Rating L-DOPA-Induced Dyskinesias in the Unilaterally 6-OHDA-Lesioned Rat Model of Parkinson’s Disease
Summary
Rodent models of L-DOPA-induced dyskinesias are invaluable tools to identify therapeutic interventions to attenuate the development or alleviate the manifestations that emerge due to the repeated administration of L-DOPA. This protocol demonstrates how to induce and analyze dyskinetic-like movements in the unilaterally 6-OHDA-lesioned rat model of Parkinson’s disease.
Abstract
L-DOPA-induced dyskinesias (LIDs) refer to motor complications that arise from prolonged L-DOPA administration to patients with Parkinson’s disease (PD). The most common pattern observed in the clinic is the peak-dose dyskinesia which consists of clinical manifestations of choreiform, dystonic, and ballistic movements. The 6-hydroxydopamine (6-OHDA) rat model of PD mimics several characteristics of LIDs. After repeated L-DOPA administration, 6-OHDA-lesioned rats exhibit dyskinetic-like movements (e.g., abnormal involuntary movements, AIMs). This protocol demonstrates how to induce and analyze AIMs in 6-OHDA-lesioned rats with 90%-95% dopaminergic depletion in the nigrostriatal pathway. Repeated administration (3 weeks) of L-DOPA (5 mg/kg, combined with 12.5 mg/kg of benserazide) can induce the development of AIMs. The time course analysis reveals a significant increase in AIMs at 30-90 min (peak-dose dyskinesia). Rodent models of LIDs are an important preclinical tool to identify effective antidyskinetic interventions.
Introduction
The dopamine precursor L-3,4-dihydroxyphenylalanine (L-DOPA) represents the most effective treatment for the motor symptoms of Parkinson's disease (PD)1. L-DOPA therapy may ameliorate motor symptoms associated with PD but loses effectiveness with time. Motor fluctuations such as "wearing-off fluctuation" or "end-of-dose deterioration" manifest clinically as a shortened duration of the effect of single L-DOPA doses2. In other cases, clinical manifestations consist of slow twisting movements and abnormal postures (dystonia)3 and occur when dopamine levels are low (off-period dystonia)4. On the other hand, L-DOPA-induced dyskinesias (LIDs) appear when dopamine levels in the plasma and the brain are high5.
LIDs produce debilitating side effects that include motor complications such as choreiform, dystonic, and ballistic6 movements. Once established, LIDs occur after every L-DOPA administration. Motor complications occur in 40%-50% of PD patients undergoing L-DOPA therapy for 5 years, and the incidence increases over the years7. Although the pathophysiological mechanisms involved in the development of LIDs in PD patients are not yet fully elucidated, the extent of dopaminergic denervation, pulsatile L-DOPA administration, downstream changes in striatal proteins and genes, and abnormalities in non-dopamine transmitter systems are factors that contribute to the development of these unwanted side effects6,8,9,10.
The neurotoxin 6-hydroxydopamine (6-OHDA) is a well-characterized tool to study PD in rodents11,12,13,14. Since 6-OHDA does not cross the blood-brain barrier, it must be injected directly into the nigrostriatal pathway. 6-OHDA-induced dopaminergic depletion is concentration- and site-dependent15. Unilateral administration of 6-OHDA at the medial forebrain bundle (MFB) can produce severe (>90%) nigrostriatal damage in rodents16,17,18,19. Chronic administration of L-DOPA to severe unilaterally 6-OHDA-lesioned rodents causes the appearance of dyskinetic-like movements named abnormal involuntary movements (AIMs). Dyskinetic-like movements in rodents share similar molecular, functional, and pharmacological mechanisms related to LIDs in PD patients5. Therefore, 6-OHDA-lesioned rats20 and mice21 are valuable preclinical models to study LIDs. When treated chronically (7-21 days) with therapeutic doses of L-DOPA (5-20 mg/kg), unilaterally 6-OHDA-lesioned rats and mice show a gradual development of AIMs that affect the forelimb, trunk, and orofacial muscles contralateral to the lesion17,18,19,20,22,23,24. These movements are presented at a time course similar to L-DOPA-induced peak-dose dyskinesias in PD patients25 and are characterized by hyperkinetic movements and dystonia5. AIMs are usually scored based on their severity (e.g., when a specific AIM is present) and amplitude (e.g., characterized by the amplitude of each movement)5,23,25.
6-OHDA-lesioned rodent models of LIDs present face validity (i.e., the model has several characteristics that look like the human condition)5,11,26,27,28. Rodent AIMs, similar to what occurs in PD patients, are seen as hyperkinetic (forelimb and orolingual) and dystonic (axial) movements29 and mimics peak-dose dyskinesia. At the molecular and functional level, rodent models share many pathological characteristics with PD patients5, such as upregulation of FosB/ΔFosB19,26,30,31,32,33 and serotonin transporter (SERT)34,35. Concerning predictive validity, drugs that reduce LIDs in PD patients (e.g., the N-methyl-D-aspartate (NMDA) receptor antagonist amantadine) present antidyskinetic efficacy in the rodent model22,36,37,38,39.
The rodent AIMs rating scale was created based on four AIMs subtypes that include AIMs affecting the head, neck, and trunk (axial AIMs), hyperkinetic forelimb movements (limb AIMs), and dyskinetic-like orolingual movements (orolingual AIMs). Although contralateral rotation (locomotive AIMs) is also present in unilaterally lesioned rodents20,22,23,25,40, it has not been scored as a dyskinetic-like movement since it may not represent a specific measure of LIDs22,37,41.
