Summary
This article provides a stepwise guide to establish a primary culture of dental pulps stem cells using the explant culture method and characterization of these cells based on ICSCRT guidelines. The cells isolated by this protocol can be considered as mesenchymal stem cells for further applications.
Abstract
The human dental pulp represents a promising multipotent stem cell reservoir with pre-eminent regenerative competence that can be harvested from an extracted tooth. The neural crest-derived ecto-mesenchymal origin of dental pulp stem cells (DPSCs) bestows a high degree of plasticity that owes to its multifaceted benefits in tissue repair and regeneration. There are various practical ways of harvesting, maintaining, and proliferating adult stem cells being investigated for their use in regenerative medicine. In this work, we demonstrate the establishment of a primary mesenchymal stem cell culture from dental tissue by the explant culture method. The isolated cells were spindle-shaped and adhered to the plastic surface of the culture plate. The phenotypic characterization of these stem cells showed positive expression of the international society of cell therapy (ISCT)-recommended cell surface markers for MSC, such as CD90, CD73, and CD105. Further, negligible expression of hematopoietic (CD45) and endothelial markers (CD34), and less than 2% expression of HLA-DR markers, confirmed the homogeneity and purity of the DPSC cultures. We further illustrated their multipotency based on differentiation to adipogenic, osteogenic, and chondrogenic lineages. We also induced these cells to differentiate into hepatic-like and neuronal-like cells by adding corresponding stimulation media. This optimized protocol will aid in the cultivation of a highly expandable population of mesenchymal stem cells to be utilized in the laboratory or for preclinical studies. Similar protocols can be incorporated into clinical setups for practicing DPSC-based treatments.
Introduction
Adult stem cells have transpired into a powerful therapeutic tool for cell-directed treatments and therapies due to their plasticity, paracrine mechanisms, and immunomodulatory properties1,2,3. The encouraging data from stem cell-based preclinical studies have inspired researchers to work for the bench to-bedside translation. The type of stem cells used for stem cell therapy plays a significant role in successful outcomes. In preclinical and clinical studies, the most widely reported source for mesenchymal stem cells (MSCs) remain bone marrow4,5. However, major drawbacks to using bone marrow-derived stem cells (BMSCs) include their rare population, highly invasive procedures for isolation, and their limited ability to expand. Therefore, alternative sources of MSCs are being explored. In this regard, dental tissues, with their ease of accessibility, enormous plasticity, high regenerative potential, and high proliferative ability, have now been deemed as a rich and potential alternative source of stem cells6,7,8,9,10.
Dental pulp stem cells (DPSCs) were the first type of dental stem cells to be isolated and characterized by Gronthos in 200011. DPSCs have grabbed the attention for tissue engineering applications because of their high proliferation rate, significant differentiation potential, ease of accessibility with effortless culturing, and, most importantly, their ability to be obtained from a discarded tooth without any ethical concern12. The limitations posed by other stem cell sources, such as BMSCs and adipose-derived stem cells (ADSCs), in their isolation and inadequate self-renewal capacities are circumvented by DPSCs13. Human DPSCs can be obtained from human primary teeth, permanent teeth, wisdom teeth, exfoliated deciduous teeth (SHEDs), and apical papillae. Moreover, DPSCs can also be isolated from supernumerary teeth, which are generally discarded14. DPSCs express neural crest-associated markers and have the potential to differentiate into neuronal cells both in vitro and in vivo15. In addition to their neurogenic potential, DPSCs can differentiate into other cell lineages, such as osteogenic, chondrogenic, adipogenic, hepatic, and myogenic, when given specific differentiation conditions13. Thus, these multipotent cells hold great potential for cell-based therapy and can be employed for the regeneration of various tissues. Studies have also reported the potential role of DPSCs in the reconstruction of the cornea16, repair of myocardial infarction17, and their potential therapeutic role in diseases like limb ischemia18, Alzheimer's 19, Parkinson's 20, and aging21. Therefore, dental tissue-derived stem cells can be used not only for dental regeneration, but also for the repair and regeneration of non-dental organs like eyes16, hearts17, livers22, bones23 etc.
There are two particular methods for the isolation of an MSC population from pulp tissue - enzymatic digestion and explant culture24,25. Successful establishment of primary cultures without any significant difference in the quantity and properties of DPSCs have been reported by both these methods26. In this study, we have focused on the isolation of DPSCs by the explant method27, since this method generates DPSCs without contamination of hematopoietic and endothelial cells, as compared to enzymatic digestion which can result in fibroblast contamination28.
Protocol
All the procedures described in the study have been approved by the Institute Ethics Committee (IEC# 9195/PG-12 ITRG/2571-72) of PGIMER, Chandigarh. All the cell culture related experiments need to be performed in a Class II biological safety cabinet (BSC) following aseptic technique. Dental pulp was obtained from healthy teeth of three (F/14, M/14, and M/20) patients undergoing third molar extractions for orthodontic reasons. Before the sample collection, written informed consent was obtained from the patient/guardian in accordance with the guidelines provided by the ethics committee of PGIMER, Chandigarh.
1. Establishment of primary culture of dental pulp stem cells (DPSCs) from human dental tissue
NOTE: Any decayed teeth should not be used.
- Dental pulp sample collection and transportation to tissue culture lab
- Perform a tooth extraction procedure under non-traumatic and aseptic conditions. Rinse the extracted tooth thoroughly with a sterile saline solution to remove remnants of blood.
- Cut the extracted tooth longitudinally into two pieces using a carbide fissure bur with constant irrigation with sterile normal saline to expose the pulp.
- Transport the extracted tooth along with the intact pulp in sterile α-minimum essential media (α-MEM) maintained on ice to the cell and tissue culture laboratory.
NOTE: Ideally, process all samples received within 2 h for cell culture. However, a recent report has suggested that processing the tooth sample even after 24 h did not affect the viability and proliferation of DPSCs, however, it did reduce their functionality29.
