Subconjunctival Administration of Adeno-associated Virus Vectors in Small Animal Models
Summary
In this manuscript, subconjunctival injection is demonstrated as a valid vector delivery method for ocular tissues in mice using an injection system consisting of an infusion/withdrawal syringe pump and a gastight removable syringe coupled with microinjection needles. This injection system is also adaptable for other intraocular administration routes.
Abstract
Ocular diseases include a wide range of inherited genetic and acquired disorders that are appealing targets for local drug delivery due to their relative ease of accessibility via multiple administration routes. Subconjunctival (SCJ) injections offer advantages over other intraocular administration routes as they are simple, safe, and usually performed in an outpatient setting. SCJ injections in small animals usually require the assistance of an operating microscope due to the size of the eye. Previous work has demonstrated that SCJ injection of specific adeno-associated virus (AAV) serotypes is a valid gene delivery strategy for targeted transduction of the ocular surface, eye muscle, cornea, and optic nerve, providing a potential approach for the treatment of many ocular diseases.
Herein, a detailed protocol is presented for SCJ injections in a mouse model using an injection system consisting of a programmable infusion/withdrawal syringe pump (which allows for consistent and precise injection speed and pressure) and a gastight removable syringe coupled with microinjection needles. The injection system is also adaptable for other intraocular administration routes such as intrastromal, intracameral, intravitreal, and subretinal injections in small animals. Although the delivery of adeno-associated viral vectors for ocular gene therapy studies is described, the protocol herein can also be adapted for a variety of ophthalmic solutions in small animal models. The key practical steps in the administration route, setup for the injection platform, preparation of the injection, and tips from direct experience will be discussed in detail. In addition, common validation techniques for AAV delivery confirmation to the desired tissues will also be briefly discussed.
Introduction
Ocular diseases encompass a broad range of both genetic and acquired disorders. In 2015, an estimated 36 million people were legally blind worldwide, and over 1 billion people suffer from at least some level of visual impairment, highlighting the need to scale up alleviation efforts at all levels1. The main methods for delivering ocular medications include both topical and local administration, such as eye drops or subconjunctival (SCJ), intracameral, intravitreal, and subretinal injections. Although noninvasive topical therapy is the most common delivery method for ophthalmic drugs and is extensively used for many anterior segment disorders, the presence of corneal anatomical barriers presents a challenge for the bioavailability, biodistribution, and efficacy of topically administrated substances, suggesting that it may not be the best candidate treatment route for many diseases of the inner eye. Local injection into the specific ocular compartment affected by the disease is likely to be a more effective and targeted drug delivery approach2. However, adverse effects resulting from repeated injections can complicate administration strategies. Ideally, a therapy should maintain long-term therapeutic efficacy following a single administration. Thus, gene therapy is a promising option for minimizing the number of required injections and providing sustained transgene expression for the treatment of ocular disease3,4.
Numerous viral and nonviral vectors are available for gene therapy; however, AAV vectors are of high interest due to their excellent safety profile. AAV is a small, single-stranded, non-enveloped DNA virus that was initially discovered as a contaminant of an adenovirus preparation in 1965 by Atchison et al.5,6 AAV was subsequently engineered as an efficient viral vector for gene delivery in the 1980s and has become the gene therapy vector of choice for many diseases, including ocular disorders, over the last few decades. The most notable of these is the first commercially available gene therapy drug, voretigene neparvovec, which was approved by the United States Food and Drug Administration to treat Leber's Congenital Amaurosis, a rare posterior eye disease. Although voretigene neparvovec has successfully overcome barriers to clinical development, challenges remain for the commercialization of additional ocular gene therapies. For example, voretigene neparvovec is administered to patients who retain viable retinal cells via subretinal injection. Thus, patients with more advanced forms of the disease who lack viable retinal cells are not eligible for treatment, as it would provide no clinical benefit. In addition, known complications associated with the subretinal injection procedure were observed, including eye inflammation, cataracts, retinal tearing, maculopathy, and pain7,8. Other concerns related to this procedure include the possibility of hemorrhage, retinal detachment, endophthalmitis, and revocation of the ocular immune privileged status through eye tissue destruction9,10,11,12. Thus, efforts to explore less invasive gene delivery routes such as SCJ injection have become increasingly important13,14,15,16,17.