Here, we will describe how to induce and analyze dyskinetic-like movements (axial, limb, and orolingual AIMs) in the severe (>90%) unilaterally 6-OHDA-lesioned rat model of PD. We organized our protocol based on the previous literature and our laboratory expertise.
Protocol
All experiments were performed in accordance with The Ethics Committee of the Faculty of Philosophy, Sciences, and Letters of Ribeirão Preto (CEUA/FFCLRP 18.5.35.59.5).
1. 6-OHDA lesion
- Use Sprague-Dawley male rats weighing 200-250 g at the beginning of the experiments (6 weeks). House the animals (2-3 per cage) under standard laboratory conditions (12:12 h light/dark cycle, lights on at 06:00 h, temperature-controlled facilities (22-24 °C), with food and water available ad libitum.
NOTE: A complete description of how to generate severe 6-OHDA-lesioned rats is described elsewhere42. - Appropriately acclimate animals before the beginning of any experimental procedure.
- Administer (intraperitoneally, i.p.) the norepinephrine transporter inhibitor imipramine (20 mg/kg, dissolved in 0.9% saline solution) 30 min before surgery.
NOTE: Imipramine administration will increase 6-OHDA selectivity for dopaminergic neurons. - Administer ketamine/xylazine (70/10 mg/kg) intraperitoneally.
- Make sure the animal is deeply anesthetized by the lack of response to toe pinch and positioned in a prone position in the stereotaxic apparatus on the top of a heating pad.
- Remove the fur on the head and disinfect the surgical area 3 times, alternating between an iodine-based scrub and alcohol.
- Use a scalpel to make an incision (approximately 1 cm) at the region where the microinjection will occur.
- Clean the skull region with cotton swabs and ensure the Bregma and Lambda are exposed.
- Ensure MFB stereotaxic coordinates are taken from bregma43,44,45: -4.3 mm anterior, 1.6 mm lateral (right side), and 8.3 mm ventral (from the dura mater).
- Administer the 6-OHDA at a rate of 0.4 µL/min (10 µg in 4 µL of saline solution containing 0.1% ascorbic acid) unilaterally in the right medial forebrain bundle (MFB) using a 50 µL Hamilton glass syringe.
- At the end of the surgery, suture the scalp incision and rehydrate the animal with warm (~37 °C) sterile 0.9% saline solution (~10 mL/kg, s.c.).
- Remove the animal from the stereotaxic frame and place it in a warmed recovery cage. Monitor until consciousness is regained.
- Assess the effectiveness of the dopaminergic lesion 4 weeks post-lesion using a stepping test46,47.
NOTE: In this test, the akinesia of the forelimb contralateral to the lesion is assessed through the number of adjusting steps with the forepaw contralateral to the lesion. Rats presenting three or fewer adjusting steps with the contralateral forelimb are included in the study as putative severely 6-OHDA-lesioned rats48.
2. L-DOPA chronic treatment
- Start the chronic treatment on Monday, 4 weeks post-6-OHDA lesion.
NOTE: Since AIMs will be videotaped for 3 consecutive days per week (Wednesday, Thursday, and Friday) for 3 weeks, it is recommended to start the treatment on Monday. - Treat the rats with freshly prepared L-DOPA (via subcutaneous (s.c.), 1 mL/kg) plus benserazide hydrochloride (5 mg/kg and 12.5 mg/kg, respectively) for 3 weeks, once daily from Monday to Friday45.
NOTE: Once established, AIMs will manifest with each administration of L-DOPA. Therefore, L-DOPA administration 2-4 times a week after the 3-week induction period is sufficient to maintain a stable expression of AIMs48.
3. AIMs recording and scoring
- Perform experiments between 09:00 h and 17:00 h. Ensure that the investigator is entirely unaware of the rats' identity and pharmacological treatment.
- Carefully put the rat inside a transparent cylinder (20 cm diameter x 40 cm tall) and allow it to acclimatize for at least 15 min. Ensure that the floor is covered with the bedding material. Mirrors must be positioned behind the cylinder so the experimenter can observe the animal from all possible angles.
NOTE: If there is more than one rat per experiment, use a cylinder for each rat. Manipulation of the animals between sessions can interfere with the behavioral analysis. - Position a high-resolution video camera in a way that favors the viewing of axial, limb, and orolingual AIMs. Since rotational behavior is present under these experimental conditions, the mirrors behind the cylinder will allow tracking AIMs at a 360° angle. Use a tripod or fix the camera directly to the bench.
NOTE: Orolingual AIMs can be challenging to score, especially if there is a high incidence of rotational behavior. Capturing videos with the camera positioned slightly below the plane where the animals are at a 15° angle will help observe this movement subtype. - Gently remove the animal from the cylinder and administer L-DOPA (5 mg/kg combined with 12.5 mg/kg of benserazide, s.c.).
- Put the animal back in the cylinder and start a timer to track AIMs after L-DOPA injection. Use a video camera to record AIMs for 180 min after the injection of L-DOPA. Although the analysis can be carried out by direct observation of the animal, offline scoring is preferred.
NOTE: Ensure the timer is also videotaped so that AIMs can be scored at precise times after L-DOPA administration. - Score AIMs at 30 min intervals up to 180 min post-L-DOPA injection (see Table 1 for definitions) as initially described20,23. Scores must be given over 1-2 min epochs and classified as axial, limb, or orolingual (Figure 1).
