LPS and ATP-induced Death of PMA-differentiated THP-1 Macrophages and its Validation
Summary
This protocol is based on LPS and ATP-induced death of PMA-differentiated THP-1 macrophages. We use flow cytometry to analyze Annexin V and 7-AAD double staining to detect cell death, using the whole cell and employing scanning electron microscopy to observe the cell membrane morphology.
Abstract
Cell death is a fundamental process in all living organisms. The protocol establishes a lipopolysaccharide (LPS) and adenosine triphosphate (ATP)-induced phorbol-12-myristate-13-acetate (PMA)-differentiated lipid deposition in human monocyte (THP-1) macrophage model to observe cell death. LPS combined with ATP is a classic inflammatory induction method, often used to study pyroptosis, but apoptosis and necroptosis also respond to stimulation by LPS/ATP. Under normal circumstances, phosphatidylserine is only localized in the inner leaflet of the plasma membrane. However, in the early stages of pyroptosis, apoptosis, and necroptosis, the cell membrane remains intact and exposed to phosphatidylserine, and in the later stages, the cell membrane loses its integrity. Here, flow cytometry was used to analyze Annexin V and 7-Aminoactinomycin D (AAD) double staining to detect the cell death from the whole cells. The results show that substantial cells died after stimulation with LPS/ATP. Using scanning electron microscopy, we observe the possible forms of cell death in individual cells. The results indicate that cells may undergo pyroptosis, apoptosis, or necroptosis after stimulation with LPS/ATP. This protocol focuses on observing the death of macrophages after stimulation with LPS/ATP. The results showed that cell death after LPS and ATP stimulation is not limited to pyroptosis and that apoptosis and necrotic apoptosis can also occur, helping researchers better understand cell death after LPS and ATP stimulation and choose a better experimental method.
Introduction
Cell death is a fundamental physiological process in all living organisms. In recent years, substantial studies have shown that cell death is involved in the immunity and balance within the organism. Studying cell death helps us better understand the onset and development of diseases. Several forms of programmed cell death have been described, and some key targets in these processes have been identified. Pyroptosis, apoptosis, and necroptosis are the three genetically defined programmed cell death pathways involved in internal balance and disease1.
Pyroptosis is characterized by the formation of membrane pores and the release of cell contents. Activated caspases or granzymes cleave gasdermins to separate their N-terminal domains, which then oligomerize, bind to the membrane, and form pores2,3,4. The gasdermin pore provides an atypical secretion channel across cellular membranes, resulting in downstream cell responses, including content release and ion influx2,3,4. Ultimately, the cells eventually experience plasma membrane rupture and pyroptotic lysis facilitated by ninjurin-15. In apoptosis, activated Bax and Bak form oligomers on the mitochondrial outer membrane and release cytochrome C, which is regulated by a balance between proapoptotic and antiapoptotic proteins of the BCL-2 family, initiator caspases (caspase-8, -9 and -10) and effector caspases (caspase-3, -6 and -7)1,6,7. The morphological changes of apoptosis include membrane blebbing, cell shrinkage, nuclear fragmentation, chromatin condensation, and apoptotic body formation6,8. The receptor-interacting serine/threonine-protein kinase 1 (RIPK3) and mixed-lineage kinase domain-like (MLKL) are two downstream core components of the necroptotic mechanism1. RIPK3 recruits and phosphorylates MLKL, p-MLKL oligomerizes, associates with the cell membrane, initiates membrane perforations, causes ion influx, increases intracellular osmolarity, and eventually cell rupture6,9. Gasdermins and MLKL bind to the plasma membrane and mediate pyroptosis and necroptosis, respectively, while BAX/BAK mediates apoptosis by binding to the outer membrane of mitochondria6.
