A Mouse Model to Investigate the Role of Cancer-Associated Fibroblasts in Tumor Growth
Summary
A protocol to co-inject cancer cells and fibroblasts and monitor tumor growth over time is provided. This protocol can be used to understand the molecular basis for the role of fibroblasts as regulators of tumor growth.
Abstract
Cancer-associated fibroblasts (CAFs) can play an important role in tumor growth by creating a tumor-promoting microenvironment. Models to study the role of CAFs in the tumor microenvironment can be helpful for understanding the functional importance of fibroblasts, fibroblasts from different tissues, and specific genetic factors in fibroblasts. Mouse models are essential for understanding the contributors to tumor growth and progression in an in vivo context. Here, a protocol in which cancer cells are mixed with fibroblasts and introduced into mice to develop tumors is provided. Tumor sizes over time and final tumor weights are determined and compared among groups. The protocol described can provide more insight into the functional role of CAFs in tumor growth and progression.
Introduction
Within the tumor microenvironment, one of the most prominent cell type is the cancer-associated fibroblast (CAF)1. These carcinoma-associated fibroblasts can play a tumor-suppressive role2,3. For example, S100A-expressing fibroblasts secrete collagens that can encapsulate carcinogens and protect against carcinoma formation4. Further, depletion of α-smooth muscle actin (SMA)-positive myofibroblasts in pancreatic cancer causes immunosuppression and accelerates pancreatic cancer progression2. CAFs can also co-evolve with cancer cells and promote tumor progression5,6,7,8. Fibroblasts can synthesize and secrete extracellular matrix proteins that create a tumor-promoting environment8. These extracellular matrix proteins can cause mechanical stiffening of the tissue, which is associated with tumor progression9,10. The deposited extracellular matrix can act as a physical barrier that inhibits immune infiltration11. Matrix deposition by CAFs has also been associated with tumor invasion as fibronectin generated by CAFs has been shown to promote tumor invasion12. CAFs promote angiogenesis and recruit immunosuppressive cells to the tumor microenvironment by secreting transforming growth factor-β (TGF- β), vascular endothelial growth factor (VEGF), interleukin-6 (IL-6), and CXC-chemokine ligand 12 (CXCL12)13,14,15. Because of their central role in promoting tumor growth, cancer-associated fibroblasts are an emerging target for anti-cancer therapy6,16,17,18.
The protocol below describes a method for testing how fibroblasts affect the growth of tumors in a well-established and widely-used mouse model of tumor growth. In order to understand the importance of fibroblasts in the tumor microenvironment, the standard protocol for introducing cancer cells into mice to monitor their growth was modified to include fibroblasts with the cancer cell introduction. The cancer cells can be introduced subcutaneously or intradermally. Intradermal introduction would result in tumors that arise from the skin itself. Xenografts in which cancer cells and fibroblasts are co-injected into mice represent an important methodological tool for dissecting the role of fibroblasts, subpopulations of fibroblasts and protein factors in the ability to promote cancer growth19,20,21. A detailed protocol for co-injection of cancer cells and fibroblasts into mice is provided. This method can be used to compare the presence or absence of fibroblasts, to compare fibroblasts from different sources20, or to compare fibroblasts with and without expression of specific proteins19. After the cancer cells and fibroblasts are introduced, tumor size can be monitored over time. At the end of the experiments, tumors can be dissected and weighed. By monitoring tumor growth over time, the importance of different factors can be dissected.
There are possible alternative approaches for studying the role of fibroblasts in tumor growth. As an example, there are Cre-loxed based models that provide for tissue-specific knockout of genes with drivers expressed preferentially in fibroblasts. Such approaches also provide opportunities to investigate the role of specific genes and pathways in fibroblasts for tumor progression. As compared with Cre-lox-based approaches, the protocol provided would represent a significantly more rapid approach to monitoring the role of fibroblasts because tumor growth would be monitored over just a few weeks. The provided approach is also significantly less expensive because it does not require generating and housing colonies of genetically engineered mice. The protocol provided can be used to rapidly test the effect of knockdown of different genes using shRNAs rather than needing to develop mouse colonies. The provided approach is also more flexible because it would allow for a comparison of different numbers of fibroblasts, different ratios of cancer cells and fibroblasts, knockdown of different genes, and even comparison of fibroblasts from different tissue sites or species. A Cre-lox approach would have the advantage that the fibroblasts are present within the mice in a more physiological context.
The protocol reported here would be valuable for scientists who seek to monitor the effects of fibroblasts on tumor growth rapidly and cost effectively. This protocol is especially valuable for scientists investigating different subsets of fibroblasts or fibroblasts from different sources on tumor growth on tumor growth. If it is important that tumor initiation occurs in a physiological context, then genetically engineered mouse models should be considered.
There are several possible approaches for performing these experiments. Immune-competent mice can be used as hosts, which would allow for investigation of fibroblast-immune cell interactions. For immune-competent mouse models, mouse cancer cells and mouse embryonic fibroblasts (MEFs) must be injected. The use of MEFs also allows the investigator to take advantage of the wide range of knockout mouse strains to test the presence or absence of a gene of interest. Alternatively, immune-deficient mice can be used to test the role of human fibroblasts in promoting the growth of tumors in mice that are derived from human cancer cells. Introduction of the cancer cells can be performed subcutaneously or orthotopically. For melanoma, as described below, the tumor-fibroblast mixture can be injected intradermally for orthotopic injection that more closely simulates the location within the skin where a melanoma would develop.
