Immunofluorescent Labeling in Nasal Mucosa Tissue Sections of Allergic Rhinitis Rats via Multicolor Immunoassay
Summary
This protocol describes a multicolor immunofluorescence technique for evaluating the rat model of allergic rhinitis.
Abstract
Allergic rhinitis (AR) is a chronic, non-infectious inflammatory disease of the nasal mucosa, primarily mediated by specific immunoglobulin E (IgE), affecting approximately 10%-20% of the world’s population. While immunofluorescence (IF) staining has long been a standard technique for detecting disease-specific protein expression, conventional IF techniques are limited in their ability to detect the expression levels of three or more proteins in the same sample. Consequently, multicolor IF techniques have been developed in recent years, which allow the simultaneous labeling of multiple targets in cells or tissues.
This protocol provides a comprehensive overview of the process for establishing a rat model of AR, obtaining nasal mucosal samples, and the technical procedures for multicolor immunofluorescence. All rats in the AR group exhibited typical symptoms such as sneezing, a runny nose, and an itchy nose, with behavioral observations scoring ≥5 points. Hematoxylin and eosin (H&E) staining revealed increased inflammatory cell counts and disrupted nasal mucosal integrity in the AR group. Multicolor immunofluorescence (mIF) demonstrated increased expression of RORγt and TICAM-1, while Foxp3 expression decreased in the nasal mucosa tissue of AR rats.
Introduction
Allergic rhinitis (AR) is a chronic, non-infectious inflammatory disease of the nasal mucosa primarily mediated by specific immunoglobulin E (IgE)1,2. It is characterized by symptoms such as sneezing, a runny nose, nasal congestion, and nasal itching. With industrialization and urbanization, the prevalence of AR is gradually increasing, affecting approximately 10%-20% of the world's population1. The immunofluorescence (IF) technique is a fluorescent staining method that utilizes an antibody-antigen binding reaction. It can be employed to detect and quantify the distribution and expression levels of specific proteins in biological tissues or cells. In AR research, IF can simultaneously detect multiple targets, including AR-related cytokines, inflammatory cells, receptors, and more, facilitating the exploration of AR pathogenesis and the effects of drugs3,4,5,6.
The multicolor immunofluorescence (mIF) staining process closely resembles traditional IF, with the addition of an antibody elution step during each round of staining. This modification enables the simultaneous detection of multiple biomarkers on the same tissue section through sequential single-labeling and multiple rounds of re-staining. mIF is based on tyramide signal amplification (TSA), allowing for repeated cycles of TSA fluorescent staining and the use of microwave heating to remove antibodies while retaining fluorescent signals7,8. In comparison to conventional IF, mIF offers several advantages: (1) it can detect weakly expressed antigens that are challenging to identify with conventional IF9,10; (2) it provides high-quality staining with an improved signal-to-noise ratio; (3) it allows for the quantification of tissue-specific structures and regions of interest11; (4) multiplexing multiple pathways efficiently utilizes tissues and conserves limited pathological resources12; (5) multi-parameter analysis through mIF offers deeper insights into tissues, uncovering hidden biological information13.
Overall, mIF allows the observation of different antigen expressions and distributions within the same sample, facilitating the study of target proteins. In the future, researchers seeking to understand the expression and distribution of multiple target proteins will find this technique a valuable choice. This study demonstrates the application of mIF for staining nasal mucosa samples from rats with AR and evaluate the establishment of a rat model of AR.
Protocol
The experimental protocol and procedures have received approval from the Administrative and Animal Research Committee of Chengdu University of Traditional Chinese Medicine (Record number: 2022DL-010). Eight-week-old male Sprague Dawley (SD) rats, weighing 180-200 g, were commercially obtained (see Table of Materials) and housed under a natural light/dark cycle with controlled temperature (23 ± 2 °C) and relative humidity (55% ± 10%). Twelve rats were randomly divided into two groups: the control group and the AR group. All rats were acclimated to these conditions and provided with free access to food and water for one week before the trial.
1. Establishment of a rat model of AR
- Preparing the sensitizing solution: suspend 30 mg of ovalbumin (OVA) and 3 g of Al(OH)3 in 100 mL of saline (see Table of Materials)14,15.
- Basic sensitization: grasp and hold the fully awake rat with its abdomen facing up. Disinfect the abdomen using 75% alcohol-soaked cotton balls. Use a 1.5 mL syringe to inject 1 mL of the sensitizing solution prepared in step 1.1 into the left abdominal cavity of the rats in the AR group. Repeat this injection once every two days for 14 days (Figure 1A).
NOTE: Ensure that the rat's head is positioned lower than its tail to prevent damage to the large and small intestines when inserting the syringe. The injection site is located 1.5 cm from the ventral midline. Rats in the control group should receive intraperitoneal injections of 1 mL of normal saline once every two days for 14 days. - Nasal challenge: Administer 50 µL of 5% OVA via nasal drops using a 100 µL pipette (Figure 1B) on the day following the basal sensitization, repeating this procedure for 7 days (Figure 1C).
NOTE: Prepare the 5% OVA solution by mixing 5 g of OVA with 100 mL of saline. For the control group, administer drops of the same volume of saline.