Although each pathway has specific mechanisms and outcomes, they lead to similar changes on the cell membrane. Under normal circumstances, phosphatidylserine (PS) is only localized in the inner leaflet of the plasma membrane. However, in the early stages of pyroptosis, apoptosis, and necroptosis, PS will be exposed, outside of the plasma membrane. Caspase-3/caspase-7 activates TMEM16 and XKR families, which leads to an asymmetrical cell membrane and externalizes PS during apoptosis10. Gasdermin D-mediated and MLKL-mediated Ca2+ influx leads to loss of the symmetry of the phospholipid bilayer on the cell membrane and the exposure of PS. The exposure occurs before the loss of cell membrane integrity11,12. Based on the similar changes in the membrane of these three types of programmed cell death, we use flow cytometry to analyze Annexin V/7-Aminoactinomycin D (7-AAD) double staining to detect cell death. Annexin V, a calcium-dependent phospholipid-binding protein, has a high affinity for PS, which can serve as a sensitive probe to detect exposed PS on the surface of the plasma membrane13. 7-AAD is a nucleic acid stain that cannot pass through the entire cell membrane. It is similar to propidium iodide (PI), a commonly used nucleic acid dye. They have similar fluorescence characteristics, but 7-AAD has a narrower emission spectrum and less interference with other detection channels. Owing to these similarities, it is not sufficient to distinguish between pyroptosis, apoptosis, and necroptosis. We used flow cytometry to detect the cell death from whole cells. A second method is used to capture the cell membrane using scanning electron microscopy (SEM) to observe the possible forms of cell death in individual cells.
We established a lipopolysaccharide (LPS) and adenosine triphosphate (ATP)-induced phorbol-12-myristate-13-acetate (PMA)-differentiated lipid deposition in human monocyte (THP-1) macrophage model to observe cell death. This protocol focuses on observing cell death rather than investigating mechanisms.
Protocol
1. Cell line and cell culture
- Grow the human monocytic cell line THP-1 in RPMI-1640 complete culture medium with 10% fetal bovine serum, 1% penicillin-streptomycin and 0.05 mM β-mercaptoethanol. Culture the cells at 37 °C in 5% CO2 humidified air. Sub-culture cells every 2-3 days.
NOTE: THP-1 is a suspension cell, select to change half of the medium for sub-culturing. When observing many cell fragments under the light microscope, centrifuge at 300 x g for 3 min at room temperature. Resuspend cells in the pre-warmed fresh complete cell culture medium. - Use cells (in passages 4-10) at the logarithmic growth phase for all experiments. When the cells are in good condition and logarithmic growth phase, they are round, bright, dense and without clumps and cell fragments under the light microscope at 100 x.
2. Differentiation of THP-1 cells
- Prepare a stock solution of PMA with a concentration of 1 mM (dissolve 1 mg of PMA in 1.62 mL of dimethyl sulfoxide). Dilute the PMA stock solution withcomplete cell culture medium to a final working concentration of 100 nM.
- Collect the cells in the culture dish into a 10 mL sterile centrifuge tube. Centrifuge at 300 x g for 3 min at room temperature, resuspend cells with 3 mL of complete culture medium containing PMA.
- Open the cell counter and the counting software. Pipette 20 µL of cell suspension to the cell counter board and insert it into the cell counter board. Click Preview B1, turn the knob at the bottom right of the instrument, and adjust the focal distance to make the cells with bright center with clear contours on the software. Click Count to start counting.
- Based on the counting results, use complete culture medium containing PMA to adjust a cell density to 1 x 106 cells/mL. Seed cells in a 6 well plate, incubate the plate for 24 h at 37 °C and 5% CO2 before starting further treatment.
3. Treatment of THP-1cells
- Prepare a stock solution of LPS with a concentration of 1 mg/mL (dissolve 1 mg of LPS in 1 mL of phosphate-buffered saline (PBS)). Dilute the LPS stock solution with serum-free medium to a final working concentration of 1 µg/mL. Prepare a stock solution of Na2ATP with a concentration of 500 mM (dissolve 0.3026 g of ATP in 1 mL of PBS).
- Aspirate the PMA-containing medium from wells and wash with PBS 1x. Add serum-free medium to the control group and serum-free medium containing LPS to the model group. Incubate the plate for 6 h at 37 °C + 5% CO2 before proceeding with further treatment.
- After 6 h, add the ATP stock solution to the model group to a final working concentration of 500 nM. Incubate the plate for 45 min at 37 °C + 5% CO2 before proceeding.
4. Flow cytometry analysis (Method 1)
- Preparation of experimental samples
- Seed and incubate cells in 6-well plates according to step 2. Establish four control groups with no treatment: one no-staining control, two single-staining controls (one for each stain), and one double-staining control. Establish one treatment group and treat according to step 3.
- After treatment, aspirate all the supernatant from the wells and wash with PBS 2x. Add 500 µL of 0.05% Trypsin (non-EDTA) to each well, and incubate in the incubator for 30 s.
- Add 1 mL of RPMI-1640 complete culture medium to each well to stop cell detaching and collect the cells into the 5 mL tube. Centrifuge at 300 x g for 3 min at room temperature.
