1.25 U) of high-fidelity DNA polymerase, 2 µL of dNTP solution (2.5 mM of each dNTP), 0.5 µL of the COX1 or ND forward primer (100 µM), 0.5 µL of the corresponding COX1 or ND reverse primer (100 µM), 1 µL of the cDNA library, 5 µL of 5x buffer solution and 16 µL of distilled H2O into a 300 µL PCR tube.
- Perform the PCR amplification using the following parameters: After an initial denaturation step at 95 °C (duration 5 min), 35 PCR reaction cycles consisting of an annealing step at 55 °C (30 s), elongation step at 72 °C (2 min), and denaturation step at 95 °C (30 s), perform one finalizing elongation step for 5 min at 72 °C.
- Use 5 µL of the PCR product to verify by agarose gel electrophoresis the quality of the obtained PCR product. Observe the amplified COX1 or ND gene as a single band at either 759 or 748 base pairs, respectively.
- Purify the rest of the PCR product with a PCR clean-up kit.
- Add 100 µL of solution 'PCR-A' (DNA binding buffer which contains high concentrations of chaotropic salts11) to the sample. Vortex briefly to mix the contents.
- Place the purification column into a 2 mL centrifuge tube. Pipette the reaction mixture of 4.4.1. into the column. Centrifuge at 14,000 x g for 1 min at room temperature.
- Discard the filtrate from the centrifuge tube. Return the column to the 2 mL centrifuge tube, add 700 µL of solution 'W2' into the column and centrifuge at 14,000 x g for 1 min at room temperature ('W2' is a washing solution which contains high concentrations of ethanol for the removal of residual chaotropic salts from the purification column).
- Discard the filtrate and return the column to the 2 mL centrifuge tube. Add 400 µL of solution 'W2' to the column and centrifuge at 14,000 x g for 1 min at room temperature.
- Pre-heat 1 mL of deionized water to 65 °C in a metal block heater. Transfer the column into a new 1.5 mL centrifuge tube. Pipette 25 µL of the 65 °C hot pre-heated deionized water to the center of the white column membrane. Let the membrane soak for 1 min at room temperature.
- Centrifuge at 14,000 x g for 1 min at room temperature and discard the column.
- Store the purified PCR product for up to 12 months at -20 °C.
5. Mitochondrial gene sequencing and comparison
- Send the purified PCR samples from step 4.5 for Sanger sequencing using the relevant COX1 or ND forward primers as sequencing primers. Optionally, the COX1 or ND reverse primers can also be used for bidirectional sequencing.
- After retrieving the sequencing results, compare the sequences with the genome sequence of the Pacific oyster reference strain (NCBI Taxonomy ID: 29159) using the NCBI Nucleotide BLAST online tool (blast.ncbi.nlm.nih.gov) (Figure 2 and Figure 3).
6. Applying the cDNA library for cloning the genes MgUGD and MgUXS
- Amplify and purify the MgUGD and MgUXS genes using the respective forward and reverse primers of MgUGD and MgUXS by PCR following steps 4.1. to 4.4.
- Transfer 2 µL of digestion buffer (10x concentrated) and 6 µL of deionized water into a 1.5 mL centrifuge tube. Add 10 µL of the purified MgUGD or MgUXS PCR products together with the restriction endonucleases Nde I and Xho I (1 µL each, 20 U). Incubate at 37 °C for 3 h.
- Prepare the predigested pET-30a vector: Transfer 500 ng of the pET-30a vector into a new 1.5 mL centrifuge tube and top up the volume to 16 µL using deionized water. Add 2 µL of digestion buffer (10x concentrated) together with the restriction endonucleases Nde I and Xho I (1 µL each, 20 U).
- After incubating the mixture at 37 °C for 3 h, add 1 µL of alkaline phosphatase (1 U) and incubate at 37 °C for an additional hour. Inactivate the alkaline phosphatase by heating at 75 °C for 10 min in a pre-heated metal block heater.
- Transfer 4 µL of the digested MgUGD or MgUXS DNA products into fresh 1.5 mL centrifuge tubes and add 4 µL of the digested pET-30a vector. Add 1 µL of Ligation Buffer (10x), 1 µL of T4 ligase (3 U) and incubate the reaction mixture at 22 °C for 3 h.