2. Behavioral scoring of rats
- Within 30 min after completing the final nasal challenge, observe and record the rats using a digital camera to identify symptoms such as nose scratching, sneezing, and a runny nose, based on the scoring criteria outlined in Table 1 (Figure 1D).
NOTE: The confirmation of successful AR modeling is based on achieving a total score of ≥5 points14.
3. Acquisition of nasal mucosa samples
- Sacrificing rats: sacrifice the rats by exposing them to an overdose of CO2. Afterward, disinfect the rats with 75% ethanol (see Table of Materials). Use a scalpel to make an incision along the two corners of the mouth to expose the muscle tissue (Figure 2A). Next, cut the joints of the zygomatic bone and the mandible on both sides of the skull using tissue scissors (Figure 2B), and remove the mandible (Figure 2C).
- Nasal cavity exposure: separate the skin of the maxilla using a hemostat (Figure 2D) to expose the nasal cavity (Figure 2E). Cut the bony connection between the nasal cavity and the maxilla with tissue scissors (Figure 2F). Then, sever the connection between the nasal cavity and the orbital bones on both sides at the infraorbital foramen (Figure 2G) and remove the nasal cavity (Figure 2H).
- Fixation: place the extracted nasal cavity in 4% paraformaldehyde for fixation and store it at room temperature for 24-72 h (see Table of Materials).
NOTE: Ensure that the amount of paraformaldehyde fixative used is sufficient to completely cover the sections for proper fixation.
4. Pre-processing of nasal mucosa samples
- Decalcification: begin by fixing the removed tissues from step 3.1. Wash them three times with PBS for 20 min each. Follow this by washing them three times with distilled water, each time for 20 min. Subsequently, decalcify the tissues in a decalcification solution with a volume of 20-30 times that of the tissues at room temperature.
- The decalcification solution (see Table of Materials) should maintain a pH of 7.2-7.4. Finally, place the decalcified tissue into a dehydration box.
NOTE: The decalcifying solution needs to be changed weekly. The decalcification process can be concluded when the tissue softens (approximately 2 months).
- The decalcification solution (see Table of Materials) should maintain a pH of 7.2-7.4. Finally, place the decalcified tissue into a dehydration box.
- Dehydration and waxing: transfer the dehydration box to a dehydrator (see Table of Materials). Begin with 75% alcohol for 4 h, followed by 85% alcohol for 2 h, 90% alcohol for 2 h, and 95% alcohol for 1 h.
- Subsequently, immerse the tissue in anhydrous ethanol for 30 min, repeat the process for another 30 min, use xylene for 5-10 min, repeat this step for another 5-10 min, immerse the tissue in wax for 1 h, repeat this step for another hour, and finally, immerse the tissue in wax for an additional 1 h.
- Embedding: place the melted wax block into the embedding box (see Table of Materials) and store it in a -20 °C freezer. Once the wax solidifies, remove it and trim the wax block.
- Slicing: insert the trimmed wax block into a paraffin microtome (see Table of Materials) and cut it into slices with a thickness of 3 µm. Place the slices in a 40 °C water bath on a slide spreader. Flatten the tissue, then lift it with a glass slide and bake it in a 60 °C oven.
- After the water has evaporated, and the wax is properly set, remove the slides and store them at a temperature of 25 °C.
5. H&E staining of nasal mucosal tissue
- Dewaxing and hydration: place the slices in xylene for 20 min, repeat the process for another 20 min, absolute ethanol for 5 min, repeat this step for another 5 min, 75% alcohol for 5 min, and finally, wash for 5 min.
- Hematoxylin staining: dispense 100 µL of hematoxylin staining solution (see Table of Materials) for 3-5 min using a 100 µL pipette, rinse for 5-10 s, add 100 µL of 0.5% hydrochloric acid ethanol for 10-30 s, rinse for 5-10 s, add 100 µL of 0.2% ammonia water for 10-30 s, and rinse for 30 s.
NOTE: After hematoxylin staining, differentiation with 0.5% hydrochloric acid ethanol is necessary to remove excessive hematoxylin dye bound to the nucleus and absorbed by the cytoplasm. When eosin staining is performed, the nuclei and cytoplasm will be clearly stained. The slices will appear red or pink after differentiation with 0.5% hydrochloric acid ethanol, so rinse immediately after differentiation to remove the acid from the tissue slices to halt the differentiation. Then, use 0.2% ammonia water to enhance nuclear staining. - Eosin staining: use a 100 µL pipette to dispense 100 µL of eosin staining solution (see Table of Materials) for 5 min and rinse for 30 s.
- Dehydration and sealing: immerse the slices in absolute ethanol for 5 min, repeat the process for another 5 min, repeat once more for 5 min, dimethyl for 5 min, and xylene for 5 min. Finally, seal with neutral resin (see Table of Materials).
- Placing the stained section on the microscope: position the stained section on the microscope, adjust the microscope's focus until the image is clearly visible, set the magnification to 40x, and capture the image.