Protocol
All experiments described were approved by the Animal Care Committee at the University of California, Los Angeles.
NOTE: Select cancer cells and fibroblasts that match the host mice for mouse strain. Select cancer cells and fibroblasts that match the sex of the host mouse. Obtain mice from breeding colonies or purchase them from reputable vendors. Introduce tumors into mice that are ~8-10 weeks of age. Mice with fur will be in the telogen or resting phase of the hair follicle cycle. Plan for a ratio of 0.5 to 3 fibroblasts to cancer cell.
1. Determine the appropriate number of mice to be used for experimentation
- Prior to performing the studies, perform a power analysis to determine the appropriate number of mice to use for the studies. Use the following formula to determine sample size for an experiment with the specified power:
n = (Z(1-α/2)+Z(1-β)/ES)2
If α is the selected level of significance (usually 0.05), then 1-α/2 = 0.975 and Z=1.960.
β is the power and if 80% power is desired, then Z=0.84.
ES, the effect size = μ1-μ0/σ
where μ0 is the mean under hypothesis H0, μ1 is the mean under hypothesis H1, and σ is the standard deviation of the outcome of interest22.
NOTE: More information on calculating sample size can be found22.
2. Generate cancer cells for injection
- Determine the relevant growth rate with the following equation:
Gr = (ln (N(t)/N(0)))/t
NOTE: An example for cells that double in 24 hours is shown
Doubling time = ln(2)/growth rate
24 hours = ln(2)/growth rate
24*growth rate = .693
Growth rate = .693/24= .028875 - Determine the number of cancer cells to plate and the amount of time the cells need to be grown prior to injection.
NOTE: 0.25-1 million cancer cells are injected for each tumor formed. - Calculate the number of days required to generate a sufficient number of cells according to the equation
N(t)=N(0)egr*t
where N(t) is the number of cells at time t, N(0) is the number of cells at time 0, gr is the growth rate, and t is the time.
NOTE: An example of these calculations for growing 500,000 cells to generate 106 cells into each of 12 tumors. Based on these calculations, 5 days is sufficient to grow the necessary number of cells:
N(t)=N(0)egr*t
12 x 106 = 500,000e(gr*t)
12 x 106=500,000e(.028875*t)
24=e(.028875*x)
ln(24)=0.028875x
3.178=0.028875x
X=110 hours= 4.59 days
Allow extra time for cells to attach to the plate and for cells that are lost during collection. - Plate cancer cells.
NOTE: Wear gloves and lab coats when working with cultured cells.
NOTE: Perform cell manipulations in a laminar flow hood to minimize the introduction of microbes from the air into the samples. Treat the tissue culture hood with ultraviolet light prior to use. Use sterile technique and only sterile cell culture plasticware and consumables such as tissue culture-treated plates, pipets, and conical tubes.- Calculate the correct number and size of tissue culture plates based on the number of cells in the vial and the desired cell concentration once plated.
- Label tissue culture plates.
- Prepare a water bath with water at 37 °C.
- Aliquot medium into conical tubes and place in a water bath at 37 °C. Many cancer cells can be grown in Dulbecco's Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum (FBS).
- Remove the required number of vials of frozen cells from the liquid nitrogen freezer.
NOTE: Use freezer gloves when handling cells in a liquid nitrogen freezer. - Quickly thaw the cell vials in the water bath while gently shaking.
NOTE: Add ethanol to gloves while handling vials to maintain sterility. - Using sterile technique in a tissue culture hood, pipet the cells into a 15 mL conical tube containing 10 mL of DMEM + 10% FBS.
- Centrifuge the cell mixture at 180 x g for 5 minutes.
- Gently remove the supernatant with sterile suction or a sterile 10 mL pipet.
- Gently resuspend the cells in the pellet into 1 mL of DMEM + 10% FBS.
- Pipet the resuspended cells onto a labeled tissue culture plate with a sterile p1000.
- Using sterile 10 mL pipets, pipet medium with serum into tissue culture plates.
- Incubate tissue culture plates in a tissue culture incubator at 37 °C with 5% CO2.
- Expand cancer cells to generate sufficient cancer cells for injection.
NOTE: Monitor cells with light microscopy for confluency. Trypsinize every two to three days or as needed to prevent the cells from reaching 100% confluence. If a large number of cells are required, use hyperflasks (Table of Materials) to grow a large number of cells in a space- and medium-efficient way. Each time the cells need to be passaged, follow this procedure:- Remove the medium from the plate with sterile suction or a sterile 10 mL pipet.
- Gently wash the plate by pipetting on warm phosphate buffered saline (PBS). Then remove the PBS with sterile suction or a pipet.
- Pipet 5 mL of 1x Trypsin-ethylenediaminetetraacetic acid (EDTA) in PBS for a 10 cm plate onto the cells and incubate for 5 minutes at 37 °C.
- Remove cells from the plate with gentle taps to dislodge the cells from the plate.