- Transform electrocompetent E. coli Mach1 Competent Cells by electroporation using the ligation products. Spread the transformed cells on LB agar plates containing 50 μg/mL kanamycin. Incubate cells at 37 °C for 16 h.
- Verify colonies for the desired insertion by Sanger sequencing using the plasmid-specific T7 promoter and terminator primers. Prepare plasmids from the validated bacterial clones.
7. Expression and activity tests of MgUGD and MgUXS
- Transform E. coli BL21 (DE3) Competent Cells with plasmids bearing the MgUGD and MgUXS genes and spread the transformed cells on LB agar plates containing 50 μg/mL kanamycin. Incubate cells at 37 °C for 16 h.
- Cultivate a single colony in 5 mL LB medium with 50 μg/mL kanamycin overnight. Transfer the culture into 400 mL LB medium and continuously shake at 200 rpm at a temperature of 37 °C until the optical density at a wavelength of 600 nm (OD600) reaches an absorption of approximately 0.5.
- Reduce the incubation temperature to 20 °C and add 400 µL of isopropyl β-D-thiogalactopyranoside (concentration 1 M). Induce the expression of the recombinant proteins for 3 h.
- Harvest cells by centrifugation at 4,500 x g for 15 min at 4 °C. Suspend pellets in 10 mL of lysis buffer (100 mM NaCl, 50 mM Tris/HCl, 1% Triton X-100, 1 mM phenylmethylsulfonyl- fluoride (PMSF), pH 8.0).
- Disrupt cells by sonication for 20 min (40 on/off cycles with 20 μm amplitude for 15 s at 4 °C). Centrifuge at 14,000 x g at 4°C for 20 min and collect the supernatant for the activity test.
- Perform the activity assay of MgUGD by incubating 2 µL of the cell lysate with 2 µL of UDP-glucose (10 mM), 4 µL of NAD+ (10mM), 4 µL of MgCl2 (10 mM), 2 µL of Tris/HCl buffer (500 mM, pH 7.5), and 6 µL of deionized H2O in a new 1.5 mL centrifuge tube and incubate at 37 °C for 30 min.
- Perform the activity assay of MgUXS by incubating 2 µL of the cell lysate with 2 µL of UDP-glucuronic acid (10 mM), 2 µL of Tris/HCl buffer (500 mM, pH 7.5), and 14 µL of deionized H2O in a new 1.5 mL centrifuge tube and incubate at 37 °C for 30 min.
- Quench the reactions in step 7.5 and 7.6 by adding 20 μL of methanol and 40 μL of chloroform to each mixture. After vortexing the sample mixtures, centrifuge at 14,000 x g for 6 min at 4 °C and collect the upper aqueous layer of each tube.
- Analyze the reaction products using MALDI-TOF mass spectrometry in negative ionization mode in the m/z range from 500-700. Mix 1 μL of sample mixture with 1 μL of 2,5-dihydroxybenzoic acid sample matrix (1% w/V in 50% aqueous acetonitrile). Observe the expected m/z values at 579 and 535 for UDP-glucuronic acid and UDP-xylose, respectively (Figure 4).
Representative Results
Figure 1 shows a schematic overview of the described preparation method of the convergent cDNA library derived from Pacific oyster individuals. Figure 2 shows the sequences of the COX1 and ND genes of a distantly related oyster specimen with high divergence from the COX1 and ND gene sequences of the reference material. Figure 3 shows the sequences of the COX1 and ND genes of a closely related oyster specimen with low divergence from the COX1 and ND gene sequences of the reference material. Figure 4 shows the successful application of the cDNA library to clone the industrially relevant genes MgUGD and MgUXS.
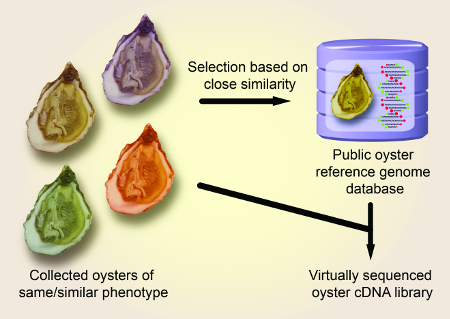
Figure 1: Schematic overview of the described analysis method for molecular identification of Pacific oyster specimen using COX1 and ND as reference genes. Please click here to view a larger version of this figure.