The conjunctiva is a thin membrane containing 3-5 layers of cells and connecting the anterior eye to the interior eyelid. SCJ injections are used clinically for ophthalmic drug delivery to both the anterior and/or posterior segments of the eye for the treatment of ocular diseases such as age-related macular degeneration, glaucoma, retinitis, and posterior uveitis18,19. They are relatively simple to perform, employed routinely for ophthalmic drug delivery in an outpatient setting20, somewhat painless, do not compromise ocular immune privilege, and allow administered drugs to spread through a large periorbital region that encompasses the optic nerve. Hence, SCJ injections are an attractive route of administration for AAV gene therapy applications. Natural AAV serotypes administered via SCJ injection in mice have previously been characterized for safety, transduction efficiency, serum immunogenicity, biodistribution, and tissue specificity13,16,21. These data demonstrated that gene delivery to individual ocular tissues via SCJ administration is a formal possibility.
This paper describes a simple and adaptable protocol for SCJ injection to deliver AAV vectors in a mouse model. To ensure the reproducibility of this approach, an injection system consisting of a stereomicroscope, a programmable infusion/withdrawal syringe pump (which allows consistent and precise injection speed and pressure), and a gastight removable syringe coupled with microinjection needles is described. This system is adaptable for other intraocular administration routes such as intrastromal, intracameral, intravitreal, and subretinal injections in small animals. In addition, a fluorescein dye is often utilized to allow for visualization of the AAV injection site. The key practical steps in the administration route, setup for the injection platform, preparation of the injection, and tips from direct experience will be discussed in detail. Finally, common validation techniques for confirmation of AAV delivery to the desired tissues will be briefly discussed.
Protocol
All animal procedures were performed in accordance with the regulations of the Institutional Animal Care and Use Committee at the University of North Carolina at Chapel Hill. The use of AAV vectors is a Biosafety Level 1 biohazard risk. Wear proper personal protective equipment, including a lab coat, gloves, and goggles when handling AAV. For the experiment described herein, a recombinant AAV vector packaged with the serotype 8 capsid and encoding a generic ubiquitous cytomegalovirus (CMV) promoter controlling the expression of green fluorescence protein (GFP) was utilized.
1. AAV vector handling and storage
- Store the virus in a -80 °C freezer in 100 µL aliquots in siliconized or low-retention microcentrifuge tubes.
- Thaw all vector stock solutions on ice before use.
NOTE: Dyes such as sodium fluorescein solution (at a final concentration of 0.1-2%) are often mixed with the AAV vectors to visualize the injected solution. Additionally, visualization of injected solutions helps detect air bubbles and monitor AAV distribution and/or leakage after injection.
2. Subconjunctival (SCJ) injection
- Assemble the injection system.
- To assemble the injection system, place a stereomicroscope and a syringe pump in a biosafety cabinet.
NOTE: An infusion pump is needed to perform injections with high precision. Herein, a Standard Infuse/Withdraw Programmable Syringe Pump is utilized (see the Table of Materials), which includes a tight grip and a secure syringe clamp for syringes ranging in volume from 0.5 µL to 60 mL. This pump also offers enhanced flow performance with high accuracy and smooth flow rates from 1.28 pl/min to 88.28 ml/min. - Cut the polyethylene tubing to a length of approximately 50 cm (see the Table of Materials).
- Insert the hub end of a 36 G needle into one of the ends of the tubing.
NOTE: Slide the needle hub end into the tube for ~3 mm to ensure that no leakage occurs. The 36 G needle is used for the subsequent SCJ injection. Needles ranging between 32 G and 36 G are the most commonly used sizes for SCJ injections. The use of a hemostat to assist this step is highly recommended to avoid the potential risk of sharps injury. - Fill a disposable 3 mL syringe with sterile water; insert this disposable syringe into the side of the tube opposite the needle and flush the water throughout the tubing/needle. Repeat this step with 70% alcohol.
- Repeat step 2.1.4 three more times, alternating flushes with sterile water and 70% alcohol, to disinfect the tubing and ensure no leaks, clogs, or damage are observed throughout the tubing.
- Use the disposable 3 mL syringe to fill the tubing with sterile water and leave the tubing attached to the disposable syringe.
- Place a piece of parafilm on the bench surface and add a pool of sterile water to it (~1 mL). Submerge the portion of the tubing connected to the needle into the pool of sterile water. Pull the disposable syringe out from the tube opening at the opposite end to prevent any air from entering the tubing/needle system upon removal of the syringe. Leave the portion of the tubing connected to the needle submerged in the pool of water.
