Using Live-Cell Imaging to Measure the Effects of Pathological Proteins on Axonal Transport in Primary Hippocampal Neurons
Summary
Here, we demonstrate how to combine transfection of primary hippocampal rodent neurons with live-cell confocal imaging to analyze pathological protein-induced effects on axonal transport and identify mechanistic pathways mediating these effects.
Abstract
Bidirectional transport of cargos along the axon is critical for maintaining functional synapses, neural connectivity, and healthy neurons. Axonal transport is disrupted in multiple neurodegenerative diseases, and projection neurons are particularly vulnerable because of the need to transport cellular materials over long distances and sustain substantial axonal mass. Pathological modifications of several disease-related proteins negatively affect transport, including tau, amyloid-β, α-synuclein, superoxide dismutase, and huntingtin, providing a potential common mechanism by which pathological proteins exert toxicity in disease. Methods to study these toxic mechanisms are necessary to understand neurodegenerative disorders and identify potential therapeutic interventions.
Here, cultured primary rodent hippocampal neurons are co-transfected with multiple plasmids to study the effects of pathological proteins on fast axonal transport using live-cell confocal imaging of fluorescently-tagged cargo proteins. We begin with the harvest, dissociation, and culturing of primary hippocampal neurons from rodents. Then, we co-transfect the neurons with plasmid DNA constructs to express fluorescent-tagged cargo protein and wild-type or mutant tau (used as an exemplar of pathological proteins). Axons are identified in live cells using an antibody that binds an extracellular domain of neurofascin, an axon initial segment protein, and an axonal region of interest is imaged to measure fluorescent cargo transport.
Using KymoAnalyzer, a freely available ImageJ macro, we extensively characterize the velocity, pause frequency, and directional cargo density of axonal transport, all of which may be affected by the presence of pathological proteins. Through this method, we identify a phenotype of increased cargo pause frequency associated with the expression of pathological tau protein. Additionally, gene-silencing shRNA constructs can be added to the transfection mix to test the role of other proteins in mediating transport disruption. This protocol is easily adaptable for use with other neurodegenerative disease-related proteins and is a reproducible method to study the mechanisms of how those proteins disrupt axonal transport.
Introduction
Neurons depend on the bidirectional transport of cargo along the axon to maintain functional synapses and neural connectivity. Axonal transport deficits are thought to be critical contributors to the pathogenesis of several neurodegenerative diseases, including Alzheimer's disease (AD) and other tauopathies, Parkinson's disease, amyotrophic lateral sclerosis, and Huntington's disease1,2,3. Indeed, pathological modifications to several disease-related proteins negatively affect transport (reviewed in 4). Developing methods to investigate the mechanisms by which pathological proteins exert toxicity in disease is necessary to understand neurodegenerative disorders and identify potential targets for therapeutic intervention.
Several important insights into axon transport, including the discovery of conventional kinesin, the kinase- and phosphatase-dependent pathways regulating motor proteins, and mechanisms by which pathological proteins disrupt the regulation of axon transport, were made using the isolated squid axoplasm model4,5. Perfusion of squid axoplasm with pathological forms of tau protein inhibits anterograde fast axonal transport (FAT), an effect dependent on the exposure of the phosphatase-activating domain of tau, which activates protein phosphatase 1 (PP1)6,7,8. PP1 activates glycogen synthase kinase 3 (GSK3), which in turn phosphorylates kinesin light chains causing the release of cargo. Another pathological protein in AD is amyloid-β. Oligomeric forms of amyloid-β inhibit bidirectional FAT through casein kinase 2, which phosphorylates kinesin light chains9. Furthermore, pathological huntingtin protein harboring a polyglutamine expansion and a familial ALS-linked SOD1 mutant each disrupt axonal transport in the squid axoplasm through c-Jun N-terminal kinase and p38 mitogen-activated protein kinase activity, respectively10,11.
While the squid axoplasm model continues to be a valuable tool in understanding the effects of pathological proteins on axonal transport, limited access to the equipment and specimens prevents it from being more widely used. We developed a transport assay using live-cell confocal microscopy imaging of rodent (mouse and rat) primary neurons. This model represents an easily adaptable and manipulatable mammalian neuron-based approach using widely available cell sources and microscopy systems. For example, a variety of pathological proteins (e.g., harboring disease-related modifications) are expressed to identify how specific modifications of these proteins affect transport in axons. Similarly, a variety of fluorescently-tagged cargo proteins can be used to examine cargo-specific changes. Moreover, the underlying molecular mechanisms are studied relatively easily by targeting expression (i.e., knockdown or overexpression) of selected proteins that may mediate these effects. This method also is easily adapted for primary neurons derived from a wide variety of animal models.
We present a detailed protocol describing the live-cell axon transport assay that was previously used in primary hippocampal neurons to show that mutant tau proteins associated with frontotemporal lobar dementias (FTLD; P301L or R5L tau) increase the pausing frequency of fluorescently-labeled cargo proteins bidirectionally12. Furthermore, the knockdown of the PP1γ isoform rescued the pausing effects12. This provides support for the model of pathological tau-induced disruption that is mediated through aberrant activation of a signaling pathway initiated by PP1 as described above6,7,12. In a separate study, we showed that pseudophosphorylation of tau at S199/S202/T205 (the pathogenic AT8 phosphoepitope relevant to tauopathies) increased cargo pause frequency and anterograde segment velocity. These effects were dependent on the N-terminal phosphatase activating domain of tau13. These examples highlight the utility of this model for identifying the mechanisms of how pathological proteins disrupt axon transport in mammalian neurons.
This paper provides a detailed description of the method beginning with the harvest, dissociation, and culturing of primary mouse hippocampal neurons, followed by the transfection of the neurons with cargo proteins fused with a fluorescent protein, and finally, the live-cell imaging and image analysis approach. We demonstrate how this method is used to study the effects of modified tau on the bidirectional transport of the vesicle-associated protein, synaptophysin, as an example. However, there is flexibility in the pathogenic protein and transport cargo protein of interest, which makes this a versatile approach to study axonal transport.