2. Removal of pulp tissue from the tooth and cell culture of DPSCs (time: 60-120 min)
NOTE: All the steps after tooth transportation have been performed in the cell and tissue culture lab inside a biosafety cabinet level 2.
- Place the tooth sample in sterile culture Petri dishes and thoroughly rinse (three or four times) with 1x sterile phosphate-buffered saline (PBS) solution.
- Carefully remove the pulp tissue from the pulp cavity with the help of a sharp excavator and forceps.
- Once the pulp is removed, clean it thoroughly with sterile PBS to remove any remnants of blood.
- Cut the pulp tissue into small pieces of approximately 2-3 mm with the help of a surgical blade in a Petri dish.
- Divide the surface area of the dish into four parts by putting 1-2 mL of sterile PBS in each area. Put the pulp tissue sample in one area with PBS and cut the tissue into the smallest possible pieces with a surgical blade (the smaller the piece, the easier it will be for stem cells to migrate out of the tissue). After this, transfer each piece by lifting it into another area with PBS, and so on till the fourth area, so that the tissue pieces are properly washed and free of any contaminants.
- Meanwhile, pour approximately 0.8-1 mL of α-MEM containing non-essential amino acids with 10% fetal bovine serum (FBS) and an antibiotic cocktail (i.e., penicillin and streptomycin) in the wells of a 6-well plate. Distribute the media uniformly by moving the plate, so as to spread it evenly on the well surface.
- Seed the small pieces of pulp tissue or explants with the help of forceps into a single well (four to six pieces per well) of the 6-well plate and keep the plate undisturbed for 5 min in a biosafety hood.
NOTE: This step allows/aids in the adherence of tissue/explants to the treated surfaces of culture plates. - Maintain the culture plate in a humidified atmosphere with 37 °C temperature and 5% CO2 level.
- The next day, carefully aspirate the media from the wells without disturbing the explants and add fresh media to it. Change the media every other day at this step to prevent drying. Make sure the incubator has sufficient water in the tray so that extra evaporation does not cause the media in the plate wells to dry up.
NOTE: This is a very crucial step, and disturbance can cause the explants to dislodge from their position. If the explant pieces are dislodged from their position during media transfer, try to reattach them by removing the media completely and letting them adhere to the well surface. The media should be carefully added along the wall of the well without disturbing the explants, otherwise they might detach again. Observe the cell culture plates regularly under an inverted microscope (4x or 10x magnification). Identify DPSCs by their ability to adhere to the plastic surface and their spindle-shaped morphology. - Once sufficient cells migrate from the explants, remove the tissues and allow the cells to proliferate and reach the optimum 70%-75% cell confluence before subculturing. The volume of α-MEM containing 10% FBS can be increased to 2 mL at this step so that the cells can divide faster. Change the media every third day at this stage.
3. DPSC expansion
- Wash the confluent T75 culture flask (confluency: 70%-80%) of DPSCs twice with PBS (10 mL).
- Add 2.5 mL of 0.25% trypsin-ethylenediaminetetraacetic acid (EDTA) to the side of the T75 flask and gently swirl it to ensure that the trypsin-EDTA covers the entire cell sheet or monolayer of DPSCs.
- Keep the flask inside the hood at 37 °C for 3-5 min. After 3 min, gently tap the flask to ensure the dissociation of cells.
- Add 5 mL of complete media (α-MEM with 10% FBS) or 250 µL of FBS (10% of the trypsin volume used) as a neutralizing agent to inactivate the trypsin activity. Transfer the whole cell suspension of dissociated DPSCs in a fresh 15 mL conical centrifuge tube.
- Centrifuge the dissociated cells at 500 x g for 2 min at room temperature to get the cell pellet.
- Aspirate off all supernatant using the pipette, add 5-10 mL of PBS to the flask to collect remnant cells, wash down the cell in PBS with gentle pipetting, and centrifuge the cells again at 500 x g for 2 min. Repeat if needed.
- Resuspend the cell pellet in 1 mL of complete media (α-MEM with 10% FBS) after carefully removing the supernatant and performing gentle pipetting to obtain a single-cell suspension.
- Split the cells and seed them (equal volume) into two T75 culture flasks, each containing 10 mL of full complete media. Keep the flask inside the hood at 37 °C.
- The next day, aspirate off the media and replace it with the fresh complete media.
NOTE: Prolong trypsin treatment can compromise cell integrity, viability, and cell function by cleaving the cell surface protein and extracellular matrix protein.
4. Identification of stem cell phenotypic markers (time: 90 – 120 min)
NOTE: For the characterization of cells harvested from the pulp tissue, use cells between the third and fifth passage.
- Use two 70%-80% (~3-4 x 106 cells; count the cells using an image-based cytometer or hemocytometer) confluent T75 culture flasks of DPSCs and wash the cells twice with 10 mL of PBS.
- Add 2.5 mL of 0.25% trypsin-EDTA and keep the flask inside the hood at 37 °C for 3-5 min. Add 5 mL of complete media or 250 µL of FBS (10% of the trypsin volume used) as a neutralizing agent to inactivate the trypsin activity.
- Collect the cells using a pipette in a 15 mL conical centrifuge tube. Centrifuge the dissociated cells at 500 x g for 2 min at room temperature to get the cell pellet.
- Aspirate off all supernatant using the pipette, add 5 mL of PBS to the flask to collect remnant cells, wash down the cell in PBS with gentle pipetting, and centrifuge the cells again at 500 x g for 2 min.
- Resuspend the pellet in 1 mL of PBS after carefully removing the supernatant and perform gentle pipetting to obtain a single-cell suspension.
- Count the cells using an image-based cytometer or hemocytometer, depending on the availability, and distribute them in microcentrifuge tubes at 1 x 104 cells/tube. Incubate the cells in 0.5%-1% bovine serum albumin (BSA) for 0.5-1 h.