- Carefully observe each AIM and attribute scores for severity and amplitude. Use Table 1 for instructions. Do not include normal behaviors, such as rearing, sniffing, grooming, and gnawing in the rating. There will be a total of six observation periods (30, 60, 90, 120, 150, and 180 min).
| SUBTYPES |
| Axial: torsion of head, neck, and trunk |
| Limb: involuntary movements of both distal and proximal forelimb |
| Orolingual: involuntary movements of the orofacial muscles |
| SEVERITY |
| 0: Absent |
| 1: Occasional (with interruptions, present in less than half of the observation time) |
| 2: Frequent (with interruptions but present in more than half of the observation time) |
| 3: Continuous but interrupted by external sensory stimuli |
| 4: Continuous and not interrupted by external sensory stimuli |
| AMPLITUDE |
| Axial |
| 1: Torsion of head and neck at approximately 30° angle |
| 2: Torsion of head and neck at approximately 30° < angle ≤ 60° |
| 3: Torsion of the head, neck, and upper trunk at approximately 60° < angle ≤ 90° |
| 4: Torsion of head, neck, and trunk at > 90° angle, often causing the rat to lose balance |
| Limb |
| 1: Small involuntary movements of the distal forelimb |
| 2: Movements of low amplitude causing translocation of both distal and proximal forelimb |
| 3: Involuntary movements of the whole limb, including shoulder muscles |
| 4: Strong limb and shoulder movements, often similar to ballism |
| Orolingual |
| 1: Small involuntary movements of the orofacial muscles |
| 2: Orofacial movements of high amplitude with tongue protrusion |
Table 1: AIMS definition and rating criteria.

Figure 1: Sequence of photos showing each AIM subtype. (A to A'") Axial AIM showing dystonic-like movements in different angles (A: 30° angle, A': 30° < angle ≤ 60°, A": 60° < angle ≤ 90°, and A'": > 90° angle). (B to B") Limb AIM (black arrows) showing involuntary movements of the distal (B and B') and whole forelimb (including shoulder, B"). (C) Orolingual AIM (red arrow) of high amplitude with tongue protrusion. Please click here to view a larger version of this figure.
4. Data analysis
- Compute axial, limb, or orolingual AIMs subtypes by multiplying the severity and amplitude scores for each observation period after L-DOPA administration (i.e., six observation periods) as previously described20. The theoretical maximum score that one animal could accumulate in one testing session is 240 (maximum score per observation period is 40, see Table 1 for details).
- Plot AIMs scores for all the six observation periods recorded on Wednesday, Thursday, and Friday over the 3 weeks of chronic L-DOPA administration (Figure 2). AIMs can be summed up to indicate each animal's maximum score on each scoring day (Figure 3) or each week (Figure 4) over the 3 weeks of chronic L-DOPA administration.
- Verify if the data has a close-to-normal distribution before selecting the statistical analysis approach.
NOTE: AIMs rating scales consist of ordinal data and can be analyzed with nonparametric statistics. Parametric tests are widely used when comparing AIMs scores across experimental groups following repeated testing sessions40.
Representative Results
Although the AIMs patterns observed in rats are simpler and limited compared to those observed in humans and nonhuman primates, this model reproduces both hyperkinetic and dystonic-like movements induced by chronic L-DOPA administration. Here we present data collected from a group (n = 10) of unilaterally 6-OHDA-lesioned rats chronically treated with L-DOPA (5 mg/kg combined with 12.5 mg/kg of benserazide) for 3 weeks (Monday to Friday). Note that the data presented in Figure 2, Figure 3, and Figure 4 are from the same animals.
Figure 2 shows the time course analysis for scores applied to axial, limb, and orolingual AIMs over 3 weeks of chronic L-DOPA administration. Each AIM score subtype at a specific observation period is reported as the multiplication of severity and amplitude scores in this analysis. This type of analysis can be convenient to explore the effect of drugs that interfere with the temporal profile of AIMs.Note that L-DOPA-induced peak-dose dyskinesias occur between 30-90 min with a gradual decrease after 120 min post-injection (Figure 2).
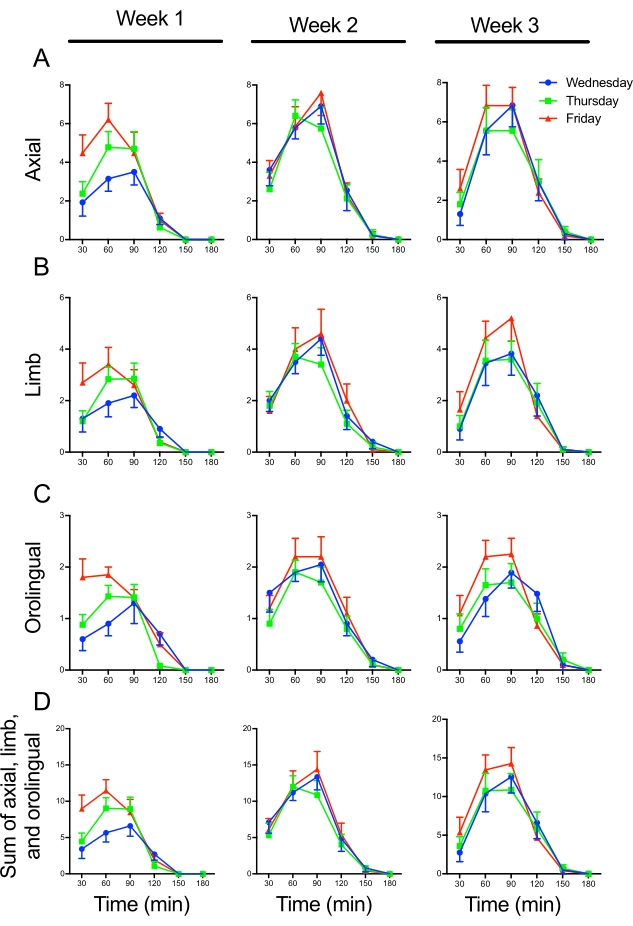
Figure 2: Time course analysis showing axial, limb, and orolingual AIMs over 3 weeks of chronic L-DOPA administration (5 mg/kg combined with 12.5 mg/kg of benserazide) to 6-OHDA-lesioned rats. (A) Axial, (B) limb, and (C) orolingual AIMs were scored on Wednesday, Thursday, and Friday over 3 weeks of chronic L-DOPA administration. Each AIM subtype is reported as the multiplication of severity times amplitude on each of the six observation periods. Each AIM subtype can be analyzed individually or summed up as indicated in (D). Note that the peak severity of the AIMs will gradually increase during the first week of chronic L-DOPA administration. Data are expressed as mean ± SEM (n = 10). Please click here to view a larger version of this figure.