NOTE: Perform procedures 2.1.4 to 2.1.7 in a laminar hood. - Fill a 10 µL Hamilton syringe/needle with sterile water and avoid air in the syringe. Connect the Hamilton syringe/needle to the remaining open end of the tubing by submerging the tubing and needle tip of the Hamilton syringe into the pool of sterile water on the parafilm.
- Press the fast reverse button on the pump screen to move the pusher block to the approximate length of the syringe. Unscrew the bracket clamping knobs to loosen the retaining brackets on the pusher and the syringe holder blocks. Load the Hamilton syringe onto the syringe holder block and secure the syringe following the manufacturer's instructions.
NOTE: To secure the syringe, the syringe barrel clamp should be tight against the syringe barrel; however, do not overtighten, especially when using glass syringes. The syringe plunger should be secured by the pusher block retaining bracket. - Adjust the parameters in the pump settings screen.
- Press the Force button, and set the force level at 30%. Accept the changes to go back to the settings screen.
- Press the quick start button and select Method | Infuse/withdraw.
- For the Syringe, select Hamilton 1700, glass, 10 µL. Select the infusion and withdraw rate and the injection volume.
NOTE: The Force level is set depending on the syringe type/material/capacity/manufacturers; see factory manufacturer instructions for suggested force for each syringe. The injection speed used in this experiment was 200 nL/s. SCJ injections are relatively safe, and there is less of a concern for the induction of elevated intraocular pressure (IOP) resulting from the injection. A slower injection speed is often desirable for certain applications to avoid reflux into the needle and maintain consistency in injections among animals.
- Eject the water from the Hamilton syringe but leave the tubing and injection needle full of water. Slightly pull back on the Hamilton syringe by pressing the Reverse button to introduce a small air bubble in the tubing/needle.
NOTE: The air bubble will serve as a barrier between the water in the tube and the therapeutic drug (in this case, AAV), ensuring the accuracy of the administered dose. - Withdraw the virus by placing the injection needle into an aliquot of the virus stock. Ensure that a visible air bubble remains between the virus and the water in the tubing.
NOTE: AAV vectors can bind to the plastic tubing and metal needle, leading to a loss of virus and/or inaccurate dosing regimens. Thus, to ensure rigor, reproducibility, and an accurate dose of AAV, precoating the surfaces that subsequently come into contact with the AAV is recommended. To coat the tubing/needle system with the virus, draw the viral vector solution into the tubing/needle and incubate it at room temperature for 10 min to allow for saturation of virus binding to the wall of the needle and/or tubing. Discard the virus.
- To assemble the injection system, place a stereomicroscope and a syringe pump in a biosafety cabinet.
- Virus injection
- Anesthetize the mouse with inhaled anesthesia (isoflurane) or intraperitoneal injection of ketamine/xylazine/acepromazine. Confirm the surgical plane of anesthesia by a lack of response to firm toe pinches.
NOTE: Use female and/or male C57BL/6J or BALB/c mice that are at least 6 weeks old. Ketamine/xylazine/acepromazine doses are as follows: ketamine at 70 mg/kg, xylazine at 7 mg/kg, and acepromazine at 1.5 mg/kg. - Apply topical anesthesia to the eye that will receive the injection.
NOTE: Use 0.1% proparacaine hydrochloride and/or tetracaine hydrochloride ophthalmic solution (0.5%) for topical anesthesia. - Apply topical ointment to the other eye that will not receive an injection to prevent dryness and injury.
- Position the mouse on the microscopic stage and expose the mouse eye under the stereomicroscope.
- Place two fingers on the eyelid and slightly pull it away from the mouse's eye to expose the conjunctiva, which is the inner membrane connecting the eyelid to the sclera.
- Grab the conjunctiva with forceps.
- Release the eyelid and hold the needle with the bevel facing upwards using the dominant hand.
- Insert the needle into the conjunctiva. Insert the needle until the bevel is completely covered by the conjunctival membrane. Lay the needle against the globe.
NOTE: As the conjunctiva is a clear membrane, the needle tip/bevel is easily visible. - Start the injection by pressing the Start button using the footswitch.
NOTE: The movement of the air and the Hamilton plunger are synchronized; any delay indicates excess air in the injecting system or possibly a loose connection between the tubing, needle, and/or syringe components. - After the injection is finished, hold the needle in place for 10 s before withdrawing the needle from the conjunctiva to decrease the chances of backflow.