Protocol
These protocols were approved by the Michigan State University Institutional Animal Care and Use Committee. This protocol has been successfully applied to Tau Knockout mice in the C57BL/6J background and wild-type Sprague Dawley rats. Other strains should be acceptable as well.
1. Primary hippocampal neuron harvest
- Coat a 4-well glass bottom chamber slide with filtered 0.5 mg/mL poly-d-lysine (PDL) in borate buffer (12.5 mM sodium borate decahydrate and 50 mM boric acid). Add 750 µL/well and incubate at room temperature overnight.
- Wash the chamber slide 4x with sterile deionized water. Air dry after the last wash and keep at 4 °C for short-term storage (<1 week).
NOTE: Do not allow the PDL to dry on the slide prior to and between washes as this is toxic to the cultures. We find that longer-term storage of PDL-coated slides may affect neuronal health and that maintaining consistent intervals between coating and plating reduces inter-run variability in culture viability. - Obtain a timed-pregnant mouse or rat.
NOTE: Timed-pregnant animals can be ordered from various companies or bred in-house. Embryonic day zero refers to the day the vaginal plug is found, and we have had success culturing E16-18 mouse primary hippocampal and cortical neurons with this approach. Rat neurons are also harvested on embryonic day 18. - On E18, euthanize the pregnant female by an approved method. Cut through the abdominal wall and remove the two uterine horns. Transfer them to a Petri dish containing ice-cold 0.9% saline. Rinse until the saline is clean.
NOTE: We administer an overdose of sodium pentobarbital (at least 100 mg/kg) via intraperitoneal injection. - Decapitate the pups. Beginning at the foramen magnum (the opening in the back of the skull), use microdissection scissors to cut the skin and skull along the midline, angling the lower tip of the scissors up against the internal part of the skull to avoid damage to the brain tissue.
- Remove the overlying skin and carefully remove the skull to expose the brain.
- Use forceps to hold the mouth/nose area and turn the skull so the brain is facing downward over a Petri dish filled with 0.9% saline. Flip the brain out of the skull and cut the nerves and brain stem to release the brain into the saline.
- Place the Petri dish under a dissecting microscope for dissection of the hippocampus. Insert scissors into the brain's midline, angle the scissors so that the top blade is pushing one of the cerebral hemispheres outward, and cut to separate the hemisphere away from the subcortical regions.
- Make a vertical cut through the cortex so that the frontal cortex is anterior to, and the hippocampus is posterior to, the cut site. Insert the lower tip of the scissors into the resulting hole and carefully cut along the apex of the cortex stopping at the posterior end.
- Using the forceps, carefully swing out the cortex and finish the posterior cut. The hippocampus should resemble a crescent shape. Carefully trim the remaining cortex so the hippocampus maintains its rounded, crescent shape.
- Using forceps and scissors, gently remove the meninges from the hippocampus.
NOTE: It is important to remove all the meninges from the brain to reduce the number of non-neuronal cells that are collected while refraining from damaging or tearing the hippocampal tissue. - Cut the hippocampus into four equal pieces and place the tissue into a 15 mL conical tube filled with ice-cold calcium- and magnesium-free buffer (CMF; Dulbecco's PBS with 0.1% glucose, 2.5 µg/mL Amphotericin B, and 50 µg/mL Gentamicin).
NOTE: For best results, collect the hippocampi from 5-10 pups per dissociation. Each hippocampus should provide approximately 300,000 cells for mice and 500,000 cells for rats.
2. Primary hippocampal neuron dissociation and plating
- Remove the CMF with a Pasteur pipette and rinse the tissue with fresh cold CMF 4x, swirling gently in between rinses.
- Remove the CMF and add 3 mL of filtered 0.125% trypsin solution (dilute 2.5% trypsin in prewarmed [37 °C] CMF). Incubate the tissue for exactly 15 min at 37 °C; gently swirl every 5 min.
NOTE: Make the trypsin solution immediately before use. - Prepare 5 mL of trypsin inactivation solution (TIS; Hanks' Balanced Salt Solution, 20% newborn calf serum and 1x DNase I solution [10x stock: 5 mM sodium acetate, 1 µM calcium chloride, 0.5 mg/mL DNase I]).
- Remove the trypsin solution and wash 2x with cold CMF. Add 3 mL of cold TIS.
- Dissociate cells through trituration using a 3 mL syringe with progressively smaller diameter needles: triturate 30x using a 14 G needle, 30x using a 15 G needle, 20x using a 16 G needle, 20x using an 18 G needle, and 15x using a 21 G needle. Use 2 s draws with the first four needles and 3 s draws with the 21 G needle.
NOTE: Other methods of trituration (e.g., fire-polished glass pipets14,15) may also be used. We find that using the standardized metal syringe needles substantively reduces variability in cell clumping and viability. - Using the 21 G needle, place a droplet of the cell suspension on a microscope slide and check for cell clumps. If several clumps are observed, triturate three more times through a 22 G needle and re-check. Repeat this process until a homogeneous single-cell suspension is obtained, being careful to triturate gently throughout the process.
- Filter 5 mL of fetal bovine serum (FBS) using a 0.22 µm syringe filter. Gently layer the cell suspension onto the FBS in a 15 mL conical tube. Centrifuge the cells (200 × g at 4 °C) for 5 min.
- Remove as much FBS as possible without disrupting the pellet. Resuspend in 1 mL of warm (37 °C) antibiotic-free neurobasal media (NBM; supplemented with 1x glutamine substitute and 1x B-27).
- Dilute the cell suspension in Trypan blue (0.4%) and obtain a cell count.
NOTE: A 1:1 dilution works well for an automatic cell counter and use a 1:60 dilution for manual counting with a hemacytometer. To continue onto cell plating, the neuron viability should be higher than 90% at this point. This significantly enhances the likelihood of a healthy neuron culture after dissociation. - Plate the cells at a density of 175,000 cells/well (70,000 cells/mm2) in a prewarmed PDL-coated four-well chamber slide in 750 µL of antibiotic-free NBM. Additionally, plate 200,000 cells/well in four wells of a PDL-coated 24-well plate in 1.5 mL of antibiotic-free NBM for use as conditioned medium in the transfection described in section 3. Incubate the cells in a humidity-controlled, 37 °C, and 5% CO2 chamber.
