Electroporation of Plasmid DNA into Mouse Skeletal Muscle
Summary
Electroporation of plasmid DNA into skeletal muscle is a viable method to modulate gene expression without compromising muscle contractility in mice.
Abstract
Transient gene expression modulation in murine skeletal muscle by plasmid electroporation is a useful tool for assessing normal and pathological physiology. Overexpression or knockdown of target genes enables investigators to manipulate individual molecular events and, thus, better understand the mechanisms that impact muscle mass, muscle metabolism, and contractility. In addition, electroporation of DNA plasmids that encode fluorescent tags allows investigators to measure changes in subcellular localization of proteins in skeletal muscle in vivo. A key functional assessment of skeletal muscle includes the measurement of muscle contractility. In this protocol, we demonstrate that whole muscle contractility studies are still possible after plasmid DNA injection, electroporation, and gene expression modulation. The goal of this instructional procedure is to demonstrate the step-by-step method of DNA plasmid electroporation into mouse skeletal muscle to facilitate uptake and expression in the myofibers of the tibialis anterior and extensor digitorum longus muscles, as well as to demonstrate that skeletal muscle contractility is not compromised by injection and electroporation.
Introduction
Plasmid DNA electroporation into skeletal muscle in vivo is an important tool for assessing changes in skeletal muscle physiology and molecular signaling by modulating gene expression in a variety of physiological and pathophysiological conditions1,2,3,4,5,6,7,8,9. Experimental gene transfer into skeletal muscle was demonstrated as early as 1990 by Wolff et al., where both RNA and DNA were successfully transferred without electroporation, and luciferase expression was maintained for at least 2 months10. The relatively low transfection efficiency with injection only is problematic, and Aihara and Miyazaki demonstrated increased gene transfer with electroporation in 1998 by electroporating a pCAGGS-IL-5 construct into the tibialis anterior (TA) muscle and measuring serum IL-5 expression11. Since that time, many studies have investigated the efficacy of different DNA concentrations, volumes, and electroporation parameters to ensure maximal gene transfer efficiency. Mir et al. tested different electroporation parameters, including voltage, pulse number, pulse duration, and frequency, as well as DNA concentration, and determined that greater voltage, pulse number, and DNA concentration all contributed to increased electroporation efficiency12. A major caveat to high electroporation voltage is that, while it facilitates increased DNA uptake into myofibers, it also causes muscle damage, which can confound results. Schertzer et al. showed that electroporation at 200 V caused damage in around 50% of myofibers 3 days after electroporation, whereas only 10% of myofibers were damaged at 50 V13. We have taken into consideration the variables affecting efficient DNA transfer versus muscle damage and found that a voltage of 125 V per centimeter of caliper width is sufficient to accomplish effective gene transfer.
Analysis of muscle fiber cross-sectional area and whole muscle contractility after electroporation are important aspects of the method for measuring changes in muscle size and function due to gene modulation. We and others have previously demonstrated that electroporation of control vectors alone does not cause a decrease in myofiber area. The green fluorescent protein (EGFP) construct was a useful fluorescent indicator of DNA transfection in these studies13,14. A number of studies have investigated in situ contractility of the TA after electroporation and found varying results. One study showed that 75 V/cm electroporation caused about a 30% reduction in tetanic force 3 days post-electroporation, and by 7 days post-electroporation, tetanic force was back to the control level, while 50 V/cm electroporation did not compromise force13,15. Another study showed that there was a 30% loss of tetanic force 3 h after 180 V/cm electroporation, which recovered to the sham force levels after 7 days16.
In the following detailed procedure, we demonstrate injection and electroporation of a pcDNA3-EGFP plasmid in the TA and extensor digitorum longus (EDL) muscles of mice. We also demonstrate that this method does not affect EDL whole-muscle contractility. The aim is to demonstrate efficient plasmid uptake into myofibers without causing loss of function.
Protocol
All experiments using animals were performed at the Penn State College of Medicine, approved by Penn State University's Institutional Animal Care and Use Committee, and performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. 12-week-old female C57BL/6 mice were used for this procedure. All surgical tools were autoclaved for sterility prior to experimentation.
1. TA and EDL injection/electroporation preparation
NOTE: These steps are identical for TA and EDL injection/electroporation preparation.