NOTE: It is common for a bleb to appear at the site of the SCJ injection. Such blebs normally resolve completely within a few hours post injection. - Put a drop of the topical lubricant gel on the mouse's eyes to prevent ocular dryness/injury, and then place the mouse on a heating pad to recover.
- Perform ocular examinations such as tear production, IOP, and slit-lamp examination in conjunction with corneal fluorescein staining to assess ocular abnormities following the injection.
NOTE: Tear production is measured by a Phenol Red Thread test, and a tonometer is often used to examine the IOP of the mouse eye. It is reported that some intraocular injections, such as intravitreal injections, might result in a significant increase in IOP; however, IOP changes after SCJ injection are not obvious13,22,23,24.
- Anesthetize the mouse with inhaled anesthesia (isoflurane) or intraperitoneal injection of ketamine/xylazine/acepromazine. Confirm the surgical plane of anesthesia by a lack of response to firm toe pinches.
- AAV biodistribution and transduction efficiency examination following subconjunctival injection
- To investigate the viral genome biodistribution and/or transduction profile of AAV vectors delivered via SCJ, euthanize the mice by AVMA approved method.
NOTE: In this experiment, the mice were sacrificed 8 weeks post injection. - For biodistribution and transgene expression in targeted ocular compartments, dissect the relevant tissue of interest such as the eyelids, cornea, conjunctiva, eye muscle, retina, and optic nerve. Flash-freeze all tissues and store at -80 °C. To examine whole-body AAV biodistribution, collect organs such as the submandibular lymph nodes and liver, and flash-freeze and store them at -80 °C.
- Using a DNA/RNA extraction kit, collect gDNA and RNA from the same sample to examine transgene expression and AAV biodistribution, respectively. If only vector biodistribution is desired, use a DNA extraction kit to extract gDNA.
- Perform standard qPCR and RT-qPCR to determine the AAV vector biodistribution and cDNA abundance using vector transgene-specific primers/probes13,25.
- For histology analysis, fix the eyes, embed them in paraffin, and section them at a thickness of 5 µm. Perform standard immunofluorescence staining to reveal transgene expression26.
- To investigate the viral genome biodistribution and/or transduction profile of AAV vectors delivered via SCJ, euthanize the mice by AVMA approved method.
Representative Results
Solution injected into the subconjunctival space presents as a bleb depending on the injection volume.
In this experiment, 7 µL of AAV (7 × 109 viral genomes (vg)/eye) mixed with fluorescein at a final concentration of 0.1% was injected with a 36 G needle under a stereomicroscope, and the injection speed/pressure was held constant using a programmable syringe pump at 1 µL/s. A bleb can appear upon injection (arrow). A microscopic view of AAV vector administration to the murine SCJ compartment is shown in Figure 1.
Substances injected into the subconjunctival space diffuse around the globe of the eye and are distributed throughout the periocular tissues.
To define the distribution of AAV administered via SCJ injection, 7 µL of diluted India ink was injected into the subconjunctival space of a 10-month-old mouse after anesthetization. No bleeding, leakage, or backflow was detected during or following the SCJ injection. Thirty minutes post injection, the ocular and surrounding tissues were harvested and subsequently stained with Hematoxylin and Eosin (H&E) to visualize the distribution of India ink. The representative sagittal sections depicted in Figure 2 demonstrate that the dispersion of India ink occurred mainly adjacent to the extraocular muscles, in the outer surface of the sclera, and the periocular loose connective tissues (Figure 2).
Self-complementary AAV8 successfully transduces the cornea and periocular muscles following SCJ.
To determine the transduction profile of self-complementary AAV8 at eight weeks post injection, GFP abundance in whole-globe cross-sections was examined via immunofluorescence staining using an anti-GFP antibody at a dilution of 1:500. Images were taken under a fluorescence microscope (Figure 3). These results revealed that AAV vectors administered via SCJ injection efficiently transduce the periocular muscles posterior to the eye and the cornea.
Abundant vector genome and transgene expression in distinct eye compartments following SCJ injection
To quantitatively analyze the vector biodistribution and transgene expression, vector genome copy numbers in distinct eye compartments and organs such as the liver and heart were examined by qPCR (Figure 4A), while the transgene expression was tested by qRT-PCR (Figure 4B). These results suggest that SCJ injection of AAV8 results in transgene expression in the eyelid, conjunctiva, cornea, and optic nerve.