- To each tube, add 1-2 µL of fluorescein isothiocyanate (FITC)- and phycoerythrin (PE)-conjugated antibodies for cluster of differentiation (CD) markers (i.e., CD90 [0.4 µg], CD73 [0.8 µg], CD105 [0.4 µg], CD34 [0.8 µg], CD45 [0.4 µg], and HLA-DR [0.08 µg])6,7,8,9 and incubate for 0.5 h in the dark at room temperature.
- After 0.5 h, pellet down the cells by centrifuging at 500 x g for 5 min. Aspirate the supernatant, wash down the pellet of cells with PBS, and centrifuge again.
- After centrifugation, resuspend the pellet in PBS (200 µL/tube), transfer the cell suspension to flow cytometry tubes, and acquire them using a flow cytometer. Any commercial software can be used to analyze the percentage of positive cells further for various antibodies.
NOTE: Steps 4.7 to 4.9 are to be performed in the dark at room temperature. Proper isotype controls and unstained samples should be used to omit the background fluorescence of the cells.
5. DPSC differentiation into multiple lineages
NOTE: Use 75%-80% confluent cultures of DPSCs at the third to fifth passage for the assessment of multipotency. A control cell group containing α-MEM should be used for all types of differentiations described below.
- Osteogenic differentiation of DPSCs, alizarin red staining, and quantification
- Make a single-cell suspension using 0.25% trypsin-EDTA, as described above in step 4.2, and resuspend the cell pellet in complete α-MEM.
- Count the cells and seed them in 24-well tissue culture-treated plates at a 2 x 104 cells/well cell density containing 0.4-0.5 mL of complete media for 48 h at 37 °C and 5% CO2.
- After 48 h, remove the media from the wells and add 0.4-0.5 mL of osteogenic inductive media (i.e., α-MEM with osteogenic inductive factors such as beta glycerophosphate [5 mM], monopotassium phosphate [1.8 mM], and dexamethasone disodium phosphate [0.01 mM])8. Prepare control wells (uninduced cells) containing complete α-MEM only without any osteogenic inductive factors (control media).
NOTE: The Addition of 50 µM ascorbate is also reported to enhance osteogenic differentiation 30,31. After every third day, replace the induction media and control media with fresh media and culture for 21 days (3 weeks). At the end of induction period, remove the media from the culture plates, add 0.5 mL of PBS, and wash two times. - After PBS washing, fix the cells in 0.5 mL of 10% neutral buffered formalin (NBF) solution for 20-30 min at room temperature. Perform fixation in a fume/chemical hood.
- Alizarin red staining
- Prepare the alizarin red S stain (2%) by dissolving 1 g of alizarin red S in 50 mL of double distilled water (ddH2O) and adjusting the pH to 4.0-4.2 with the help of hydrochloric acid (HCL) or ammonium hydroxide (NH4OH). Filter the stain with filter paper. Store the stain in the dark (4 °C).
- Wash the cells twice with double distilled water (ddH2O), add alizarin red S stain, and incubate at room temperature for 30-45 min.
- Remove the stain from all the wells and wash the cells four or five times with ddH2O to remove any unbound stain. Capture the images using a bright field microscope.
- Alizarin red stain extraction for quantitative analysis
- Add cetyl pyridinium chloride (CPC) buffer (pH 7.0) to the stained wells (0.4 mL/well) and incubate the cells for 2 h at 37 °C. Additionally, the cells can also be incubated with 10% acetic acid for the proper dissolution of alizarin red crystals32. Keep the plates on a plate rocker or shaker.
- After the incubation, collect the supernatant (extracted dye), transfer it to a 96-well plate, and read the plate or samples at 405 nm. To obtain a blank value, add CPC (same volume) buffer only and read32,33 .
6. Adipogenic differentiation of DPSCs, oil red O staining, and quantification
NOTE: The initial steps of seeding are the same as above (i.e., osteogenic differentiation step 5.1).
- After 48 h, remove the media from the wells and add 0.4-0.5 mL of adipogenic inductive media (i.e., α-MEM with adipogenic inductive factors such as isobutyl-methylxanthine [IBMX; 0.5 mM], dexamethasone [1 µM], indomethacin [200 µM], and insulin [10 µM])8. Prepare control wells containing uninduced cells (i.e., cultured in α-MEM only without inductive factors {control media]).
- Replenish the media every third day and culture the cells for 3 weeks. After 3 weeks, aspirate the media from the culture plates and add 0.5 mL of PBS to wash the cells twice.
- After PBS washing, fix the cells in 10% NBF solution for 20-30 min at room temperature.
- Wash the cells twice with double distilled water (ddH2O), add oil red O stain, and incubate at room temperature for 30-40 min.
- Remove the stain from all the wells and rinse the wells containing cells repeatedly with ddH2O until clear (two to four times). Observe under the microscope and capture images.
- Oil red O extraction for quantitative analysis
- Add 0.4 mL of 100% 2-propanol to the stained wells and incubate at room temperature by properly covering the plate for 30 min. Keep the plate on a plate rocker or shaker.
- After the incubation, collect the supernatant, transfer it to 96-well plate, read the plate or samples at 510 nm. Use 100% 2-propanol for blanking.
7. Chondrogenic differentiation of DPSCs, Alcian blue staining, and quantification
NOTE: Chondrogenic differentiation was induced in the monolayer culture of DPSCs. The initial steps of seeding are the same as above (i.e., osteogenic differentiation step 5.1).
- After 48 h, remove the media from the wells and add 0.4-0.5 mL of chondrogenic induction media (i.e., α-MEM with inductive factors specific for chondrogenic differentiation, ascorbic acid [0.2 M], sodium pyruvate [1 mM], transforming growth factor-beta 3 [TGF-β3; 10 µM], dexamethasone [1 mM], and 1x ITS premix [insulin, transferrin, and selenous acid)10. Prepare control wells containing uninduced cells (i.e., cultured in α-MEM only without inductive factors [control media]).