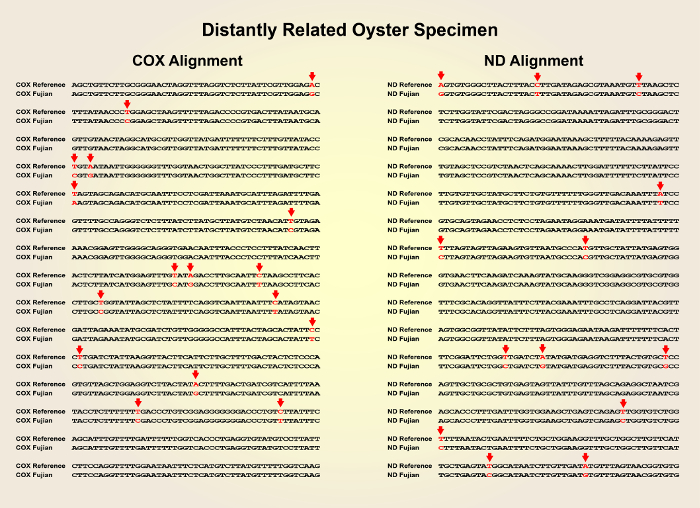
Figure 2: Sequence alignment of the COX1 and ND gene sequences of a highly-divergent specimen compared with the COX1 and ND gene sequences from the reference Pacific oyster strain. Please click here to view a larger version of this figure.
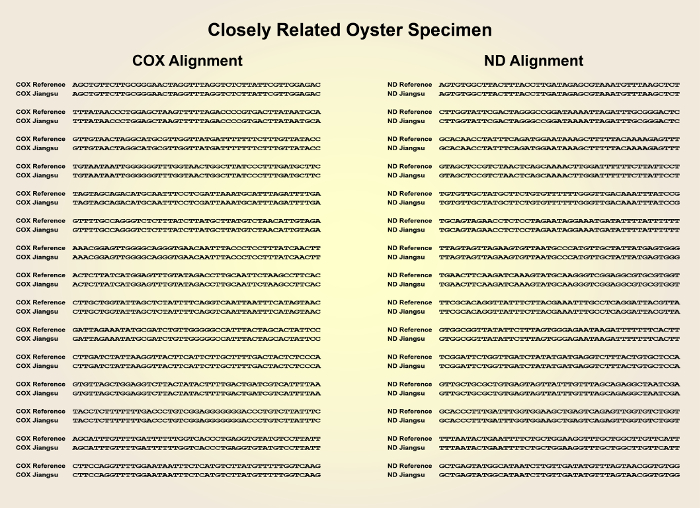
Figure 3: Sequence alignment of the COX1 and ND gene sequences of a closely related specimen compared with the COX1 and ND gene sequences from the reference Pacific oyster strain. Please click here to view a larger version of this figure.
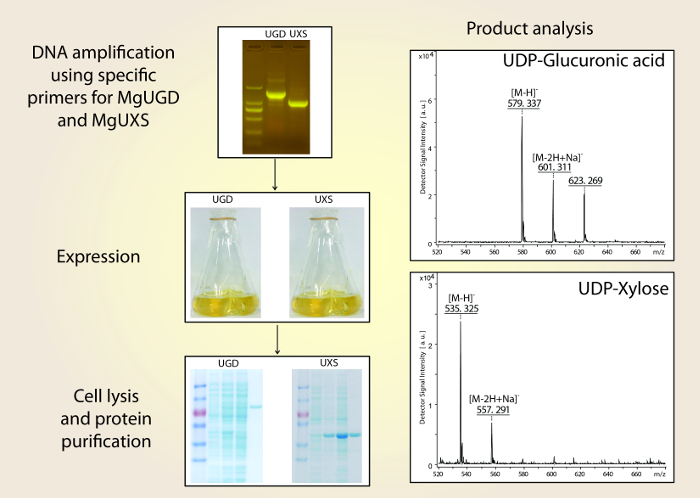
Figure 4: Schematic overview of molecular cloning, recombinant expression and detection of the reaction products of MgUGD and MgUXS. Please click here to view a larger version of this figure.