- Add 1 mL of PBS to resuspend cells. Place tubes with cells in a water bath at 37 °C for 3 min. Centrifuge all samples at 300 x g for 3 min at room temperature.
- Dilute 200 µL of 5x binding buffer with 800 µL of distilled water to 1x binding buffer. Add 100 µL of 1x binding buffer to resuspend cells and transfer the cells into the 5 mL tube.
- Add 5 µL of Annexin V-PE solution to one tube of control group cells for 10 min and 5 µL of 7-AAD solution to another tube of control group cells for 5 min (as single staining controls separately). Gently vortex each tube to mix dye and incubate the samples at room temperature protected from light. Add 400 µL of PBS to each tube and vortex gently to terminate incubation.
- Add 5 µL of Annexin V-PE solution to the remaining tubes and incubate for 5 min at room temperature. Gently vortex each tube to mix dye and incubate the samples at room temperature protected from light. After 5 min, add 5 µL of 7-AAD solution for 5 min at room temperature. Add 400 µL of PBS to each tube and vortex gently to terminate the incubation.
- Filter the cell suspension using a 35 µm nylon mesh that are part of the 5 mL polystyrene round-bottom tubes. Place the tube on ice and wait for testing. Gently mix before testing and then insert it into the instrument.
- Flow cytometry
- Use fluorescence-activated cell sorting flow cytometer for testing. Open the flow cytometer data analysis software and confirm the performance of the detector and laser. Click the Cytometer and choose Fluidics startup, wait for the system to be ready. Exclude flow chamber bubbles by clicking on Cytometer > Clean Modes > De-gas Flow cell.
- Click Experiment, select in sequence New folder, Experiment, Specimen, Tube and Edit Name. A pentagonal shaped arrow in front of the tube, turns green when light up.
- Click on the icon of Cytometer FACSCelesta (a shortcut key icon), select Parameter, and then select FSC, SSC and PE signal. Click on the icon of Global Worksheet (a shortcut key icon) to establish PE histogram. For the flow cytometer used in this study, use PE for Annexin V at 586 nm and plot on the X-axis; PerCP-Cy5.5 for 7-AAD at 700 nm and plot on the Y-axis.
- Insert unstained control sample first into the instrument. Click on the icon of Acquisition Dashboard (a shortcut key icon), acquire a minimum of 10,000 events from the desired population. Exclude debris using a gate (P1) on an FSC-A and SSC-A dot plot. Adjust flow rate to Middle speed and Record Data. Adjust voltage to place cell population on the diagonal of the FSC/SSC histogram and click Restart. Adjust flow rate to Middle speed and Record Data.
- Click Next tube, run single staining controls of Annexin V and 7-AAD to adjust compensation. Run the remaining tubes and collect data.
5. SEM imaging (Method 2)
- Preparation of experimental samples
- Seed and incubate cells in 6-well plates according to step 2 and treat according to step 3.
- After treatment, aspirate all the supernatant from the wells and wash 2x with PBS. Add 500 µL of 0.05% Trypsin (non-EDTA) to each well, and incubate in the incubator for 30 s.
- Add 1 mL of RPMI-1640 complete culture medium to each well to stop cell detaching and collect the cells into the 5 mL tube. Centrifuge at 300 x g for 3 min at room temperature.
- Fix cells with 500 µL of electron microscope fixative (2.5% glutaric dialdehyde, 100 mM phosphorous salts) for 2 h at room temperature, followed by overnight at 4 °C.
- Prepare chromium alum solution: Add 1.0 g of gelatin to 100 mL of ultrapure water, 70 °C water-bath heating, and stir evenly. Add 0.05 g of chromium alum, stir evenly and filter with filter paper. Dip the clean cover glass in chromium alum solution for 2 h at 37 °C and dry it in a 37 °C oven for later use.
NOTE: The purpose is to prevent cell detachment during the experiment. - Collect cells from fixed solution. Centrifuge at 300 x g for 3 min at room temperature. Add 100 µL of PBS and gently blow away the cells. Drop the cell suspension onto the cover glass, allow to stand for 5 min. Aspirate all the supernatant and wash 3x with PBS for 5 min each time.
NOTE: The operation must be performed gently to prevent cell detachment. Do not shake vigorously, move slowly, and add liquid slowly along the wall. - Add 500 μL of 1% osmic acid fixation for 1 hour. Aspirate all the supernatant and wash 3x with PBS for 5 min each time.