Figure 3 shows the sum of AIMs scores presented for all the six observation periods depicted in Figure 2 (datasets are color-coded for easy comparisons). This analysis indicates each animal's maximum score on each scoring day (Wednesday, Thursday, and Friday) over the 3 weeks of chronic L-DOPA administration. This type of analysis can be advantageous to explore the effect of compounds with potential antidyskinetic profiles, especially because AIMs scores are stable over weeks 2 and 3 of chronic L-DOPA administration. Furthermore, weekly AIMs scores (Figure 4) can be generated from the sum of scores attributed to 6-OHDA-lesioned rats on Wednesday, Thursday, and Friday.
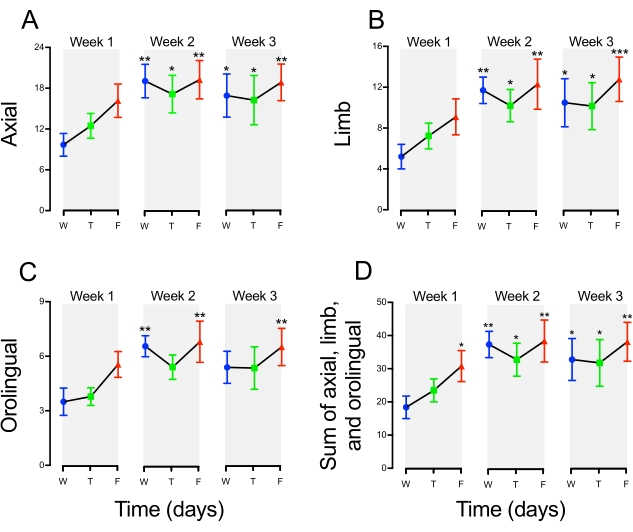
Figure 3: Sum of axial, limb, and orolingual AIMs on individual scoring days over 3 weeks of chronic L-DOPA administration (5 mg/kg combined with 12.5 mg/kg of benserazide) to 6-OHDA-lesioned rats. In this figure, AIMs scores presented in each of the six observation periods (see details in Figure 2 legend) were summed. Individual datasets were color-coded to compare with the data displayed in Figure 2. This analysis indicates each animal's maximum score on each scoring day (Wednesday – W, Thursday – T, and Friday – F) following 3 weeks of chronic L-DOPA administration. (A) Axial, (B) Limb, and (C) orolingual AIMs. (D) Sum of axial, limb, and orolingual AIMs. One-way repeated measures ANOVA and Holm-Sidak post-hoc test revealed that AIMs increase gradually over the chronic administration of L-DOPA. Note that AIMs scores are stable over weeks 2 and 3 of chronic L-DOPA administration (*p < 0.05, **p < 0.01, ***p < 0.001 vs. Wednesday (W) on Week 1). Data are expressed as mean ± SEM (n = 10). Please click here to view a larger version of this figure.
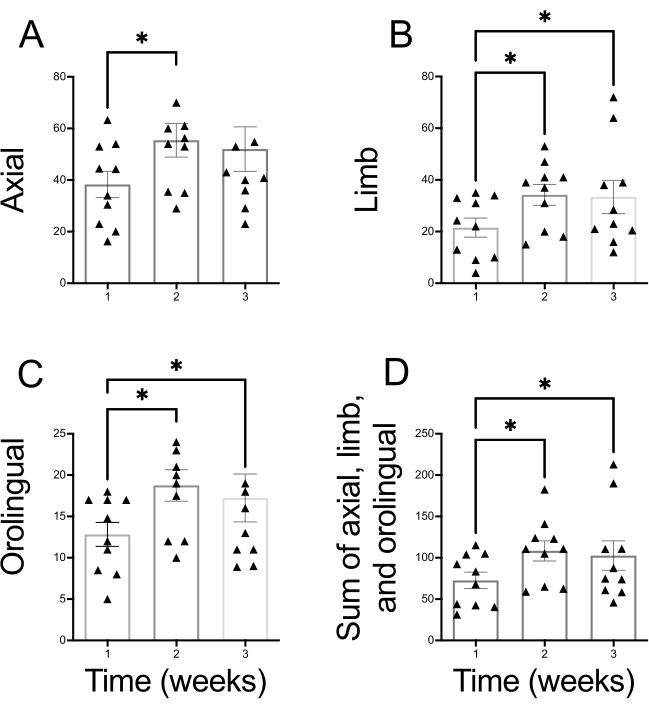
Figure 4: Sum of weekly axial, limb, and orolingual AIMs throughout chronic L-DOPA administration (5 mg/kg combined with 12.5 mg/kg of benserazide) to 6-OHDA-lesioned rats. In this figure, AIMs scored each week (Wednesday, Thursday, and Friday – see Figure 3) were summed. This analysis indicates each animal's maximum score on each week of chronic L-DOPA administration. (A) Axial, (B) Limb, and (C) orolingual AIMs. (D) Sum of axial, limb, and orolingual AIMs. One-way repeated measures ANOVA and Holm-Sidak post-hoc test revealed that AIMs increase over the chronic administration of L-DOPA (*p < 0.05 vs. Week 1). Data are expressed as mean ± SEM (n = 10). Please click here to view a larger version of this figure.
