Obtaining Quality Extended Field-of-View Ultrasound Images of Skeletal Muscle to Measure Muscle Fascicle Length
Summary
This study describes how to obtain high quality musculoskeletal images using the extended field-of-view ultrasound (EFOV-US) method for the purpose of making muscle fascicle length measures. We apply this method to muscles with fascicles that extend past the field-of-view of common traditional ultrasound (T-US) probes.
Abstract
Muscle fascicle length, which is commonly measured in vivo using traditional ultrasound, is an important parameter defining a muscle’s force generating capacity. However, over 90% of all upper limb muscles and 85% of all lower limb muscles have optimal fascicle lengths longer than the field-of-view of common traditional ultrasound (T-US) probes. A newer, less frequently adopted method called extended field-of-view ultrasound (EFOV-US) can enable direct measurement of fascicles longer than the field-of-view of a single T-US image. This method, which automatically fits together a sequence of T-US images from a dynamic scan, has been demonstrated to be valid and reliable for obtaining muscle fascicle lengths in vivo. Despite the numerous skeletal muscles with long fascicles and the validity of the EFOV-US method for making measurements of such fascicles, few published studies have utilized this method. In this study, we demonstrate both how to implement the EFOV-US method to obtain high quality musculoskeletal images and how to quantify fascicle lengths from those images. We expect that this demonstration will encourage the use of the EFOV-US method to increase the pool of muscles, both in healthy and impaired populations, for which we have in vivo muscle fascicle length data.
Introduction
Fascicle length is an important parameter of skeletal muscle architecture, which overall is indicative of a muscle’s ability to produce force1,2. Specifically, a muscle’s fascicle length provides insight into the absolute range of lengths over which a muscle can generate active force3,4. For example, given two muscles with identical values for all isometric force-generating parameters (i.e., average sarcomere length, pennation angle, physiological cross sectional area, contraction state, etc.) except for fascicle length, the muscle with the longer fascicles would produce its peak force at a longer length and would produce force over a wider range of lengths than the muscle with shorter fascicles3. Quantification of muscle fascicle length is important for understanding both healthy muscle function and changes in a muscle’s force-generating capacity, which can occur as a result of altered muscle use (e.g., immobilization5,6, exercise intervention7,8,9, high heel wearing10) or a change in the muscle’s environment (e.g., tendon transfer surgery11, limb distraction12). Measurements of muscle fascicle length were originally obtained through ex vivo cadaveric experiments that allow for direct measurement of dissected fascicles13,14,15,16. The valuable information provided by these ex vivo experiments led to an interest in implementing in vivo methods17,18,19 to address questions that could not be answered in cadavers; in vivo methods allow for quantification of muscle parameters in a native state as well as at different joint postures, different muscle contraction states, different loading or unloading states, and across populations with differing conditions (i.e. healthy/injured, young/old, etc.). Most frequently, ultrasound is the method employed to obtain in vivo muscle fascicle lengths18,19,20; it is quicker, less expensive, and easier to implement than other imaging techniques, such as diffusion tensor imaging (DTI)18,21.
Extended field-of-view ultrasound (EFOV-US) has been demonstrated to be a valid and reliable method for measuring muscle fascicle length in vivo. While commonly implemented, traditional ultrasound (T-US) has a field-of-view which is limited by the ultrasound transducer’s array length (typically between 4 and 6 cm, although there are probes that extend to 10 cm10)18,20. To overcome this limitation, Weng et al. developed an EFOV-US technology that automatically acquires a composite, two-dimensional “panoramic” image (up to 60 cm long) from a dynamic, extended distance scan22. The image is created by fitting together, in real-time, a sequence of traditional, B-mode ultrasound images as the transducer dynamically scans the object of interest. Because sequential T-US images have large overlapping regions, the small differences from one image to the next can be used to calculate the probe motion without the use of external motion sensors. Once the probe motion between two consecutive images is calculated, the “current” image is merged successively with the preceding images. The EFOV-US method allows direct measurement of long, curved muscle fascicles and has been demonstrated to be reliable across muscles, trials, and sonographers23,24,25 and valid for both flat and curved surfaces23,26.
Implementing ultrasound to measure muscle fascicle length in vivo is not trivial. Unlike other imaging techniques that involve more automated protocols (i.e., MRI, CT), ultrasound is dependent on sonographer skill and anatomical knowledge27,28. There is concern that probe misalignment with the fascicle plane may cause substantial error in fascicle measures. One study demonstrates little difference (on average < 3 mm) in measures of fascicle length taken using ultrasound and DTI MRI but also shows that measurement precision is low (standard deviation of difference ~12 mm)29. Still, it has been shown that a novice sonographer, with practice and guidance from an experienced sonographer, can obtain valid meaures using EFOV-US23. Thus, efforts should be made to demonstrate appropriate protocols to reduce human error and improve accuracy of measurements obtained using EFOV-US. Ultimately, developing and sharing appropriate protocols may expand the number of experimenters and laboratories that can reproduce fascicle length data from the literature or obtain novel data in muscles which have not yet been studied in vivo.
In this protocol, we demonstrate how to implement the EFOV-US method to obtain high quality musculoskeletal images that can be used to quantify muscle fascicle length. Specifically, we address (a) collecting EFOV-US images of a single upper limb and a single lower limb muscle (b) determining, in real-time, the “quality” of the EFOV-US image, and (c) quantifying muscle architecture parameters offline. We provide this detailed guide to encourage the adoption of the EFOV-US method for obtaining muscle fascicle length data in muscles that have gone unstudied in vivo due to their long fascicles.