NOTE: The density of cells can be adjusted based on the results. High densities can make it more difficult to image the neurons while low densities can negatively affect the health of the culture and neuron survival. In our experience, rat neurons can be plated at a lower density compared to mouse neurons.
3. Neuron transfection
NOTE: Neurons can be transfected on DIV 7 or DIV 8 without notable changes in transfection efficiency or transport results. These volumes are for four wells of a four-well glass-bottom chamber slide in 750 µL volume/well. This protocol describes transfection using lipid transfection reagents. Allow media and transfection reagents to warm to room temperature prior to use.
- Prepare transfection stock reagent by adding 4.4 µL of transfection reagent to 127.6 µL of reduced-serum medium. Rock gently by hand to mix and incubate at room temperature for 30 min.
- Prepare DNA stocks during the lipid transfection incubation. Add 200 ng of fluorescent-cargo DNA (pFIN mApple-synaptophysin; 12,112 base pairs) and 200 ng of DNA to express the protein of interest (tau plasmids used were pCMV tau-Flag constructs; 5,036 base pairs) into reduced-serum or serum-free medium to a final volume of 32 µL12. Add 0.8 µL of transfection enhancer reagent (2 µL per µg of DNA). Mix well by hand.
NOTE: We find that a derivative of RFP works best (mApple or mCherry), but GFP can be used as well. Additionally, a DNA construct to express a gene-silencing small hairpin RNA (shRNA) can be included to test mechanisms of phenotypes observed with the expression of the protein of interest. To do this, include 150 ng of each of the three DNA constructs in the DNA mix and adjust the amount of transfection enhancer reagent accordingly. Changing the parameters of DNA or lipofection reagent can affect the transfection efficiency and can be adjusted based on the need for more or less efficient transfections. - Add 32 µL of lipid transfection mix to each tube of DNA. Rock gently by hand to mix. Incubate at room temperature for 30 min.
- Slowly add 64 µL of DNA/transfection reagent mix to the cells by placing the tip of the pipette just below the surface of the medium and moving in a serpentine motion throughout the well. Gently rock the chamber slide by hand 3x after addition to all wells to mix further and place the chamber slide in the incubator. Incubate at 37 °C and 5% CO2 for 2 h.
NOTE: Minimize the amount of time neurons are outside of the incubator as they are very sensitive to changes in temperature and pH, which can occur quickly. - Perform a half medium change 2 h after transfection with conditioned media + Neurofascin antibody. Immediately prior to the medium change, pool 1.5 mL of conditioned antibiotic-free NBM from the cells cultured on the 24-well plate. Add 0.75 µL of Neurofascin 186 (NF-186) antibody (500 ng/mL) to the conditioned medium.
NOTE: The NF-186 antibody detects an extracellular domain of neurofascin that is enriched in the axon initial segment (AIS) to definitively identify the axon for imaging. - Perform a full medium change 18 h after transfection with conditioned medium containing the secondary antibody. Add 1 µL of goat-anti-rabbit secondary antibody conjugated to AlexaFluor 647 to 3 mL of conditioned NBM (antibiotic-free) pooled from the 24-well plate. Mix well.
4. Neuron imaging
- Prepare for imaging 2 days after transfection. Warm the live cell chamber attachment for the confocal microscope to 37 °C (including the 60x objective and lens oil) and equilibrate the chamber to 5% CO2.
- Use a high magnification objective and live-cell chamber to image the cells.
NOTE: We use a 60x 1.40 NA objective on a laser scanning confocal microscope but other confocal (e.g., spinning disc) or widefield fluorescence microscopy systems can also be used if they are able to achieve high imaging frame rates. - Visually identify transfected neurons in the channel containing the cargo protein using the eyepieces. Next, image the neurons to identify the AIS in the Cy5 channel (Figure 1).
- Set the field of view to a rectangular box of 32 pixels by 128 pixels and move it to image a region of the axon ~50-150 µm distal to the AIS. Note and record the directionality of the cargo transport and the orientation of the ROI. The dots on the box will appear as the top and right, respectively, of any subsequent images and movies. Record which side of the image is closest to the cell body and which is closest to the axon terminus so the kymograph polyline in step 5.1 can be drawn with the correct orientation.
NOTE: When selecting the region of interest, the imaged area of an isolated axon should be relatively straight and intersect the field of view at each end. The entire segment should be within the same focal plane. The area should clearly display active transport of cargo. Excessively bright neurons typically express the cargo protein and proteins of interest at levels that are likely to be toxic to the neuron and should be avoided. - Set the 561 laser power a 1.40, the pinhole a 7.8, the Size a 128 px2, and the Speed a 32 frames/s. Adjust the gain to visualize the transport of individual cargo vesicles without it being obscured by background signal (typically between 10 and 50 results in high-quality kymographs).
NOTE: The extremely low laser power is achievable because of highly sensitive GaAsP detectors and prevents potential phototoxic effects and photobleaching of moving cargo. The pinhole size is set as wide as possible to increase the focal depth to ensure that the entire ROI is imaged as the axon may not lay perfectly flat on the surface of the slide. - Set the ROI by selecting Draw Rectangular ROI… from the ROI dropdown menu. Draw the ROI so that it covers the entire field of view. Right-click on the ROI and select Use as Stimulation ROI: S1.
- Prepare the ND Setup by including the following steps in the ND Setup tab.
NOTE: This is the five-step acquisition protocol that is set up once and then will be performed by the software each time the movies are collected.- Acquire Image to collect the prestimulation reference image.
- Stimulation; ROI: S1; Interval: NoDelay; Duration 1.64 s, Loops: 7. This step bleaches background fluorescence with several pulses of the laser.