- Dehydrate the samples successively in a series of ethanol (EtOH) concentrations (30%, 50%, 70%, 80%, 90%, 95%, 100%, 100%) for 15 min per EtOH solution.
NOTE: The cover glasses can be kept in 100% EtOH for several days at 4 °C but it should dry as soon as possible to avoid excessive dehydration. - Switch on the critical point dryer, open the CO2 tank, cool the chamber to 10 °C. Quickly wrap the sample with filter paper when the chamber pressure is 0 psi and put it into the chamber.
- Open the inlet valve, observe through CO2 observation window, raise the liquid CO2 level between the two red lines (Do not exceed the upper limit), close the inlet valve. Soak the sample for 10 min. Open the inlet and exhaust valves simultaneously to equalize the CO2 for 1 min (liquid CO2 displaces EtOH). Close the exhaust valve to raise the liquid CO2 level to the specified position, close the inlet valve.
- Set the temperature to 35 °C and pressure at 1250 psi. Once the temperature and pressure stabilize, depressurize at 100 psi/min. Take out the sample when the chamber pressure is zero and switch off the instrument.
- Mount specimens on SEM aluminum specimen holders using conductive tape. Employ a sputter coater tocover the samples with a layer (2-5 nm) of gold.
- Imaging
- Use high resolution cold field emission SEM for imaging. Set the accelerating voltage of SEM to 3 kV. Set the working distance to 10 mm. Set the magnification to 1,000x to observe the panorama. Set the magnification to 5,000x and 20,000x to locate the plasma membrane and image the sample.
NOTE: The process of visualizing PMA-differentiated THP-1 macrophages via SEM is similar to other types of cells and tissues and depends on the instruments used. The reference14 provides a detailed SEM protocol with accompanying videos.
- Use high resolution cold field emission SEM for imaging. Set the accelerating voltage of SEM to 3 kV. Set the working distance to 10 mm. Set the magnification to 1,000x to observe the panorama. Set the magnification to 5,000x and 20,000x to locate the plasma membrane and image the sample.
Representative Results
The cell samples were treated as described in the protocol and flow cytometry detection was done. Normal cells cannot be stained with Annexin V and 7-AAD (Annexin V-/7-AAD-). In the early stages of pyroptosis, apoptosis, and necroptosis, PS was exposed and bound to Annexin V, but the cell membrane was still intact and excluded 7-AAD from the extracellular space (Annexin V+/7-AAD-). In the later stages, the cell membrane loses its integrity, cells are simultaneously stained by Annexin V and 7-AAD, showing double positive results (Annexin V+/7-AAD+). The necrotic cells were only stained with 7-AAD (Annexin V-/7-AAD+). Figure 1 shows that after LPS/ATP stimulation, the cells in the Q2 region increased significantly, indicating cell death.
We use SEM to capture the plasma membrane surface, directly observing the characteristics of membrane from a single cell. Figure 2A shows the control cells, Figure 2B-D show the cells after LPS/ATP stimulation. We observed cells with characteristics of pyroptosis, including membrane blebbing and rupture membrane (Figure 2B), cells with apoptotic characteristics, including membrane blebbing and cell shrinkage (Figure 2C), and cells with necroptosis characteristics, including cell swelling and rupture membrane (Figure 2D).
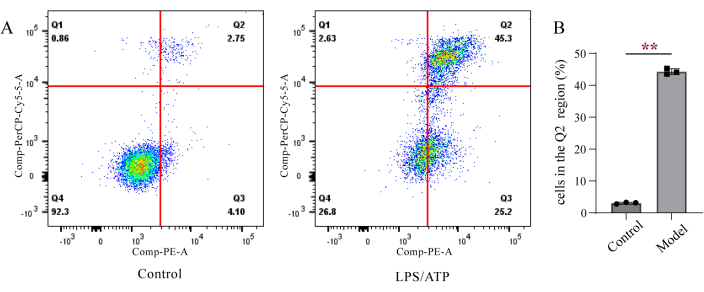
Figure 1: Detection of Annexin V/7-AAD double staining using flow cytometry. (A) Annexin V negative and 7-AAD negative staining (Q4), Annexin V positive and 7-AAD negative double staining (Q3), Annexin V positive and 7-AAD positive staining (Q2), and Annexin V negative and 7-AAD positive staining (Q1). (B) Quantitative data for (A). Data are presented as mean ± SD (n=3), unpaired t test, **p < 0.01. Please click here to view a larger version of this figure.