- Using a sterile 10 mL pipet, collect the cells into DMEM with 10% FBS serum in conical tubes.
- Centrifuge conical tubes at 180 x g for 5 minutes.
- Remove the supernatant by pouring it off. The cell pellet should be visible. Watch it to make sure it remains in the tube.
- Resuspend the pelleted cells by adding 1 mL of DMEM + 10% FBS and pipetting up and down gently.
- Aliquot resuspended cells onto new tissue culture plates of the appropriate size with DMEM + 10% FBS (10 mL of media is suitable for a 10 cm tissue culture plate).
3. Generate fibroblasts for injection
NOTE: Primary fibroblasts will senesce after too many doublings/passages. It is important to use primary fibroblasts after a limited number of passages or doublings. Keep track of the number of passages or doublings that the fibroblasts have grown from the mouse or human skin. Use fibroblasts with fewer than 15 passages for primary human dermal fibroblasts. Use fibroblasts with fewer than 9 passages for mouse embryonic fibroblasts. Fibroblasts are altered when they become confluent. Trypsinize the fibroblasts when they are approximately 90% confluent. Fibroblasts will have different properties depending on how they are cultured. Many scientists promote culturing on more physiologically relevant substrates than tissue culture plates such as 3D collagen matrices that more effectively capture tissue-like environments23,24,25.
- Use the equation in section 2.1 to determine the growth rate for the fibroblast cells.
- Use the equation in section 2.2 to determine the number of days required to expand the fibroblasts.
- Plate the fibroblasts as described in section 2.3 using the appropriate medium on the appropriate day to ensure cancer cells and fibroblasts will be ready for injection on the same day.
- Expand the fibroblasts in the appropriate medium to generate a sufficient number of fibroblasts needed following the procedures described in section 2.4.
4. Shave mice to prepare mice for injection
NOTE: Wear lab coats, hair nets, shoe covers, and gloves when working with mice.
- One to two days prior to injection, in accordance with the rules of the Institutional Animal Care Committee, anesthetize the mice. If using isoflurane for anesthesia, use a mixture of isoflurane and O2. Place the mice in an anesthesia chamber and introduce an isoflurane (5%)/ O2 mixture to induce anesthesia. Then, place the nose cone on the anesthetized animal to maintain the surgical plane of anesthesia with a constant flow of isoflurane (2%)/O2 mixture.
- Check the animals to ensure they are in the appropriate surgical plane of anesthesia by pinching the mouse's toe and confirming the mouse does not move.
- Using an animal clipper with surgical blade #40, gently remove the fur from the appropriate positions on the flanks of the mice by passing the animal clipper over the skin multiple times until the fur is removed.
- Monitor the mice until they recover from anesthesia.
5. Prepare cancer cells and fibroblasts for injection
NOTE: Cancer cells and fibroblasts should be injected as soon as possible after collection, preferably within 30 minutes. On the morning of the injection, harvest the cancer cells and fibroblasts separately from tissue culture plates. For each cell type perform the following steps:
- Remove the medium from the tissue culture plate with gentle sterile suction or by pipetting off the medium.
- Gently pipet on 5 mL of PBS without disrupting the cells. Swirl the PBS. Gently aspirate the PBS with suction or pipet off the PBS.
- Gently pipet 5 mL of Trypsin-EDTA in PBS onto the cells layer and incubate for 5 minutes at 37 °C.
- Remove cells from the plate with gentle taps to dislodge the cells. Pipet the cells several times with a 10 mL pipet and transfer the cells to a conical tube containing DMEM with 10% FBS.
- Centrifuge the conical tubes at 180 x g for 5 minutes.
- Remove the supernatant by gently pouring it off, retaining the cell pellet. Wash the cell pellet twice with PBS. Each time, pipet 10 mL of PBS onto the cells. Gently mix with a p1000 pipet.
- Centrifuge the cells as in step 5.5. Gently remove the PBS with a pipette and repeat.
- Resuspend the washed, pelleted cells in 1 mL of PBS with a p1000.
- Clean a hemocytometer slide with alcohol and dry.
- Pipet 100 µL of the cell mixture into a tube and add 400 µL of 0.4% Trypan blue for a final Trypan blue concentration of 0.32%.
- Gently fill the chambers of a glass hemocytometer underneath the coverslip by pipetting the cell mixture until the chamber is full.
- Using a microscope, focus on the grid lines of a 10x objective.
- Count the live, unstained cells and blue cells in one set of 16 squares.
- Count all 4 sets of 16 squares.
- Average the cell count for the clear and blue cells from the 4 sets of 16 squares and multiply by 10,000. Multiply by 5 to correct for the 1:5 dilution from the Trypan blue.
- The final counts are the concentration in cells/mL for the viable and dead cells. Multiply by the volume to determine the total number of cancer cells and fibroblasts. Confirm that the fraction of cells that are viable is >80%.
- Confirm that there are a sufficient number of melanoma cells and fibroblasts for all of the injections planned, assuming at least 20% loss of sample during the injections.
- Transfer the required number of cells to a new conical tube and pellet the cells by centrifugation (180 x g for 5 minutes).
- Remove the supernatant with a 10 mL pipet.