- Acquire Image. This step collects a poststimulation reference image.
- Waiting; No Action; 2 min. This step provides a recovery period for fluorescent cargo to repopulate the bleached ROI.
- Acquisition; Interval: NoDelay; Duration 5 min, Loops: 8,615. This step collects images at the maximum rate for the 5 min imaging period.
- After the ND Setup protocol is set up, click the Apply Stimulation Settings button on the ND Setup tab. Click Run Now when ready to image cargo transport. This will initiate the ND Acquisition protocol from step 4.7.
NOTE: Although the frame rate is set to 32 frames/s, the practical frame rate may be slower (~28.7 frames/s). This value should be recorded for accurate kymograph analysis in section 5. Ideally, live-cell transport imaging is done with the fastest frame rate possible (e.g., >25 frames/s) to better decipher cargo movements. - Reset the field of view to the entire image; then, collect and save an image of the full neuron in the 561 and 647 channels with the ROI box included. Record the XY coordinates where the image was taken for postfixation confirmation of the expression of the protein of interest.
- Collect transport videos from at least two neurons for analysis. Ensure that each independent replicate comes from the means of variables measured from an individual primary neuron harvest (i.e., not individual neurons).
NOTE: For a given primary neuron harvest, we collect videos from 2-4 neurons per condition. - Fix the cells by removing the medium and incubating with prewarmed 4% paraformaldehyde for 20 min at room temperature. Remove the buffer and wash cells for 3 x 5 min each in TBS.
NOTE: For paraformaldehyde fixation, we use cytoskeleton buffer, a common buffer that maintains cytoskeletal structure (10 mM 2-(N-morpholino)ethanesulfonic acid [MES], 138 mM KCl, 3 mM MgCl2, and 4 mM EGTA, pH 6.1). Other buffers (e.g., TBS, PBS) can also be used for fixation. - Confirm that the analyzed neurons express both the protein of interest and the fluorescently tagged cargo protein through immunocytofluorescence staining. Stain for human tau (Tau12) to confirm exogenous tau expression and β-III tubulin (Tuj1) to verify that the neurons remained healthy throughout the imaging process.
NOTE: We observe co-expression of both DNA constructs in nearly all transfected cells using these methods.
5. Generate and analyze Kymographs
NOTE: Kymographs can be generated and analyzed using a variety of different programs. We briefly describe the freely-available KymoAnalyzer (v. 1.01) software using six ImageJ (v. 1.51n) plugins16. These plugins can be downloaded from the developers at the Encalada lab website (https://www.encalada.scripps.edu/kymoanalyzer). More detailed instructions on the use of this software can be found at this site.
- Group the movies generated in step 4.9 based on experimental conditions and replicate. Run the BatchKymographGeneration macro and select the appropriate folder. Draw a kymograph polyline to trace the axon beginning with the proximal end in that particular image. The resulting kymographs are stored in the selected folder (Figure 2).
- Assign tracks manually by running the Tracks macro, selecting an individual movie folder, selecting yes to add a track, and then clicking to track each particle's trajectory. Perform the initial click at the top of the track and click again at every point the particle changes velocity or direction so that the polyline overlays the particle's trace on the kymograph. After the polyline is laid over the entire track, click OK. Repeat until all tracks are assigned.
- Run the remaining macros, assigning frame rate and pixel size based on imaging parameters. First, run CargoPopulation, then NetCargoPopulation, and then Segments. Enter the pixel size and frame rate of the movies when prompted (0.22 µm and 28.7 frames/s, respectively, with these settings). Finally, run the poolData macro to calculate and pool transport parameter data from all kymographs in the selected folder.
- Plot the results from the data files and compare the various conditions with the independent replicates represented by the mean values of all analyzed neurons within an experimental condition for an individual neuron harvest.
Representative Results
Using these methods, we characterized axonal transport in the presence of wild-type or disease-related forms of tau protein to examine potential mechanisms of pathological tau-induced neurotoxicity in disease12,13. The KymoAnalyzer software calculates and pools a variety of different parameters from all kymographs within a given folder. Transport rates are calculated only when cargo is in motion (segmental velocity) as well as the overall rate, including pauses (net velocity). The amount of cargo is calculated as a total, density (#tracks/µm), or flux (#tracks∙µm-1∙s-1). The run length is calculated individually or combined to determine the mean distances traveled between pauses. The cargo pauses are characterized by duration and frequency as well as the percent of time the cargo is not in motion (i.e., moving < 0.1 mm/s). Most measurements are calculated in the anterograde and retrograde directions, as well as in totals (both directions combined). Additionally, the frequency and timing of reversals in direction are calculated. Together, this provides a robust characterization of cargo transport within a specific region of the axon in the presence of a protein of interest.
We expressed wild-type tau and found that these transport parameters did not typically change compared to GFP-expressing control neurons (Figure 3). However, expression of FTLD mutant tau (Figure 3) or pseudophosphorylated tau (data not shown) induced little to no impact on velocity in either direction (Figure 3A,B) but increased total cargo pause frequency compared to WT tau and GFP neurons (Figure 3C)12,13. This effect occurred in cargo traveling in the anterograde and retrograde direction (Figure 3D,E).
This protocol can be adapted to include gene silencing techniques to knock down the expression of other proteins and identify their role in mediating potentially toxic effects. We co-transfected the neurons with plasmids to express shRNAs targeted toward specific isoforms of PP112. Pathological forms of tau disrupt fast axonal transport in a squid axoplasm model of transport and the effect is dependent on PP1 and GSK3β activity6,7,12,17,18. In our model, pathological tau aberrantly activates a PP1-GSK3β-kinesin light chain phosphorylation-based signaling pathway that disrupts normal transport7,19. We found that knocking down the PP1γ1 isoforms, but not the PP1α isoform, rescued the increased pause frequency caused by mutant tau protein (Figure 4)12. In our hands, shRNA-mediated knockdown of GSK3β was toxic to transport in all conditions demonstrating the need for proper controls to accurately interpret results.