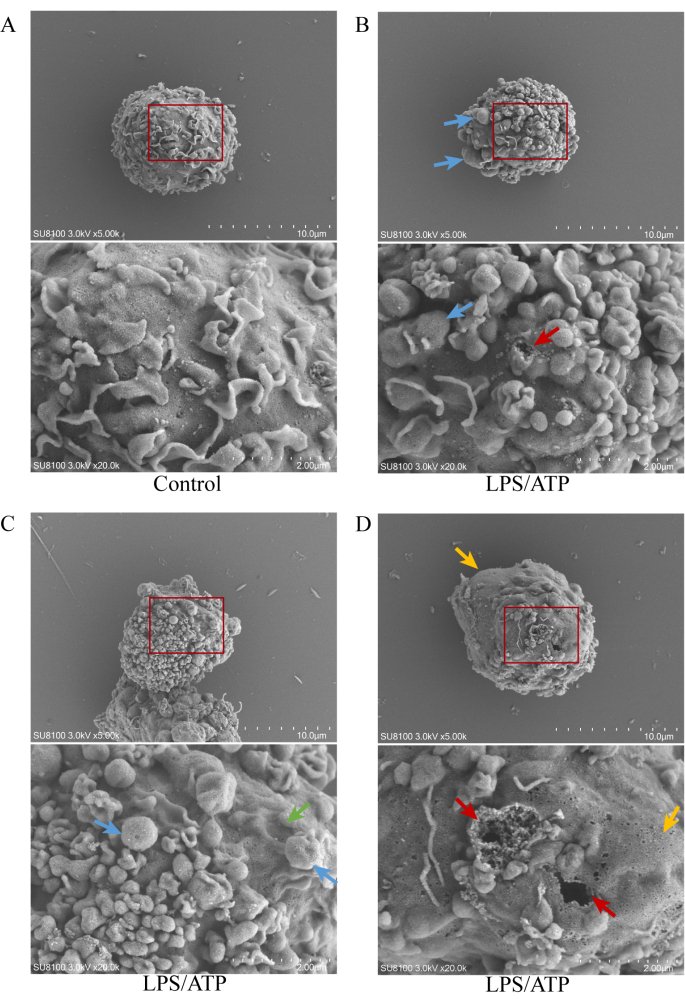
Figure 2: Scanning electron microscopy images of PMA-differentiated THP-1 macrophages. (A) Control group, 5,000x/20,000x magnification. (B, C, D) LPS/ATP group show membrane blebbing, rupture of membrane (↑), cell swelling (↑), and cell shrinkage (↑) at 5,000x/20,000x magnification. Please click here to view a larger version of this figure.
Discussion
In this manuscript, two methods were used to detect LPS and ATP-induced death of PMA-differentiated THP-1 macrophages. Annexin V/7-AAD double staining was used, and the results were analyzed by flow cytometry from overall staining. As with other flow cytometric analysis, a group of unstained cells and two groups of single-stained cells were set up to exclude false positive and false negative results. The results show that after LPS/ATP stimulation, several cells lost membrane integrity, indicating that cell death may have occurred. This detection method allows us to evaluate the cell death from whole cells. It is worth noting that the Annexin V/7-AAD double staining is not sufficient to distinguish between pyroptosis, apoptosis, and necroptosis. In the early stages of these three forms, PS will be exposed outside the plasma membrane, and Annexin V staining is positive. The integrity of membrane is lost during the late stage of these three forms, and the staining results show double-positive staining. Therefore, we used SEM to further observe the possible forms of cell death that might occur.
In apoptosis, BAX/BAK pore formation on the mitochondrial outer membrane, subsequent cell shrinkage and cell membrane blebbing occurs. Necroptosis employs MLKL to open pores in the cell membrane, while pyroptosis signal activates gasdermin to form pores in the cell membrane. It is worth noting that both pyroptosis and necroptosis are accompanied by cell membrane rupture in the final stage. However, gasedermin pores are much larger than MLKL pores, so gasdermin pores do not have ion selectivity. Necroptotic cells exhibit significant swelling in morphology15. They ultimately end in membrane rupture. Regardless of the signaling pathway upstream of cell membrane permeability activation, changes in the cell membrane accompany these three forms of cell death. We used SEM to further observe the cell membrane, which provide an intuitive way to examine cell death. We observed the characteristics of pyroptosis, including membrane blebbing and the rupture membrane, and necroptosis, including cell swelling and the rupture membrane, consistent with characteristics studied by previous researchers15. We observed membrane blebbing and cell shrinkage, consistent with the characteristics of apoptosis studied by previous researchers16.