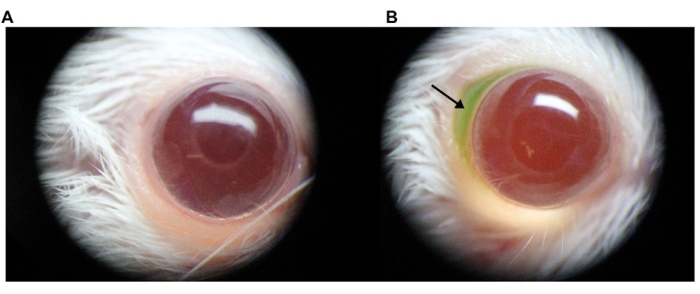
Figure 1: Microscopic view of AAV vector administration into the murine SCJ space. To allow for visualization of the formation of a bleb during the procedure, 1% fluorescein was directly added to the AAV vector preparation. Images were taken using a digital camera attached to a stereomicroscope. (A) Representative image of a noninjected eye; (B) representative image of an injected eye. The arrow indicates the injected AAV solution containing fluorescein in the SCJ space. Abbreviations: AAV = Adeno-associated virus; SCJ = subconjunctival. Please click here to view a larger version of this figure.
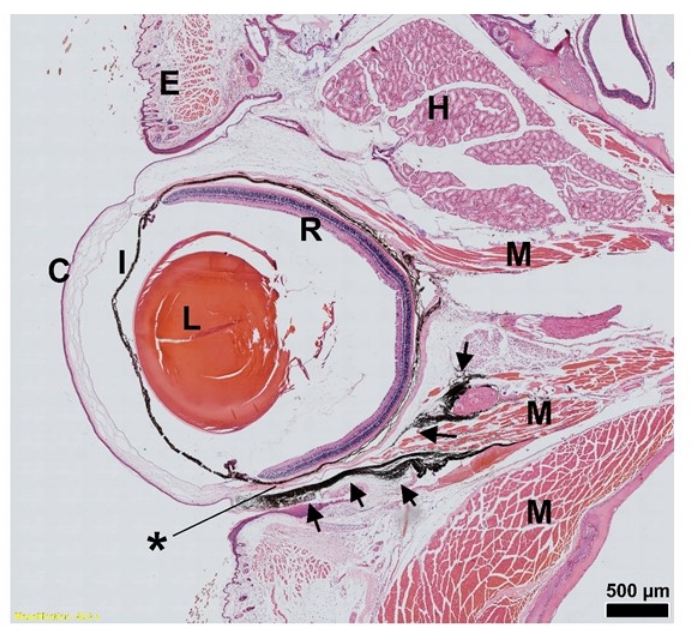
Figure 2: H&E staining of India ink distribution (arrow) after SCJ injection in the mouse eye. Sagittal sections of an eye injected with India ink are presented; 7 µL of India ink was injected at the indicated site. *, Injection site. Scale bar = 500 µm. Abbreviations: SCJ = subconjunctival; H&E = hematoxylin and eosin; C = cornea; I = iris; L = lens; R = Retina; E = eyelid; H = Harderian gland; M = muscle. Please click here to view a larger version of this figure.
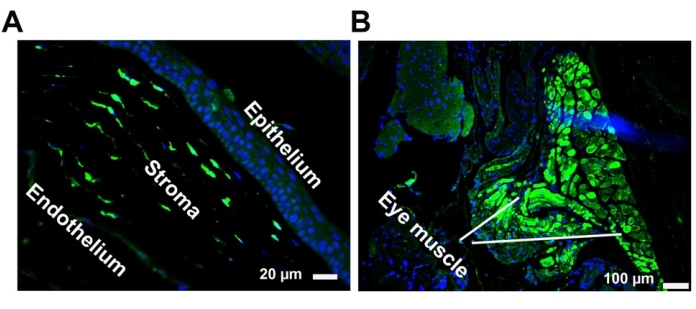
Figure 3: Representative GFP histology images of self-complementary AAV8 after SCJ injection. Transduction of the cornea (A) and eye muscles (B) following SCJ injection. GFP expression (green) was visualized via immunostaining in paraffin-embedded tissue sections with an anti-GFP antibody. Nuclei were stained with DAPI (blue). Scale bar = 100 µm (eye muscle), 20 µm (cornea). Abbreviations: GFP = green fluorescent protein; AAV = adeno-associated virus; SCJ = subconjunctival; DAPI = 4',6-diamidino-2-phenylindole. Please click here to view a larger version of this figure.