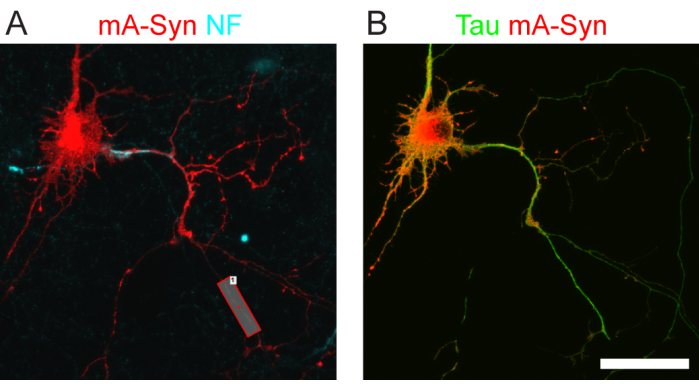
Figure 1: Fluorescent cargo and tau proteins expressed by transfected neurons. (A) A live-cell image of a transfected neuron expressing mApple-synaptophysin (red). The NF-186 antibody and a fluorescent secondary antibody label the external domain of neurofascin in the axon initial segment (cyan)12. The rectangular box labels the axonal region of interest for imaging of cargo transport. (B) After imaging, the cells are fixed and stained via immunofluorescence to confirm co-expression of the protein of interest (tau, green) and cargo protein (mApple-synaptophysin; red). Scale bar = 50 µm (A,B). Abbreviations: mA-Syn = mApple-synaptophysin; NF = neurofascin. This figure is modified from Combs et al.12. Please click here to view a larger version of this figure.
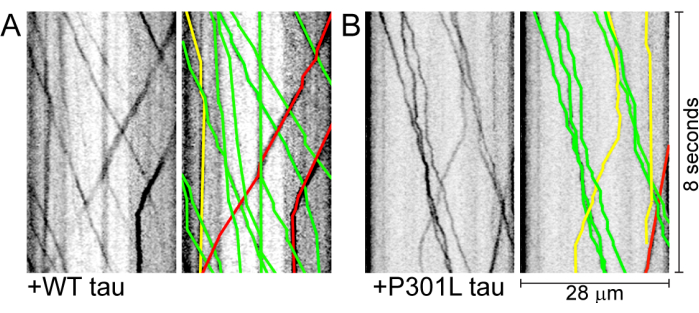
Figure 2: Kymographs generated and analyzed using the KymoAnalyzer software. (A) A kymograph of a representative transport movie from a neuron expressing mApple-synaptophysin and wild-type (WT) full-length tau (left). Tracks show cargo moving in the anterograde (green) and retrograde (red) directions, as well as those that underwent reversals (i.e., tracks containing anterograde and retrograde movements; yellow; right)12. (B) A kymograph displaying transport of mApple-synaptophysin in a neuron expressing tau with the pathological P301L mutation and tracks of the cargo after KymoAnalysis (right). Vertical scale bar = 8 s; horizontal scale bar = 28 µm of length. Abbreviation: WT = wild-type. This figure is modified from Combs et al.12. Please click here to view a larger version of this figure.
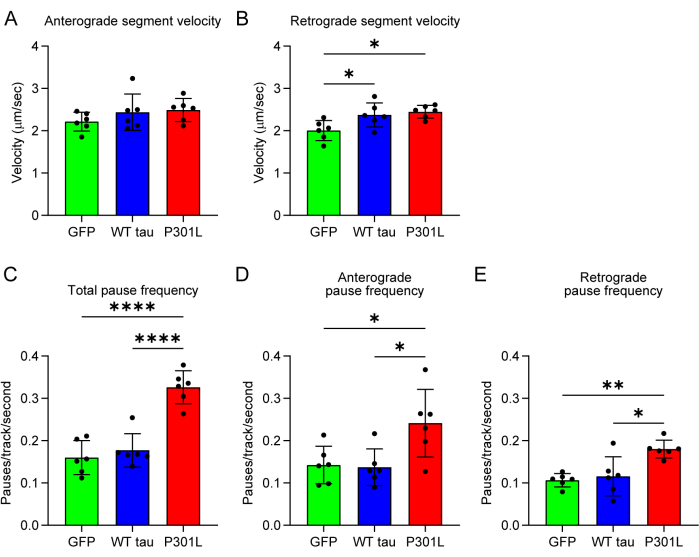
Figure 3: Sampling of parameters characterizing transport of mApple-synaptophysin in the presence of tau or GFP. (A) Anterograde segment velocity represents the rate of cargo transport when it is in motion toward the synapse. WT or P301L tau expression did not change anterograde velocity when compared to GFP expression. (B) Retrograde segment velocity increased slightly in tau-expressing neurons (WT and P301L tau) compared to GFP-expressing neurons. (C) Total pause frequency, which is defined as the number of pauses per track per second, increased with P301L tau expression compared to GFP and WT tau. Directional pause frequency can also be measured. P301L tau expression increased pause frequency in the (D) anterograde and (E) retrograde directions compared to GFP and WT tau-expressing neurons. Each data point represents an independent replicate (n = 6). The data are represented as mean ± SD. Data are analyzed using a one-way ANOVA with Tukey's multiple comparison test to compare each group. * p < 0.05, ** p ≤ 0.01, *** p ≤ 0.001, **** p ≤ 0.0001. Abbreviations: GFP = green fluorescent protein; WT = wild-type. This figure is modified from Combs et al.12. Please click here to view a larger version of this figure.