- Purify expression plasmid constructs to a concentration of 1 µg/µL diluted in sterile PBS prior to the experiment. For this procedure, a commercially available endotoxin-free purification kit was used to purify pcDNA3-EGFP (GFP) or pcDNA3 empty vector (control).
- Calculate the plasmid concentration to ensure adequate injection volume (TA 50 µg of DNA in 50 µL of sterile PBS; EDL 10 µg of DNA in 10 µL of sterile PBS) and aliquot samples.
- Program the electroporator settings by using the select wheel and depressing the wheel to choose the parameters as follows: Mode – LV, Voltage – 12.5 V/mm (to be adjusted at the time of injection), pulse length – 20 ms, number of pulses – 5, interval – 200 ms, polarity – unipolar.
- Anesthetize the mice using isoflurane gas. Place the mice in an induction box with 5% isoflurane. Confirm the surgical plane of anesthesia by the absence of the toe pinch reflex using forceps and reduce isoflurane to 2% maintenance dose. Transfer mice to an appropriate nose cone resting on a circulating water plate at 37 °C for the rest of the procedure.
- Remove hair from both hindlimbs using small hair clippers. Scrub the lower limbs with alternating 70% ethanol/betadine to sanitize the injection area.
2. TA injection/electroporation
- With the mouse in a supine position, locate the TA tendon visible through the skin on the lateral side of the lower leg. Using a 50 µL micro-syringe with a detachable 30 G needle, insert the needle 1-2 mm superior to the myotendinous junction at a shallow 5° angle until the needle reaches the superior end of the muscle.
NOTE: Injection into the middle of the muscle is the goal. - Slowly depress the plunger while slowly retracting the needle along the injection path to deliver 50 µL of plasmid solution. The muscle should swell.
- Set the timer for 1 min and measure the thickness of the leg at the TA. Depending on the size of the mouse, this can be from 5-10 mm. Set the caliper electrodes to the measured thickness and set the electroporator voltage to 12.5 V/mm.
- After 1 min, place the caliper electrodes around the lower limb. The electrodes should be snug but not overly tight. Deliver 5 square-wave pulses, with 20 ms duration and 200 ms intervals (the muscle should twitch with each pulse).
- Repeat with the alternate limb using the control vector.
- Remove the mouse from anesthesia and allow it to recover on a heating pad set to 37 °C. Once recovered, return the mouse to its cage.
3. EDL injection/electroporation
- With the mouse in a supine position, locate the tibia bone anterior crest visually and through gentle palpation.
- Using a scalpel, make a shallow incision through the skin on the lateral side of the tibia anterior crest 5 mm inferior to the knee to 2 mm superior to the TA myotendinous junction.
- Using small scissors, blunt dissect the fascia, exposing the TA muscle.
- Again, using blunt dissection with scissors, separate the TA muscle from the tibia gently by pulling the muscle laterally, exposing the EDL. Small, curved forceps can be used to keep the TA clear from the EDL during the procedure.
- Using a 50 µL micro-syringe with a detachable 30 G needle, insert the needle into the EDL longitudinally until the needle reaches the superior end of the muscle.
- Slowly depress the plunger while slowly retracting the needle along the injection path to inject 10 µL of the plasmid solution (muscle should swell).
- Set a timer for 1 min and measure the thickness of the leg at the EDL. Depending on the size of the mouse, this can be from 5-10 mm. Set the caliper electrodes to the measured thickness and set the electroporator voltage to 12.5 V/mm.
- After 1 min, place the caliper electrodes around the lower limb. The electrodes should be snug but not overly tight. Deliver 5 square-wave pulses, with 20 ms duration and 200 ms intervals (muscle should twitch with each pulse).
- Close the incision using disposable 4/0 non-absorbable nylon sutures.
- Repeat with the alternate limb or leave the limb untouched as absolute control.
- Remove the mouse from anesthesia and allow it to recover on a heating pad set to 37 °C. Administer an appropriate subcutaneous analgesic immediately after surgery and 12-24 h following surgery. Once recovered, return the mouse to its cage.
NOTE: Previous studies have shown a muscle contractility deficit in the TA immediately after electroporation and that contractile recovery occurs over the course of 3 days13,15. For this reason, the analysis of both the TA and EDL muscles for histology, protein expression, or muscle contractility is conducted 3-10 days following electroporation. Prolonged expression of EGFP has been observed up to 3 weeks following the procedure13.
