Profiling Luminal pH in Three-Dimensional Gastrointestinal Organoids Using Microelectrodes
Summary
The present protocol describes pH measurements in human tissue-derived gastric organoids using microelectrodes for spatiotemporal characterization of intraluminal physiology.
Abstract
The optimization and detailed characterization of gastrointestinal organoid models require advanced methods for analyzing their luminal environments. This paper presents a highly reproducible method for the precise measurement of pH within the lumina of 3D human gastric organoids via micromanipulator-controlled microelectrodes. The pH microelectrodes are commercially available and consist of beveled glass tips of 25 µm in diameter. For measurements, the pH microelectrode is advanced into the lumen of an organoid (>200 µm) that is suspended in Matrigel, while a reference electrode rests submerged in the surrounding medium in the culture plate.
Using such microelectrodes to profile organoids derived from the human gastric body, we demonstrate that luminal pH is relatively consistent within each culture well at ~7.7 ± 0.037 and that continuous measurements can be obtained for a minimum of 15 min. In some larger organoids, the measurements revealed a pH gradient between the epithelial surface and the lumen, suggesting that pH measurements in organoids can be achieved with high spatial resolution. In a previous study, microelectrodes were successfully used to measure luminal oxygen concentrations in organoids, demonstrating the versatility of this method for organoid analyses. In summary, this protocol describes an important tool for the functional characterization of the complex luminal space within 3D organoids.
Introduction
Organoids-miniature multicellular structures derived from stem cells-have revolutionized our ability to study human physiology and are starting to replace animal models, even in regulatory settings1. Since the initial description of intestinal organoids by Sato et al. in 2009, organoid technology has become immensely popular2. A large number of studies have characterized the cellular composition and function of organoid models in great detail3,4,5,6. However, the luminal space of these 3D multicellular structures remains largely undefined7,8. The lumen is the central cavity of organoids derived from mucosal tissues that is surrounded by the apical portions of polarized epithelial cells. Since cellular secretion and absorption predominantly occur at the apical epithelial surface, the luminal microenvironment of organoids is controlled by these important physiological processes. Currently used organoid models demonstrate variations in cell signaling patterns, overall stemness, metabolite concentration gradients, and environmental conditions9. Understanding organoid luminal physiology is therefore necessary for the accurate modeling of organ function and pathology. Unfortunately, the relative inaccessibility of the lumen significantly hinders functional analyses of luminal physiology in 3D organoids10.
The ability to examine pH profiles is especially important in the stomach, which is notorious for having the steepest proton gradient in the body, ranging from approximately 1-3 in the lumen, to near neutral at the epithelium11,12,13.There remains a significant gap in our understanding of the microscale maintenance of the gastric pH gradient, and the relevance of organoid models in recapitulating this dynamic milieu across the gastric mucus layer. Traditional approaches for the analysis of organoid pH have involved the use of pH-sensitive dyes, which can be fluorescent or colorimetric indicators. McCracken et al. used a luminal injection of SNARF-5F-a ratiometric pH indicator-into organoids to analyze a drop in luminal pH in response to histamine treatment. Such dyes can be incorporated into the culture media, allowing for real-time, non-invasive monitoring of pH. Not only do pH-sensitive dyes require complex calibration steps that contribute to poor reliability and accuracy with measurements, but such dyes also tend to operate within specific detection ranges that may not be representative of the full pH range within the microenvironment of interest14,15. It could be considered reasonable, however, to use indicator dyes for confirmatory experiments. Optical nanosensors that use fluorescent optode-based, pH-sensing approaches have also been developed; however, such sensing techniques require microscopic imaging and are also susceptible to photobleaching, phototoxicity, as well as imaging bias16,17. Additionally, Brooks et al. have 3D-printed multiwell plates containing microelectrodes on top of which organoids may be plated18. This approach, however, does not allow for measurements directly inside the organoid lumen.