Discussion
This protocol demonstrates how to induce and analyze AIMs in the rat model of PD induced by unilateral microinjection of 6-OHDA in the MFB. Chronic daily administration of low doses of L-DOPA (5 mg/kg, combined with 12.5 mg/kg of benserazide) produced the development of AIMs over the 3 weeks of treatment. Temporal analysis revealed a significant increase of AIMs, and the peak-dose dyskinesia is observed between 30 and 90 min after L-DOPA administration. AIMs are repetitive and purposeless movements affecting axial, limb, and orolingual muscles. The protocol and the data presented here agree with the previous literature22,23,49,50,51.
The axial, limb, and orolingual AIMs rating scale is very sensitive because it rates the amount of time (i.e., severity) that the rodents display dyskinetic-like movements and indicates the amplitude of these movements20. AIMs scores can be expressed in different temporal resolutions (see Figure 2, Figure 3, Figure 4)17,18,19,52. Several studies have addressed the role of L-DOPA dosage on the development of AIMs. Chronic administration of lower doses of L-DOPA (5-20 mg/kg) favors a gradual development of AIMs over the treatment, while high doses of L-DOPA (25 mg/kg or more) result in the rapid and robust development of AIMs23,25,27,53. Interestingly, a proportion of severe 6-OHDA-lesioned animals may not develop AIMs (or have very low AIMs scores) following chronic administration of low doses of L-DOPA40,49,53. It is also essential to consider the L-DOPA's route of administration. Although AIMs share similar onset, time profile, and severity grade40, it has been demonstrated that dose-failure episodes frequently occurred in rats treated intraperitoneally27. Also, i.p. administration of L-DOPA produced inter-individual variations in plasma levels, an effect that was not observed after s.c. injections27.
In this study, AIMs were evident in animals with severe nigrostriatal damage (over 90% of dopaminergic depletion). Massive loss of striatal dopaminergic innervation is characterized by a reduction in the number of adjusting steps performed with the forelimb contralateral to the lesion48. Therefore, we recommend the stepping test to select animals with putative severe 6-OHDA lesions at the nigrostriatal pathway. The amphetamine-induced rotation test is commonly used to select putative 6-OHDA-lesioned rats. Amphetamine increases dopamine release in the intact striatum, and thus it increases the existing imbalance between the innervated and denervated striatum. However, an interesting study conducted in 312 6-OHDA-lesioned rats11 revealed no correlation between amphetamine-induced rotation and tyrosine hydroxylase positive cells in the ipsilateral substantia nigra pars compacta. This same study showed a poor correlation between amphetamine-induced rotation and the severity of AIMs.
There is a clear relationship between the phenomenology of AIMs and the pattern of nigrostriatal dopaminergic depletion20. The 6-OHDA rat model of PD we used in this protocol develops severe unilateral depletion of nigrostriatal dopamine terminals following 6-OHDA infusion in the MFB. Fully dopamine-depleted 6-OHDA-lesioned rats have a higher incidence of AIMs compared to partially dopamine-depleted animals. A detailed analysis of striatal expression of the neuropeptides dynorphin and enkephalin following 6-OHDA lesion and chronic L-DOPA administration is described elsewhere45 and is consistent with previous literature23,25. At the molecular level, dyskinetic rodents share molecular markers associated with the dyskinesia evident in nonhuman primate models of PD and PD patients, such as striatal upregulation of ΔFosB transcription factor26,32,33,54. Also, the rodent model of PD shows increased microvessel density in the striatum in response to L-DOPA treatment55 in a similar way observed in the putamen of PD patients56. Rodent models of LIDs respond to antidyskinetic pharmacological approaches used in the clinic, such as the NMDA receptor antagonist amantadine22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37, 38,39. Altogether, these pieces of evidence confirm the face validity and predictive validity of the 6-OHDA rat model to characterize the pathophysiology of LIDs and for the screening of compounds with antidyskinetic properties. The mice model of LIDs is also a valuable tool to study several aspects of LIDs in transgenic mice. Protocols for the mice model of LIDs can be found in papers from different research groups30,57,58.
It is important to note that the 6-OHDA-lesioned rodent model of LIDs has limitations. 6-OHDA-induced dopaminergic depletion is acute compared to the progressive nature of nigrostriatal depletion in the PD patient5. Also, it can be challenging to find an excellent therapeutic window for using an antidyskinetic drug in patients because increasing the dosage of a drug may interfere with the beneficial effect produced by L-DOPA or induce side effects that may not have been observed in the animal model59,60. Another limitation of the rat model of LIDs is the lack of physical similarity between AIMs in the rodents and the manifestation of the choreiform movements observed in patients and nonhuman primates10,61,62. Another potential limitation of the protocol presented here is that AIMs are scored every 30 min after L-DOPA administration, but this issue can be managed if the experimenter videotapes all animals for 180 min after L-DOPA administration. The experimenter can then adjust the scoring protocol for more frequent scoring (e.g., every 10 or 20 min). This approach is advantageous in experiments that require a more dynamic picture of AIMs time course (e.g., the combination of AIMs scoring and in vivo electrophysiological or neurochemical recordings).