Representative Results
Electroporation to facilitate gene transfer in skeletal muscle is a useful technique used to evaluate changes in muscle physiology. We have demonstrated a detailed, step-by-step procedure to accomplish efficient gene transfer in both the TA and EDL muscles. Differences in transfection efficiency occur due to a number of variables. Among these variables are electroporation parameters (pulses, voltage, pulse duration, etc.), gene construct size, and concentration/volume of DNA injected. We have previously shown that electroporation parameters of 5 pulses at 125 V/cm, with 20 ms duration separated by 200 ms intervals, are sufficient to accomplish efficient gene transfer in the TA14. We also demonstrate in the current study that injection/electroporation of DNA does not cause loss of muscle contractility in the EDL 3-days post-experimentation.
In order to visualize gene transfer, a pcDNA3-EGFP or pcDNA3 (control) construct were electroporated into mouse TA or EDL muscle. 3 days after the procedure, the TA or EDL was carefully dissected, placed in optimal cutting temperature (OTC) media, and snap-frozen in liquid nitrogen-cooled isopentane (2-methylbutane), as previously described14,17,18,19. 10 µm muscle sections were obtained using a cryostat taken from the mid-belly of each muscle. Sections were then incubated in 1% paraformaldehyde for 5 min, followed by 3-5 min washes in PBS. Sections were then incubated in wheat-germ agglutinin conjugated to Texas Red diluted 1:100 in PBS for 90 min in the dark. Sections were again washed in PBS 3 times for 5 min each. Muscle sections were then coverslipped in aqueous mounting media and imaged in the 594 nm wavelength (red) to visualize the individual muscle fibers and in 480 nm wavelength to detect GFP (Figure 1A). The control muscles injected with pcDNA3 were imaged in both channels with the same exposure settings to control for potential autofluorescence. Muscles injected/electroporated with EGFP showed positive green fibers, demonstrating uptake of the pcDNA3-EGFP construct, while muscles injected/electroporated with pcDNA3 (control) showed no green positive fibers. We and others have previously shown that myofiber cross-sectional area is not compromised by GFP expression by comparing GFP positive fibers (green) to non-GFP expressing fibers (black) within the same muscle sections4,6,19. When imaged at a lower magnification, EDL muscle transfection efficiency is visualized through the appearance of green fibers (positive) versus black fibers (negative) (Figure 1B). Transfection efficiency using this procedure was 56.6% ± 4.7%, as measured in 3 EDL muscles. This data shows that the injection and electroporation of the TA muscle is sufficient for efficient gene transfer. Additionally, injection and electroporation of the EDL elicits efficient uptake of gene constructs.
An important tool for the evaluation of skeletal muscle physiology is the measurement of muscle contractility. Previous investigators have shown that the contractility of the TA measured in situ may be compromised early after injection and electroporation of the hind limb13. In order to test whether EDL contractility is compromised after injection and electroporation, we surgically exposed the EDL, injected it with pcDNA3-EGFP or construct, and electroporated the hindlimb. As a control, the alternate limb was left untouched for comparison. The mice were euthanized 3 days later, and the EDL whole muscle contractility was measured using field stimulation in a physiological bath, as previously described14,19,20,21. Briefly, the EDL was dissected, and the tendons were tied via 4/0 silk suture to stainless steel hooks and suspended between a force transducer and static base. The muscles were bathed in Tyrode buffer, which is a physiological bath solution (121 mM NaCl, 5.0 mM KCl, 1.8 mM CaCl2, 0.5 mM MgCl2, 0.4 mM NaH2PO4, 24 mM NaHCO3, 0.1 mM EDTA, 5.5 mM glucose) and bubbled with 100% oxygen throughout the procedure. Muscle contraction was stimulated using platinum electrodes to deliver a supramaximal stimulus. Muscle optimal length (Lo) was adjusted to yield the maximum force at the beginning of the procedure. The force-frequency relationship was determined using stimulation frequencies between 1-150 Hz (0.5 ms pulses at supramaximal voltage). The muscle was allowed to relax for 3 min between each stimulation. After the stimulation procedure was completed, the weight and length of the muscle were measured. The specific force was calculated by normalizing the absolute force to the whole muscle cross-sectional area, which is calculated as the weight divided by the length and using the previously determined muscle density constant (1.056 kg/m−3)21. We have shown that representative injected and electroporated EDLs have similar tetanic responses at 100 Hz compared to control non-injected or electroporated muscles (Figure 2A and Figure 2B). We found muscle tetanic force, specific tetanic force, time to peak tension, and half relaxation time were not compromised in the injected and electroporated EDL compared to the untouched control (Table 1). Our data demonstrate that protein expression that affects contractility may be modulated using electroporation in the EDL without causing adverse effects.