- Replenish the media every third day and culture the cells for 3 weeks. After the end of the induction period, aspirate the media from the culture plates and add 0.5 mL of PBS to wash the cells twice.
- After PBS washing, fix the cells in 10% NBF solution for 15-20 min at room temperature. Wash the cells two or three times with double distilled water (ddH2O), add Alcian blue stain, and incubate at room temperature for 45 min.
- Remove the stain from all the wells, followed by rinsing with 0.1 N HCL two or three times. Wash the cells four or give times with ddH2O.
- Observe the cells for proteoglycans (blue color) under the microscope and capture the images.
- For Alcian blue quantification, extract the dye using 6 M guanidine-HCl and read the samples at an optical density (OD) of 595 nm. The Alcian blue kit used in this study stains acidic proteoglycans in cartilage and specially binds sulfated glycosaminoglycans (GAGs) present in the matrix of chondrocytes10.
8. Differentiation of DPSCs toward hepatic-like lineage and characterization
- Make a single-cell suspension of cells by trypsin treatment, as described above, and resuspend the cell pellet in complete α-MEM.
- Count the cells and seed them in 24-well-treated culture plates at 2 x 104 cells/well containing 0.4-0.5 mL of complete media for 48 h.
- After 48 h, remove the media from the wells and incubate the cells with hepatic induction medium for 14 days. The induction media consists of low glucose Dulbecco's modified eagle medium (DMEM) containing dexamethasone (0.5 µM), epidermal growth factor (EGF; 2 ng/mL), ITS+ premix (50 mg/mL), and hepatocyte growth factor (HGF; 20 ng/mL) 6.
- After 14 days, replace the media with maturation medium for another 14 days (α-MEM supplemented with Oncostatin M [OSM; 20 ng/mL], ITS premix [50 mg/mL], and dexamethasone [0.5 µM]) 6. Replenish the media with fresh media after every third day.
- Perform immunohistochemistry analysis and a low-density lipoprotein (LDL) assay for confirming hepatic-like differentiation.
- Uptake of LDL and immunofluorescent staining for the LDL receptor
NOTE: Hepatocyte-like cells post-differentiation were characterized by the LDL uptake assay kit as per the kit instructions. LDL-550 fluorochrome was used to assess the LDL uptake in differentiated cells under the live state and the LDL receptor antibody was used for the detection of LDL receptors in differentiated cells post-fixation.- After differentiation, stain the live cells with LDL-550 (1:100) for 3-4 h. After 3-4 h, replace the staining solution with culture media and observe the LDL uptake under a fluorescent microscope capable of propidium iodide (PI) excitation and emission wavelengths at 537 and 617 nm, respectively.
- Fix the cells in the fixative solution for 10 min. After cell fixation, block the cells in the blocking solution for 30 min.
- Incubate the cells with a primary LDL receptor antibody for 1 h, wash with 1x PBS three times, and then incubate with 488 secondary antibody (1:100) for 1 h in dark.
- Wash the cells with PBS three times to remove excess secondary antibody. Add 4′,6-diamidino-2-phenylindole (DAPI) as a nuclear stain.
- Observe the cells under a fluorescence microscope. Take images using a fluorescent microscope capable of measuring wavelengths in excitation/emission range of DAPI, PI, and FITC.
NOTE: Single-cell intensity parameters can be recorded to quantify the LDL receptor antibody expression and LDL uptake. Proper secondary antibody controls should be used for immunofluorescence staining to omit background fluorescence resulting from the secondary antibody only.
- Uptake of LDL and immunofluorescent staining for the LDL receptor
9. Neural differentiation of DPSCs toward neuronal-like lineage and characterization
- After the plating of the cells, as described above, culture the cells in α-MEM for 48 h.
- Aspirate the media and replace it with neurobasal media containing growth factors such as 0.5% G5 supplement, 0.2% N2 supplement, 20 ng/mL epidermal growth factor, 20 ng/mL basic fibroblast growth factor, and B27 supplement (1%). Culture the cells in this media for 25-41 days7.
- Change the media every second or third day, depending on its metabolism by the cells. To attain efficient neuronal maturation and more microtubule-associated protein-2 (MAP-2+) neurons, remove EGF and fibroblast growth factor 2 (FGF2) after the neuronal induction stage (14 days).
NOTE: Over time and after repeated media change, differentiated neurons might show detachment from the plate wells due to low adherence, resulting in neuron loss in long-term culture. To avoid this, coat the plate wells with laminin or fibronectin (optional). - Perform immunohistochemistry analysis for confirming neuronal-like differentiation.
- Immunostaining of differentiated DPSCs
NOTE: To confirm the neuronal-like differentiation of DPSCs, immunocytochemistry was performed in the differentiated cells using MAP-2 and neurofilament (NFM) antibodies, which are neurogenic lineage-specific proteins7.- Remove the culture media after differentiation is complete. Add PBS, gently swirl the plate, and remove the PBS with the help of a pipette. Repeat the step.
- Fix the cells using 10% NBF or 4% paraformaldehyde for 20-30 min at room temperature.
- Remove the fixative and wash the cells with 1x PBS two or three times. Add permeabilization solution (Triton-X-100; 0.1%) and incubate the cells for 5 min.
- Perform PBS wash two or three times and block the cells with 0.5%-1% blocking agent (BSA) for 1 h at 37 °C or overnight at 4 °C. Remove the blocking agent and wash the cells with 1x PBS.
- Incubate the cells overnight with primary antibody (prepared in 0.1% BSA, NFM [1:50], and MAP-2 [1:200]).
- Remove the primary antibody and wash the cells with 1x PBS three or four times to remove unbound antibodies.
- Add secondary antibody (rabbit anti-mouse 1:2,000) and incubate the cells at room temperature for 1.5-2 h in the dark. Wash the cells with PBS (two or three times).