Protocol
Northwestern University’s Institutional Review Board (IRB) approved the procedures of this study. All participants enrolled in this work gave informed consent prior to beginning the protocol detailed below.
NOTE: The specific ultrasound system used in this study had EFOV-US capabilities and was adopted because we were able to review details about and validity assessments for the algorithm in the scientific literature22,26; multiple other systems with EFOV-US also exist18,20,30. A linear array transducer 14L5 (frequency bandwidth 5-14 MHz) was utilized. The muscles imaged in this protocol are just a small subset of muscles for which US images have been captured and fascicle lengths measured (e.g., triceps25, extensor carpi ulnaris23, medial gastrocnemius10, vastus lateralis24, biceps femoris8,31). This protocol is intended to provide pointers and describe the necessary standards so that that it may be applied to muscles beyond the two examples we provide.
1. Collecting EFOV-US images of muscles
Preparation
- Sonographer Preparation
- Prior to operating the ultrasound system, read through the system’s manual to become familiar with system safety, care for maintaining the system, system setup and controls, etc. In addition, review the system’s instructions for obtaining EFOV-US images and be familiar with the method implemented to obtain the EFOV-US images.
NOTE: Different ultrasound systems name the EFOV-US mode using different terminology. For example, in the system used here, the EFOV mode is referred to as “Panoramic Imaging”. While the technical details of the algorithm implemented in various commercial systems are usually intellectual property and therefore not freely available, from a cursory review, many commercial systems with panoramic ultrasound capabilities describe an approach similar to the one described by Weng et al.22. Evaluating the general validity of measurements acquired from any system, either by obtaining more detailed information directly from the company who manufactures the system, by using an imaging phantom26,32, or by other means (e.g., comparison to animal dissection24) is recommended as an important step before initiating research involving human participants. - Take time to become familiar with the anatomy of the muscle(s) of interest as well as the surrounding anatomy. It is suggested that the sonographer use an anatomy textbook or preferably an interactive online 3D anatomy model to become familiar with the anatomy of interest.
- Prior to operating the ultrasound system, read through the system’s manual to become familiar with system safety, care for maintaining the system, system setup and controls, etc. In addition, review the system’s instructions for obtaining EFOV-US images and be familiar with the method implemented to obtain the EFOV-US images.
- Participant Preparation
- Explain the protocol of the study to the participant and acquire IRB approved consent prior to beginning the imaging protocol.
- Ask the participant to wear appropriate clothing to enable access to the muscle of interest. For example, if the sonographer plans to image a forearm muscle, the participant should be asked to wear a short-sleeved shirt.
- Seat the participant in an adjustable chair that can be locked in place. Take time to adjust the chair to make the participant as comfortable as possible while still providing access to the muscle of interest.
NOTE: If an adjustable chair which can lay completely flat is not available, some study designs may require the use of a table to access the muscle of interest (i.e., hamstrings). - Place the joint(s) that the muscle of interest spans in a posture that can be controlled and repeated. Use clinical guidance33 for locating anatomical landmarks and implementing goniometry; use ISB standards for defining the joint coordinate system34,35. In general, to measure joint angle, mark anatomical landmarks with skin safe marker (Table of Materials) and then align the center of a handheld goniometer up with the axis of rotation of the joint and the arms of the goniometer up with the joint segments.
NOTE: If imaging passive muscle, placing the muscle of interest in a relatively lengthened position is recommended to avoid imaging slack muscle.- To replicate the biceps brachii as imaged in this study, seat participants with feet supported, back straight, shoulder at 85° of abduction and 10° of horizontal flexion, elbow at 25° flexion, and forearm, wrist, and fingers at neutral.
- To replicate the tibialis anterior as imaged in this study, seat participants with knee at 60° of flexion and the ankle at 15° of plantar flexion.
- Secure the participants limb using cloth straps to minimize movement during the imaging protocol.
Image Acquisition
- Plug in and turn on the ultrasound system. Ensure that the exam is set to Musculoskeletal, the transducer in use is selected (here we used 14L5), and the transmit frequency is set between 5-17 MHz (here 11MHz was used), a typical frequency range for musculoskeletal imaging. Higher frequencies are generally used for more superficial imaging as they improve resolution but decrease wave penetration.
- Go into the system settings to adjust the footswitch settings. For the purposes of this protocol, we recommend setting the footswitch to start/stop the imaging. If the footswitch in use has multiple pedals, set additional pedals to “Freeze” or “Pause”, and “Print” or “Store” the image.
- Apply a generous amount of ultrasound gel to the head of the transducer.
- Place the transducer on the participant’s skin on the approximate region of interest.
- Move the transducer in the short axis plane of the muscle. Note that the transducer has a small protuberance on one side, called an indicator. The side of the transducer that has the indicator corresponds with the left side of the ultrasound image. When imaging in the short axis, have the sonographer keep the indicator pointed laterally and when the sonographer is in long axis, point the indicator distally.
- Identify the muscle of interest in the short axis plane (perpendicular to muscle fiber direction) and move the transducer distal and proximal to get a full visualization of the muscle path.