- Resuspend the pellet at the required concentration in the appropriate amount of United States Pharmacopeia (USP) GRADE PBS (50 µL per tumor for intradermal injection and 100 µL per tumor for subcutaneous injection).
6. Inject cancer cells and fibroblasts into mice
NOTE: If approved by the institutional Animal Care Committee, inject two tumors into each mouse, one on each flank. Randomize which mouse will receive which injection on the right and left flanks. Depending on the number of mice to be injected, anesthetize the mice and inject cancer cells and fibroblasts into the mice in batches.
- For subcutaneous injection of cancer cells
- Anesthetize mice as described in section 4.1.
- Wipe the shaved skin with alcohol swabs.
- Fill a sterile syringe with the desired volume of cells or cell mixture and cap the syringe with a 27-gauge needle.
NOTE: Make sure no air bubbles are present. Flick the syringe as needed to remove bubbles. Once cells have been introduced into the syringe, keep the cells within the syringes well-mixed by continuously inverting the syringes to prevent clumping. - Insert the needle into the subcutaneous region on the mouse's flank and gently inject the cells into the mouse. Use 100 μL cell suspension per tumor for subcutaneous injection(using volume more than 100 μL per injection is not recommended).
- Monitor the mice until they recover from anesthesia. If needed, provide the mice with extra insulating materials like huts and/or heating pads to help the mice to fully recover.
- For intradermal injections of cancer cells
- Anesthetize mice as described in section 4.1.
- Wipe the shaved skin with alcohol swabs.
- Fill a sterile syringe with the desired volume of cells or cell mixture and cap the syringe with a 27-gauge needle.
NOTE: Make sure no air bubbles are present. Flick the syringe as needed to remove bubbles. Once cells have been introduced into the syringe, keep the cells within the syringes well-mixed by continuously inverting the syringes to prevent clumping. - Insert a 27-gauge needle into the intradermal region of the skin on the mouse's flank and gently inject the cells into the intradermal region in the mouse. Use 50μL cell suspension per tumor for intradermal injection (using volume more than 50 μL per injection is not recommended).
- Monitor the mice until they recover from anesthesia. If there are signs that the mice are in distress, consult a veterinarian. If needed, provide the mice with extra insulating materials like huts and/or heating pads to help the mice to fully recover.
7. Monitor mice during tumor growth
- Monitor mice daily for any signs of pain or distress (hunched posture, writhing, vocalization, stretching out along the cage, pressing their abdomen to the bottom of the cage floor, or reluctance to move around the cage) or abscess formation. If there are signs that the mice are in distress, consult a veterinarian.
- Once tumors are formed, measure tumor volume approximately every two to three days. Measure tumor length (the longest side) and width (perpendicular to length) with calipers.
NOTE: Best practices are for the same person to perform these measurements throughout the experiment to reduce variability in the data. For measurements, anesthetize mice as described in section 4.1 - Calculate tumor volumes with the formula Volume= 0.5 x (Length x Width2).
8. Harvest tumors and measure tumor weights
- Euthanize mice when tumor growth reaches an experimental endpoint established by the institution Committee on Animal Care. Place mice in a chamber and euthanize them with slow (20-30% per minute) displacement of chamber air with compressed CO2 for one minute past cessation of respiration.
- Perform cervical dislocation on the mice. Restrain the rodent on a firm, flat surface. Place a sturdy pen or another object against the back of the neck at the base of the skull. Quickly push forward and down with the object restraining the head while pulling backward with the hand holding the base of the tail. Confirm with palpation that the spinal cord is severed.
NOTE: Cervical dislocation should be performed by experienced lab personnel. - For each mouse in the experiment, confirm that the mouse is no longer alive by checking that there is no breathing, no heartbeat and no response to a toe pinch.
- Using a sterile scalpel and surgical scissors, excise the entire tumor from the mouse. With surgical scissors, trim non-tumor tissue from the tumor.
- Weigh the tumor on an analytical balance and record the tumor weight.
9. Statistical analysis of tumor volumes and tumor weights
- Compare tumor volumes over time in each group with repeated measures analysis of variance (ANOVA). Perform paired analysis if two tumors were injected per mouse.
- Compare tumor weights among the groups with ANOVA using a two-tailed test. Tukey's post-hoc test can then be used to determine which groups are significantly different from each other. Perform paired analysis if two tumors were injected per mouse.
Representative Results
A2058 human melanoma cells and primary human dermal fibroblasts were cultured under sterile conditions. Cells were collected and washed three times with PBS. Immunodeficient mice (NU/J – Foxn1 nude strain) were injected subcutaneously on one flank with 0.25 million A2058 melanoma cells alone. On the other flank, mice were injected with a mixture of 0.25 million A2058 melanoma cells and 0.75 million fibroblasts. Cells were injected into 12 immune-deficient mice. Injections into left and right flanks were randomized. Tumor volumes were monitored on days 12, 14, 16, 19, and 21 days post injection (Figure 1A). Upon euthanasia, tumors were excised, imaged (Figure 1B) and weighed (Figure 1C). The presence of the fibroblasts results in significantly larger tumors.