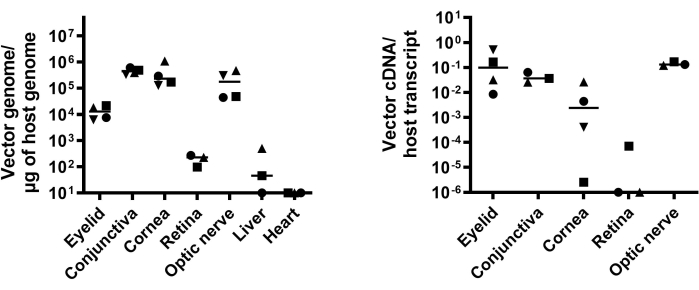
Figure 4: Quantitative analysis of vector biodistribution and transgene expression. (A) Vector biodistribution in eye compartments (eyelid, conjunctiva, cornea, optic nerve, and retina) and other organs (liver and heart) following SCJ is presented as vector genome copy number/µg of host genome DNA. (B) GFP abundance determined by qRT-PCR is presented as vector cDNA copy number/host transcript. This figure is modified from 13. Abbreviations: SCJ = subconjunctival; qRT-PCR = quantitative revere-transcription PCR; GFP =green fluorescent protein. Please click here to view a larger version of this figure.
Discussion
AAV-mediated gene therapy holds great potential for the treatment of ocular diseases. Current ocular gene therapy relies on two major local administration routes, intravitreal and subretinal injections. Unfortunately, both routes are invasive and can cause serious complications, including retinal detachment, cataract formation, and endophthalmitis. Thus, the investigation of relatively less invasive routes, such as SCJ injection, is of great interest.
Although this technique is relatively straightforward and significantly less invasive, there are several important aspects of AAV delivery that must be highlighted. It is recommended that the AAV vector be stored at -80 °C in aliquots of the desired volume (100 µL here) in siliconized or low-retention microcentrifuge tubes to avoid multiple freeze-thaw cycles and prevent a reduction in viral titer. The vector used in this experiment was stored at -80 °C for ~6 years without a significant loss of titer. Additionally, the vehicle composition may affect the stability of the vector. In this experiment, the virus vehicle was PBS with 350 mM NaCl + 5% D-sorbitol. The titer of the virus used in this study was 5.1 × 109 vg/µL (determined by qPCR), and a total of 7 × 109 vg/eye was administered. However, a range of 1 × 108-1 × 1010 vg/eye is appropriate, depending on the targeted tissue and the transgene.
SCJ injections are relatively less restrictive regarding the administered volume (1-100 µL for a mouse). However, volume differences are thought to play a role in AAV biodistribution and transduction, and larger injection volumes have been reportedly used to create a conjunctival scarring model27. One important aspect is the vascular nature of the conjunctiva, which may lead to substantial systemic clearance of the AAV vector, thus resulting in the generation of neutralizing antibodies to the AAV vector. Ongoing research is actively studying potential strategies to reduce the systemic clearance of AAV therapeutic vectors.
In addition, the in vivo distribution following SCJ injection is a critical consideration for the potential application of this injection route. In the current protocol, the distribution is examined by using a dye (India ink) at one time point rather than multiple time points or in real time. Although these data provide some indication of how the AAV vector solution spreads immediately following the injection, the distribution and kinetic properties of the AAV vector and other substances may vary significantly over time. Although the biodistribution of the AAV vector was detected in different eye compartments by qPCR, as mentioned in the above methods section at the experimental endpoint, ideally, a technique that could monitor the trafficking of the therapeutic reagents throughout the eye in real time would be used; however, this is often challenging in practice. Lastly, the AAV transduction profile observed in mice following SCJ injection may differ in human eyes due to the obvious size, anatomical, and physiological differences between a mouse eye and a human eye.
Retinal diseases are also a major target of gene therapy28; thus, the determination of whether AAV administered via SCJ injection can reach the retinal tissues is worth further examination13,17,29. Previous studies have demonstrated that nanoparticles administered via SCJ injections can reach the inner retina. However, the specific trafficking pathways are unclear. The distribution data of India ink (Figure 2) demonstrates that most of the solution spreads into the periocular connective tissues, indicating that AAV may reach the inner retina through periocular permeation by penetrating the sclera, choroid, and retinal pigmented epithelium.