- Mark important anatomical landmarks (i.e., the lateral and medial edges of the muscle, the muscle tendon junction, and muscle insertion) using skin safe ink markers (Table of Materials).
- Once the location of the muscle has been identified and properly marked, have the sonographer move the ultrasound transducer in the long axis plane (parallel to the muscle fiber direction).
- Beginning at either the distal or proximal end of the muscle, rotate and tilt the transducer to identify the fascicle plane at that point. Make a mark on the skin when the correct transducer position has been established.
- Once the approximate fascicle plane has been established along the entire desired length to be scanned, have the sonographer practice following this path.
- To begin collecting images, put the ultrasound system in EFOV-US mode.
- Starting at one end of the muscle, click the footswitch to start image acquisition and slowly and continuously move the ultrasound transducer in the long axis. Once the end of the muscle has been reached, click the footswitch to end image acquisition.
- Practice and ensure the correct transducer path. This may take several practice images before consistently obtaining “quality” EFOV-US images (See section 2 for explanation of quality images).
- To optimize image visibility and clarity, consider adjustments to the following parameters.
- Depth: If image acquisition ends before the desired length of the muscle can be captured, increase the depth of the image (in the system used here, increasing image depth increases the absolute length the scan can be).
- Focus: Place the focus arrow in the lower half of the Image just below the muscle of interest.
- Gain: Ensure the gain is balanced through the depth of the image.
- Speed: Image at the optimum speed as guided by the indicator (in most systems a speed indicator displays on the monitor during panoramic imaging).
- Once qualitatively good images have been collected (step 2.1), hit the Print/Store footswitch pedal or a synonymous button on the control panel to save the image.
- Repeat steps 1.13-1.16 until 3 quality EFOV-US images of the muscle are obtained.
- Repeat steps 1.6-1.17 until all muscles of interest are obtained.
- Use a towel to gently wipe the gel from the participant’s skin. Then have the participant rinse off the area of the skin or use a damp towel to wipe the skin that was exposed to the gel. Dry.
- Wipe gel from the head of the transducer and disinfect.
- Export images as uncompressed DICOM images onto a CD-DVD, flash drive, or through the local network onto a computer.
2. Determining “quality” of the EFOV-US image
- Following step 1.13, have the sonographer identify and evaluate the quality of key anatomical features of the muscle of interest and its surrounding anatomy. This is a qualitative assessment based on the sonographer’s knowledge of anatomy and musculoskeletal tissue echogenicity (ability of a tissue to reflect ultrasonic waves). For an EFOV-US images to be considered qualitatively “good” the following should be met:
- In any long-axis image of a muscle, check that the sonographer can clearly identify the muscle as a hypoechoic (dark) shape with hyperechoic (bright) boundaries which represent the deep and superficial muscle fascia.
- Between the muscle boundaries, check that the sonographer can identify the connective tissue surrounding a muscles fascicle as hyperechoic (bright) lines.
NOTE: When imaging multi-pennated muscles, the image should also contain central tendon(s) that show up in the muscle belly, between the deep and superficial muscle fascia, as a hyperechoic (bright) structure. - Check that the image does not have excessive bending. This is usually indicated by shadows or gaps in the image or a jagged flexible ruler line over the image.
- If the image is missing one or more of the tissue structures described in 2.1, deem the image “qualitatively poor” and return to live 2D-mode.
3. Quanitfying Muscle Fascicle Length
- To quantify muscle fascicle length, use ImageJ, an open source image processing platform. ImageJ can be downloaded at https://imagej.net/Downloads.
NOTE: Though ImageJ is frequently implemented24,25,31,36,37,38, quantification of muscle fascicle length may be measured using other image processing software8,39 or custom codes40,41. - Once downloaded, open the ultrasound images as DICOM images in ImageJ by clicking File | Open and selecting the image to analyze.
- To ensure that the DICOM image properties have been preserved, click on the Straight Line tool in the Tools menu and draw a straight line from 0 to 1 cm on the ruler on the side of the ultrasound image. Then go to Analyze | Measure to measure the line made. If the image properties have been preserved, the length of the straight line should be 1 cm.
- To measure fascicle lengths in the image, complete the following.
- Right click on the Straight Line tool.
- Select Segmented Line.
- Move the cursor onto the image and click at one end of the fascicle that has been chosen to be measured.
NOTE: Only make measurements on fascicles that the entire fascicle path (i.e., from one aponeurosis to the next aponeurosis or aponeurosis to central tendon) can convincingly be seen. - Click along the path to ensure curvature in the fascicle path is captured.
- Once the end of the fascicle path is reached, double click to end the line and go to Analyze | Measure to measure the length of the line.
NOTE: A new window, “Results”, will pop up the first time a measurement is made. What values are displayed can be managed in the Results window by going to Results | Set Measurements.
- Repeat steps 3.4.3-3.4.5 until multiple fascicle measures are made in a single image.
- Save fascicle measurements by clicking File | Save on the results tab or the values can be copy and pasted into another document/spreadsheet.