- Incubate the cells in DAPI for 10 min and wash again with PBS. Keep the residual volume to avoid drying of the sample. Observe under the fluorescence microscope.
NOTE: Additionally, karyotype stability10 and senescence characterization can also be performed to get a better idea of the purity and functionality of the cells.
- Immunostaining of differentiated DPSCs
Representative Results
Here, we describe how researchers can establish a pure culture of DPSCs via the explant method6,7,8,9,10 and induce them toward multiple lineages to establish the purity of culture for downstream applications.
We established a primary culture of DPSCs from the small tissue of pulp extracted from the third molar tooth of patients, as shown in Figure 1A. Initially, very few rounded cells were observed in the immediate vicinity of explants or tissue on the fourth and fifth day post-explant seeding. However, by the 12th day, the count of cells that emerged out of the explants increased, and by the end of second week (14th-16th day), most of the explants got surrounded by many adherent cells with spindle-shaped morphology. At this time, the explant could be removed from the plates and the cells were allowed to grow in the presence of complete media. After reaching 70%-75% confluence (third week), the cells were sub-cultured and expanded for further experimental setup. Through the explant method, we obtained a uniform culture of stem cells with positivity of MSC CD markers CD90, CD105, CD73, and an almost negligible cell population of another cell type, endothelial cell marker CD34 and hematopoietic cell marker CD45 (Figure 1B).
We also differentiated DPSCs successfully into cells of osteogenic lineage, as evident by positive alizarin red stain uptake in calcified granules in osteocytes, adipogenic lineage, as demonstrated by the uptake of oil red O staining by lipid droplets in differentiated adipocytes, and chondrogenic lineage, as shown by positive Alcian blue staining (Figure 2). DPSCs also showed efficient differentiation toward hepatic-like lineage, as evident by the typical cuboidal morphology and uptake of LDL-550 by live cell (Figure 3A) staining. The differentiated hepatocyte-like cells were fixed and processed for staining with the LDL receptor antibody. Figure 3B shows the positive expression of the LDL receptor in differentiated hepatocyte- like cells along with LDL-550. DPSCs also demonstrated a marked ability to differentiate into neuronal-like cells, as shown by the positive staining of MAP-2 and NFM, typical neuronal proteins (Figure 4). In a nutshell, our study shows that DPSCs can be isolated and cultured as a pure population, with a >95% purity for stem cell markers, and differentiated into various lineages for further downstream applications like cell therapy, drug testing, and disease modeling.
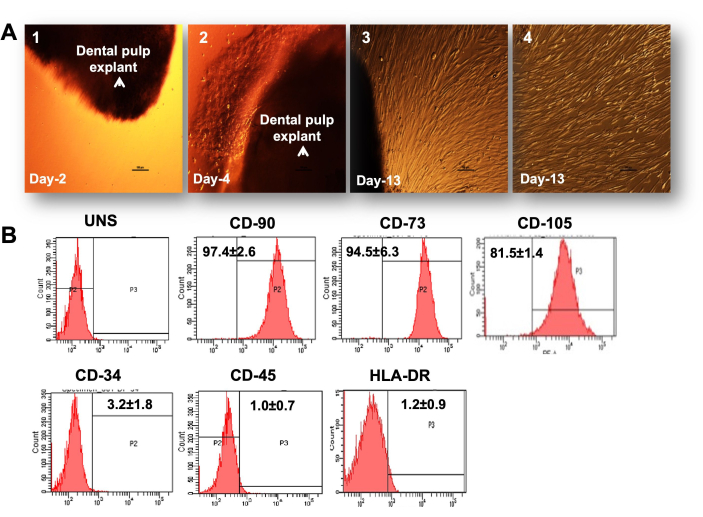
Figure 1: Primary culture of DPSCs. (A). Differential interference contrast (DIC) images showing various stages in development of DPSC primary culture: 1) explant at the initial budding stage with rounded bubble type cells coming out of dental tissue on the second day of seeding; 2) establishment of a mess-like network of cells coming out of tissue on the fourth day; 3) cells with full spindle-shaped morphology at confluence on the 13th day and cells covering the entire surface of the culture dish after the removal of tissue. Scale bar = 100 µm, (B). Stem cell marker characterization by flow cytometry with histograms and a bar graph showing the positive expression of stemness markers and negative expression of non-stem cell markers in DPSCs (n = 3). Each experiment was repeated in triplicate at least three times. Please click here to view a larger version of this figure.
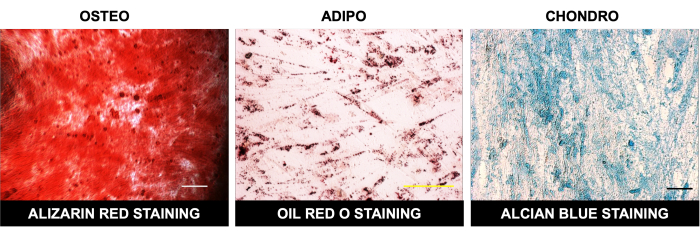
Figure 2: Multilineage differentiation of DPSCs. Bright field microscopic images showing alizarin red staining after osteogenic differentiation, oil red O staining post-adipogenic differentiation, and Alcian blue staining after chondrogenic differentiation in DPSCs. Scale bar = 100 µm (n = 3). Each experiment was repeated in triplicate at least three times. Please click here to view a larger version of this figure.