6. Multicolor immunostaining of nasal mucosal tissue
- Dewaxing and hydration: follow the same as mentioned in step 5.1.
- Citrate-phosphate buffer treatment: place the citrate-phosphate buffer (pH 6.0) (see Table of Materials) into a microwave-safe container. Heat it until boiling, remove it, and then add the slides to the container. Microwave on medium heat for 8 min until boiling, turn off the heat for 8 min, and then switch to medium-low heat for 7 min.
- After natural cooling, transfer the slices to PBS (pH 7.4), shake, and wash them three times using a decolorizing shaker, each time for 5 min.
NOTE: Ensure that the buffer does not evaporate during this process, and prevent the slices from drying out.
- After natural cooling, transfer the slices to PBS (pH 7.4), shake, and wash them three times using a decolorizing shaker, each time for 5 min.
- Blocking endogenous peroxidase: dry the slices, draw a circle around the tissue with an immunohistochemical pen (to prevent the antibody from flowing away), and apply 3% hydrogen peroxide solution (hydrogen peroxide: pure water = 1:9).
- Incubate at room temperature in the dark for 15 min. After natural cooling, place the slices in PBS (pH 7.4) and shake them on a decolorizing shaker for three cycles of 5 min each.
- Blocking: apply 5% goat serum within the circle and incubate for 30 min.
- Primary antibody addition: gently remove the blocking solution, add FOXP3 (dilution: 1:200, see Table of Materials) to the slice, and place it in a humid chamber. Incubate at 4 °C overnight.
NOTE: Add a small amount of water to the humid chamber to prevent antibody evaporation. - Secondary antibody addition: place the slices in PBS (pH 7.4) and shake them on a decolorizing shaker for three cycles of 5 min each. Gently dry the slices and apply the secondary antibody (Goat Anti-Rabbit IgG H&L, see Table of Materials) within the circle. Incubate at room temperature for 50 min in the dark.
- Preparation of Tyramide dilution solution: combine 100 µL of 3% H2O2 with 100 mL of 1x TBST (see Table of Materials).
- Addition of TSA working solution: Mix 1 mL of Tyramide diluent with 2 µL of CY3-Tyramide to prepare TSA working solution. Apply 100 µL of TSA working solution to each slice and incubate at room temperature in the dark for 10 min. Then, wash the slices with PBS three times, 5 min each time.
NOTE: Prepare and use the TSA working solution immediately, and store it at 4 °C in the dark; it is valid for up to 24 h. - Repeat Steps 6.2-6.8: switch to a 1:200 dilution of RORγt (FITC fluorescent dye) and then to a 1:200 dilution of TICAM1 (TYP620 dye) (see Table of Materials).
- DAPI counterstained nuclei: shake the slices gently to dry them, apply DAPI staining solution, and incubate at room temperature for 10 min in the dark.
- Quenching autofluorescence: place the slices in PBS (pH 7.4) and shake them on a decolorizing shaker for three cycles of 5 min each. Apply the autofluorescence quencher (see Table of Materials) to the slices, wait for 5 min, and then wash for 10 min.
- Blocking: place the slices in PBS (pH 7.4) and shake them on a decolorizing shaker for three cycles of 5 min each. Gently shake the slices to dry them and seal them with the anti-fluorescence quencher (see Table of Materials).
- Microscopy: place the stained section on the microscope, adjust the microscope's focus for clear visibility, set the magnification to 20x and 40x, and capture the image.
NOTE: DAPI has a UV excitation wavelength of 330-380 nm and an emission wavelength of 420 nm (blue light). FITC has an excitation wavelength of 465-495 nm and an emission wavelength of 515-555 nm (green light). CY3 has an excitation wavelength of 510-560 nm and an emission wavelength of 590 nm (red light). TYP-690 has an excitation wavelength of 630 nm and an emission wavelength of 690 nm (pink light).
Representative Results
Six SD rats were successfully induced into the AR model through OVA intraperitoneal injection and nasal challenge. AR was induced in all rats in the AR group, accounting for 100% of the group. All rats in the AR group exhibited typical symptoms such as sneezing, a runny nose, and an itchy nose. All behavioral observations scored ≥5 points (Table 2).
H&E staining results on the 21st day of modeling revealed that in the control rats, the nasal mucosa's epithelial cells and cilia were well-arranged, with no signs of inflammatory cell infiltration. Conversely, in the AR group, the nasal septum's mucosa was damaged and detached, with notable neutrophil infiltration (Figure 3).
When comparing the nasal mucosa tissue of AR rats with the control group, it was observed that the expression of RORγt (a Th17 cell-specific transcription factor) and TICAM-1 (Toll-like receptor linker molecule-1) was increased, while Foxp3 (a Treg cell-specific transcription factor) was decreased (Figure 4).