In previous studies, the combination of LPS and ATP was often used to induce pyroptosis. Interestingly, we observed features of apoptosis and necroptosis. Although all three forms may occur in LPS/ATP-induced cell death, previous studies have shown that pyroptosis remains the dominant mode of death. LPS is one of the most characterized pathogen-associated molecular patterns. ATP released from dying cells, has a high concentration within the cell. Extracellular ATP is one of the danger-associated molecular patterns17. LPS/ATP is a classic two-step stimulation method to induce pyroptosis. Research has shown that the addition of ATP to LPS-stimulated cells can rapidly activate caspase-118 while silencing the caspase-1 gene can significantly reverse LPS/ATP-induced cell death19. In previous studies, we have also found LPS/ATP-induced activation of apoptotic and necroptotic target proteins20,21. Although LPS/ATP is a classic pyroptosis inducer, it may also induce apoptosis and necroptosis.
In fact, these three forms often do not exist independently, and crosstalk between signal stages. For example, gasdermin proteins target the mitochondria to promote cytochrome c release to enhance the mitochondrial apoptotic pathway22. Activation of caspase-3 can cleave gasdermin E and then trigger pyroptosis23. A study suggests that MLKL/PIPK3 signaling is involved in the activation of NLRP3 inflammasomes24. This complex cross-talk led to the concept of PANoptosis, defined as an inflammatory programmed cell death pathway regulated by the PANoptosome complex with key features of pyroptosis, apoptosis, and/or necroptosis that cannot be accounted for by any of these programmed cell death pathways alone25. As the concept of PANoptosis was proposed, scientists described the occurrence of programmed cell death more comprehensively, this also highlights the complexity of programmed cell death. Meanwhile, this also indicates the limitations of this research method. Although pyroptosis is considered dominant in LPS/ATP-induced cell death, one cannot determine the proportion of these death forms or whether they all exist, even involving other forms of programmed death. This can be an area to investigate in the future.
This protocol is based on LPS and ATP-induced death of macrophages. We have described a method of testing from the whole to the individual, which is also applicable to other models and cells. The results indicate that cells may have undergone pyroptosis, apoptosis, or necroptosis, which is not limited to pyroptosis, helping researchers better understand cell death after LPS and ATP stimulation and choose a better experimental method. In previous studies, Annexin V/7-AAD or Annexin V/PI double staining was commonly used to indicate the occurrence of apoptosis26, 27, some researchers have also used it to study apoptosis28. In fact, we cannot distinguish between the three types of cell death through these double staining. This protocol reminds researchers, especially beginners, that Annexin V/7-AAD or Annexin V/PI double staining cannot qualitative the type of cell death. It can only show cell damage and death as a whole or indicate that the cell may have undergone some form of death. In more targeted research, it is necessary to introduce a control group that inhibits other deaths to show that conclusively program death is at hand. Similarly, further mechanistic research will investigate the critical targets of different programmed deaths.
In addition, during the preparation of the experiment, we found that there are currently many detailed experimental procedures for SEM of adherent cells, with little description of suspended cells. Here, we provide a more detailed description of the sample preparation method for suspended cells. Step 5.5 is a necessary step to assist in immobilizing suspended cells onto cell slides. Subsequent operations must be performed gently to prevent cell detachment. In addition to the chromium alum solution, poly-L-lysine can also immobilize cells.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We express our great appreciation to Jiayi Sun and Lu Yang at Innovative Institute of Chinese Medicine and Pharmacy, Chengdu University of Traditional Chinese Medicine, for the assistance with flow cytometry, Cuiping Chen at State Key Laboratory of Southwestern Chinese Medicine Resources, for the help with scanning electron microscopy. This work was supported by the National Natural Science Foundation of China [82104491], the Natural Science Foundation of Sichuan [2023NSFSC0674], and the Post-doctoral Science Foundation of China [2021M693789].