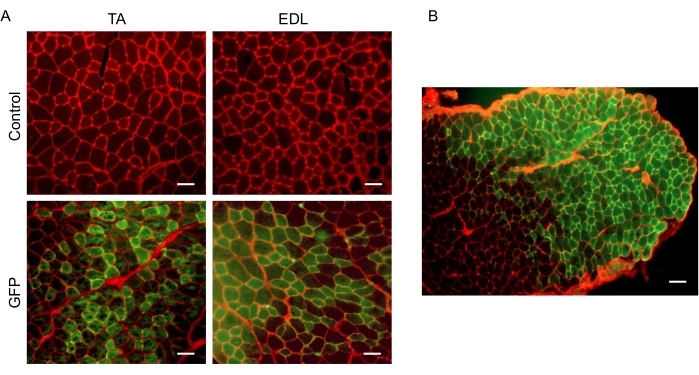
Figure 1: Electroporation of pcDNA3-EGFP is sufficient for DNA uptake in TA and EDL muscle. A) Representative cross-sections from TA and EDL controls (injected with control vector and electroporated 125 V/cm) and GFP (injected with pcDNA3-EGFP and electroporated 125 V/cm). Scale bar 50 µm. B) Representative cross-section from the EDL demonstrating transfection efficiency. Scale bar 100 µm. Please click here to view a larger version of this figure.
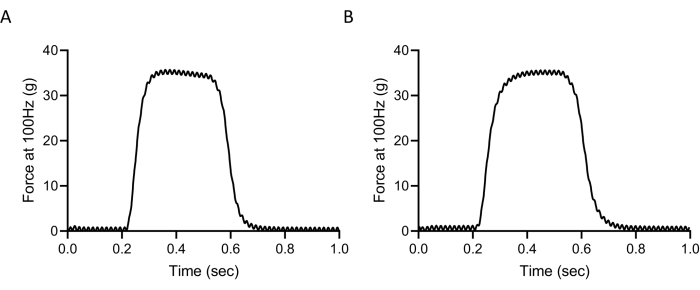
Figure 2: Injection and electroporation does not compromise muscle function. Representative tetanic force curves at 100 Hz from A) non-injected/electroporated EDL (control) and B) injected/electroporated EDL (GFP). Please click here to view a larger version of this figure.
| Control (n=3) | GFP (n=3) | p-value | |
| FO (g) | 34.13 ± 1.15 | 35.87 ± 1.55 | 0.1918 |
| sFO (kN/m2) | 691.56 ± 45.80 | 660.00 ± 33.61 | 0.3917 |
| TTP (sec) | 0.249 ± 0.0203 | 0.247 ± 0.0197 | 0.9084 |
| RT½ (sec) | 0.035 ± 0.0035 | 0.033 ± .0031 | 0.6458 |
| Control -non-injected/non-electroporated; GFP-injected with pcDNA3-EGFP and electroporated. Values are mean ± SD. p=0.05 considered significant. F0– Raw Tetanic Force at 100Hz, sF0-Tetanic Specific Force at 100Hz, TTP- Time to Peak at 1Hz, RT½- Time to half relaxation at 1Hz. | |||
Table 1: Whole muscle contractility from control versus electroporated EDLs. Table showing that different muscle force parameters are not compromised in injected/electroporated EDLs (GFP) compared to non-injected/non-electroporated controls. F0 = tetanic force at 100 Hz, sF0 = specific tetanic force at 100 Hz, TTP = time to peak tension at 1 Hz, RT½ = half relaxation time. Student's t-test; Significance at p = 0.05. n = 3.
Discussion
In vivo gene transfer in skeletal muscle enhanced by electroporation is a useful and relatively simple tool for modulating protein expression in muscle. We have shown the steps required to achieve efficient gene transfer in the EDL and TA muscles and demonstrated that contractility measurement of the EDL is viable following the procedure. This technique does not require more complicated viral vectors and allows for the comparison of transfected and non-transfected muscle fiber cross-sectional area in a single muscle. A limitation to this procedure is that the construct uptake efficiency was not complete and some myofibers remained non-transfected.