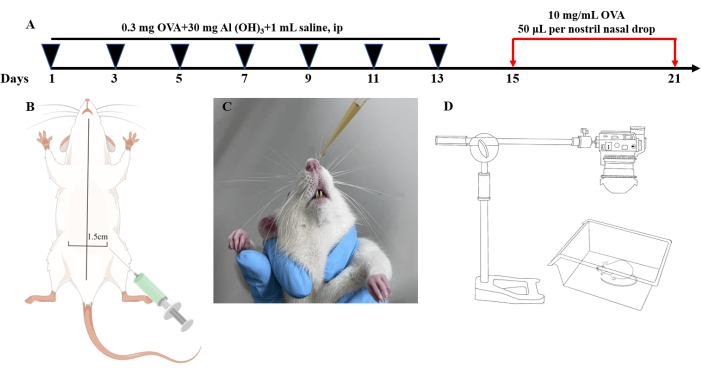
Figure 1: The experimental workflow. (A) Schematic diagram of intraperitoneal injection. (B) Schematic diagram of nasal drip. (C) Flowchart of modeling of allergic rhinitis rats. (D) Schematic diagram of behavioral observation. Please click here to view a larger version of this figure.
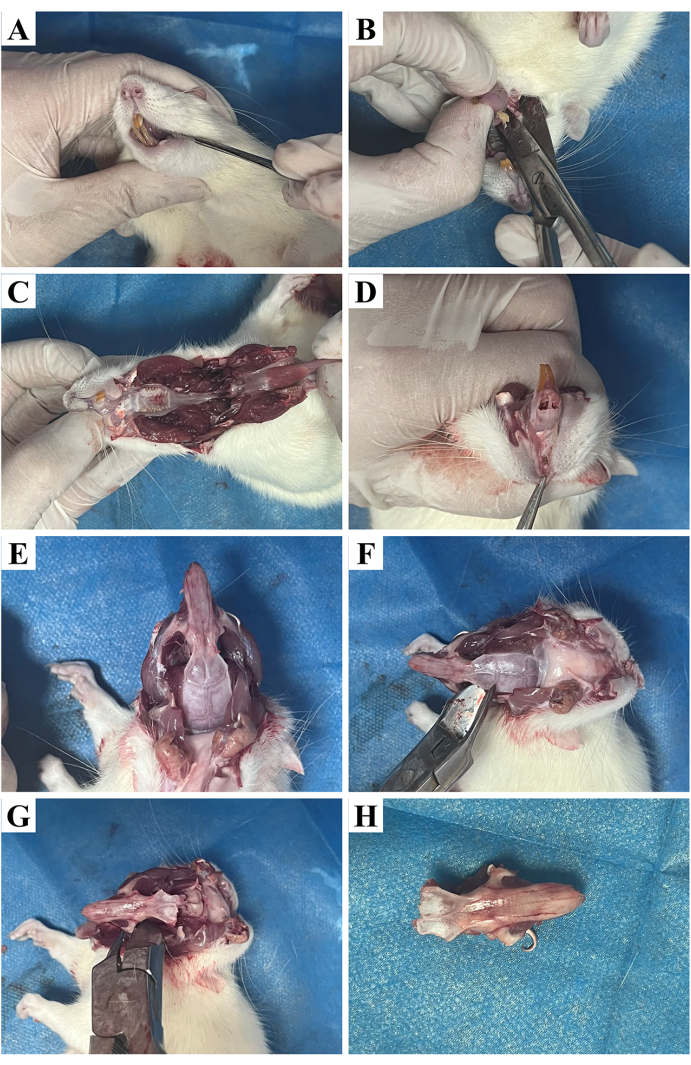
Figure 2: Nasal removal process. (A) Cutting of the muscle tissue at the corners of the mouth. (B) Cutting the connection between the cheekbone and the mandible. (C) Separation of the mandible. (D) Removal of the skin of the maxilla. (E) Exposing the nasal cavity. (F) Cutting the connection between the nasal cavity and the maxilla. (G) Cutting the connection between the nasal cavity and the orbital bone. (H) The removed nasal cavity. Please click here to view a larger version of this figure.
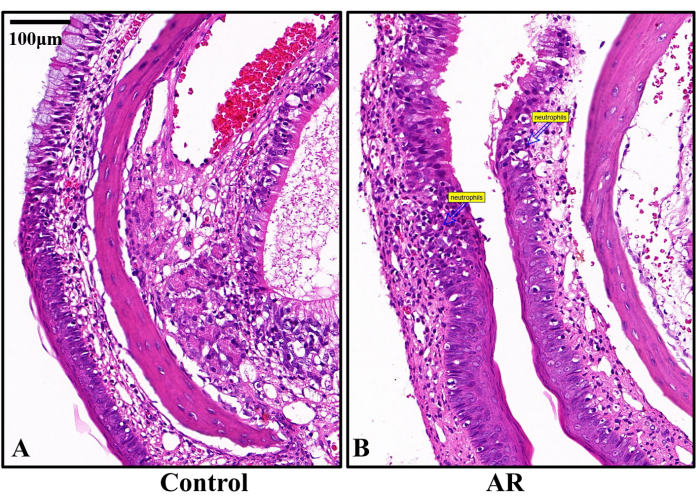
Figure 3: Representative histopathological H&E staining images on day 14 after AR modeling (n = 6). (A) The epithelial cells and cilia in the nasal mucosa of the control rats were well arranged, and no inflammatory cell infiltration was observed. (B) The mucosa of the nasal septum of the AR group was damaged and detached with neutrophil infiltration. Scale bars = 100 µm. Please click here to view a larger version of this figure.