Materials
| 0.25% pancreatic enzyme solution (excluding EDTA) | BOSTER Biological Technology co.ltd | PYG0068 | |
| 5 mL Polystyrene Round-Bottom Tube | CORNING | 352235 | |
| 5 mL centrifuge tube | Labgic Technology Co., Ltd. | BS-50-M | |
| 6-well plate | Sorfa Life Science Research Co.,Ltd | 220100 | |
| Annexin V-PE/7-AAD apoptosis analysis kit | Absin (Shanghai) Biological Technology co.ltd | abs50007 | Annexin V-PE, 7-AAD, 5×Binding buffer, Apoptosis Positive Control Solution |
| celculture CO2 incubator | Esco (Shanghai) Enterprise Development Co., Ltd. | N/A | |
| cell culture dish, 100 mm | Sorfa Life Science Research Co.,Ltd | 230301 | |
| Cellometer K2 Fluorescent Cell Counter | Nexcelom Bioscience LLC | Cellometer K2 | |
| Cellometer SD100 Counting Chambers | Nexcelom Bioscience LLC | CHT4-SD100-002 | |
| centrifuge machine | Hunan Xiangyi Laboratory Instrument Development Co., Ltd | L530 | |
| chromium alum | Guangdong Wengjiang Chemical Reagent Co., Ltd. | PA04354 | |
| cover glasses, 9 mm | Labgic Technology Co., Ltd. | BS-09-RC | |
| critical point dryer | Quorum Technologies | K850 | |
| dimethyl sulfoxide | BOSTER Biological Technology co.ltd | PYG0040 | |
| electron microscope fixative | Servicebio Technology co.ltd | G1102 | 2.5% glutaric dialdehyde, 100 mM phosphorous salts |
| electronic balance | SHIMADZU | ATX124 | |
| ethanol absolute | Chengdu Kelong Chemical Co., Ltd | 2021033102 | |
| flow cytometer | Becton,Dickinson and Company | FACSCanto  |
|
| flow cytometry analysis software | Becton,Dickinson and Company | BD FACSDivaTM Software | |
| gelatin | Guangdong Wengjiang Chemical Reagent Co., Ltd. | PA00256 | |
| High resolution cold field emission scanning electron microscope | TITACHI | Regulus 8100 | |
| human monocytic cell line THP-1 | Procell Life Science&Technology Co.,Ltd. | CL0233 | |
| inverted microscope | Leica Microsystems Co., Ltd | DMi1 | |
| IR Vortex Mixer | VELP Scientifica Srl | ZX4 | |
| lipopolysaccharide | Beijing Solarbio Science & Technology Co.,Ltd. | L8880 | LPS is derived from Escherichia coli 055:B5 |
| Na2ATP | Beijing Solarbio Science & Technology Co.,Ltd. | A8270 | |
| phorbol-12-myristate-13-acetate | Beijing Solarbio Science & Technology Co.,Ltd. | P6741 | |
| phosphate-buffered saline | Servicebio Technology co.ltd | G4202 | |
| Pipette | Eppendorf AG | N/A | |
| pipette tips, 10 μL | Servicebio Technology co.ltd | T-10PL | |
| pipette tips, 1 mL | Servicebio Technology co.ltd | T-1250L | |
| pipette tips, 200 μL | Servicebio Technology co.ltd | T-200L | |
| RPMI-1640 complete culture media | Procell Life Science&Technology Co.,Ltd. | CM0233 | RPMI-1640 + 10% FBS + 0.05mM β-mercaptoethanol + 1% P/S |
| RPMI-1640 culture media | Shanghai BasalMedia Technologies Co., LTD. | K211104 | |
| sheath fluid | BECKMAN COULTER | 8546733 | |
| sputter coater | Cressington Scientific Instruments Ltd | 108 | |
| thermostatic water bath | GUOHUA Electric Appliance CO.,Ltd | HH-1 |
Riferimenti
- Bertheloot, D., Latz, E., Franklin, B. S. Necroptosis, pyroptosis and apoptosis: an intricate game of cell death. Cell Mol Immunol. 18 (5), 1106-1121 (2021).
- Rao, Z., et al. Pyroptosis in inflammatory diseases and cancer. Theranostics. 12 (9), 4310-4329 (2022).
- Huston, H. C., Anderson, M. J., Fink, S. L. Pyroptosis and the cellular consequences of gasdermin pores. Semin Immunol. 69, 101803 (2023).
- Kovacs, S. B., Miao, E. A. Gasdermins: Effectors of pyroptosis. Trend Cell Biol. 27 (9), 673-684 (2017).
- Kayagaki, N., et al. NINJ1 mediates plasma membrane rupture during lytic cell death. Nature. 591 (7848), 131-136 (2021).