The discussed procedures are not limited to the electroporation of expression vectors. We have previously shown that protein knockdown can be achieved by following the same procedures, but using either siRNA or shRNA constructs in vivo14. Additionally, reporter constructs can be utilized to measure the transcriptional activity of target genes2,4,22. These tools allow for the manipulation of multiple proteins concurrently in a single skeletal muscle, while also giving the investigator the option to easily measure transcriptional changes. Skeletal muscle can also be co-transfected with an expression or sh/siRNA vector and fluorescent reporter. Studies have shown that upwards of 95% of muscle fibers that take up one vector will also take up another vector23. This is useful when tagged constructs are not viable. Similar experiments can be conducted in various muscles, including the soleus, gastrocnemius, quadriceps, and flexor digitorum brevis, among others24. The procedure is not limited to mice. Rats have been used extensively for muscle gene transfer, but changes in injection volume, DNA concentration, and electroporation parameters should be considered. Importantly, gene transfer efficiency decreases when electroporation is applied to large areas, and larger muscles may require multiple injections24.
Post-gene transfer animals can be used to study a variety of physiological and pathophysiological conditions. The main determinants of the efficacy of this procedure to take into consideration are the transfection efficiency of the desired construct and the duration of the study. While increased protein expression has been observed as long as 270 days after electroporation12, studies have shown that there is a reduction in expression over time13,25. The simplicity and efficacy of this procedure make it highly applicable to the study of skeletal muscle physiology.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
None
Materials
| 4-0 Nylon suture (non-absorbable) | Ethicon | 662G | Suture to close skin incision |
| 50µl Hamilton syringe | Hamilton | 80501 | microsyringe |
| C57BLl/6NHsd mice | Envigo | 044 | 12 week-old female mice used for experimentation |
| Caliper Electrode | BTX | 45-0102 | 1.0cm x 1.0cm stainless steel |
| Dynamic Muscle Control Data Acquisition/analysis | Aurora Scientific | 605A | Software used for muscle contractility measurement and analysis |
| ECM 830 Electroporation System | BTX | 45-0662 | electroporator |
| EndoFree Plasmid Maxi Kit | Qiagen | 12362 | Plasmid purification kit |
| Extra Narrow Scissors | Fine Science Tools | 14088-10 | Scissors for blunt dissection |
| Force Transducer | Aurora Scientific | 407A | To measure force from EDL |
| Micro-Masquito Hemastats | Fine Science Tools | 13010-12 | Hemastats for surturing |
| pcDNA3.1 mammalian expression vector | Fisher Scientific | V79020 | Control Vector |
| pcDNA3-EGFP expression plasmid | Addgene | 13031 | Plasmid for GFP expression |
| Semken curved forceps | Fine Science Tools | 11009-13 | Forceps for surgery |
| Surgical blades stainless steel no. 10 | Becton Dickinson | 37 1210 | Scalpel blades |
| Tissue-Tek O.C.T. media | VWR | 25608-930 | Freezing media for histology |
| Wheat Germ Agglutinin- Texas Red | Thermo-Fisher Scientific | W21405 | Membrane staining for muscle cross section |
Riferimenti
- Dodd, S., Hain, B., Judge, A. Hsp70 prevents disuse muscle atrophy in senescent rats. Biogerontology. 10, 605-611 (2009).
- Dodd, S. L., Gagnon, B. J., Senf, S. M., Hain, B. A., Judge, A. R. Ros-mediated activation of NF-kappaB and Foxo during muscle disuse. Muscle and Nerve. 41 (1), 110-113 (2010).
- Dodd, S. L., Hain, B., Senf, S. M., Judge, A. R. Hsp27 inhibits IKKβ-induced NF-κB activity and skeletal muscle atrophy. The FASEB Journal. 23 (10), 3415-3423 (2009).
- Hain, B. A., Dodd, S. L., Judge, A. R. IkappaBalpha degradation is necessary for skeletal muscle atrophy associated with contractile claudication. American Journal of Physiology Regulatory, Integregrative and Comparative Physiology. 300 (3), 595-604 (2011).