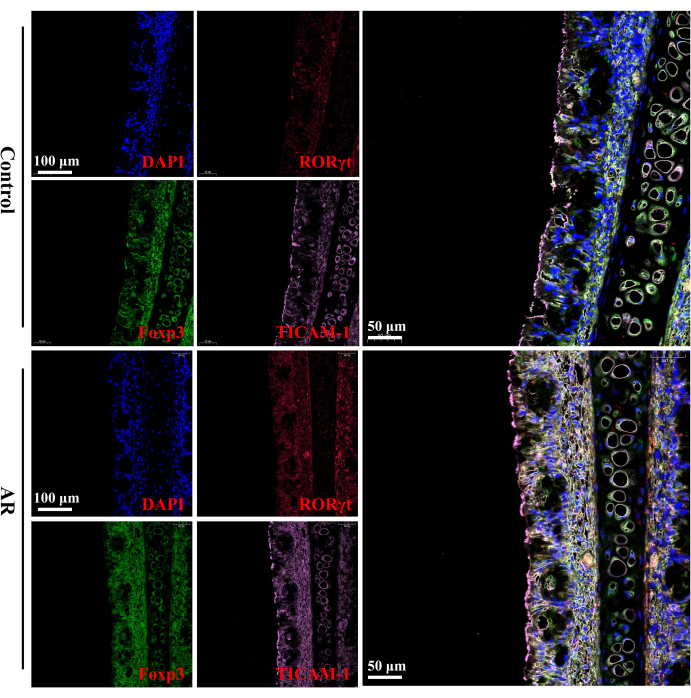
Figure 4: MIF staining analysis of RORγt, Foxp3, and TICAM-1 (n = 6). Compared with the control group, the expression of RORγt and TICAM-1 in the nasal mucosa tissues of rats in the AR group was elevated, while the expression of Foxp3 was decreased. Scale bars = 100 µm (left panels); 50 µm (right panels). Please click here to view a larger version of this figure.
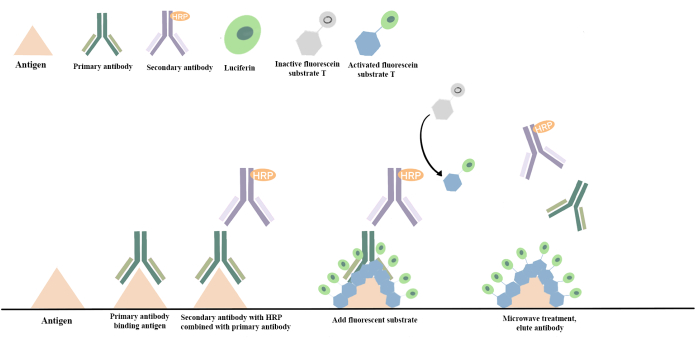
Figure 5: Principle of mIF technique. The mIF technique is based on the TSA technique, which involves covalently binding the fluorescent signal to the antigen. In this process, horseradish peroxidase-labeled on the secondary antibody catalyzes the transition of the fluorescein substrate from an inactive to an activated state. This activated state can covalently bind to the tyrosine on the antigen, resulting in a stable covalent attachment of fluorescein to the sample. Subsequently, non-covalently bound antibodies are removed through thermal repair. The procedure is then repeated with additional primary antibodies, secondary antibodies, and fluorescein to enable the detection of a completely different antigen. This image was redrawn and color-matched with reference to the mIF schematic diagram17. Please click here to view a larger version of this figure.
| Scores | Sneezing frequency | Rhinorrhea | Nasal rubbing |
| 1 | <3 | watery discharge within the nasal cavity | slight and occasional nasal rubbings |
| 2 | 4~10 | watery discharge spilling out of the anterior naris | repeated nasal rubbings |
| 3 | ≥11 | the face covered with abundant watery discharge | rubbings from nose to face |
Table 1: Quantistive scale table of rat behavioral test.
| Individiuals | Day 1 | Day 21 |
| Control 1 | 0 | 0 |
| Control 2 | 0 | 0 |
| Control 3 | 0 | 0 |
| Control 4 | 0 | 0 |
| Control 5 | 0 | 0 |
| Control 6 | 0 | 0 |
| AR 1 | 0 | 7 |
| AR 2 | 0 | 6 |
| AR 3 | 0 | 5 |
| AR 4 | 0 | 5 |
| AR 5 | 0 | 5 |
| AR 6 | 0 | 6 |
Table 2: Results of behavioral scoring.