Representative Results
Extended field-of-view ultrasound (EFOV-US) was implemented to obtain images from the long head of the biceps brachii and the tibialis anterior in 4 healthy volunteers (Table 1). Figure 1 shows what EFOV-US images of both muscles imaged in this representative imaging session and highlights important aspects of each image such as muscle aponeurosis, central tendon, fascicle path, etc. After the imaging session was over, 3 qualitatively “good” images (Figure 2) were analyzed for each muscle in each individual. ImageJ was implemented to measure 4 fascicles per image. In each image, fascicles with paths that could be convincingly visualized from origin to insertion and that were located in different portions of the muscle selected were measured. The average fascicle lengths obtained in this study for the biceps brachii (14.6 ± 1.7 cm) and the tibialis anterior (7.3 ± 0.6 cm) are within the range of fascicle lengths reported previously25,42 (Table 1).
As most challenging and subjective parts of this protocol is determining factors which lead to correctly deeming an image as qualitatively “good” or qualitatively “bad”. We provide several examples of “good” and “bad” images (Figure 2) and how image landmarks and quality vary across people (Figure 3). In addition we have highlighted the portions of the images which are specifically “bad”.
| Subject | Gender | Height (m) | Age | Bicep Side | Bicep Fascicle Length (cm) | Tibialis Anterior Side | Tibialis Anterior Fascicle length (cm) |
| 1 | M | 1.78 | 24 | L | 16.4 ± 0.3 | L | 7.6 ± 0.1 |
| 2 | F | 1.8 | 23 | R | 12.2 ± 0.2 | L | 7.5 ± 0.2 |
| 3 | M | 1.82 | 24 | L | 14.9 ± 0.2 | R | 7.7 ± 0.1 |
| 4 | F | 1.79 | 28 | R | 14.7 ± 0.2 | L | 6.4 ± 0.3 |
| Average | 14.6 | 7.3 | |||||
| SD | 1.7 | 0.6 |
Table 1: Participant Demographics and Data. Measurements of fascicle length are represented as average ± standard deviation.
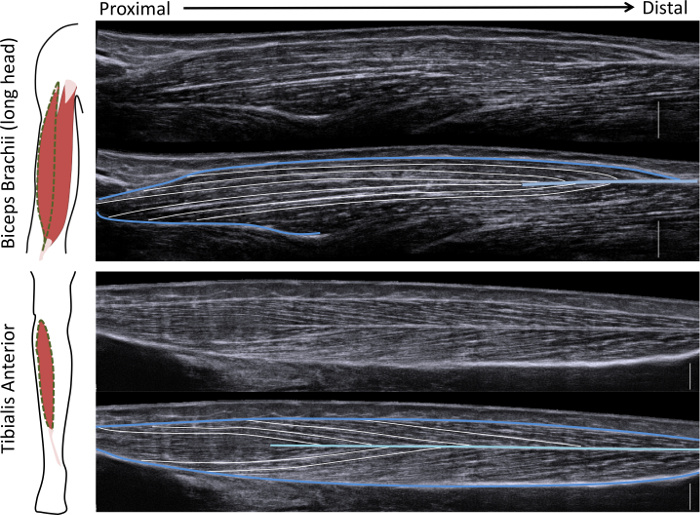
Figure 1: Schematic and EFOV images of two example muscles. (left) Illustration of the muscle being studied. (right) Example of “good” images on top and the same image with whole muscle (dark blue), central tendon (light blue), and muscle fascicles (white) outlined. Each image has a corresponding 1 cm scale bar (white) on the bottom right of the image. Please click here to view a larger version of this figure.
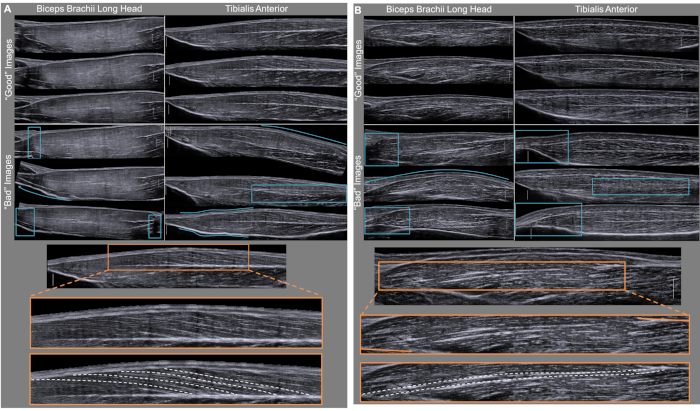
Figure 2: Demonstration of Image Quality. Demonstration of three qualitatively “good” and three qualitatively “bad” images obtained from the biceps brachii and the tibialis anterior of participants 1 and 2. (Top A & B) In all the qualitatively “good” images fascicles which extend from internal tendon to muscle aponeurosis can be visualized. We illustrate images which are qualitatively “bad” and should not be analyzed. Portions of the image which qualify it is as “bad” are emphasized (blue boxes and arrows) and include jagged or broken images, excessive or non-anatomically relevant bending, images which exclude the entire fascicle, and images with blurred central tendons. Each image has a scale bar (white vertical line) which represents 1 cm. This portion of the figure is highlighting the variability among images due mainly to the sonographer’s inconsistency across separate imaging sweeps. (Bottom A & B) One “good” biceps and one “good” tibialis anterior muscle are shown. The orange box on the original image is then blown up to illustrate more accurately the zoom that is seen when measuring fascicles in ImageJ. The bottom image shows representative outlined fascicles (white dashed lines). These images are deemed “good” because fascicles can be followed from origin to insertion and the zoomed portion of the image doesn't have substantial distortions or artifacts. Please click here to view a larger version of this figure.