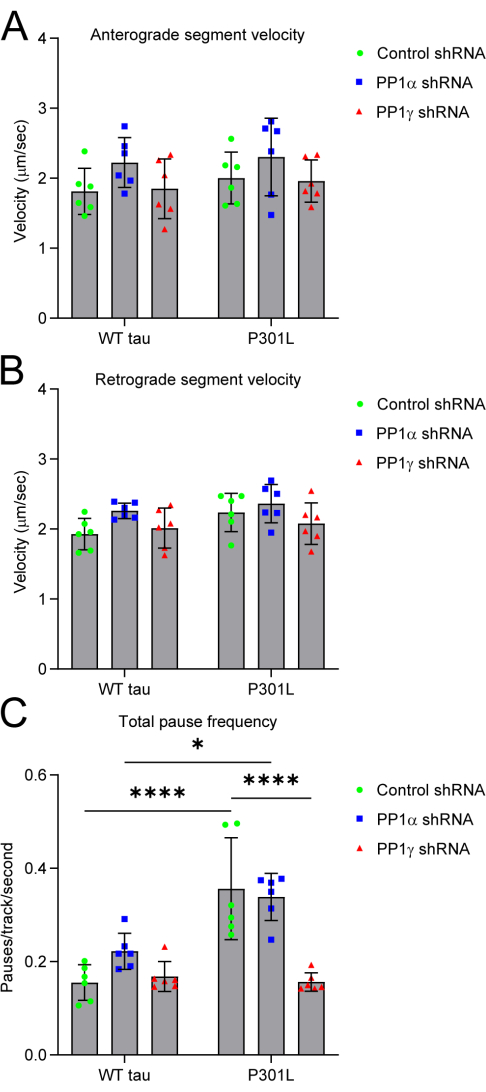
Figure 4: Knockdown of gene expression to identify molecular mechanisms of transport disruptions12. Co-transfection of neurons with isoform-specific protein phosphatase 1 shRNAs or a control shRNA was used to test the role of PP1 in P301L tau-mediated transport disruptions. (A) Anterograde and (B) retrograde segment velocities were unaffected by all shRNAs. (C) Knockdown of PP1γ, but not PP1α, rescued P301L tau-induced increases in total pause frequency. Each data point represents an independent replicate (n = 5). The data are represented as the mean ± SD. Data are analyzed using a two-way ANOVA with Tukey's multiple comparison test to compare the three tau groups within each shRNA treatment group. * p < 0.05, ** p ≤ 0.01, *** p ≤ 0.001, **** p ≤ 0.0001. Abbreviations: PP1 = protein phosphatase 1; shRNA = short hairpin RNA. This figure is modified from Combs et al.12. Please click here to view a larger version of this figure.
Discussion
There is growing evidence that multiple pathological proteins associated with a variety of neurodegenerative disorders disrupt fast axonal transport in neurons. This represents a potential common mechanism of neurotoxicity across these diseases. To better understand the process by which these proteins disrupt transport, we need tools and models that allow us to address specific questions. The method described here allows the examination of mechanisms engaged by pathological proteins to negatively affect cargo transport in primary hippocampal neurons from rodents.
Primary neurons provide a system that is easily manipulatable while still maintaining all the functional aspects of fast axonal transport regulation. Liposome-based transfections of neurons are not particularly efficient (we typically observe 5-15 transfected neurons/well) but do provide several advantages over other methods. First, the relatively low efficiency allows the user to visually follow processes projecting from an individual cell body of an isolated neuron. When used in conjunction with the live-cell marker of the AIS (i.e., neurofascin) the axon can be clearly identified for imaging. Prior work demonstrated that live-cell imaging of the AIS using this approach does not affect AIS functionality20.
Second, lipofection also allows for co-transfection of multiple DNA plasmids in nearly all cells expressing the fluorescent cargo protein. These plasmids are easily interchangeable, making it simple to prepare plasmids for multiple modified forms of a protein and compare their effects within the same cell preparations or test multiple cargo proteins. We have successfully imaged Rab proteins, amyloid precursor protein, and TrkB, among other cargo proteins. The choice of specific cargo proteins may be an important factor in experimental design. Cargo proteins can differ in the subcellular locations they are trafficked to and in their affinity for individual motor protein subunits among many other potential differences. Care should be taken to choose an appropriate cargo marker for the specific experimental questions that are being asked.
Third, lipofection also provides the user with greater flexibility to test multiple protein modifications or cargo proteins in a way that is less labor-intensive than purifying lentiviruses. However, we have also successfully adapted the protocol to use lentiviruses for use with older neurons (DIV21) that are less tolerant of lipofection. Older neurons can also be transfected with a reduced efficiency.
Other adaptations to the protocol are easily made based on the user's needs. We have successfully used rat and mouse neurons isolated from the hippocampus and cortex. Additionally, constructs that modulate the expression of endogenous genes can be added to the mix to identify mechanistic pathways involved in changes to axonal transport. For example, we successfully used shRNA-mediated gene knockdown to determine that a pathological tau-based effect was dependent on the PP1γ isoform12.
There are limitations to transfections that should be considered as well. Neurons are sensitive to lipid-based transfections and may experience some toxicity. Paying particular attention to the gentle mixing of reagents and slow pipetting can help reduce this toxicity. There are also considerations related to the relatively high and sometimes variable levels of protein expression. Care should be taken to choose cells that display enough expression to effectively detect fluorescent cargoes, but not those that are extremely weak or bright (i.e., expressing too little or too much of the exogenous proteins). The highly transfected neurons may experience toxicity due to the high levels of protein expression making it difficult to isolate protein-based effects. Additionally, these cells are more difficult to image due to excess background signal. Ideally, the investigator will utilize the same inclusion and exclusion parameters for all conditions to help maintain comparability and consistency.
Care should be taken to choose constructs that can control for the effects of overexpressing proteins. We used GFP as a non-related protein and then confirmed that overexpression of WT tau did not alter cargo transport when compared to GFP12,13. The inclusion of a control protein and the wild-type version of the protein of interest provides a robust means to identify effects induced by the pathological protein and helps to rule out protein overexpression as a cause. There is some potential that overexpressing the cargo protein may alter behavior to a certain extent, but this can be mitigated by reducing the levels of overexpression and testing other cargo proteins as well.