Discussion
Allergic Rhinitis (AR) is a non-infectious inflammatory disease of the nasal mucosa resulting from a combination of environmental and genetic factors. It has become a global health concern, impacting work efficiency, diminishing the quality of life, impairing sleep, cognitive function, and causing irritability and fatigue. AR affects 10%-20% of the world's population¹ and carries substantial economic costs, causing annual losses of up to 30-50 billion euros in EU countries18. Moreover, several studies have established a strong association between AR and asthma18,19,20, with individuals having AR being seven times more likely to develop asthma than those without AR21. Current research suggests that an imbalance between type 1 helper T cells (Th1) and type 2 helper T cells (Th2) with a bias towards Th2 cells is a significant contributor to AR. Th2 cells, stimulated by interleukin-4, produce Th2-like cytokines such as IL-5 and IL-13, triggering inflammation in B lymphocytes, mast cells, eosinophils, and dendritic cells22. Furthermore, recent AR research indicates a strong connection between an imbalance in helper Th17/regulatory T cell (Treg) populations23. Th17 and Treg cells, both derived from CD4+ T cells, play critical roles in the immune system. RORγt is essential for Th17 cell differentiation24, while Treg cells, with immunosuppressive functions, are key to maintaining immune tolerance. Changes in Foxp3 expression influence Treg cell function25,26. Thus, alterations in RORγt and Foxp3 levels reflect Th17/Treg imbalances, potentially inducing an inflammatory response primarily driven by Th17 cells27. Toll-like receptors (TLRs) are vital proteins in the body's innate immune system, mediating intracellular signaling pathways to activate specific gene expression28. Research by He Shan et al.29 found that variations in the TLR4 gene, specifically CT heterozygous and TT pure mutations at the rs10759930 locus, were associated with AR development. Given TICAM-1's role as the bridging molecule between TLR3 and TLR4, it was hypothesized that AR might be linked to TICAM-1. Indeed, the results showed elevated TICAM-1 expression in the AR group, consistent with Xu et al.'s study30.
The mIF (Multiplex immunofluorescence) technique employed in this study is a relatively new detection method developed over the past 20 years. It offers several advantages over conventional immunofluorescence (IF) techniques, including increased specificity, sensitivity, and throughput, along with the capability for visual analysis. This technique is based on the TSA (Tyramide Signal Amplification) method, which covalently bonds fluorescent signals to antigens and remains unaffected by microwave heating. This allows for the multicolor labeling of tissues through repeated cycles of TSA fluorescent staining, followed by microwave heating to remove antibodies while retaining the fluorescent signal17,31,32 (Figure 5). Advanced algorithms are then applied for the automatic identification and segmentation of target tissue regions displaying specific structures in multilabeled images. This enables quantitative statistical analysis of regions of interest, allowing for the quantitative assessment of cellular immune phenotypes and the functional localization of immune cells. It also provides information on tissue context and spatial distribution33,34.
However, technically, mIF still relies on traditional IF techniques and faces challenges such as antibody cross-reactivity and optical crosstalk. The use of multiple antibodies can lead to cross-reactivity and false-positive results, while overlapping fluorescence spectra may result in the blending of fluorescence signals from multiple targets, making naked-eye differentiation challenging and increasing reliance on specialized image analysis algorithms35. Furthermore, as fluorescent dyes are used to label antibodies, mIF can be affected by fluorescence quenching and bleaching, leading to diminished signal intensity. Notably, while conventional IF techniques evaluate the entire tissue, mIF techniques focus on specific regions of interest within the tissue section for analysis. This may introduce deviations from reality due to tissue heterogeneity, potentially resulting in inaccurate histological quantification or the omission of rare events.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Sichuan Provincial Department of Science and Technology (2021YJ0175).
Materials
| Al(OH)3 | Sollerbauer Biotechnology Co., Ltd | A7130 | |
| 75% ethanol | Anhui Yiren An Co., Ltd | 20210107 | |
| Ammonia | Chengdu Kolon Chemical Co., Ltd | 2021070101 | |
| Anhydrous ethanol | Chengdu Kolon Chemical Co., Ltd | 2022070501 | |
| Anti-fluorescence quenching sealer | SouthernBiotech | 0100-01 | |
| Automatic dyeing machine | Thermo scientific | Varistain Gemini ES | |
| Carrier slides | Nantong Mei Wei De Experimental Equipment Co., Ltd | 220518001 | |