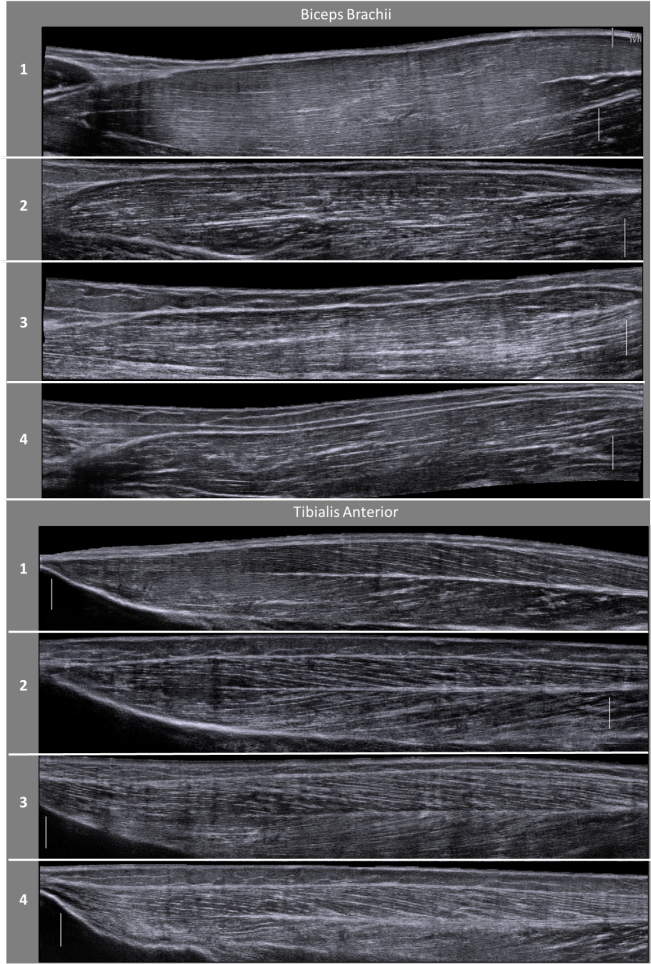
Figure 3: Variability in image quality across individuals. Variability in image quality and visibility exists between participants, largely due to anatomical variation (i.e. muscle size, muscle length, subcutaneous fat content) and differences in muscle content (i.e. amounts of intramuscular fat, connective tissue, fibrosis). Specifically, variations in muscle content and layers of tissue above the muscle can affect the echo intensity of the imaged muscle43. Natural anatomical differences across individuals will result in muscle architectural features varying in location and/or relative size across US images of different individuals. This demonstration of muscles in different participants stresses the importance of a thorough understanding of anatomy and sufficient practice obtaining images on various individuals for gaining confidence in the quality and accuracy of the images being obtained. Please click here to view a larger version of this figure.
Discussion
Critical steps in the protocol.
There are a few critical components to obtaining quality EFOV-US images that yield valid and reliable fascicle length measures. First, as indicated in method 1.1.2 it is essential that the sonographer take time to become familiar with the anatomy of the muscle being imaged as well as surrounding muscles, bones, and other soft tissue structures. This will improve the sonographer’s ability to image the correct muscle and determine if multiple images are capturing the same muscle plane. Second, the sonographer should practice the protocol on phantoms and multiple pilot participants before collecting data for publication. Ultrasound is known to result in measurement error if the sonographer does not properly identify the fascicle plane, a task which is challenging and can improve with practice. Last, it is strongly recommended to ensure that the validity of the measurements made by the EFOV-US algorithm in the ultrasound system being used has been established. If the method’s accuracy has not been demonstrated, validation can be done easily using an ultrasound phantom23,26 or through comparison to another imaging tool44 or cadaveric dissection45.
Modifications and troubleshooting of the method.
If image visibility is poor or the probe motion is uneven during dynamic scanning, adding ultrasound gel may enhance image quality by improving transducer-to-skin coupling. If image acquisition is cut-off by the algorithm before the entire object of interest is captured, the depth of the image should be increased. Increasing the depth of the image expands the available scanning distance, thus enabling longer objects to be captured within a single EFOV-US image. In general, it is best to refer to the ultrasound system’s manual when attempting to improve or troubleshoot image quality or image acquisition.
Here, we demonstrate how to capture EFOV-US images of the entire muscle from the muscle tendon junction of the origin tendon to the insertion tendon. Capturing the entire muscle is necessary for some muscles, such as the biceps brachii, whose fascicles span nearly the entire length of the muscle. However, for other muscles, such as the tibialis anterior or other pennated muscles, shorter scans that do not include the full muscle belly may still capture entire muscle fascicles. For novice sonographers, acquiring images from shorter scans that still capture full fascicle lengths may decrease the chances of probe misalignment with the fascicle plane and improve image quality, decreasing the potential for fascicle measurement error.
Limitations of the Method
Notably, muscle activation can change muscle fascicle length. Due to the nature of the scanning method, the major limitation of EFOV-US is that it cannot be implemented to study muscle fascicle changes due to dynamic muscle contraction (e.g., during walking46,47). Additionally, due to the time required to capture an EFOV-US image, imaging a muscle at maximum contraction is likely infeasible due to muscle fatigue. Instead, the EFOV-US method is beneficial for sub-maximal or passive imaging. One way to ensure muscle activity is constant across participants, limbs, or sessions is to simultaneously measure EMG during imaging and analyze only images which are taken when the muscle is at some desired activity level. Though recommended, particularly if studying populations with altered neural drive, measures of EMG were not taken in the population studied here.