Discussion
The presented protocol allows the genetic identification of unreferenced oyster specimens with similar phenotype from regional seafood markets by comparison of the COX1 and ND genes with a publicly available oyster DNA genome database. The significance of this method lies in its simplicity, as only a single PCR reaction is needed for the evaluation of the virtual cDNA library. The two conserved mitochondrial COX1 and ND genes were amplified from a cDNA library which was generated by reverse transcription of RNA extracts from each oyster. The method of RNA isolation (step 2.1) was simplified by directly grinding the oyster tissue in liquid nitrogen. After sequencing the COX1 and ND genes of each specimen, sequence alignments revealed that some samples show high similarity to the reference strain. The closest relative showed complete identity of both the COX1 and the ND gene sequences.
The most critical steps of this procedure are the RNA extraction step; in order to minimize RNA degradation, it is essential to reduce the time between harvesting the oyster tissue and the RNA extraction.
Successful cloning was recently exemplified by cloning the oyster UGE gene12 and herein by cloning the MgUGD and MgUXS genes13, which validated the practicality of the generated cDNA library, allowing cloning of any number of genes of interest without the need for cumbersome cloning strategies using degenerated primers. This method of molecular identification by amplification of the COX1 and ND genes to generate virtually sequenced cDNA libraries may also be used in future applications for other biological materials that do not have physical samples of referenced genomes available.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work was supported in part by the Natural Science Foundation of China (grant numbers 31471703, A0201300537 and 31671854 to J.V. and L.L., grant number 31470435 to G.Y.), and the 100 Foreign Talents Plan (grant number JSB2014012 to J.V.).
Materials
| Chemicals: | |||
| 1% Triton X-100 | Solarbio | 9002-93-1 | *Alternative distributors possible |
| 2,5-Dihydroxybenzoic acid | Alfa Aesar | 490-79-9 | *Alternative distributors possible |
| Acetonitrile | Merck | 75-05-8 | *Alternative distributors possible |
| Agarose for molecular biology | Biowest Chemicals | 111860 | *Alternative distributors possible |
| Ampicilin | Solarbio | 69-52-3 | *Alternative distributors possible |
| Chloroform | Lingfeng, Shanghai | 67-66-3 | *Alternative distributors possible |
| DEPC water | Thermo Scientific | R0601 | |
| Ethanol | Jinhuada, Guangzhou | 64-17-5 | *Alternative distributors possible |
| Guanidinium thiocyanate-phenol reagent | Invitrogen | 15596018 | TRIzol reagent |
| Imidazole | Energy Chemical | 288-32-4 | *Alternative distributors possible |
| Isopropyl alcohol | Nanjing Chemical Reagent | 67-63-0 | *Alternative distributors possible |
| Isopropyl β-D-thiogalactopyranoside | Solarbio | 367-93-1 | *Alternative distributors possible |
| Kanamycin | Solarbio | 25389-94-0 | *Alternative distributors possible |
| LB Agar | Thermo Fisher | 22700025 | *Alternative distributors possible |
| LB Broth | Thermo Fisher | 10855021 | *Alternative distributors possible |
| Methanol | Jinhuada, Guangzhou | 67-56-1 | *Alternative distributors possible |
| MgCl2 hexahydrate | Xilong Huagong | 7791-18-6 | *Alternative distributors possible |
| NaCl | Xilong Huagong | 7647-14-5 | *Alternative distributors possible |
| NAD+ | Duly Biotech | 53-84-9 | *Alternative distributors possible |
| Phenyl-methylsulfonyl fluoride | Macklin | 329-98-6 | *Alternative distributors possible |
| Tris | Solarbio | 77-86-1 | *Alternative distributors possible |
| UDP-glucose | Wuhu Nuowei Chemicals | 28053-08-9 | *Alternative distributors possible |
| UDP-glucuronic acid | SIGMA | 63700-19-6 | *Alternative distributors possible |
| Tools/Instruments: | |||