The KymoAnalyzer software16 was used with high success in our group, but other methods may also be used to generate and analyze kymographs from the transport movies. Additionally, there are other software methods to track cargo movement that do not use kymographs21. KymoAnalyzer provides a robust characterization of cargo transport but with some limitations due to the low throughput nature of the manual addition of tracks over the images. Other methods using automatic detection miss some critical aspects of transport such as the increased pause frequency we identified in response to pathological tau expression. However, it is important to reduce the potential introduction of biases by the user. Determine, in advance, the level of detail in assigning tracks. Too little detail misses potential pausing but too great of detail introduces noise that is beyond the level of resolution in the movies and can affect the transport rates in the final analysis. Additionally, a protocol to blind the user to experimental conditions should be included.
While a strength of this method is its adaptability to user needs, there are some critical parts of the protocol that will help ensure success. Neuron cultures should have high viability and very few clumps during the plating step. Use antibiotic-free media throughout the culturing of the neurons. For transfections, either reduced- or serum-free media can be used to dilute reagents and DNA. The transfection reagents are highly diluted when added to the wells, which also reduces any toxic effects from the transfection. Liposome/DNA mixtures should be handled and added to cultures gently to minimize toxicity to the cells. An extracellular marker of the AIS should be used to positively identify the axon to help avoid mistakenly imaging dendrites. Furthermore, conditioned media, not fresh media, from cells grown in parallel should be used during the primary and secondary antibody addition steps to limit stress to the cells. An axon selected for imaging should be isolated and relatively straight. Make sure that the depth of field is wide enough to capture the full extent of transport in the ROI (i.e., increase the pinhole size in a confocal system). This will prevent issues deciphering the traces from individual cargo on the kymograph. The direction of transport moving into the ROI must be noted, as well as the actual frame rate, as this information will be important for the accurate assignment of transport direction and velocity, respectively. Finally, low laser power should be used during movie collection to prevent phototoxicity to the cell and photobleaching of the fluorescently-tagged cargo. Highly sensitive GaAsP detectors on our system allow extremely low laser powers of ~1.5% typically. By avoiding excessive imaging of the neuron and limiting imaging to the small ROI of the axon, phototoxicity can be avoided. We are able to successfully image neurons beyond 10 min without observing phototoxicity. Again, it is important to confirm neuronal health throughout the imaging process by choosing only neurons displaying healthy transport and confirming their health by postimaging fixation and immunostaining.
This protocol provides a powerful tool to analyze the molecular mechanisms of fast axonal transport disruption in primary neurons. These disruptions are linked to multiple pathological proteins found in a variety of neurodegenerative diseases, including AD. It is easily adaptable to identify the effects of specific disease-related protein modifications, elucidate mechanistic details of these effects, and test potential therapeutic interventions.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We thank Chelsea Tiernan and Kyle Christensen for their efforts in developing and optimizing aspects of these protocols. This work was supported by National Institutes of Health (NIH) Grants R01 NS082730 (N.M.K.), R01 AG044372 (N.M.K.), R01 AG067762 (N.M.K.), and F31 AG074521 (R.L.M.); NIH/National Institute on Aging, Michigan Alzheimer's Disease Research Center Grant 5P30AG053760 (N.M.K. and B.C.); Office of the Assistant Secretary of Defense for Health Affairs through the Peer Reviewed Alzheimer's Research Program Award W81XWH-20-1-0174 (B.C.); Alzheimer's Association Research Grants 20-682085 (B.C.); and the Secchia Family Foundation (N.M.K.).
Materials
| 0.4% Trypan blue | Gibco | 15250-061 | |
| 1.7 mL microcentrifuge tubes | DOT | RN1700-GMT | |
| 2.5% trypsin | Gibco | 15090-046 | |
| 3 mL syringe with 21 G needle | Fisher | 14-826-84 | |
| 10 mL plastic syringe | Fisher | 14-823-2A | |
| 14 G needle | Fisher | 14-817-203 | |
| 15 G needle | Medline | SWD200029Z | |
| 16 G needle | Fisher | 14-817-104 | |
| 18 G needle | Fisher | 14-840-97 | |
| 22 G needle | Fisher | 14-840-90 | |
| 32% paraformaldehyde | Fisher | 50-980-495 | |
| AlexaFluor 647 goat anti-rabbit IgG (H+L) | Invitrogen | A21244 | RRID:AB_2535813 |
| Amphotericin B | Gibco | 15290-026 | |
| Arruga Micro Embryonic Capsule Forceps, Curved; 4" | Roboz | RS-5163 | autoclave |
| B-27 Supplement (50x), serum free | Gibco | A3582801 | |
| BioCoat 24-well Poly D lysine plates | Fisher | 08-774-124 | |
| boric acid | Sigma | B6768-1KG | |
| Calcium chloride | Sigma | C7902 | |
| Castroviejo 3 1/2" Long 8 x 0.15 mm Angle Sharp Scissors | Roboz | RS-5658 | autoclave |
| Cell counting device | automatic or manual | ||
| Confocal microscope with live cell chamber attachment | |||
| Confocal imaging software | |||
| D-(+)-glucose | Sigma | G7528 | |
| DNase I (Worthington) | Fisher | NC9185812 | |