Electrode-based pH measurements can achieve improved accuracy compared to other methods, as well as provide real-time pH monitoring. In addition, pH electrodes mounted on micromanipulators allow for superior spatial resolution of pH measurements as the precise location of the electrode tip can be finely controlled. This enables the highest possible flexibility and reproducibility in the analyses of organoid models. The electrodes used here are miniaturized pH microelectrodes that operate based on the diffusion of protons through selective pH glass that surrounds a thin platinum wire. The microelectrode is connected to an external Ag-AgCl reference electrode and then connected to a high-impedance millivolt meter. The electrical potential between the two electrode tips when submerged in the same solution will reflect the pH of the solution19. Such microprofiling systems have been used in the metabolic analysis of biofilms20,21, planktonic algae22, human sputum samples23, and even in mesenchymal stem cell spheroids24. Both our lab and Murphy et al. have previously used micromanipulator-controlled O2 microelectrodes to evaluate the oxygen concentrations in the luminal spaces of organoids. Murphy et al. paired this method with mathematical modeling to reveal an oxygen gradient within their spheroids. Our group was able to find reduced luminal oxygen levels in tissue-derived gastric organoids compared to the surrounding extracellular matrix25.
Here, we provide a detailed method for the manual microelectrode profiling of the luminal pH in spherical gastrointestinal tract organoids that will enable enhanced physiological understanding of their complex luminal microenvironment. We anticipate that this technique will add a new dimension to the exploration of organoid physiology through real-time, high-resolution measurements of pH levels at a microscale. Furthermore, the following protocol could be easily adapted for the analysis of O2, N2O, H2, NO, H2S, redox, and temperature in various types of organoid models. Physiological profiling serves as a valuable tool for optimizing organoid culture conditions to better mimic in vivo environments, thereby enhancing the relevance and utility of organoid models in biomedical research.
Protocol
This protocol requires 3D organoids of at least 200 µm in diameter that have a distinct lumen and that are embedded in an artificial extracellular matrix (ECM, e.g., Matrigel). Human gastric tissues for organoid derivation were obtained with approval from the Institutional Review Board of Montana State University and informed consent from patients undergoing upper endoscopy at Bozeman Health (protocol # 2023-48-FCR, to D.B.) or as exempt whole stomach or sleeve gastrectomy specimens from the National Disease Research Interchange (protocol #DB062615-EX). Information about the organoid lines and passage numbers used for this study is provided in Table 1, and the media composition is listed in Table 2. Refer to previously published protocols for the generation and maintenance of gastrointestinal organoid lines6,26,27.
1. Preparation of human gastric organoids for pH profiling
- Initiate and maintain gastric organoid cultures using standard protocols. Maintain organoids on 24-well plates in 500 µL of expansion media per well (Table 2). Passage established organoid lines every 5-7 days, transferring to a 35 mm glass-bottom dish in preparation for pH profiling experiments.
NOTE: Organoid cultures for our experiments are derived from gastric gland preparations, which are obtained from adult tissue as described above. We use a collagenase tissue digestion method to isolate these glands before they are suspended in ECM, as previously described25,28,29. - From actively growing organoid cultures at passages 1-15, select wells that contain at least 100 organoids (approximately 2 million cells) with diameters between 200 and 700 µm for transfer and expansion on 3.5 mm Petri dishes.
- While preparing organoids for plating (steps 1.4-1.8), allow extracellular matrix (ECM) aliquot(s) to thaw on ice for at least 45 min. Maintain ECM on ice throughout this protocol to prevent gelation. Prewarm cell culture plates by placing them in a 37 °C, 5% CO2 incubator.
- Remove the media from the wells and harvest the gastric organoid cultures by pipetting ice-cold PBS onto each ECM droplet and scratching the gel with the tip of a P1000 pipette. Pipette the PBS with the ECM fragments containing organoids into a 15 mL conical tube.