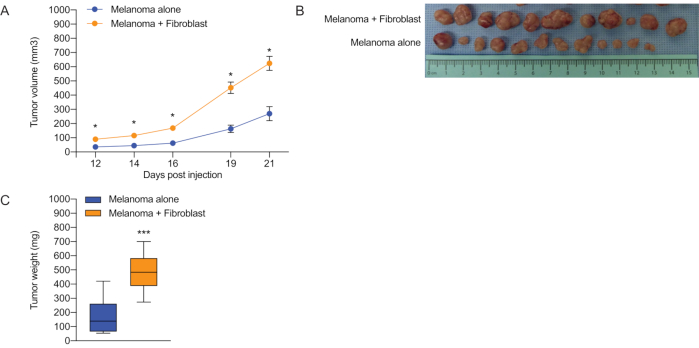
Figure 1: Comparison of melanoma growth in the presence and absence of co-injected fibroblasts. Melanoma cells were introduced into nude mice either with or without primary skin fibroblasts. (A) Plot of tumor volume over time for melanomas with and without co-injected fibroblasts. Volume was calculated as 0.5*length*width2. Mean volumes and standard error of the mean (SEM) are plotted. ANOVA was performed to determine significance. (B) Images of excised tumors. (C) Final weights of tumors at the end of the experiment on day 21. Mean weights and SEM are plotted. An unpaired, two-tailed t-test was used to compare final weights. * indicates p < 0.05, *** indicates p < 0.001. Please click here to view a larger version of this figure.
Discussion
In the experiment in Figure 1, co-introducing human dermal fibroblasts with human A2058 melanoma cells resulted in larger tumors than when the melanoma cells were introduced without co-injected fibroblasts. This difference could be easily detected based on tumor volume and tumor weight. The results are consistent with multiple reports that cancer-associated fibroblasts can promote tumor growth5,6,7,8. In addition to the endpoints discussed here such as tumor volume and tumor weight, additional endpoints can also be monitored. Additional analyses are also possible. For instance, the tumors that develop can be collected into formalin for fixation, paraffin-embedded and analyzed with hematoxylin and eosin for histology. Alternatively, the tumors developed can be collected into optimal cutting temperature compound (OCT) for further analysis with immunofluorescence for specific proteins.
One important limitation of these experiments is that the fibroblasts that are co-injected with the cancer cells may senesce or die after a few cell divisions. Alternatively, co-injected fibroblasts may migrate away from the tumor, in which case, they may not affect tumor growth. If the fibroblasts senesce, die or migrate away from the tumor, then they will become less abundant over time and the effect of the co-introduced fibroblasts will become less important for tumor growth. Indeed, the introduced cancer cells are expected to recruit additional fibroblasts from the host. For experiments in which a specific gene is knocked down or knocked out in the introduced fibroblasts, over time, recruitment of host fibroblasts that express the encoded protein will be expected to reduce any phenotype that might be observed. To monitor the presence of the introduced fibroblasts, the fibroblasts can be genetically engineered to express a fluorescent protein such as GFP26,27. If the fibroblasts express GFP, then flow cytometry can be used to monitor the number of fibroblasts that are present within the tumor on different days after they are introduced. As an alternative, the luciferase enzyme can be introduced in the fibroblasts and bioluminescence imaging can be used to monitor the amount of fibroblasts in the mice over time. If the fibroblasts are rapidly eliminated from the tumor microenvironment, Cre-lox genetically engineered mouse models would address this concern and complement the studies described.
If the experiments are performed by introducing human cancer cells and fibroblasts into nude mice, then the limited immune system is expected to have a significant impact on the results. Athymic nude mice (NU/J) lack T cells and therefore lack cell-mediated immunity. They also have a partial defect in B cell development. Cell mediated immunity, that is the killing of cells via activated CD8+ T cells, can play an important role in the suppression of tumor growth28,29. In recent studies, interplay between cancer-associated fibroblasts and CD8+ T cells has been reported30,31,32. Introducing mouse cancer cells and mouse fibroblasts into mice with an immune system can be considered as an alternative approach that would address this issue.
Another alternative approach is to use genetically engineered mouse models in which the Cre-lox system is used to modulate the levels or activity of specific proteins in host fibroblasts. One such approach is to use fibroblast-enriched Cre-lox models with drivers such as FSP1, Col1a1, Col1a2, or PDGFRa33,34,35. With a Cre/lox recombinase model, there would not be concern that the injected fibroblasts will die or migrate away from the tumor.
There are a few steps in the protocol provided that are critical for its success. The growth of the fibroblasts is an important part of the protocol and is likely to contribute significantly to the outcome. Primary fibroblasts senesce easily, and the passage or doublings must be carefully monitored to ensure the fibroblasts are in the exponential growth phase and have not senesced. Senescent fibroblasts are expected to secrete high levels of cytokines that can have a dramatic effect on tumor growth36,37,38. It is consequently of high importance that passage number is carefully documented and that the fibroblasts injected are not senescent unless the protocol calls for analysis of senescent fibroblasts.