| Dulbecco's Phosphate Buffered Saline | Gibco | 14200-075 | |
| EGTA | Fisher | O2783-100 | |
| Fatal-Plus Solution | Vortech Pharmaceuticals, LTD | NDC 0298-9373-68 | sodium pentobarbital; other approved methods of euthanasia may be used |
| Fetal bovine serum | Invitrogen | 16000044 | |
| Gentamicin Reagent Solution | Gibco | 15710-072 | |
| GlutaMAX | Gibco | 35050-061 | glutamine substitute |
| Hanks' Balanced Salt Solution | Gibco | 24020-117 | |
| ImageJ version 1.51n | ImageJ | Life-Line version 2017 May 30: https://imagej.net/software/fiji/downloads | |
| KymoAnalyzer (version 1.01) | Encalada Lab | Package includes all 6 macros: https://www.encalada.scripps.edu/kymoanalyzer | |
| Lipofectamine 3000 | Invitrogen | 100022050 | Use with P3000 transfection enhancer reagent |
| Magnesium chloride | Fisher | AC223211000 | |
| MES hydrate | Sigma | M8250 | |
| Micro Dissecting Scissors 3.5" Straight Sharp/Sharp | Roboz | RS-5910 | autoclave |
| Neurobasal Plus medium | Gibco | A3582901 | |
| Neurofascin (A12/18) Mouse IgG2a | UC Davis/NIH NeuroMab | 75-172 | RRID:AB_2282826; 250 ng/mL; Works in rat neurons, NOT in mouse neurons |
| Neurofascin 186 (D6G60) Rabbit IgG | Cell Signaling | 15034 | RRID:AB_2773024; 500 ng/mL; Works in mouse neurons, we have not tested in rat neurons |
| newborn calf serum | Gibco | 16010-167 | |
| Opti-MEM | Gibco | 31985-062 | |
| P3000 | Invitrogen | 100022057 | |
| Petri dish, 100 x 10 mm glass | Fisher | 08-748B | For dissection; autoclave |
| Petri dish, 100 x 20 mm glass | Fisher | 08-748D | To place uterine horns in; autoclave |
| Poly-D-lysine | Sigma | P7886-100MG | |
| Polypropylene conical centrifuge tubes (15 mL) | Fisher | 14-955-238 | |
| Polypropylene conical centrifuge tubes (50 mL) | Fisher | 14-955-238 | |
| Potassium chloride | Fisher | P217-500 | |
| Sodium acetate | Sigma | S5636 | |
| sodium borate decahydrate | VWR | MK745706 | |
| Straight-Blade Operating Scissors Blunt/Sharp | Fisher | 13-810-2 | autoclave |
| Syringe Filters, 0.22 µm | VWR | 514-1263 | |
| Thumb dressing forceps, serrated, 4.5" | Roboz | RS-8100 | autoclave |
| µ-Slide 4 Well Glass Bottom | Ibidi | 80427 |
Riferimenti
- Kneynsberg, A., Combs, B., Christensen, K., Morfini, G., Kanaan, N. M. Axonal degeneration in tauopathies: disease relevance and underlying mechanisms. Front Neurosci. 11, 572 (2017).
- Combs, B., Mueller, R. L., Morfini, G., Brady, S. T., Kanaan, N. M. Tau and axonal transport misregulation in tauopathies. Adv Exp Med Biol. 1184, 81-95 (2019).
- Brady, S. T., Morfini, G. A. Regulation of motor proteins, axonal transport deficits and adult-onset neurodegenerative diseases. Neurobiol Dis. 105, 273-282 (2017).
- Morfini, G. A., et al. Axonal transport defects in neurodegenerative diseases. J Neurosci. 29 (41), 12776-12786 (2009).
- Brady, S. T., Lasek, R. J., Allen, R. D. Fast axonal transport in extruded axoplasm from squid giant axon. Science. 218 (4577), 1129-1131 (1982).
- LaPointe, N. E., et al. The amino terminus of tau inhibits kinesin-dependent axonal transport: implications for filament toxicity. J Neurosci Res. 87 (2), 440-451 (2009).
- Kanaan, N. M., et al. Pathogenic forms of tau inhibit kinesin-dependent axonal transport through a mechanism involving activation of axonal phosphotransferases. J Neurosci. 31 (27), 9858-9868 (2011).
- Kanaan, N. M., et al. Phosphorylation in the amino terminus of tau prevents inhibition of anterograde axonal transport. Neurobiol Aging. 33 (4), 826.e15-826.e30 (2012).
- Pigino, G., et al. Disruption of fast axonal transport is a pathogenic mechanism for intraneuronal amyloid beta. Proc Natl Acad Sci U S A. 106 (14), 5907-5912 (2009).
- Morfini, G. A., et al. Pathogenic huntingtin inhibits fast axonal transport by activating JNK3 and phosphorylating kinesin. Nat Neurosci. 12 (7), 864-871 (2009).
- Morfini, G. A., et al. Inhibition of fast axonal transport by pathogenic SOD1 involves activation of p38 MAP kinase. PLoS One. 8 (6), e65235 (2013).
- Combs, B., et al. Frontotemporal lobar dementia mutant tau impairs axonal transport through a protein phosphatase 1gamma-dependent mechanism. J Neurosci. 41 (45), 9431-9451 (2021).
- Christensen, K. R., et al. Phosphomimetics at Ser199/Ser202/Thr205 in tau impairs axonal transport in rat hippocampal neurons. Mol Neurobiol. 60 (6), 3423-3438 (2023).
- Kaech, S., Banker, G. Culturing hippocampal neurons. Nat Protoc. 1 (5), 2406-2415 (2006).
- Seibenhener, M. L., Wooten, M. W. Isolation and culture of hippocampal neurons from prenatal mice. J Vis Exp. (65), 3634 (2012).
- Neumann, S., Chassefeyre, R., Campbell, G. E., Encalada, S. E. KymoAnalyzer: a software tool for the quantitative analysis of intracellular transport in neurons. Traffic. 18 (1), 71-88 (2017).
- Cox, K., et al. Analysis of isoform-specific tau aggregates suggests a common toxic mechanism involving similar pathological conformations and axonal transport inhibition. Neurobiol Aging. 47, 113-126 (2016).
- Tiernan, C. T., et al. Pseudophosphorylation of tau at S422 enhances SDS-stable dimer formation and impairs both anterograde and retrograde fast axonal transport. Exp Neurol. 283 (Pt A), 318-329 (2016).
- Mueller, R. L., et al. Tau: a signaling hub protein. Front Mol Neurosci. 14, 647054 (2021).
- Hedstrom, K. L., Ogawa, Y., Rasband, M. N. AnkyrinG is required for maintenance of the axon initial segment and neuronal polarity. J Cell Biol. 183 (4), 635-640 (2008).
- Basu, H., Schwarz, T. L. QuoVadoPro, an autonomous tool for measuring intracellular dynamics using temporal variance. Curr Protoc Cell Biol. 87 (1), e108 (2020).

.