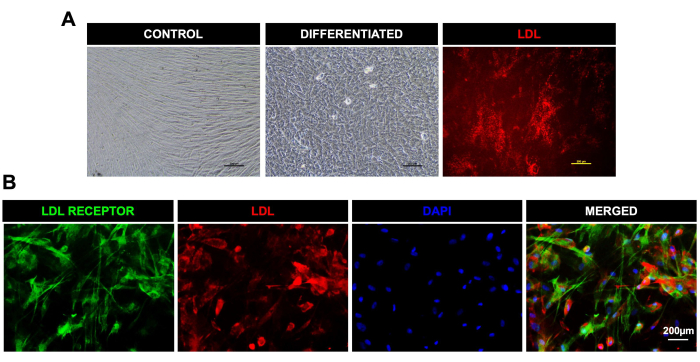
Figure 3: Differentiation of DPSCs toward hepatocyte-like cells and uptake of LDL. (A). Light microscopic images for control and hepatic-like cells differentiated from DPSCs; the last image on the extreme right shows LDL uptake in the live differentiated cells. (B). Fluorescent microscopic images showing the expression of the LDL receptor (green) and uptake of LDL-550 (red) (fixed cells) in DPSCs post-hepatic differentiation. DAPI was used as a nuclear stain. Scale bar = 200 µm (n = 3). Each experiment was repeated in triplicate at least three times. Please click here to view a larger version of this figure.
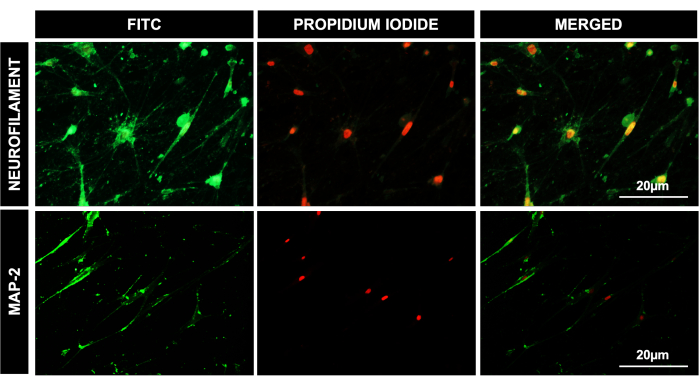
Figure 4: Differentiation of DPSCs to neuronal lineage. Fluorescent microscopic images for DPSCs after 41 days of neural differentiation, showing a positive expression of neural lineage-specific antibodies' neurofilament and MAP-2. PI was used as a nuclear stain. Scale bar = 20 µm (n = 3). Each experiment was repeated in triplicate at least three times. Please click here to view a larger version of this figure.
Discussion
Stem cells have pinned the hopes of curing numerous diseases, owing to their plasticity, robustness, immunomodulatory properties, paracrine mechanisms, and homing efficiencies. Dental pulp tissue is considered the most potent and valuable source of stem cells, with eminent plasticity and a regenerative capability. Here, we demonstrate the isolation of DPSCs, utilizing the widely adopted explant culture method, in which the cells migrate from pieces of pulp tissue or explants to grow into a homogenous cell culture that morphologically resembles spindle-shaped fibroblast-like cells. The explant method yields more uniform cultures of DPSCs, devoid of other cell types such as endothelial cells and pericytes, which remain during the enzymatic procedure for DPSC culture establishment34. We showed that DPSCs derived by the explant method express ISCT-recommended mesenchymal cell surface CD markers, such as CD90, CD73, and CD105. They are also devoid of CD45, CD34, and HLA-DR marker-expressing cells, which indicate the homogeneity and purity of DPSC culture. The lack of HLA-DR also makes them suitable for transplantation purposes, with minimum rejection possibilities. There are a few critical steps in the protocol to ensure the efficiency and purity of DPSC culture. The explants should be washed at least four times with PBS before being put in the culture plates to avoid any possible contamination. Also, the explants should adhere well to the culture well surface, as detached explants would not yield any cells. Hence, utmost care should be taken while handling explants during media change. The media should be added carefully, dropwise along the corners of the well, to not disturb the explants. Once the cells grow in the plate wells, tissue pieces should be removed carefully using the tip, in order not to damage any surrounding cells. During the retrieval of DPSCs at the first passage from plate wells, there might be very few cells, making it impossible to see any visible cell pellet in the tube after centrifugation. Some cells might also stick to the walls of the centrifuge tube, making it hard to get any cells during the upscaling process. Care should be taken to rinse the culture plate and 15 mL tube well with PBS to retrieve as many cells as possible. All cell-based differentiation experiments should be performed in DPSCs below passage number eight, as higher passages might lead to the accumulation of genetic aberrations and spontaneous differentiation, hence affecting the results.
There are also a few limitations of the protocol. The explant culture is a slow protocol and yields enough cells after 15-20 days of culture only. In between, there is a risk of contamination, and the explant (dental pulp pieces) can get detached from the plate surface, which would not yield any cells later. Hence, utmost care should be taken while changing the media. Also, repeated PBS washings are important before seeding the explants, as teeth are rich in oral micro flora and there are very high chances of contamination in the culture if proper washing is not performed. Human DPSCs are ectodermal in origin and during tooth development; they migrate from the neural crest and later differentiate into mesenchymal cells. This confers special properties of both mesodermal and ectodermal lineage in them34. Arthur et al. postulated that the neural crest-derived cells are predisposed to neural differentiation35; the spontaneous neural differentiation of DPSCs was also reported by Kim et al.36. Another study has indicated the potential role of the distal C-terminus L-type voltage-gated calcium channel in directing DPSCs to attain a neuronal phenotype37. Previously, our lab demonstrated the cryopreservation efficiency of these DPSCs by preserving them at -80 °C for 5 years10. These cells were able to maintain stemness characteristics, proliferation, and differentiation ability, as well as a stable karyotype without any structural or numerical abnormalities. In addition, in a comparative analysis, our lab also demonstrated the superior tendency of DPSCs toward neurogenic7 and osteogenic8 lineages, as compared to BMSCs. Their superior competence and inherit tendency toward osteogenic and neural lineage make these cells the best-suited candidate for stem cell therapies in bone disorders and neurodegenerative diseases.