The corneal transduction data presented in Figure 3A suggests that AAV injected into the SCJ space can penetrate the sclera and cornea. This further suggests that AAV administration via SCJ injection may reach the anterior chamber or even the posterior and vitreous chambers, although a significantly higher dose may be required to achieve desirable effects in the retina30. Nevertheless, SCJ injection is one of the simplest ocular administration routes and holds great promise for AAV delivery to treat multiple ocular diseases, including but not limited to ocular surface diseases such as limbal stem cell deficiency, dry eye disease, and/or diseases of the ocular muscles, such as oculopharyngeal muscular dystrophy.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
The authors thank the Vector Core at the University of North Carolina for providing the scAAV8-GFP vectors used in this study, the CGIBD Histology Core, and the laboratory of Dr. Brian C. Gilger for their assistance with the clinical assessment aspects of this study. This study was supported by the Pfizer-NC Biotech Distinguished Postdoctoral Fellowship and a Career Development Award from the American Society of Gene & Cell Therapy and the Cystic Fibrosis Foundation. The content is solely the responsibility of the authors and does not necessarily represent the official views of the American Society of Gene & Cell Therapy or the Cystic Fibrosis Foundation.
Materials
| 36 G NanoFil Needles | World Precision Instruments | NF36BV-2 | |
| AAV vector | University of North Carolina at Chapel Hill | / | |
| Acepromazine | Henry Schein | NDC 11695-0079-8 | |
| anti-GFP antibody | AVES labs Inc. | ||
| Digital camera | Cannon | Cannon EOS T5i | |
| DNA/RNA extraction kit | Qiagen | 80204 | |
| Forceps | Fine Science Tools | F6521 | |
| Hamilton syringe | Hamilton | 7654-01 | |
| India ink | StatLab | NC9903975 | |
| Ketamine hydrochloride injection solution | Henry Schein | NDC 0409-2051-05 | |
| Moisture-resistant film | Parafilm | 807-6 | |
| Polyethylene tubing | Becton Dickinson and Company | 427401 | |
| Proparacaine 0.1% | Bausch Health US | NDC 24208-730-06 | |
| Rebound tonometer | Tonovet | / | |
| Sodium fluorescein solution | Sigma-Aldich | 46960 | |
| Standard Infuse/Withdraw Pump 11 Pico Plus Elite Programmable Syringe Pump | Harvard Bioscience | 70-4504 | |
| Stereo microscopye | Leica | Mz6 | |
| Tetracaine Hydrochloride Ophthalmic Solution 0.5% | Bausch and Lomb | Rx only | |
| Topical ointment | GenTeal | NDC 0078-0429-47 | |
| Xylazine | Akorn | NDC 59399-110-20 | |
| Zone-Quick Phenol Red Thread Box 100 Threads | ZONE-QUICK | PO6448 |
Riferimenti
- Bourne, R. R. A., et al. Magnitude, temporal trends, and projections of the global prevalence of blindness and distance and near vision impairment: a systematic review and meta-analysis. The Lancet Global Health. 5 (9), 888-897 (2017).
- Swetledge, S., Jung, J. P., Carter, R., Sabliov, C. Distribution of polymeric nanoparticles in the eye: implications in ocular disease therapy. Journal of Nanobiotechnology. 19 (1), 10 (2021).
- Petit, L., Khanna, H., Punzo, C. Advances in gene therapy for diseases of the eye. Humam Gene Therapy. 27 (8), 563-579 (2016).
- Russell, S., et al. Efficacy and safety of voretigene neparvovec (AAV2-hRPE65v2) in patients with RPE65-mediated inherited retinal dystrophy: a randomised, controlled, open-label, phase 3 trial. Lancet. 390 (10097), 849-860 (2017).
- Atchison, R. W., Casto, B. C., Hammon, W. M. Adenovirus-associated defective virus particles. Science. 149 (3685), 754-756 (1965).
- Atchison, R. W., Casto, B. C., Hammon, W. M. Electron microscopy of adenovirus-associated virus (AAV) in cell cultures. Virology. 29 (2), 353-357 (1966).
- Peng, Y., Tang, L., Zhou, Y. Subretinal injection: a review on the novel route of therapeutic delivery for vitreoretinal diseases. Ophthalmic Research. 58 (4), 217-226 (2017).
- Gaudana, R., Jwala, J., Boddu, S. H., Mitra, A. K. Recent perspectives in ocular drug delivery. Pharmacological Research. 26 (5), 1197-1216 (2009).