Though traditional ultrasound has been shown to be valid and reliable for measuring in vivo muscle fascicle lengths, some fascicle measurement error will occur if the sonographer’s alignment of the ultrasound transducer deviates from the fascicle plane27,29,48. Due to the nature of the EFOV-US’s dynamic scan, there is concern that the EFOV-US method may have more error than T-US21,24. While a recent study demonstrated that fascicle measurement error from probe misalignment was not larger in EFOV-US than in the well-established, T-US method23 in a single wrist muscle, a general limitation of B-mode ultrasound is that you are only able to capture a relatively small, 2-dimensional (2D) view of the muscle. The true path of individual fascicles may be 3D; concerns remain that errors associated with measuring lengths of potentially 3D paths from 2D views may be bigger for longer fascicles.
Significance of the Method with Respect to Existing/Alternative Methods
Static, B-mode ultrasound is a widely accepted method for measuring muscle fascicle lengths in vivo. However, the field-of-view of T-US probes limits the length of fascicles that can be directly measured. Instead, measurement of fascicles longer than the field-of-view of T-US requires trigonometric estimation methods, diffusion tensor imaging (DTI), or EFOV-US20. In general, ultrasound imaging is favored over magnetic resonance imaging (MRI) techniques such as DTI because MRI is more expensive and challenging to implement18. Fascicle lengths captured with EFOV-US have been shown to be more accurate than trigonometric estimation methods24,36, which is expected since muscle fascicles regularly follow a curved path, but trigonometric estimation methods assume linearity in their calculation of muscle fascicle length.
It should be noted that though most ultrasound probes are 4-6 cm in length, ultrasound probes up to 10 cm have been used9,10. The 10 cm probes enable a wider field-of-view, enabling the capture of longer, straight fascicles. Still, the longer probe length decreases frame rate, would require the imaging surface (the body) to also be straight to avoid uneven compression of the imaged tissue, and may not be able to capture longer curved fascicles (without the use of EFOV)20.
Future Applications or Directions of the Method
The guide detailed here for obtaining quality EFOV-US images for measuring muscle fascicle length is intended to encourage the use of the EFOV-US method to expand the pool of muscles for which the field has in vivo muscle architecture data. The expectation is that this method be applied to both healthy and impaired populations (e.g., individuals post-stroke38,49 or post-orthopedic surgery) to better understand muscle function and muscle adaptation. In addition, these in vivo data are important for development of models that more accurately predict human movement as well as the development of subject specific musculoskeletal models.
Notably, the EFOV-US method is not limited to measurements of muscle fascicle length. The method has been used for measurement of tendon length50,51 and muscle anatomical cross-sectional area,52,53 as well as for documentation of various superficial lesions54,55. Thus, there is opportunity to develop guides, similar to the one presented here, for obtaining high quality images with the EFOV-US method for various applications.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We would like to thank Vikram Darbhe and Patrick Franks for their experimental guidance. This work is supported by the National Science Foundation Graduate Research Fellowship Program under Grant No. DGE-1324585 as well as NIH R01D084009 and F31AR076920. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation or NIH.
Materials
| 14L5 linear transducers | Siemens | 10789396 | |
| Acuson S2000 Ultrasound System | Siemens | 10032746 | |
| Adjustable chair (Biodex System) | Biodex Medical Systems | System Pro 4 | |
| Skin Marker Medium Tip | SportSafe | n/a | Multi-color 4 Pack recommended |
| Ultrasound Gel – Standard 8 Ounce Non-Sterile Fragrance Free Glacial Tint | MediChoice, Owens &Minor | M500812 |
Riferimenti
- Gans, C., Bock, W. J. The functional significance of muscle architecture: a theoretical analysis. Advances in Anatomy, Embryology and Cell Biology. 38, 115-142 (1965).
- Gans, C. Fiber architecture and muscle function. Exercise and Sports Sciences Reviews. 10, 160-207 (1982).
- Lieber, R. L., Fridén, J. Functional and clinical significance of skeletal muscle architecture. Muscle & Nerve. 23 (11), 1647-1666 (2000).
- Zajac, F. E. Muscle and tendon: properties, models, scaling, and application to biomechanics and motor control. Critical Reviews in Biomedical Engineering. 17 (4), 359-411 (1989).
- Williams, P. E., Goldspink, G. The effect of immobilization on the longitudinal growth of striated muscle fibres. Journal of Anatomy. 116 (1), 45 (1973).
- Williams, P. E., Goldspink, G. Changes in sarcomere length and physiological properties in immobilized muscle. Journal of Anatomy. 127 (3), 459-468 (1978).
- Blazevich, A. J., Cannavan, D., Coleman, D. R., Horne, S. Influence of concentric and eccentric resistance training on architectural adaptation in human quadriceps muscles. Journal of Applied Physiology. 103 (5), 1565-1575 (2007).
- Seymore, K. D., Domire, Z. J., DeVita, P., Rider, P. M., Kulas, A. S. The effect of Nordic hamstring strength training on muscle architecture, stiffness, and strength. European Journal of Applied Physiology. 117 (5), 943-953 (2017).