| Citrate-phosphate buffer | Servicebio biotechnology co., Ltd | G1201 | |
| Citric acid antigen repair solution (PH 6.0) | Xavier Biotechnology Co., Ltd | G1201 | |
| Coverslip | Nantong Mei Wei De Experimental Equipment Co. | 220518001 | |
| Coverslip | Nantong Mewtech Life Science Co., Ltd | CS01-2450 | |
| CY3-Tyramide | Sawell Biotechnology Co., Ltd | G1223-50UL | |
| DAPI | Sawell Biotechnology Co., Ltd | G1012 | |
| Decoloring shaker | SCILOGEX | S1010E | |
| EDTA decalcification solution | Wuhan Xavier Biotechnology Co., Ltd | CR2203047 | |
| Electric heating blast dryer | Shanghai Yiheng Scientific Instruments Co., Ltd | DHG-9240A | |
| Embedding box marking machine | Thermo scientific | PrintMate AS | |
| Embedding machine | Wuhan Junjie Electronics Co., Ltd | JB-P5 | |
| Fast tissue dewatering machine | Thermo scientific | STP420 ES | |
| Film sealer | Thermo scientific | Autostainer 360 | |
| FITC-Tyramide | Sawell Biotechnology Co., Ltd | G1222-50UL | |
| Fluorescence microscope | Sunny Optical Technology Co.Ltd | CX40 | |
| Foxp3 | Affinity Biosciences Co., Ltd | bs-10211R | |
| Freezing table | Wuhan Junjie Electronics Co., Ltd | JB-L5 | |
| Goat Anti-Rabbit IgG H&L (HRP) | Liankebio Co., Ltd | GAR0072 | |
| Goat serum | Biosharp | BL210A | |
| H&E staining kit | Leagene | DH0020 | |
| Hemostatic forceps | Shanghai Medical Devices Co., Ltd | J31010 | |
| Hydrochloric acid | Sichuan Xilong Science Co., Ltd | 210608 | |
| Immunohistochemical pen | Biosharp | BC004 | |
| Microwave oven | Midea | M1-L213B | |
| Neutral gum | Sinopharm Group Chemical Reagent Co., Ltd | 10004160 | |
| Ovalbumin | Sollerbauer Biotechnology Co., Ltd | A804010 | |
| Oven | Shanghai Yiheng Scientific Instruments Co., Ltd | DHG-9240A | |
| Palm centrifuge | SCILOGEX | D1008E | |
| Paraformaldehyde | Beyotime Biotechnology Co., Ltd | P0099-100ml | |
| Pathology section scanner | 3DHISTECH Kft | Pannoramic SCAN | |
| PBS buffer | Biosharp | G4202 | |
| Pipette | Dragon | KE0003087/KA0056573 | |
| Rorγt | Affinity Biosciences Co., Ltd | DF3196 | |
| Scalpel | Quanzhou Excellence Medical Co., Ltd | 20170022 | |
| Self-fluorescent quenching agent Sudan Black B | Bioengineering Co., Ltd | A602008-0025 | |
| Slicer | Thermo scientific | HM325 | |
| Slicing machine | Thermo scientific | HM325 | |
| Slide | Nantong Mewtech Life Science Co., Ltd | PC2-301 | |
| Sprague Dawley rats | Sichuan Academy of Traditional Chinese Medicine | SYX  2023-0100 2023-0100 |
|
| TICAM-1 | Affinity Biosciences Co., Ltd | DF6289 | |
| Tissue scissors | Shanghai Medical Devices Co., Ltd | J22120 | |
| Tissue spreading baking sheet machine | Wuhan Junjie Electronics Co., Ltd | JK-6 | |
| TYR-690 fluorescent dyes | Shanghai Rutron Biotechnology Co., Ltd | RC0086-34RM | |
| Vortex mixer | SCILOGEX | SLK-O3000-S | |
| Water bath-slide drier | Wuhan Junjie Electronics Co., Ltd | JK-6 | |
| Wax trimmer | Wuhan Junjie Electronics Co., Ltd | JXL-818 | |
| Xylene | Chengdu Kolon Chemical Co., Ltd | 2022051901 |
Riferimenti
- Brożek, J. L., et al. Allergic rhinitis and its impact on asthma (ARIA) Guidelines-2016 revision. The Journal of Allergy and Clinical Immunology. 140 (4), 950-958 (2017).
- Maurer, M., Zuberbier, T. Undertreatment of rhinitis symptoms in Europe: findings from a cross-sectional questionnaire survey. Allergy. 62 (9), 1057-1063 (2007).
- Ding, Y., et al. Alpha-linolenic acid improves nasal mucosa epithelial barrier function in allergic rhinitis by arresting CD4+ T cell differentiation via IL-4Rα-JAK2-STAT3 pathway. Phytomedicine: International Journal of Phytotherapy and Phytopharmacology. 116, 154825 (2023).
- Zhou, H., et al. Activation of NLRP3 inflammasome contributes to the inflammatory response to allergic rhinitis via macrophage pyroptosis. International Immunopharmacology. 110, 109012 (2022).
- Watts, A. M., Cripps, A. W., West, N. P., Cox, A. J. Modulation of allergic inflammation in the nasal mucosa of allergic rhinitis sufferers with topical pharmaceutical agents. Frontiers in pharmacology. 10, 294 (2019).
- Patel, N., et al. Fungal extracts stimulate solitary chemosensory cell expansion in noninvasive fungal rhinosinusitis. International Forum of Allergy & Rhinology. 9 (7), 730-737 (2019).
- Granier, C., et al. Tim-3 expression on tumor-infiltrating PD-1(+)CD8(+) T cells correlates with poor clinical outcome in renal cell carcinoma. Ricerca sul cancro. 77 (5), 1075-1082 (2017).
- Badoua, C., et al. PD-1-ex pressing tumor-infiltrating T cells are a favorable prognostic biomarker in HPV-associated head and neck cancer. Ricerca sul cancro. 73 (1), 128-138 (2013).
- Fedchenko, N., Reifenrath, J. Different approaches for interpretation and reporting of immunohistochemistry analysis results in the bone tissue – a review. Diagnostic Pathology. 9, 221 (2014).
- Faget, L., Hnasko, T. S. Tyramide signal amplification for immunofluorescent enhancement. Methods in Molecular Biology. 1318, 161-172 (2015).