- Centrifuge the tube at 200 × g at 4 °C for 5 min. ECM fragments containing the organoids will be visible as a layer at the bottom of the tube. Carefully aspirate the supernatant and pipette 350 µL of 0.25% trypsin-EDTA into each tube, mixing gently by pipetting up and down. Incubate the tubes in a 37 °C water bath for 2-5 min.
- Following incubation with trypsin-EDTA, add 600 µL of ice-cold DMEM with penicillin/streptomycin to each tube and vigorously pipette up and down at least 40x. Centrifuge at 200 × g at 4 °C for 5 min. Aspirate the supernatant. To set up cultures for pH measurements, resuspend the cell pellet in ice-cold, liquid ECM at a 1:4 v/v ratio of organoid pellet to ECM for plating.
- For each sample, plate 40 µL of liquid ECM containing organoids/organoid fragments in a thin horizontal line along the diameter of a 35 mm glass-bottom dish.
NOTE: Dispensing the gel onto the plate in a line instead of a round droplet enables easier access to individual organoids by thinning out the culture. - Allow the gel to polymerize for 15-30 min; then, carefully add 2 mL of organoid expansion media to the plate by pipetting along the edge of the well to avoid disturbing the gel.
- Replace media every other day. Allow 4-8 days for a minimum of 10 organoids to grow to a minimum diameter of 400 µm.
- To proceed with a pH profiling experiment, ensure that each culture contains at least 10 organoids that meet the following criteria: >200 µm diameter, healthy appearance (little to no dark material visible within organoids observed with a brightfield microscope), and not overcrowded by other organoids.
2. Unpacking and calibration of microelectrodes
NOTE: To enable microscale measurements, a separate reference electrode is used in addition to the pH sensor microelectrode rather than using an integrated (and hence bulkier) design. Both pH microelectrode and reference electrode must be stored wet. Do not allow exposure to air for more than 10 min at a time. Determine the appropriate tip size for the application. Here, we used a potentiometric pH microelectrode with a beveled tip diameter of 25 µm.
- With the microelectrode still in its protection tube, visually inspect the tip for any damage.
- Connect the reference electrode to the pH electrode cable via the connector. Then, connect the microelectrode to the amplifier and the amplifier to a grounded PC with the software via a USB cable (Figure 1A).
- Fill two 50 mL conical tubes 2/3 with 70% ethanol and deionized H2O (diH2O). Place the microelectrode and the reference electrode in the diH2O tube, ensuring that both tips appear submerged at least 1 cm in the liquid.
NOTE: Tall beakers can also be used for this and similar steps. - Allow the electrodes to equilibrate for ~10 min.
NOTE: The procedure can also be paused at this step and resumed later as the electrodes may be stored in diH2O. - While the electrodes are equilibrating, open the software (red icon) on the computer workstation. Under the Settings tab, ensure that the box next to the microelectrode is checked, indicating that it is properly connected and recognized by the software. Click Start Experiment on the top left of the window and enter your desired file name and destination. In the Sensor calibration & experiment settings window, click Clear all points to prepare the software for a new calibration.
- Fill two 50 mL conical tubes 2/3 with pH 4.01 and pH 9.21 calibration buffers. Gently blot the protective tubing and the reference electrode dry with a delicate laboratory wipe.
- Starting with the electrodes in the pH 4.01 buffer, enter 4.01 as the known pH value. Once the mV reading has stabilized, select add point. Confirm that the signal is stable at ~380 mV. After the point has been added, place both electrodes back in diH2O to rinse, then blot dry again.
- Repeat the previous step with the 9.21 buffer and click add point when the signal is stable (~83 mV). Check that the microelectrodes respond linearly between pH 4.01 and 9.21; therefore, only a two-point calibration curve is necessary. Ensure that the resulting line between these points has a negative slope of 50-70 mV/pH-unit. Click Save & use calibration.
NOTE: You are now ready to begin recording measurements30.