Altogether, we describe here an efficient protocol to obtain a pure population of DPSCs by the explant method, as evident by their stem cell marker expression and multilineage differentiation capability.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We acknowledge the funding support to AK from the Department of Health Research (DHR), ICMR, Govt. of India (DHR-NRI Grant # R.12015/01/2022-HR). SR has received funding from ICMR, Govt. of India (Grant # 2020-7593/SCR-BMS) and PS has received fellowship from CSIR, Govt. of India. We are also thankful to Ms. Sandhya Tokhi and Ms. Bhupinder Kaur for assistance in flow cytometry, and central sophisticated instrumentation core (CSIC) and PGIMER, Chandigarh for providing infrastructural support.
Materials
| 6 well cell culture plate | Costar | 3516 | For cell culture |
| Alcian blue stain | EZstain chondrocyte staining kit, HiMedia | CCK029 | |
| alizarin red S stain | Sigma-Aldrich | TMS-008 | Osteogenic stain |
| Antibiotic cocktail | Himedia | A002-5X50ML | To prevent culture contamination |
| Ascorbic Acid | Himedia | TC094-25G | Chondrogenic induction |
| B27 supplement | Gibco | 17504044 | For neural induction |
| bFGF ( basic Fibroblast Growth Factor) | Gibco | PHG0024 | For neural induction |
| CD 105 | BD-Pharmingen | 560839 | |
| CD 35 | Biolegend | 343604 | |
| CD 45 | Biolegend | 304006 | |
| CD 73 | Biolegend | 344016 | |
| CD 90 | Biolegend | 328107 | Characterization |
| cetyl pyridinium chloride (CPC) | Sigma-Aldrich | 1104006 | For Alizarin Red extraction |
| Dexamethasone 21-phosphate disodium | Sigma-Aldrich | D1159-100MG | |
| Dulbecco's Phosphate Buffered Saline | Himedia | TS1006-5L | For washing purpose |
| EGF (Epidermal Growth Factor) | Gibco | PHG0311 | For hepatic and neural induction |
| EVOS LED microscope | Invitrogen | For fluorescence imaging | |
| EZ stain Chondrocyte staining kit | Himedia | CCK029-1KT | Chondro stain Kit |
| FACS Canto flow cytometer | BD Biosciences | For cell characterization | |
| Fetal Bovine Serum | Gibco | 16000044 | For primary culture |
| Fetal Bovine Serum | Sigma-Aldrich | F2442 | For cell culture |
| G5 supplement | Gibco | 17503012 | For neural induction |
| HGF( Hepatocyte Growth Factor) | Sigma-Aldrich | H1404 | For hepatic Induction |
| HLA-DR | Biolegend | 307605 | |
| Human TGF-β3 | Peprotech | #100-36E-10U | |
| Insulin-Transferrin-Selenous acid premix | Sigma-Aldrich | I3146 | For hepatic Induction |
| ITS premix | Corning | 354350 | |
| LDL Uptake Assay kit | Abcam | ab133127 | For hepatic characterization |
| Low glucose DMEM | Gibco | 11885-084 | For hepatic induction |
| MAP2 antibody | Sigma-Aldrich | M4403 | For neural characterization |
| N2 supplement | Gibco | 17502048 | For neural induction |
| Neural Basal Media | Gibco | 21103049 | For neural induction |
| NFM antibody | Sigma-Aldrich | N4142 | For neural characterization |
| Nikon Elipse TS100 microscope | Nikon | For fluorescence imaging | |
| Oil Red O | Sigma-Aldrich | 01391-250Ml | Adipogenic stain |
| Oncostatin M | R&D Systems | 295-OM-010/CF | For hepatic Induction |
| Petridish | Tarson | 460090-90MM | For tissue cutting |
| Potassium phosphate monobasic | Sigma-Aldrich | 15655-100G | Osteogenic induction |
| Propan-2-ol | Thermo Fisher | Q13827 | For Oil Red O extraction |
| Sodium pyruvate solution | Sigma life sciences | S8636-100ML | |
| Trypsin-EDTA | Sigma-Aldrich | T4049 | For cell passaging |
| Whatman filter paper | merck | WHA1001325 | filter paper |
| α- Minimum Essential Media (α-MEM) | Sigma-Aldrich | M0643-10X 1L | Media for primary culture |
| β-glycerophosphate disodium salt hydrate | Sigma-Aldrich | G9422-50G |
Riferimenti
- Bhattacharyya, S., Kumar, A., Lal Khanduja, K. The voyage of stem cell toward terminal differentiation: a brief overview. Acta Biochimica et Biophysica Sinica. 44 (6), 463-475 (2012).
- Prentice, D. A. Adult stem cells. Circulation Research. 124 (6), 837-839 (2019).
- Raik, S., Kumar, A., Bhattacharyya, S. Insights into cell-free therapeutic approach: Role of stem cell "soup-ernatant". Biotechnology and Applied Biochemistry. 65 (2), 104-118 (2018).
- Rodriguez-Fuentes, D. E., et al. Mesenchymal stem cells current clinical applications: a systematic review. Archives of Medical Research. 52 (1), 93-101 (2021).
- Yamazaki, K., Kawabori, M., Seki, T., Houkin, K. Clinical trials of stem cell treatment for spinal cord injury. International Journal of Molecular Sciences. 21 (11), 3994 (2020).
- Kumar, A., et al. Molecular spectrum of secretome regulates the relative hepatogenic potential of mesenchymal stem cells from bone marrow and dental tissue. Scientific Reports. 7 (1), 15015 (2017).
- Kumar, A., Kumar, V., Rattan, V., Jha, V., Bhattacharyya, S. Secretome cues modulate the neurogenic potential of bone marrow and dental stem cells. Molecular Neurobiology. 54 (6), 4672-4682 (2017).
- Kumar, A., Kumar, V., Rattan, V., Jha, V., Bhattacharyya, S. Secretome proteins regulate comparative osteogenic and adipogenic potential in bone marrow and dental stem cells. Biochimie. 155, 129-139 (2018).
- Kumar, A., Bhattacharyya, S., Rattan, V. Effect of uncontrolled freezing on biological characteristics of human dental pulp stem cells. Cell Tissue Bank. 16 (4), 513-522 (2015).