When fibroblasts become confluent, they can be expected to undergo a large number of changes, some of which will likely be reversed when they re-enter the cell cycle39,40,41,42,43,44. Maintaining the fibroblasts in a proliferative state and splitting them when they are 90% confluent is important for ensuring that the studies are as consistent as possible. Culturing fibroblasts on tissue culture plates likely also affects their behavior and alternatives such as 3D collagen matrices should be considered23,24,25. Fibroblasts cultured in 3D matrices show substantially different migration patterns than fibroblasts grown on 2D surfaces45,46,47,48,49, which would likely affect their tumor-promoting capacity when introduced into tumors, where the fibroblasts will play an active role in establishing the tumor extracellular matrix microenvironment50.
Injecting melanoma cells into the mouse via intradermal injection can yield more reproducible tumor growth than subcutaneous injections. This is a tricky step as there is very little space in the intradermal region. It is important to carefully position the needle. If intradermal injections are performed, then only 50 µL of solution is injected. Even for experienced researchers, these injections can leak. It is important to take note of whether each injection leaked so that, if no tumor forms, it can be eliminated from the final analysis.
Preparing syringes for injection is another critical step. It is important to remove all bubbles from the syringe, which can require flicking the syringe to eliminate the bubbles. It is also important that the cells do not clump. Repeatedly invert the syringes to prevent clumping.
Another tricky aspect of these experiments is that measuring tumor volume can be variable from person to person. In order to limit variability in tumor volume measurements, it is helpful to assign a single lab member responsibility for taking all of the tumor volume measurements in all mice and at all timepoints throughout an experiment.
It is preferable to inject mice with cancer cells from the same sex as the host mouse whenever possible. It is possible to use both sexes and include sex as a variable in ANOVA models to determine whether sex affects tumor size over time.
Understanding the tumor microenvironment is essential for gaining insight into tumorigenesis. The protocol can provide a basis for understanding the role of fibroblasts, different types of fibroblasts, and different pathways within fibroblasts that can affect tumor growth. Findings from these experiments may identify new therapeutic targets that will prevent fibroblasts from promoting tumor growth18.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
The authors would like to acknowledge all of the members of the Coller laboratory for helpful input. H.A.C. was the Milton E. Cassel scholar of the Rita Allen Foundation. We acknowledge NIH/NCI 1 R01 CA221296-01A1, NIH 1 R01 AR070245-01A1, Melanoma Research Alliance Team Science Award, Cancer Research Institute Clinical Laboratory Integration Program Award, the Iris Cantor Women's Health Center/UCLA CTSI NIH Grant UL1TR000124, University of California Cancer Research Coordinating Committee, David Geffen School of Medicine Metabolism Theme Award, the Clinical Translational Science Institute and Jonsson Comprehensive Cancer Center, Innovation Awards from the Broad Stem Cell Research Center (Rose Hills and Ha Gaba), an Award from the UCLA SPORE in Prostate Cancer (National Cancer Institute of the National Institutes of Health under Award Number P50CA092131), an Innovation Award from the Broad Stem Cell Center, the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA, the Tumor Cell Biology Training Program (USHHS Ruth L. Kirschstein Institutional National Research Service Award # T32 CA009056), the Dermatology T32 Program at UCLA AR071307, and the UCLA Muscle Cell Biology, Pathophysiology and Therapeutics T32 Training Program 5 T32 AF 65972.
Materials
| 26G Needles | Fisher Scientific | 14-826-10 | |
| Alcohol swabs | Fisher Scientific | 326895 | |
| Animal clipper miniARCO with surgical blade #40 | WAHL Professional | 8787-450A | |
| Athymic nude mice (NU/J) | The Jackson labs | 002019 | These mice are immunocompromised and can be used for experiments in which human cells are introduced. Immunocompetent mice can also be used if mouse cancer cells and fibroblasts will be introduced. |
| Cancer cells | ATCC | ATCC® CRL-11147™ | This is the catalog number for a primary human melanoma cell line. Other cancer cell types can also be used. |
| Cell Culture Multi Flasks | Fisher Scientific | 14-826-95 | |
| Centrifuge for conical tubes capable of reaching 180 x g | Fisher Scientific | 14-432-22 | |
| Countess Cell Counting Chamber | Fisher Scientific | C10228 | |
| Dulbecco's Modified Eagle Medium | Fisher Scientific | 11965-118 | |
| Fetal bovine serum | Fisher Scientific | MT35010CV | |
| Fibroblasts | ATCC | PCS-201-012 | We isolate fibroblasts from skin in our lab. This is a catalog number for an adult primary human dermal fibroblast cell line. MEFs and fibroblasts derived from other sites can also be used. |
| Isoflurane | Henry Schein Animal Health | NDC 11695-6776-2 | |
| PBS USP grade for injection into mice | Fisher Scientific | 50-751-7476 | |
| Sterile 10 ml serological pipet | Celltreat | 667210B | |
| Sterile 5 ml serological pipet | Celltreat | 229005B | |
| Sterile 50 ml centrifuge tubes | Genesee Scientific | 28-108 | |
| Sterile Syringe Filters pore size 0.2 microns | Fisher Scientific | 09-740-61A | |
| Sterile tissue culture-grade Trypsin-EDTA | Fisher Scientific | 15400054 | |
| Sterile tissue-culture grade PBS | Fisher Scientific | 50-751-7476 | |
| Sterle 25 ml serological pipet | Celltreat | 667225B | |
| TC treated 100 x 20 mm dishes | Genesee Scientific | 25-202 | |
| TC treated 150 x 20 mm dishes | Genesee Scientific | 25-203 | |
| TC treated 60 x 15 mm dishes | Genesee Scientific | 25-260 | |
| Trypan blue | Fisher Scientific | C10228 |
Riferimenti
- Liu, T., et al. Cancer-associated fibroblasts: an emerging target of anti-cancer immunotherapy. Journal of Hematology and Oncololgy. 12 (1), 86 (2019).