3. pH profiling of human gastric organoids
NOTE: The following protocol is described for a right-handed user. CAUTION: Disable all power-saving options on your PC as ongoing measurements will be disrupted if the PC enters sleep mode.
- Assemble a ring stand with a clamp on the left-hand side of a stereomicroscope to hold the reference electrode. Position a micromanipulator attached to a heavy lab stand on the right side of the microscope (Figure 1A).
- Carefully remove the microelectrode from its protection case by laying it flat on the bench and pulling the case off in a swift, quick motion.
CAUTION: The microelectrodes are incredibly fragile and care should be taken to ensure that the tip does not come into contact with stiff, solid material. For best results, perform measurements on a sturdy benchtop free of equipment that could cause vibration of the bench or unwanted movement of the electrodes. - Mount the microelectrode on a micromanipulator and arrange the stereoscope and micromanipulator in such a way that the microelectrode may advance toward the culture dish without hitting the objective or other parts of the microscope.
- Position the culture dish containing the organoids to have adequate visualization of the first organoid that is to be profiled (Figure 1B).
- Lightly secure the reference electrode to the clamp on the ring stand to the left of the stereoscope. Position the reference electrode so that it rests in the medium surrounding the ECM. Take care to ensure that it will not disrupt the organoids as the stage is moved around between measurements.
- Looking directly at the culture plate (not into the microscope), advance the tip of the microelectrode until it is sufficiently submerged in the media. Under the Data logger tab in the software, click the Start button (single triangle pointing to the right) to begin recording. Ensure that the Calibrated checkbox is selected on the right side of the screen.
NOTE: Allow ~10 s for each reading to stabilize before advancing to the next region. - Before entering the ECM, ensure that the electrode tip is visible under the microscope and that it is positioned to advance linearly toward the first organoid of interest. Slowly advance the electrode into the gel, without making contact with any organoids. Record at least three pH readings and calculate the average.
- Position the microelectrode so that it is prepared to advance toward the first organoid of interest along the axis perpendicular to the surface. Allow the electrode to make a minor indentation on the epithelial surface without penetrating (Figure 1C).
NOTE: This step will also provide insight into whether the organoid will remain in place or whether it may move about more freely in the ECM. - With one swift motion, advance the microelectrode into the organoid. If the organoid moves around the microelectrode or moves away, attempt a slightly different angle, or back it up against another organoid or the plastic rim surrounding the coverglass bottom of the dish. Measure the luminal pH (three individual times) of 10 different organoids for each experimental condition. When finished recording measurements, click the square Stop button.
CAUTION: Estimate the diameter of the organoid to decide how far to advance the electrode into the organoid lumen, taking care not to pierce through it. For increased accuracy, measure the diameter with your microscope's camera software, if available. NOTE: If the tip of the microelectrode becomes noticeably coated with debris following one or multiple measurements, wash the microelectrode in cell dissociation solution, PBS, EtOH, and then diH2O before proceeding.
4. Motorized profiling (optional)
NOTE: This option requires a micromanipulator that is mounted on a mechanical motor stage, which is ultimately controlled by computer software via a motor controller31.
- Open the Profiling software (brown icon) and start a new experiment under the Profiling tab.
- The settings for the z-axis will automatically recognize Motor when a motor apparatus is properly connected.
NOTE: Movement in the x and y directions is still controlled manually with this setup. - Locate the Profile interaction tab30. Before hitting start, ensure that it is possible to quickly transition to looking at the microscope eyepiece to ensure organoid entry at the desired distance. Go to Profile settings and do the following:
- Indicate that the Start distance is 0 µm.
CAUTION: If the epithelial shell is particularly tough on a given organoid, the organoid may be pushed away or deformed instead of penetrated. If pushing is the case, continue advancing the electrode but note the point at which entry occurs, as the software cannot record this automatically. - Judging the size of the organoid being profiled, indicate an End distance that is no more than ¾ way through an organoid's estimated diameter.