- Ketelut-Carneiro, N., Fitzgerald, K. A. Apoptosis, pyroptosis, and necroptosis-Oh my! The many ways a cell can die. J Mol Biol. 434 (4), 167378 (2022).
- Kakarla, R., Hur, J., Kim, Y. J., Kim, J., Chwae, Y. J. Apoptotic cell-derived exosomes: messages from dying cells. Exp Mol Med. 52 (1), 1-6 (2020).
- Imre, G. Cell death signalling in virus infection. Cell Signal. 76, 109772 (2020).
- Zhan, C., Huang, M., Yang, X., Hou, J. MLKL: Functions beyond serving as the Executioner of Necroptosis. Theranostics. 11 (10), 4759-4769 (2021).
- Lemke, G. How macrophages deal with death. Nat Rev Immunol. 19 (9), 539-549 (2019).
- Yang, X., et al. Bacterial endotoxin activates the coagulation cascade through gasdermin D-dependent phosphatidylserine exposure. Immunity. 51 (6), 983-996 (2019).
- Gong, Y. N., et al. ESCRT-III acts downstream of MLKL to regulate necroptotic cell death and its consequences. Cell. 169 (2), 286-300 (2017).
- Dong, H. P., et al. Evaluation of cell surface expression of phosphatidylserine in ovarian carcinoma effusions using the annexin-V/7-AAD assay: clinical relevance and comparison with other apoptosis parameters. Am J Clin Pathol. 132 (5), 756-762 (2009).
- JoVE, Scanning Electron Microscopy (SEM), JoVE Science Education Database, Analytical Chemistry. JoVE. , (2024).
- Chen, X., et al. Pyroptosis is driven by non-selective gasdermin-D pore and its morphology is different from MLKL channel-mediated necroptosis. Cell Res. 26 (9), 1007-1020 (2016).
- Herr, D. R., et al. Ultrastructural characteristics of DHA-induced pyroptosis. Neuromol Med. 22 (2), 293-303 (2020).
- Xie, Q., et al. Lipopolysaccharide/adenosine triphosphate induces IL-1β and IL-18 secretion through the NLRP3 inflammasome in RAW264.7 murine macrophage cells. Int Mol Med. 34 (1), 341-349 (2014).
- Gurung, P., et al. FADD and caspase-8 mediate priming and activation of the canonical and noncanonical Nlrp3 inflammasomes. J Immunol. 192 (4), 1835-1846 (2014).
- Liu, W., et al. Ablation of caspase-1 protects against TBI-induced pyroptosis in vitro and in vivo. J Neuroinflam. 15 (1), 48 (2018).
- Wang, S. H., et al. GSK-3β-mediated activation of NLRP3 inflammasome leads to pyroptosis and apoptosis of rat cardiomyocytes and fibroblasts. Euro J Pharmacol. 920, 174830 (2022).
- Chen, J., et al. RIP3 dependent NLRP3 inflammasome activation is implicated in acute lung injury in mice. J Transl Med. 16 (1), 233 (2018).
- Rogers, C., et al. Gasdermin pores permeabilize mitochondria to augment caspase-3 activation during apoptosis and inflammasome activation. Nat Comm. 10 (1), 1689 (2019).
- Wang, Y., et al. Chemotherapy drugs induce pyroptosis through caspase-3 cleavage of a gasdermin. Nature. 547 (7661), 99-103 (2017).
- Kang, S., et al. Caspase-8 scaffolding function and MLKL regulate NLRP3 inflammasome activation downstream of TLR3. Nat Comm. 6, 7515 (2015).
- Wang, Y., Kanneganti, T. D. From pyroptosis, apoptosis and necroptosis to PANoptosis: A mechanistic compendium of programmed cell death pathways. Comput Str Biotechnol J. 19, 4641-4657 (2021).
- Hou, Y., et al. Rhodiola crenulata alleviates hypobaric hypoxia-induced brain injury by maintaining BBB integrity and balancing energy metabolism dysfunction. Phytomedicine. 128, 155529 (2024).
- Shuwen, H., et al. Cholesterol induction in CD8+ T cell exhaustion in colorectal cancer via the regulation of endoplasmic reticulum-mitochondria contact sites. Cancer Immunol Immunother. 72 (12), 4441-4456 (2023).
- Luo, X., et al. Cyclophosphamide induced intestinal injury is alleviated by blocking the TLR9/caspase3/GSDME mediated intestinal epithelium pyroptosis. Int Immunopharmacol. 119, 110244 (2023).