- Franchi, M. V., et al. Architectural, functional and molecular responses to concentric and eccentric loading in human skeletal muscle. Acta Physiologica. 210 (3), 642-654 (2014).
- Csapo, R., Maganaris, C. N., Seynnes, O. R., Narici, M. V. On muscle, tendon and high heels. The Journal of Experimental Biology. 213 (15), 2582-2588 (2010).
- Takahashi, M., Ward, S. R., Marchuk, L. L., Frank, C. B., Lieber, R. L. Asynchronous muscle and tendon adaptation after surgical tensioning procedures. Journal of Bone and Joint Surgery. 92 (3), 664-674 (2010).
- Boakes, J. L., Foran, J., Ward, S. R., Lieber, R. L. Case Report: Muscle Adaptation by Serial Sarcomere Addition 1 Year after Femoral Lengthening. Clinical Orthopaedics and Related Research. 456, 250-253 (2007).
- Cutts, A., Alexander, R. M., Ker, R. F. Ratios of cross-sectional areas of muscles and their tendons in a healthy human forearm. Journal of Anatomy. 176, 133-137 (1991).
- Lieber, R. L., Friden, J. Functional and clinical significance of skeletal muscle architecture. Muscle Nerve. 23, 1647-1666 (2000).
- Lieber, R. L., Fazeli, B. M., Botte, M. J. Architecture of Selected Wrist Flexor and Extensor Muscles. Journal of Hand Surgery-American. 15 (2), 244-250 (1990).
- Brand, P. W., Beach, R. B., Thompson, D. E. Relative tension and potential excursion of muscles in the forearm and hand. Journal of Hand Surgery. 6 (3), (1981).
- Fukunaga, T., Kawakami, Y., Kuno, S., Funato, K., Fukashiro, S. Muscle architecture and function in humans. Journal of Biomechanics. 30 (5), 457-463 (1997).
- Kwah, L. K., Pinto, R. Z., Diong, J., Herbert, R. D. Reliability and validity of ultrasound measurements of muscle fascicle length and pennation in humans: a systematic review. Journal of Applied Physiology. 114, 761-769 (2013).
- Lieber, R. L., Ward, S. R. Skeletal muscle design to meet functional demands. Philosophical Transactions of the Royal Society B: Biological Sciences. 366 (1570), 1466-1476 (2011).
- Franchi, M. V., et al. Muscle architecture assessment: strengths, shortcomings and new frontiers of in vivo imaging techniques. Ultrasound in Medicine & Biology. 44 (12), 2492-2504 (2018).
- Cronin, N. J., Lichtwark, G. The use of ultrasound to study muscle-tendon function in human posture and locomotion. Gait & posture. 37 (3), 305-312 (2013).
- Weng, L., et al. US extended-field-of-view imaging technology. Radiology. 203 (3), 877-880 (1997).
- Adkins, A. N., Franks, P. F., Murray, W. M. Demonstration of extended field-of-view ultrasound’s potential to increase the pool of muscles for which in vivo fascicle length is measurable. Journal of Biomechanics. 63, 179-185 (2017).
- Noorkoiv, M., Stavnsbo, A., Aagaard, P., Blazevich, A. J. In vivo assessment of muscle fascicle length by extended field-of-view ultrasonography. Journal of Applied Physiology. , (2010).
- Nelson, C. M., Dewald, J. P. A., Murray, W. M. In vivo measurements of biceps brachii and triceps brachii fascicle lengths using extended field-of-view ultrasound. Journal of Biomechanics. 49, 1948-1952 (2016).
- Fornage, B. D., Atkinson, E. N., Nock, L. F., Jones, P. H. US with extended field of view: Phantom-tested accuracy of distance measurements. Radiology. 214, 579-584 (2000).
- Bénard, M. R., Becher, J. G., Harlaar, J., Huijing, P. A., Jaspers, R. T. Anatomical information is needed in ultrasound imaging of muscle to avoid potentially substantial errors in measurement of muscle geometry. Muscle & Nerve. 39 (5), 652-665 (2009).
- Pinto, A., et al. Sources of error in emergency ultrasonography. Critical Ultrasound Journal. 5 (1), 1 (2013).
- Bolsterlee, B., Veeger, H. E. J., van der Helm, F. C. T., Gandevia, S. C., Herbert, R. D. Comparison of measurements of medial gastrocnemius architectural parameters from ultrasound and diffusion tensor images. Journal of Biomechanics. 48 (6), 1133-1140 (2015).
- VanHooren, B., Teratsias, P., Hodson-Tole, E. F. Ultrasound imaging to assess skeletal muscle architecture during movements: a systematic review of methods, reliability, and challenges. Journal of Applied Physiology. 128 (4), 978-999 (2020).
- Pimenta, R., Blazavich, A. J., Frietas, S. R. Biceps Femoris Long-Head Architecture Assessed Using Different Sonographic Techniques. Medicine & Science in Sports & Exercise. 50 (12), 2584-2594 (2018).
- Adkins, A. N., Franks, P. W., Murray, W. M. Demonstration of extended field-of-view ultrasound’s potential to increase the pool of muscles for which in vivo fascicle length is measurable. Journal of Biomechanics. 63, 179-185 (2017).