- Parra, E. R., Villalobos, P., Rodriguez-Canales, J. The multiple faces of programmed cell death ligand 1 expression in malignant and nonmalignant cells. Applied Immunohistochemistry & Molecular Morphology.: AIMM. 27 (4), 287-294 (2019).
- Welsh, A. W., et al. Standardization of estrogen receptor measurement in breast cancer suggests false-negative results are a function of threshold intensity rather than percentage of positive cells. Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology. 29 (22), 2978-2984 (2011).
- Parra, E. R., et al. Validation of multiplex immunofluorescence panels using multispectral microscopy for immune-profiling of formalin-fixed and paraffin-embedded human tumor tissues. Scientific Reports. 7 (1), 13380 (2017).
- Miao, M., Xiang, L., Miao, Y. Specification for preparation of allergic rhinitis animal model (draft). Chinese Traditional and Herbal Drugs. 49 (1), 50-57 (2018).
- Xu, J., et al. Astragalus polysaccharides attenuate ovalbumin-induced allergic rhinitis in rats by inhibiting nlrp3 inflammasome activation and nod2-mediated nf- κ b activation. Journal of Medicinal Food. 24 (1), 1-9 (2021).
- Bousquet, J., et al. Allergic rhinitis. Nature reviews. 6 (1), 95 (2020).
- Huang, C., et al. Expression of activated signal transducer and activator of transcription-3 as a predictive and prognostic marker in advanced esophageal squamous cell carcinoma. World Journal of Surgical Oncology. 13, 314 (2015).
- Leynaert, B., Bousquet, J., Neukirch, C., Liard, R., Neukirch, F. Perennial rhinitis: An independent risk factor for asthma in nonatopic subjects: results from the European Community Respiratory Health Survey. The Journal of Allergy and Clinical Immunology. 104 (2), 301-304 (1999).
- Guerra, S., et al. Rhinitis is an independent risk factor for developing cough apart from colds among adults. Allergy. 60 (3), 343-349 (2005).
- Greisner, W. A., Settipane, R. J., Settipane, G. A. Co-existence of asthma and allergic rhinitis: a 23-year follow-up study of college students. Allergy and Asthma Proceedings. 19 (4), 185-188 (1998).
- Pawankar, R., Mori, S., Ozu, C., Kimura, S. Overview on the pathomechanisms of allergic rhinitis. Asia Pacific Allergy. 1 (3), 157-167 (2011).
- Van Nguyen, T., et al. Anti-allergic rhinitis activity of α-lipoic acid via balancing th17/treg expression and enhancing nrf2/ho-1 pathway signaling. Scientific Reports. 10 (1), 12528 (2020).
- Ivanov, I. I., et al. The orphan nuclear receptor rorγt directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell. 126 (6), 1121-1133 (2006).
- Fontenot, J. D., Gavin, M. A., Rudensky, A. Y. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nature Immunology. 4 (4), 330-336 (2003).
- Zhou, X., et al. Instability of the transcription factor Foxp3 leads to the generation of pathogenic memory T cells in Vivo. Nature Immunology. 10 (9), 1000-1007 (2009).
- Ziegler, S. F., Buckner, J. H. Foxp3 and the regulation of Treg/Th17 differentiation. Microbes and Infection. 11 (5), 594-598 (2009).
- Yaling, Y., Gui, Y., Shuqi, Q. Recent research advances of toll-like receptors and allergic rhinitis. Medical Recapitulate. 23 (7), 1273-1276 (2017).
- Shan, H., Jie, C. Correlation between TLR4 gene polymorphisms and allergic rhinitis. Journal of Clinical Otorhinolaryngology, Head, and Neck Surgery. 31 (13), 991-994 (2017).
- Xu, O., et al. Immunoregulatory mechanism of Treg cells and TICAM-1 patients with severe allergic rhinitis. Chinese Journal of Coal Industry Medicine. 23 (3), 303-306 (2020).
- Granier, C., et al. Tim-3 expression on tumor-infiltrating PD-1+CD8+ T cells correlates with poor clinical outcome in renal cell carcinoma. Ricerca sul cancro. 77 (5), 1075-1082 (2017).
- Badoual, C., et al. PD-1 expressing tumor-infiltrating T cells are a favorable prognostic biomarker in hpv-associated head and neck cancer. Ricerca sul cancro. 73 (1), 128-138 (2013).
- Gustavson, M. D., et al. Development of an unsupervised pixel-based clustering algorithm for compartmentalization of immunohistochemical expression using automated quantitative analysis. Applied Immunohistochemistry & Molecular Morphology. 17 (4), 329-337 (2009).
- Lu, S., et al. Comparison of biomarker modalities for predicting response to PD-1/PD-L1 checkpoint blockade: a systematic review and meta-analysis. JAMA Oncology. 5 (8), 1195-1204 (2019).
- Hernandez, S., et al. Multiplex immunofluorescence tyramide signal amplification for immune cell profiling of paraffin-embedded tumor tissues. Frontiers in Molecular Biosciences. 8, 667067 (2021).