- Amado, D., et al. Safety and efficacy of subretinal readministration of a viral vector in large animals to treat congenital blindness. Science Translational Medicine. 2 (21), (2010).
- Li, Q., et al. Intraocular route of AAV2 vector administration defines humoral immune response and therapeutic potential. Molecular Vision. 14, 1760-1769 (2008).
- Ausayakhun, S., Yuvaves, P., Ngamtiphakom, S., Prasitsilp, J. Treatment of cytomegalovirus retinitis in AIDS patients with intravitreal ganciclovir. Journal of Medical Association of Thailand. 88, 15-20 (2005).
- Miyadera, K., et al. Intrastromal gene therapy prevents and reverses advanced corneal clouding in a canine model of mucopolysaccharidosis I. Molecular Therapy. 28 (6), 1455-1463 (2020).
- Song, L., et al. Serotype survey of AAV gene delivery via subconjunctival injection in mice. Gene Therapy. 25 (6), 402-414 (2018).
- Cheng, H. C., Yeh, S. I., Tsao, Y. P., Kuo, P. C. Subconjunctival injection of recombinant AAV-angiostatin ameliorates alkali burn induced corneal angiogenesis. Molecular Vision. 13, 2344-2352 (2007).
- Veneziale, R. W., et al. SCH 412499: biodistribution and safety of an adenovirus containing P21(WAF-1/CIP-1) following subconjunctival injection in Cynomolgus monkeys. Cutaneous and Ocular Toxicology. 26 (2), 83-105 (2007).
- Liu, G. S., et al. Gene delivery by subconjunctival injection of adenovirus in rats: a study of local distribution, transgene duration and safety. PLoS One. 10 (12), 0143956 (2015).
- Igarashi, T., et al. Direct comparison of administration routes for AAV8-mediated ocular gene therapy. Current Eye Research. 38 (5), 569-577 (2013).
- Gaudana, R., Ananthula, H. K., Parenky, A., Mitra, A. K. Ocular drug delivery. AAPS Journal. 12 (3), 348-360 (2010).
- Short, B. G. Safety evaluation of ocular drug delivery formulations: techniques and practical considerations. Toxicologic Pathology. 36 (1), 49-62 (2008).
- Stevens, S. Administering a subconjunctival injection. Community Eye Health. 22 (69), 15 (2009).
- Song, L., Bower, J. J., Hirsch, M. L. Preparation and administration of adeno-associated virus vectors for corneal gene delivery. Methods in Molecular Biology. 2145, 77-102 (2020).
- de Vries, V. A., Bassil, F. L., Ramdas, W. D. The effects of intravitreal injections on intraocular pressure and retinal nerve fiber layer: a systematic review and meta-analysis. Scientific Reports. 10 (1), 13248 (2020).
- Hartman, R. R., Kompella, U. B. Intravitreal, subretinal, and suprachoroidal injections: evolution of microneedles for drug delivery. Journal of Ocular Pharmacology and Theraputics. 34 (1-2), 141-153 (2018).
- Nuzzi, R., Scalabrin, S., Becco, A. Reduction of intraocular pressure spikes due to intravitreal bevacizumab injections by scleral indentation with cotton swab or digital ocular massage: innovative techniques compared. Clinical Ophthalmology. 14, 2533-2541 (2020).
- Crabtree, E., et al. AAV-mediated expression of HLA-G1/5 reduces severity of experimental autoimmune uveitis. Scientific Reports. 9 (1), 19864 (2019).
- Song, L., et al. Gene delivery to human limbal stem cells using viral vectors. Human Gene Therapy. 30 (11), 1336-1348 (2019).
- Reichel, M. B., et al. New model of conjunctival scarring in the mouse eye. British Journal of Ophthalmology. 82 (9), 1072-1077 (1998).
- Barnard, A. R., Rudenko, A. N., MacLaren, R. E. Vector shedding and immunogenicity sampling for retinal gene therapy. Methods in Molecular Biology. 1715, 359-371 (2018).
- Gilger, B. C., et al. A fixed-depth microneedle enhances reproducibility and safety for corneal gene therapy. Cornea. 39 (3), 362-369 (2020).
- Cheruvu, N. P., Kompella, U. B. Bovine and porcine transscleral solute transport: influence of lipophilicity and the Choroid-Bruch’s layer. Investigative Ophthalmology and Visual Science. 47 (10), 4513-4522 (2006).