In conclusion, the 6-OHDA rat model of LID reproduces the pathophysiology of disease and develops AIMs that can be modulated by drugs clinically used. Although it has limitations, the 6-OHDA rat model of LID still represents an invaluable preclinical tool to identify effective antidyskinetic interventions with translational potential.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work was supported by São Paulo Research Foundation (FAPESP, grant 2017/00003-0). We are grateful for the Coordination for the Improvement of Higher Education Personnel (CAPES). We thank Dr. Anthony R. West, Dr. Heinz Steiner, and Dr. Kuei Y. Tseng for support and mentoring.
Materials
| 6-hydroxydopamine hydrobromide | Sigma-Aldrich, USA | H6507 | Neurotoxin that produces degeneration of catecholaminergic terminals |
| Benzerazide hydrochloride | Sigma | B7283 | Peripheral dopa-decarboxylase inhibitor |
| Camera Bullet IR Turbo HD (HD-TVI) 2.8mm B | HIKVISION | DS-2CE16C0T-IRP | Camera used to record all behavior |
| Imipramine hidrochloride | Alfa Aesar | J63723 | Norepinephrine transporter inhibitor (NET) used to protect noradrenergic neurons from 6-OHDA |
| Ketamine hydrochloride | Ceva Animal Health | Anesthesia for surgical intervention | |
| L-3,4-dihydroxyphenylalanine (L-DOPA) methyl ester (hydrochloride) | Cayman Chemical Company | 16149 | Dopamine precursor |
| Mirrors | Used to observe the behavior of animals during experiments in all directions | ||
| Needles 0.30 x 13 mm | PrecisionGlide | Needles used to inject drugs | |
| Sodium chloride (NaCl) | Samtec | Salt | |
| Syringes 1 ml Sterile | BD Plastipak | Syringes used to inject drugs | |
| Transparent cylinders | Used to record animal behavior during experiments | ||
| Xylazine hydrochloride | Ceva Animal Health | Sedative, analgesic and muscle relaxant for surgical intervention |
Riferimenti
- Jenner, P. Molecular mechanisms of L-DOPA-induced dyskinesia. Nature Reviews. Neuroscience. 9 (9), 665-677 (2008).
- Nutt, J. G. Levodopa-induced dyskinesia: review, observations, and speculations. Neurology. 40 (2), 340-345 (1990).
- Luquin, M. R., Scipioni, O., Vaamonde, J., Gershanik, O., Obeso, J. A. Levodopa-induced dyskinesias in Parkinson’s disease: clinical and pharmacological classification. Movement Disorders: Official Journal of the Movement Disorder Society. 7 (2), 117-124 (1992).
- Fabbrini, G., Brotchie, J. M., Grandas, F., Nomoto, M., Goetz, C. G. Levodopa-induced dyskinesias. Movement Disorders: Official Journal of the Movement Disorder Society. 22 (10), 1379 (2007).
- Cenci, M. A., Crossman, A. R. Animal models of l-dopa-induced dyskinesia in Parkinson’s disease. Movement Disorders: Official Journal of the Movement Disorder Society. 33 (6), 889-899 (2018).
- Bastide, M. F., et al. Pathophysiology of L-dopa-induced motor and non-motor complications in Parkinson’s disease. Progress in Neurobiology. 132, 96-168 (2015).
- Manson, A., Stirpe, P., Schrag, A. Levodopa-induced-dyskinesias clinical features, incidence, risk factors, management and impact on quality of life. Journal of Parkinson’s Disease. 2 (3), 189-198 (2012).
- Grandas, F., Galiano, M. L., Tabernero, C. Risk factors for levodopa-induced dyskinesias in Parkinson’s disease. Journal of Neurology. 246 (12), 1127-1133 (1999).
- Schrag, A., Quinn, N. Dyskinesias and motor fluctuations in Parkinson’s disease. A community-based study. Brain: A Journal of Neurology. 123, 2297-2305 (2000).
- Bezard, E., Brotchie, J. M., Gross, C. E. Pathophysiology of levodopa-induced dyskinesia: potential for new therapies. Nature Reviews. Neuroscience. 2 (8), 577-588 (2001).
- Tronci, E., Shin, E., Björklund, A., Carta, M. Amphetamine-induced rotation and L-DOPA-induced dyskinesia in the rat 6-OHDA model: a correlation study. Neuroscience Research. 73 (2), 168-172 (2012).
- Duty, S., Jenner, P. Animal models of Parkinson’s disease: a source of novel treatments and clues to the cause of the disease. British Journal of Pharmacology. 164 (4), 1357-1391 (2011).
- Ungerstedt, U. 6-Hydroxy-dopamine induced degeneration of central monoamine neurons. European Journal of Pharmacology. 5 (1), 107-110 (1968).
- Ungerstedt, U. Postsynaptic supersensitivity after 6-hydroxy-dopamine induced degeneration of the nigro-striatal dopamine system. Acta Physiologica Scandinavica. Supplementum. 367, 69-93 (1971).
- Kirik, D., Rosenblad, C., Bjorklund, A. Characterization of behavioral and neurodegenerative changes following partial lesions of the nigrostriatal dopamine system induced by intrastriatal 6-hydroxydopamine in the rat. Experimental Neurology. 152, 259-277 (1998).
- Zahm, D. S. Compartments in rat dorsal and ventral striatum revealed following injection of 6-hydroxydopamine into the ventral mesencephalon. Brain Research. 552 (1), 164-169 (1991).