- Raik, S., et al. Assessment of post-thaw quality of dental mesenchymal stromal cells after long-term cryopreservation by uncontrolled freezing. Applied Biochemistry and Biotechnology. 191 (2), 728-743 (2020).
- Gronthos, S., Mankani, M., Brahim, J., Robey, P. G., Shi, S. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proceedings of the National Academy of Sciences. 97 (25), 13625-13630 (2000).
- Tsutsui, T. W. Dental pulp stem cells: advances to applications. Stem Cells and Cloning. 13, 33-42 (2020).
- Huang, G. T. -. J., Gronthos, S., Shi, S. Mesenchymal stem cells derived from dental tissues vs. those from other sources: their biology and role in regenerative medicine. Journal of Dental Research. 88 (9), 792-806 (2009).
- La Noce, M., et al. Dental pulp stem cells: state of the art and suggestions for a true translation of research into therapy. Journal of Dentistry. 42 (7), 761-768 (2014).
- Xiao, L., Tsutsui, T. Characterization of human dental pulp cells-derived spheroids in serum-free medium: stem cells in the core. Journal of Cellular Biochemistry. 114 (11), 2624-2636 (2013).
- Kumar, A., Yun, H., Funderburgh, M. L., Du, Y. Regenerative therapy for the cornea. Progress in Retinal and Eye Research. 87, 101011 (2022).
- Gandia, C., et al. Human dental pulp stem cells improve left ventricular function, induce angiogenesis, and reduce infarct size in rats with acute myocardial infarction. Stem Cells. 26 (3), 638-645 (2008).
- Yong, Z., et al. Comparison of the angiogenic ability between SHED and DPSC in a mice model with critical limb ischemic. Tissue Engineering and Regenerative Medicine. 19 (4), 861-870 (2022).
- Zhang, X. M., et al. Therapeutic potential of dental pulp stem cell transplantation in a rat model of Alzheimer’s disease. Neural Regeneration Research. 16 (5), 893-898 (2021).
- Kabir, R., et al. Imperative role of dental pulp stem cells in regenerative therapies: a systematic review. Nigerian Journal of Surgery. 20 (1), 1-8 (2014).
- Kumar, A., et al. Dental pulp stem cell secretome ameliorates d-galactose induced accelerated aging in rat model. Cell Biochemistry and Function. 40 (5), 535-545 (2022).
- Hirata, M., et al. Multifaceted therapeutic benefits of factors derived from dental pulp stem cells for mouse liver fibrosis. Stem Cells Translational Medicine. 5 (10), 1416-1424 (2016).
- Fujii, Y., et al. regeneration by human dental pulp stem cells using a helioxanthin derivative and cell-sheet technology. Stem Cell Research & Therapy. 9 (1), 24 (2018).
- Ferrua, C. P., et al. How has dental pulp stem cells isolation been conducted? A scoping review. Brazilian Oral Research. 31, e87 (2017).
- Pilbauerova, N., Soukup, T., Suchankova Kleplova, T., Suchanek, J. Enzymatic isolation, amplification and characterization of dental pulp stem cells. Folia Biologica. 65 (3), 124-133 (2019).
- Tatullo, M., Marrelli, M., Shakesheff, K. M., White, L. J. Dental pulp stem cells: function, isolation and applications in regenerative medicine. Journal of Tissue Engineering and Regenerative Medicine. 9 (11), 1205-1216 (2015).
- Hilkens, P., et al. Effect of isolation methodology on stem cell properties and multilineage differentiation potential of human dental pulp stem cells. Cell and Tissue Research. 353 (1), 65-78 (2013).
- Hendijani, F. Explant culture: An advantageous method for isolation of mesenchymal stem cells from human tissues. Cell Proliferation. 50 (2), e12334 (2017).
- Aryal Ac, S., Islam, M. S., Samsudin, A. R. Investigation of the effect of a time delay on the characteristics and survival of dental pulp stem cells from extracted teeth. Archives of Oral Biology. 119, 104896 (2020).
- Langenbach, F., Handschel, J. Effects of dexamethasone, ascorbic acid and β-glycerophosphate on the osteogenic differentiation of stem cells in vitro. Stem Cell Research & Therapy. 4, 117 (2013).
- Jaiswal, N., Haynesworth, S. E., Caplan, A. I., Bruder, S. P. Osteogenic differentiation of purified, culture-expanded human mesenchymal stem cells in vitro. Journal of Cellular Biochemistry. 64 (2), 295-312 (1997).
- Bernar, A., Gebetsberger, J. V., Bauer, M., Streif, W., Schirmer, M. Optimization of the alizarin red S assay by enhancing mineralization of osteoblasts. International Journal of Molecular Sciences. 24 (1), 723 (2022).
- Coe, C. L., Lubach, G. R., Schneider, M. L., Dierschke, D. J., Ershler, W. B. Early rearing conditions alter immune responses in the developing infant primate. Pediatrics. 90, 505-509 (1992).
- Bakopoulou, A., et al. Assessment of the impact of two different isolation methods on the osteo/odontogenic differentiation potential of human dental stem cells derived from deciduous teeth. Calcified Tissue International. 88 (2), 130-141 (2011).
- Arthur, A., Rychkov, G., Shi, S., Koblar, S. A., Gronthos, S. Adult human dental pulp stem cells differentiate toward functionally active neurons under appropriate environmental cues. Stem Cells. 26 (7), 1787-1795 (2008).
- Kim, B. C., et al. Osteoblastic/cementoblastic and neural differentiation of dental stem cells and their applications to tissue engineering and regenerative medicine. Tissue Engineering. Part B, Reviews. 18 (3), 235-244 (2012).
- Ge, J., et al. Distal C terminus of CaV1.2 channels plays a crucial role in the neural differentiation of dental pulp stem cells. PLoS One. 8 (11), e81332 (2013).