NOTE: This particular step is designed to prevent damage to the electrode tip. - Indicate the desired step size—100 µm as shown in Figure 2E. Ensure that the minimum step size matches the size of the electrode tip (e.g., 25 µm for a 25 µm pH-25 electrode tip).
- Indicate a safe position to which the motor will return the electrode tip between profiles.
NOTE: This should be a comfortable height above the sample (outside of the organoid) where the sensor can be safely moved sideways in the x- and y-directions with the manual micromanipulator. Leave the Sensor angle at its default setting. - To allow the electrode to equilibrate, indicate at least 3 s under Wait before measure(s).
- Set the Measure period(s) to at least 1 s. The measurements over this period will be averaged.
NOTE: If performing measurements in an environment high in electrical noise, it may be helpful to indicate a longer period. - Set the Delay between(s) to at least 1 s.
- Set the preferred number of Replicates to be measured at each depth.
- Begin recording measurements by hitting the Start button.
- Indicate that the Start distance is 0 µm.
5. Cleaning of electrodes
- Place the electrode to be cleaned back in its protection tube.
- Flush the electrodes with diH2O after measurements.
- Flush the electrodes with 70% ethanol for a couple of minutes.
- Rinse the electrodes with pH 4.01 buffer.
- Rinse the electrodes once more with diH2O before proceeding with measurements.
6. Storage of electrodes
NOTE: Both electrodes are to be stored at room temperature in a low-activity location, safe from accidental damage.
- After using the pH microelectrode, carefully slide it horizontally back into its protection tube (sliding along the lab bench for horizontal support).
- Holding the microelectrode upright (tip pointed upward), gently fill the protection tube with sterile water. Plug the protection tube with the stopper the electrode came with. Ensure that any holes in the protection tube are sealed with electrical tape. Store in a shatter-proof box until the next use.
NOTE: It is recommended to use the original box that the electrodes came in as it will contain protective inserts designed to keep electrodes in place when stored. - Store the reference electrode in a beaker or graduated cylinder filled with a 3M KCl solution. Cover the beaker/graduated cylinder with parafilm to prevent evaporation of the solution.
NOTE: If using a graduated cylinder, it is recommended to secure it to the lab bench with tape to prevent accidental movement.
7. Methyl red injection (optional)
NOTE: Methyl red is a colorimetric indicator dye that can be used to validate the microelectrode measurements.
- Backfill a 2 µL glass capillary with sterile mineral oil, load it onto a micromanipulator-controlled nL autoinjector, and subsequently fill it with a solution containing 0.02% methyl red and 150 mM HCl.
NOTE: The solution should appear red/pink while in the capillary due to the acidity of the HCl. - Inject organoids of at least 300 µm in diameter with 9.2 µL of the solution25.
- Capture images or video using a camera adapted to the stereomicroscope (Figure 2G).
Representative Results
Secretion of acid is a crucial function of the human stomach. However, to what extent acid secretion can be modeled in organoids is still a matter of debate6,32,33,34. We therefore developed the protocol detailed above to accurately measure acid production in gastric organoids. Notably, we used unstimulated adult stem cell-derived organoids cultured under standard expansion conditions that had been passaged several times, which led to parietal cell loss35. Therefore, the presence of acid-secreting parietal cells and active acid release was not expected in our model system.