- Padovan-Neto, F. E., Echeverry, M. B., Tumas, V., Del-Bel, E. A. Nitric oxide synthase inhibition attenuates L-DOPA-induced dyskinesias in a rodent model of Parkinson’s disease. Neuroscienze. 159 (3), 927-935 (2009).
- Padovan-Neto, F. E., et al. Antidyskinetic effect of the neuronal nitric oxide synthase inhibitor is linked to decrease of FosB/deltaFosB expression. Neuroscience Letters. 541, 126-131 (2013).
- Padovan-Neto, F. E., Cavalcanti-Kiwiatkoviski, R., Carolino, R. O., Anselmo-Franci, J., Del Bel, E. Effects of prolonged neuronal nitric oxide synthase inhibition on the development and expression of L-DOPA-induced dyskinesia in 6-OHDA-lesioned rats. Neuropharmacology. 89, 87-99 (2015).
- Winkler, C., Kirik, D., Björklund, A., Cenci, M. A. L-DOPA-induced dyskinesia in the intrastriatal 6-hydroxydopamine model of parkinson’s disease: relation to motor and cellular parameters of nigrostriatal function. Neurobiology of Disease. 10 (2), 165-186 (2002).
- Francardo, V., et al. Impact of the lesion procedure on the profiles of motor impairment and molecular responsiveness to l-DOPA in the 6-hydroxydopamine mouse model of Parkinson’s disease. Neurobiology of Disease. 42, 327-340 (2011).
- Lundblad, M., et al. Pharmacological validation of behavioural measures of akinesia and dyskinesia in a rat model of Parkinson’s disease. The European Journal of Neuroscience. 15 (1), 120-132 (2002).
- Cenci, M. A., Lee, C. S., Björklund, A. L-DOPA-induced dyskinesia in the rat is associated with striatal overexpression of prodynorphin- and glutamic acid decarboxylase mRNA. The European Journal of Neuroscience. 10 (8), 2694-2706 (1998).
- Henry, B., Crossman, A. R., Brotchie, J. M. Characterization of enhanced behavioral responses to L-DOPA following repeated administration in the 6-hydroxydopamine-lesioned rat model of Parkinson’s disease. Experimental Neurology. 151 (2), 334-342 (1998).
- Lee, C. S., Cenci, M. A., Schulzer, M., Björklund, A. Embryonic ventral mesencephalic grafts improve levodopa-induced dyskinesia in a rat model of Parkinson’s disease. Brain: A Journal of Neurology. 123, 1365-1379 (2000).
- Andersson, M., Hilbertson, A., Cenci, M. A. Striatal fosB expression is causally linked with l-DOPA-induced abnormal involuntary movements and the associated upregulation of striatal prodynorphin mRNA in a rat model of Parkinson’s disease. Neurobiology of Disease. 6 (6), 461-474 (1999).
- Lindgren, H. S., Rylander, D., Ohlin, K. E., Lundblad, M., Cenci, M. A. The “motor complication syndrome” in rats with 6-OHDA lesions treated chronically with L-DOPA: relation to dose and route of administration. Behavioural Brain Research. 177 (1), 150-159 (2007).
- Lane, E. L., Dunnett, S. . Animal Models of Movement Disorders: volume I, Neuromethods. 61, (2011).
- Carlsson, T., Carta, M., Winkler, C., Björklund, A., Kirik, D. Serotonin neuron transplants exacerbate l-DOPA-induced dyskinesias in a rat model of Parkinson’s disease. Journal of Neuroscience. 27 (30), 8011-8022 (2007).
- Lundblad, M., Picconi, B., Lindgren, H., Cenci, M. A. A model of L-DOPA-induced dyskinesia in 6-hydroxydopamine lesioned mice: relation to motor and cellular parameters of nigrostriatal function. Neurobiology of Disease. 16 (1), 110-123 (2004).
- Fasano, S., et al. Inhibition of Ras-guanine nucleotide-releasing factor 1 (Ras-GRF1) signaling in the striatum reverts motor symptoms associated with L-dopa-induced dyskinesia. Proceedings of the National Academy of Sciences of the United States of America. 107 (50), 21824-21829 (2010).
- Berton, O., et al. Striatal overexpression of DeltaJunD resets L-DOPA-induced dyskinesia in a primate model of Parkinson disease. Biological Psychiatry. 66 (6), 554-561 (2009).
- Lindgren, H. S., et al. Putaminal upregulation of FosB/ΔFosB-like immunoreactivity in Parkinson’s disease patients with dyskinesia. Journal of Parkinson’s Disease. 1 (4), 347-357 (2011).
- Rylander, D., et al. Maladaptive plasticity of serotonin axon terminals in levodopa-induced dyskinesia. Annals of Neurology. 68 (5), 619-628 (2010).
- Smith, R., et al. The role of pallidal serotonergic function in Parkinson’s disease dyskinesias: a positron emission tomography study. Neurobiology of Aging. 36 (4), 1736-1742 (2015).
- Lundblad, M., et al. Pharmacological validation of a mouse model of l-DOPA-induced dyskinesia. Experimental Neurology. 194 (1), 66-75 (2005).
- Dekundy, A., Lundblad, M., Danysz, W., Cenci, M. A. Modulation of L-DOPA-induced abnormal involuntary movements by clinically tested compounds: further validation of the rat dyskinesia model. Behavioural Brain Research. 179 (1), 76-89 (2007).