- Houston, F. E., et al. Heat shock protein 70 overexpression does not attenuate atrophy in botulinum neurotoxin type A-treated skeletal muscle. Journal of Applied Physiology. 119 (1), 83-92 (2015).
- Reed, S. A., Sandesara, P. B., Senf, S. M., Judge, A. R. Inhibition of FoxO transcriptional activity prevents muscle fiber atrophy during cachexia and induces hypertrophy. The FASEB Journal. 26 (3), 987-1000 (2012).
- Senf, S. M., Dodd, S. L., McClung, J. M., Judge, A. R. Hsp70 overexpression inhibits NF-kappaB and Foxo3a transcriptional activities and prevents skeletal muscle atrophy. The FASEB Journal. 22 (11), 3836-3845 (2008).
- Blaveri, K., et al. Patterns of repair of dystrophic mouse muscle: studies on isolated fibers. Developmental Dynamics. 216 (3), 244-256 (1999).
- Fewell, J. G., et al. Gene therapy for the treatment of hemophilia B using PINC-formulated plasmid delivered to muscle with electroporation. Molecular Therapy. 3 (4), 574-583 (2001).
- Wolff, J. A., et al. Direct gene transfer into mouse muscle in vivo. Science. 247 (4949), 1465-1468 (1990).
- Aihara, H., Miyazaki, J. Gene transfer into muscle by electroporation in vivo. Nature Biotechnology. 16 (9), 867-870 (1998).
- Mir, L. M., et al. High-efficiency gene transfer into skeletal muscle mediated by electric pulses. Proceedings of the National Academy of Sciences of the United States of America. 96 (8), 4262-4267 (1999).
- Schertzer, J. D., Plant, D. R., Lynch, G. S. Optimizing plasmid-based gene transfer for investigating skeletal muscle structure and function. Molecular Therapy. 13 (4), 795-803 (2006).
- Hain, B. A., Xu, H., Waning, D. L. Loss of REDD1 prevents chemotherapy-induced muscle atrophy and weakness in mice. Journal of Cachexia, Sarcopenia and Muscle. 12 (6), 1597-1612 (2021).
- Schertzer, J. D., Lynch, G. S. Plasmid-based gene transfer in mouse skeletal muscle by electroporation. Methods in Molecular Biology. 433, 115-125 (2008).
- Roche, J. A., et al. Physiological and histological changes in skeletal muscle following in vivo gene transfer by electroporation. American Journal of Physiology: Cell Physiology. 301 (5), 1239-1250 (2011).
- Hain, B. A., et al. Zoledronic Acid Improves Muscle Function in Healthy Mice Treated with Chemotherapy. Journal of Bone and Mineral Research. 35 (2), 368-381 (2020).
- Hain, B. A., et al. REDD1 deletion attenuates cancer cachexia in mice. Journal of Applied Physiology. 131 (6), 1718-1730 (2021).
- Hain, B. A., Xu, H., Wilcox, J. R., Mutua, D., Waning, D. L. Chemotherapy-induced loss of bone and muscle mass in a mouse model of breast cancer bone metastases and cachexia. Journal of Cachexia, Sarcopenia and Muscle Rapid Communications. 2 (1), (2019).
- Waning, D. L., et al. Excess TGF-beta mediates muscle weakness associated with bone metastases in mice. Nature Medicine. 21, 1262-1271 (2015).
- Bonetto, A., Andersson, D. C., Waning, D. L. Assessment of muscle mass and strength in mice. Bonekey Reports. 4, 732 (2015).
- Senf, S. M., Dodd, S. L., Judge, A. R. FOXO signaling is required for disuse muscle atrophy and is directly regulated by Hsp70. American Journal of Physiology Cell Physiology. 298 (1), 38-45 (2010).
- Rana, Z. A., Ekmark, M., Gundersen, K. Coexpression after electroporation of plasmid mixtures into muscle in vivo. Acta Physiologica. 181 (2), 233-238 (2004).
- Sokolowska, E., Blachnio-Zabielska, A. U. A Critical Review of Electroporation as A Plasmid Delivery System in Mouse Skeletal Muscle. Integrative Journal of Molecular Science. 20 (11), (2019).
- Molnar, M. J., et al. Factors influencing the efficacy, longevity, and safety of electroporation-assisted plasmid-based gene transfer into mouse muscles. Molecular Therapy. 10 (1), 447-455 (2004).