For our experiments, we used organoids with diameters between 200 µm and 1,000 µm seeded on glass-bottom dishes. First, we tested two different tip sizes for the microelectrodes-a pH-25 with a tip diameter of 25 µm and a pH-50 with a tip diameter of 50 µm. As shown in Figure 2A, there was no significant difference between measurements obtained with the smaller compared to the larger tips. Interestingly, the baseline pH in the organoids tended to be slightly alkaline at ~pH 8. Using the 25 µm pH-25 tip, we next assessed the variation in luminal pH within different ten organoids maintained on the same culture plate, with three measurements obtained from each organoid (Figure 2B). Within one plate, the luminal pH of individual organoids showed only slight, non-significant variations (8.16 ± 0.12; p ≥0.0445-0.99) (Figure 2B). We also compared intraluminal pH measurements within five different organoid lines with passages ranging from 3 to 16 (Figure 2C). Again, we found consistent pH measurements within each culture but significant variability between organoid lines. However, luminal pH generally remained between pH 7.3 and pH 8.2, within one order of magnitude, and there was no apparent trend for average pH when comparing early to late passage numbers (Figure 2C). We next asked whether the pH of the organoid lumen was directly related to the pH of the organoid expansion media and extracellular matrix (ECM). Comparison between the media, the ECM surrounding the organoids, and the organoid lumen revealed significant differences in pH, with the luminal pH of the organoids lower than that of the ECM, and the ECM pH lower than that of the surrounding organoid expansion media (Figure 2D), suggesting that the luminal pH of the organoids was physiologically relevant rather than directly determined by the culture environment. Across six independent experiments, we measured an average luminal pH that was near neutral at 7.13 ± 0.09.
A motorized micromanipulator was used to obtain pH measurements of the organoid lumen with greater spatial resolution. This approach is relevant if pH or other gradients within the mucus-filled organoid lumen29 need to be recorded. Figure 2E shows two representative series of pH measurements in large organoids (>1,000 µm diameter), demonstrating the entry point into each organoid and ending at a depth of ~800 µm. As the two organoids profiled did not have the same diameter, the measurements are not immediately comparable. Regardless, we show evidence of a slight pH gradient of Δ0.6 between the epithelial surface and the deeper organoid lumen (Figure 2E). To determine the feasibility of performing pH measurements in the organoids over time, which would enable measurements of treatment responses in real time, we recorded the intraluminal pH inside a representative organoid for approximately 20 min and found that the reading remained highly consistent after an initial adaptation period (Figure 2F). To validate the luminal pH measurements with an independent method, we used a pH-sensitive colorimetric dye (methyl red) that we injected into the organoid lumen using a micromanipulator-mounted nanoliter autoinjector (Figure 2G). The yellow coloring of the dye confirmed that the organoid lumen had a pH >6.2, consistent with the microelectrode measurements that showed a near-neutral pH. Overall, these representative results illustrate the feasibility and reproducibility of microelectrode-based pH measurements in organoid cultures.
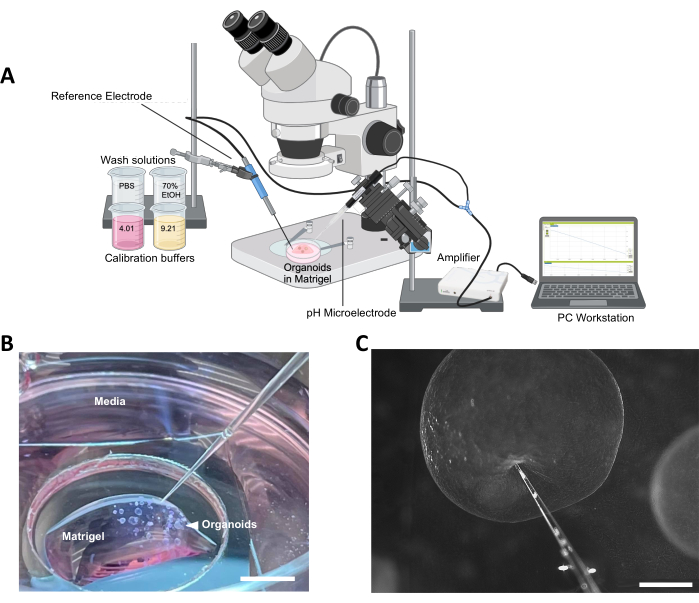
Figure 1: Overview of the method. (A) Schematic diagram of profiling setup. The microelectrode in Figure 1A was taken from the Unisense website.36 (B) Representative image of organoid culture ready for profiling, surrounding ECM, and surrounding media (scale bar = 5 mm). (C) Example of correct microelectrode positioning in preparation for organoid probing (scale bar = 500 µm). Abbreviation: ECM = extracellular matrix. Please click here to view a larger version of this figure.