- Blanchet, P. J., Konitsiotis, S., Chase, T. N. Amantadine reduces levodopa-induced dyskinesias in parkinsonian monkeys. Movement Disorders: Official Journal of the Movement Disorder Society. 13 (5), 798-802 (1998).
- Verhagen Metman, I., et al. Amantadine as treatment for dyskinesias and motor fluctuations in Parkinson’s disease. Neurology. 50 (5), 1323-1326 (1998).
- Cenci, M. A., Lundblad, M. Ratings of L-DOPA-induced dyskinesia in the unilateral 6-OHDA lesion model of Parkinson’s disease in rats and mice. Current Protocols in Neuroscience. , 25 (2007).
- Dupre, K. B., et al. Effects of coincident 5- HT1A receptor stimulation and NMDA receptor antagonism on L-DOPA-induced dyskinesia and rotational behaviors in the hemi-parkinsonian rat. Psychopharmacology (Berl). 199 (1), 99-108 (2008).
- Guimarães, P. R., Bariotto-dos-Santos, K., Ribeiro, D. L., Padovan-Neto, F. E. The 6-hydroxydopamine (6-OHDA) rat model of Parkinson’s disease. Journal of Visualized Experiments: JoVE. , (2021).
- Paxinos, G., Watson, C. . The Rat Brain in Stereotaxic Coordinates. , (1998).
- Tseng, K. Y., et al. Inhibition of striatal soluble guanylyl cyclase-cGMP signaling reverses basal ganglia dysfunction and akinesia in experimental parkinsonism. PloS One. 6 (11), 27187 (2011).
- Padovan-Neto, F. E., et al. Selective regulation of 5-HT1B serotonin receptor expression in the striatum by dopamine depletion and repeated L-DOPA treatment: Relationship to L-DOPA-induced dyskinesias. Molecular Neurobiology. 57 (2), 736-751 (2020).
- Olsson, M., Nikkhah, G., Bentlage, C., Bjorklund, A. Forelimb akinesia in the rat Parkinson model: Differential effects of dopamine agonists and nigral transplants as assessed by a new stepping test. Journal of Neuroscience. 15, 3863-3875 (1995).
- Chang, J. W., Wachtel, S. R., Young, D., Kang, U. J. Biochemical and anatomical characterization of forepaw adjusting steps in rat models of Parkinson’s disease lesions. Neuroscienze. 88, 617-628 (1995).
- Jayasinghe, V. R., Flores-Barrera, E., West, A. R., Tseng, K. Y. Frequency-dependent corticostriatal disinhibition resulting from chronic dopamine depletion: Role of local striatal cGMP and GABAAR signaling. Cerebral Cortex. 27, 625-634 (2017).
- Picconi, B., et al. Loss of bidirectional striatal synaptic plasticity in L-DOPA-induced dyskinesia. Nature Neuroscience. 6 (5), 501-506 (2003).
- Carta, M., et al. Role of striatal L-DOPA in the production of dyskinesia in 6-hydroxydopamine lesioned rats. Journal of Neurochemistry. 96 (6), 1718-1727 (2006).
- Westin, J. E., Andersson, M., Lundblad, M., Cenci, M. A. Persistent changes in striatal gene expression induced by long-term L-DOPA treatment in a rat model of Parkinson’s disease. The European Journal of Neuroscience. 14 (7), 1171-1176 (2001).
- Bariotto-Dos-Santos, K., et al. Repurposing an established drug: an emerging role for methylene blue in L-DOPA-induced dyskinesia. The European Journal of Neuroscience. 49 (6), 869-882 (2018).
- Lindgren, H. S., Lane, E. L., Lane, E., Dunnett, S. Rodent Models of l-DOPA-Induced Dyskinesia. Animal Models of Movement Disorders. Neuromethods. 61, (2011).
- Doucet, J., et al. Drug-drug interactions related to hospital admissions in older adults: A prospective study of 1000 patients. Journal of the American Geriatrics Society. 44, 944-948 (1996).
- Ohlin, K. E., et al. Vascular endothelial growth factor is upregulated by L-dopa in the parkinsonian brain: implications for the development of dyskinesia. Brain: A Journal of Neurology. 134, 2339-2357 (2011).
- Jourdain, V. A., et al. Increased putamen hypercapnic vasoreactivity in levodopa-induced dyskinesia. JCI insight. 2 (20), 96411 (2017).
- Thiele, S. L., et al. Generation of a model of l-DOPA-induced dyskinesia in two different mouse strains. Journal of Neuroscience Methods. 197 (2), 193-208 (2011).
- Dos-Santos-Pereira, M., et al. Contributive role of TNF-α to L-DOPA-induced dyskinesia in a unilateral 6-OHDA lesion model of Parkinson’s disease. Frontiers in Pharmacology. 11, 617085 (2021).
- Cenci, M. A., Ohlin, K. E., Odin, P. Current options and future possibilities for the treatment of dyskinesia and motor fluctuations in Parkinson’s disease. CNS & Neurological Disorders Drug Targets. 10 (6), 670-684 (2011).
- Cenci, M. A. Presynaptic mechanisms of l-DOPA-induced dyskinesia: The findings, the debate, and the therapeutic implications. Frontiers in Neurology. 5, 242 (2014).
- Langston, J. W., Quik, M., Petzinger, G., Jakowec, M., Di Monte, D. A. Investigating levodopa-induced dyskinesias in the parkinsonian primate. Annals of Neurology. 47 (4), 79-89 (2000).
- Chase, T. N. Levodopa therapy: consequences of the nonphysiologic replacement of dopamine. Neurology. 50 (5), 17-25 (1998).