- Norkin, C. C., White, J. D. . Measurement Of Joint Motion: A Guide To Goniometry. 5th edn. , (2016).
- Wu, G., et al. ISB recommendation on definitions of joint coordinate system of various joints for the reporting of human joint motion–part I: ankle, hip, and spine. International Society of Biomechanics. Journal of Biomechanics. 35 (4), 543-548 (2002).
- Wu, G., et al. ISB recommendation on definitions of joint coordinate systems of various joints for the reporting of human joint motion–Part II: shoulder, elbow, wrist and hand. Journal of Biomechanics. 38 (5), 981-992 (2005).
- Franchi, M. V., Fitze, D. P., Raiteri, B. J., Hahn, D., Spörri, J. Ultrasound-derived biceps femoris long-head fascicle length: extrapolation pitfalls. Medicine and Science in Sports and Exercise. 52 (1), 233-243 (2020).
- Freitas, S. R., Marmeleira, J., Valamatos, M. J., Blazevich, A., Mil-Homens, P. Ultrasonographic Measurement of the Biceps Femoris Long-Head Muscle Architecture. Journal of Ultrasound in Medicine. 37 (4), 977-986 (2018).
- Nelson, C. M., Murray, W. M., Dewald, J. P. A. Motor Impairment-Related Alterations in Biceps and Triceps Brachii Fascicle Lengths in Chronic Hemiparetic Stroke. Neurorehabilitation and Neural Repair. 32 (9), 799-809 (2018).
- Alonso-Fernandez, D., Docampo-Blanco, P., Martinez-Fernandez, J. Changes in muscle architecture of biceps femoris induced by eccentric strength training with nordic hamstring exercise. Scandinavian Journal of Medicine & Science in Sports. 28 (1), 88-94 (2018).
- Herbert, R. D., et al. In vivo passive mechanical behaviour of muscle fascicles and tendons in human gastrocnemius muscle-tendon units. The Journal of Physiology. 589 (21), 5257-5267 (2011).
- Jakubowski, K. L., Terman, A., Santana, R. V. C., Lee, S. S. M. Passive material properties of stroke-impaired plantarflexor and dorsiflexor muscles. Clinical Biomechanics. 49, 48-55 (2017).
- Ward, S. R., Eng, C. M., Smallwood, L. H., Lieber, R. L. Are Current Measurements of Lower Extremity Muscle Architecture Accurate. Clinical Orthopaedics and Related Research. 467 (4), 1074-1082 (2009).
- Pillen, S., van Alfen, N. Skeletal muscle ultrasound. Neurological Research. 33 (10), 1016-1024 (2011).
- Scott, J. M., et al. Panoramic ultrasound: a novel and valid tool for monitoring change in muscle mass. Journal of Cachexia, Sarcopenia and Muscle. 8 (3), 475-481 (2017).
- Silbernagel, K. G., Shelley, K., Powell, S., Varrecchia, S. Extended field of view ultrasound imaging to evaluate Achilles tendon length and thickness: a reliability and validity study. Muscles, Ligaments and Tendons Journal. 6 (1), 104 (2016).
- Lichtwark, G. A., Bougoulias, K., Wilson, A. M. Muscle fascicle and series elastic element length changes along the length of the human gastrocnemius during walking and running. Journal of Biomechanics. 40 (1), 157-164 (2007).
- Farris, D. J., Sawicki, G. S. Human medial gastrocnemius force-velocity behavior shifts with locomotion speed and gait. Proceedings of the National Academy of Sciences. 109 (3), 977-982 (2012).
- Bolsterlee, B., Gandevia, S. C., Herbert, R. D. Effect of Transducer Orientation on Errors in Ultrasound Image-Based Measurements of Human Medial Gastrocnemius Muscle Fascicle Length and Pennation. PLoS ONE. 11 (6), (2016).
- Adkins, A. N., Dewald, J. P. A., Garmirian, L., Nelson, C. M., et al. Serial sarcomere number is substantially decreased within the paretic biceps brachii in chronic hemiparetic stroke. bioRxiv. , (2020).
- Pang, B. S., Ying, M. Sonographic measurement of Achilles tendons in asymptomatic subjects. Journal of Ultrasound in Medicine. 25 (10), 1291-1296 (2006).
- Ryan, E. D., et al. Test-retest reliability and the minimal detectable change for achilles tendon length: a panoramic ultrasound assessment. Ultrasound in Medicine & Biology. 39 (12), 2488-2491 (2013).
- Noorkoiv, M., Nosaka, K., Blazevich, A. J. Assessment of quadriceps muscle cross-sectional area by ultrasound extended-field-of-view imaging. European Journal of Applied Physiology. 109 (4), 631-639 (2010).
- Franchi, M. V., Fitze, D. P., Hanimann, J., Sarto, F., Spörri, J. Panoramic ultrasound vs. MRI for the assessment of hamstrings cross-sectional area and volume in a large athletic cohort. Scientific Reports. 10 (1), 14144 (2020).
- Yerli, H., Eksioglu, S. Y. Extended Field-of-View Sonography: Evaluation of the Superficial Lesions. Canadian Association of Radiologists Journal. 60 (1), 35-39 (2009).
- Kim, S. H., Choi, B. I., Kim, K. W., Lee, K. H., Han, J. K. Extended Field-of-View Sonography. Journal of Ultrasound in Medicine. 22 (4), 385-394 (2003).