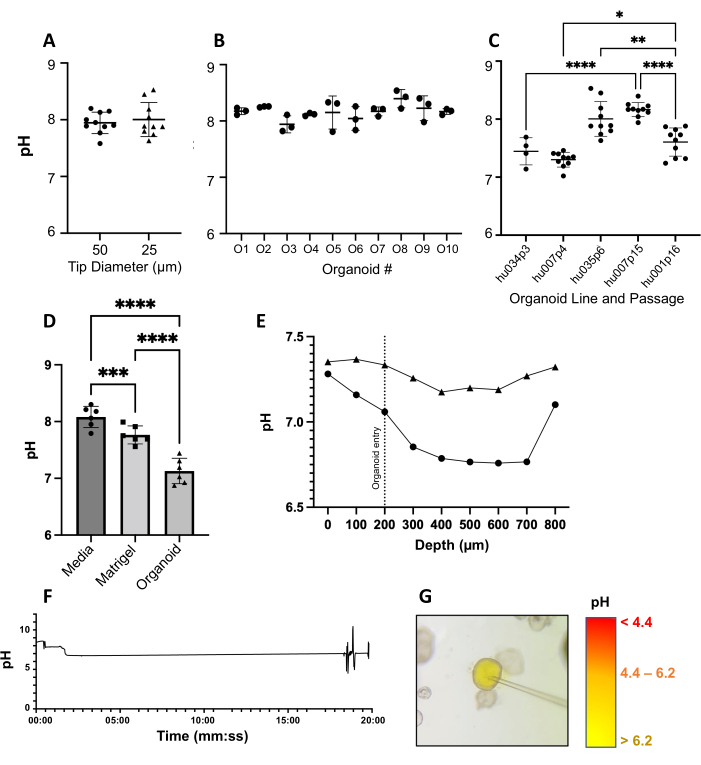
Figure 2: Validation of microsensor profiling in human gastric organoids. (A) Comparison of mid-lumen pH measurements using 25 µm (pH-25) and 50 µm (pH-50) microelectrode tips. Data from 10 individual organoids, each from a single representative experiment. (B) Three replicate pH measurements were obtained for 10 individual organoids. One-way ANOVA with multiple comparisons (p = 0.1229). (C) pH measurements obtained with a pH-25 electrode are consistent within each of five organoid lines analyzed at different passage numbers; 4-10 organoids measured per line based on size and availability. One-way ANOVA with multiple comparisons (p < 0.0001). (D) pH measurements within gastric organoids and the surrounding Matrigel and media (n = 6 independent experiments). Measurements obtained by penetrating gastric epithelial organoids with a pH-25. Each data point is the mean of 10 individual pH measurements within a single organoid. One-way ANOVA (p < 0.0001). (E) Epithelial-to-lumen pH profiles of two representative organoids using a motorized micromanipulator. (F) Stability of pH over time (~19.8 min) in one representative organoid. (G) Microinjection of methyl red pH indicator dye and HCl into the gastric organoid lumen. Please click here to view a larger version of this figure.
| Donor | |||||
| Figure | Line | Passage | Sex | Age | Ethnicity |
| 2A | 35 | 6 | F | 40 | B |
| 2B | 7 | 15 | F | 26 | B |
| 7 | 4 | F | 26 | B | |
| 1 | 16 | F | 45 | C | |
| 34 | 3 | F | 44 | Unknown | |
| 35 | 6 | F | 40 | B | |
| 2C | 7 | 15 | F | 26 | B |
| 2D | 36 | 1 | Unknown | ||
| 7 | 5 | F | 26 | B | |
| 37 | 1 | Unknown | |||
| 31 | 3 | F | 45 | H | |
| 7 | 9 | F | 26 | B | |
| 7 | 4 | F | 26 | B | |
| 2E | 10 | 15 | F | 43 | Unknown |
| 2F | 34 | 3 | F | 33 | C |
| 2G | 7 | 5 | F | 26 | B |
Table 1.