- Ozdemir, B. C., et al. Depletion of carcinoma-associated fibroblasts and fibrosis induces immunosuppression and accelerates pancreas cancer with reduced survival. Cancer Cell. 25 (6), 719-734 (2014).
- Rhim, A. D., et al. Stromal elements act to restrain, rather than support, pancreatic ductal adenocarcinoma. Cancer Cell. 25 (6), 735-747 (2014).
- Zhang, J., et al. Fibroblast-specific protein 1/S100A4-positive cells prevent carcinoma through collagen production and encapsulation of carcinogens. Ricerca sul cancro. 73 (9), 2770-2781 (2013).
- Ohlund, D., Elyada, E., Tuveson, D. Fibroblast heterogeneity in the cancer wound. Journal of Experimental Medicine. 211 (8), 1503-1523 (2014).
- Chen, X., Song, E. Turning foes to friends: targeting cancer-associated fibroblasts. Nature Reviews Drug Discovery. 18 (2), 99-115 (2019).
- Wang, W., et al. Crosstalk to stromal fibroblasts induces resistance of lung cancer to epidermal growth factor receptor tyrosine kinase inhibitors. Clinical Cancer Research. 15 (21), 6630-6638 (2009).
- Hwang, R. F., et al. Cancer-associated stromal fibroblasts promote pancreatic tumor progression. Ricerca sul cancro. 68 (3), 918-926 (2008).
- Tsujino, T., et al. Stromal myofibroblasts predict disease recurrence for colorectal cancer. Clinical Cancer Research. 13 (7), 2082-2090 (2007).
- Laklai, H., et al. Genotype tunes pancreatic ductal adenocarcinoma tissue tension to induce matricellular fibrosis and tumor progression. Nature Medicine. 22 (5), 497-505 (2016).
- Cukierman, E., Bassi, D. E. Physico-mechanical aspects of extracellular matrix influences on tumorigenic behaviors. Seminars in Cancer Biology. 20 (3), 139-145 (2010).
- Attieh, Y., et al. Cancer-associated fibroblasts lead tumor invasion through integrin-beta3-dependent fibronectin assembly. Journal of Cell Biology. 216 (11), 3509-3520 (2017).
- Ahmadzadeh, M., Rosenberg, S. A. TGF-beta 1 attenuates the acquisition and expression of effector function by tumor antigen-specific human memory CD8 T cells. Journal of Immunology. 174 (9), 5215-5223 (2005).
- Feig, C., et al. Targeting CXCL12 from FAP-expressing carcinoma-associated fibroblasts synergizes with anti-PD-L1 immunotherapy in pancreatic cancer. Proceedings of the National Academy of Science, U S A. 110 (50), 20212-20217 (2013).
- Kojima, Y., et al. Autocrine TGF-beta and stromal cell-derived factor-1 (SDF-1) signaling drives the evolution of tumor-promoting mammary stromal myofibroblasts. Proceedings of the National Academy of Science, U S A. 107 (46), 20009-20014 (2010).
- Kalluri, R. The biology and function of fibroblasts in cancer. Nature Reviews Cancer. 16 (9), 582-598 (2016).
- Ziani, L., Chouaib, S., Thiery, J. Alteration of the Antitumor Immune Response by Cancer-Associated Fibroblasts. Frontiers in Immunology. 9, 414 (2018).
- Sahai, E., et al. A framework for advancing our understanding of cancer-associated fibroblasts. Nature Reviews Cancer. 20 (3), 174-186 (2020).
- Grum-Schwensen, B., et al. Suppression of tumor development and metastasis formation in mice lacking the S100A4(mts1) gene. Ricerca sul cancro. 65 (9), 3772-3780 (2005).
- Kojima, M., et al. Human subperitoneal fibroblast and cancer cell interaction creates microenvironment that enhances tumor progression and metastasis. PLoS One. 9 (2), 88018 (2014).
- Noel, A., et al. Enhancement of tumorigenicity of human breast adenocarcinoma cells in nude mice by matrigel and fibroblasts. British Journal of Cancer. 68 (5), 909-915 (1993).
- Sullivan, L. M. Estimation from samples. Circulation. 114 (5), 445-449 (2006).
- Yamada, K. M., Cukierman, E. Modeling tissue morphogenesis and cancer in 3D. Cell. 130 (4), 601-610 (2007).
- Damianova, R., Stefanova, N., Cukierman, E., Momchilova, A., Pankov, R. Three-dimensional matrix induces sustained activation of ERK1/2 via Src/Ras/Raf signaling pathway. Cell Biology International. 32 (2), 229-234 (2008).