| MALDI-TOF mass spectrometer | Bruker | Autoflex | *Alternative distributors possible |
| Metal block heater | Long Yang Scientific Instruments | Thermoshaker HB20 | *Alternative distributors possible |
| PCR thermocycler | Hema | 9600 | *Alternative distributors possible |
| Enzyme and Kits: | |||
| 10×Ligation buffer | Thermo Scientific | B69 | *Alternative distributors possible |
| 5×PrimeSTAR buffer | Takara | 9158A | |
| Alkaline phosphatase | ThermoFisher FastAP | EF0654 | *Alternative distributors possible |
| COX forward primer | Genscript | ATGTCAACAAATCATTTAGACATTG | |
| COX reverse primer | Genscript | ACTTGACCAAAAACATAAGACATG | |
| Cutsmart Buffer | NEB | B7204S | *Alternative distributors possible |
| dNTP mix | Invitrogen | 18427088 | |
| MgUGD forward primer | Genscript | ACATATGACCCTGTCCAAGATCTGTTGT | |
| MgUGD reverse primer | Genscript | ACTCGAGACTCTGTGAGGCGGTGGAG | |
| MgUXS forward primer | Genscript | CCATATGGCAGAATCCTCACAATCAC | |
| MgUXS reverse primer | Genscript | ACTCGAGCACATTTTTGAATTTGCAGACGT | |
| ND forward primer | Genscript | ATGAGATGGCAATTATTTTTTAAT | |
| ND reverse primer | Genscript | ATGTATTTTGGAAAAATCTCCAC | |
| PCR Cleanup Kit | AxyGen | AP-PCR-250 | *Alternative distributors possible |
| pET-30a(+) vector | Merck Millipore | 69909 |
Riferimenti
- Blaxter, M. L. The promise of a DNA taxonomy. Philosophical transactions of the Royal Society of London. Series B, Biological. 359 (1444), 669-679 (2004).
- Wen, J., et al. Species identification of dried shellfish (oyster, clam and mussel) products sold on the Chinese market. Food Control. 90, 199-204 (2018).
- Zhang, H., et al. Mitochondrial cob and cox1 genes and editing of the corresponding mRNAs in Dinophysis acuminata from Narragansett Bay, with special reference to the phylogenetic position of the genus Dinophysis. Applied and Environmental Microbiology. 74 (5), 1546-1554 (2007).
- Sell, J., Spirkovski, Z. Mitochondrial DNA differentiation between two forms of trout Salmo letnica, endemic to the Balkan Lake Ohrid, reflects their reproductive isolation. Molecular Ecology. 13, 3633-3644 (2004).
- Karadjian, G., et al. Highly rearranged mitochondrial genome in Nycteria parasites (Haemosporidia) from bats. Proceedings of the National Academy of Sciences of the United States of America. 113 (35), 9834-9839 (2018).
- Morga, B., et al. Identification of genes from flat oyster Ostrea edulis as suitable housekeeping genes for quantitative real time PCR. Fish and Shellfish Immunology. 29 (6), 937-945 (2010).
- Delsuc, F., et al. Molecular systematics of armadillos (Xenarthra, Dasypodidae): contribution of maximum likelihood and Bayesian analyses of mitochondrial and nuclear genes. Molecular Phylogenetics and Evolution. 28 (2), 261-265 (2005).
- Wei, S., et al. Discovery and Biochemical Characterization of UDP-Glucose Dehydrogenase from Akkermansia muciniphila. Protein & Peptide Letters. 24 (8), 735-741 (2017).
- Gu, B., et al. Discovery and Biochemical Characterization of the UDP-Xylose Biosynthesis Pathway in Sphaerobacter thermophilus. Protein & Peptide Letters. 23 (12), 1103-1110 (2016).
- Duan, X. C., et al. Functional characterization of the UDP-xylose biosynthesis pathway in Rhodothermus marinus. Applied Microbiology and Biotechnology. 99 (22), 9463-9472 (2015).
- Vogelstein, B., Gillespie, D. Preparative and analytical purification of DNA from agarose. Proceedings of the National Academy of Sciences of the United States of America. 76 (2), 615-619 (1979).
- Song, H. B., et al. UDP-glucose 4-epimerase and β-1,4-galactosyltransferase from the oyster Magallana gigas as valuable biocatalysts for the production of galactosylated products. International Journal of Molecular Sciences. 19 (6), 1600 (2018).
- Gainey, P. A., Phelps, C. F. Uridine diphosphate glucuronic acid production and utilization in various tissues actively synthesizing glycosaminoglycans. Biochemical Journal. 128 (2), 215-227 (1972).