- Rhee, S. Fibroblasts in three dimensional matrices: cell migration and matrix remodeling. Experimental & Molecular Medicine. 41 (12), 858-865 (2009).
- Hoffman, R. M. Application of GFP imaging in cancer. Labortatory Investigation. 95 (4), 432-452 (2015).
- Orimo, A., et al. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell. 121 (3), 335-348 (2005).
- Naito, Y., et al. CD8+ T cells infiltrated within cancer cell nests as a prognostic factor in human colorectal cancer. Ricerca sul cancro. 58 (16), 3491-3494 (1998).
- Fu, C., Jiang, A. Dendritic Cells and CD8 T Cell Immunity in Tumor Microenvironment. Frontiers Immunolofy. 9, 3059 (2018).
- Lakins, M. A., Ghorani, E., Munir, H., Martins, C. P., Shields, J. D. Cancer-associated fibroblasts induce antigen-specific deletion of CD8 (+) T Cells to protect tumour cells. Nature Communications. 9 (1), 948 (2018).
- Kato, T., et al. Cancer-Associated Fibroblasts Affect Intratumoral CD8(+) and FoxP3(+) T Cells Via IL6 in the Tumor Microenvironment. Clinical Cancer Research. 24 (19), 4820-4833 (2018).
- Gorchs, L., et al. Human Pancreatic Carcinoma-Associated Fibroblasts Promote Expression of Co-inhibitory Markers on CD4(+) and CD8(+) T-Cells. Frontiers Immunology. 10, 847 (2019).
- Duscher, D., et al. Fibroblast-Specific Deletion of Hypoxia Inducible Factor-1 Critically Impairs Murine Cutaneous Neovascularization and Wound Healing. Plastic Reconstructive Surgery. 136 (5), 1004-1013 (2015).
- Zheng, B., Zhang, Z., Black, C. M., de Crombrugghe, B., Denton, C. P. Ligand-dependent genetic recombination in fibroblasts : a potentially powerful technique for investigating gene function in fibrosis. American Journal of Pathology. 160 (5), 1609-1617 (2002).
- Swonger, J. M., Liu, J. S., Ivey, M. J., Tallquist, M. D. Genetic tools for identifying and manipulating fibroblasts in the mouse. Differentiation. 92 (3), 66-83 (2016).
- Krtolica, A., Parrinello, S., Lockett, S., Desprez, P. Y., Campisi, J. Senescent fibroblasts promote epithelial cell growth and tumorigenesis: a link between cancer and aging. Proceedings of the National Academy of Science, U S A. 98 (21), 12072-12077 (2001).
- Ortiz-Montero, P., Londono-Vallejo, A., Vernot, J. P. Senescence-associated IL-6 and IL-8 cytokines induce a self- and cross-reinforced senescence/inflammatory milieu strengthening tumorigenic capabilities in the MCF-7 breast cancer cell line. Cell Communication and Signaling. 15 (1), 17 (2017).
- Coppe, J. P., et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biology. 6 (12), 2853-2868 (2008).
- Coller, H. A., Sang, L., Roberts, J. M. A new description of cellular quiescence. PLoS Biolofy. 4 (3), 83 (2006).
- Mitra, M., et al. Alternative polyadenylation factors link cell cycle to migration. Genome Biolofy. 19 (1), 176 (2018).
- Lemons, J. M., et al. Quiescent fibroblasts exhibit high metabolic activity. PLoS Biology. 8 (10), 1000514 (2010).
- Suh, E. J., et al. A microRNA network regulates proliferative timing and extracellular matrix synthesis during cellular quiescence in fibroblasts. Genome Biology. 13 (12), 121 (2012).
- Legesse-Miller, A., et al. Quiescent fibroblasts are protected from proteasome inhibition-mediated toxicity. Molecular Biology of the Cell. 23 (18), 3566-3581 (2012).
- Evertts, A. G., et al. H4K20 methylation regulates quiescence and chromatin compaction. Molecular Biology of the Cell. 24 (19), 3025-3037 (2013).
- Johnson, L. A., et al. Matrix stiffness corresponding to strictured bowel induces a fibrogenic response in human colonic fibroblasts. Inflammatory Bowel Disease. 19 (5), 891-903 (2013).
- Marinkovic, A., Liu, F., Tschumperlin, D. J. Matrices of physiologic stiffness potently inactivate idiopathic pulmonary fibrosis fibroblasts. American Journal of Respiratory Cell and Molecular Biology. 48 (4), 422-430 (2013).
- Tschumperlin, D. J. Fibroblasts and the ground they walk on. Physiology (Bethesda). 28 (6), 380-390 (2013).
- Tschumperlin, D. J., et al. Mechanotransduction through growth-factor shedding into the extracellular space. Nature. 429 (6987), 83-86 (2004).
- Doyle, A. D., Wang, F. W., Matsumoto, K., Yamada, K. M. One-dimensional topography underlies three-dimensional fibrillar cell migration. Journal of Cell Biology. 184 (4), 481-490 (2009).
- Alexander, J., Cukierman, E. Stromal dynamic reciprocity in cancer: intricacies of fibroblastic-ECM interactions. Current Opinions in Cell Biology. 42, 80-93 (2016).