| Human Gastric Organoid Expansion Medium (L-WRN) | |
| L-WRN conditioned medium | 50% |
| Advanced DMEM/F12 | 37% |
| Fetal bovine serum | 10% |
| Penicillin/streptomycin | 1% |
| L-glutamine | 1% |
| Gentamycin | 0.10% |
| Amphotericin B | 0.10% |
| HEPES buffer | 0.40% |
| Y-27632 | 0.10% |
| SB-431542 | 0.10% |
Table 2.
Supplemental Figure S1: Ion transporter expression in human gastric organoids. Relative copy numbers normalized to 18S rRNA. Please click here to download this File.
Supplemental Figure S2: Human gastric organoid with deformed architecture following puncture with the pH microsensor probe. Scale bar = 1 mm. Please click here to download this File.
Supplemental Video S1: Unsuccessful profiling attempt in a gastric organoid culture. The 25 µm pH microelectrode is manually advanced toward the organoid of interest. After a failed attempt due to an angle misjudgment, a different angle is attempted and it is revealed that the organoid lacks the structural integrity to be profiled (spherical structure is not sturdy and deforms upon penetration with the electrode). The user then redirects to attempt a different organoid. Please click here to download this File.
Discussion
Limited access to the luminal space of organoids has severely restricted our understanding of the physiological dynamics of this microenvironment. A reliable tool for functional analyses of luminal physiology will expand our ability to leverage organoids as in vitro models for physiology, pharmacology, and disease research. Organoids are highly tunable, physiologically relevant models with the added potential to replicate genetic variability within the human population. Existing methods for pH measurement inside the organoid lumen have used pH-sensitive dyes or nanoparticles, methods that often must be coupled with fluorescence microscopy or spectroscopy16,17,37. The described microsensor-based profiling method provides a new route for measuring the intraluminal pH of gastrointestinal organoids with new spatial precision. Our method demonstrates consistent and reliable measurement of gastrointestinal organoid luminal pH, as well as evidence of a pH gradient. In unpublished experiments, we confirmed the expression of SLC family genes SLC4A2 and 26A7, which are known to be responsible for bicarbonate secretion as well as SLC9A3, 9A4, and 9A8 that are responsible for proton exchange (Supplemental Figure S1)38,39,40,41. Our studies suggest a predominance of bicarbonate secretion and a lack of parietal cells in our organoids, which may explain the alkalinity.
It is important to ensure that the microelectrodes used are well-suited for organoid microprofiling – this includes selecting the correct tip diameter. We tried both 25 µm and 50 µm tips and obtained similar results (Figure 2A) but decided to move forward with the smaller tip size per the manufacturer's advice since it was expected to be less invasive and provide a higher spatial resolution, as measurements are averaged across the beveled tip area. As a downside, however, smaller tips are more fragile. Organoid size must be considered in determining the desired spatial resolution.
A critical step in the protocol was ensuring the ability to distinguish between the organoid intraluminal pH from the pH in the ECM, and subsequently, in the surrounding culture medium. One would expect a reasonable amount of metabolite flux into and out of the organoids42. We expect an organoid's pH to be influenced by its environment and found that the organoids were consistently less alkaline than their surroundings in six independent experiments (Figure 2D). In addition, stability is just as important in profiling as it is during sensor calibration.
Tissue-derived organoids are expected to exhibit a certain degree of donor-to-donor variability, so it is essential to profile any organoid line at baseline before applying any experimental interventions such as drug treatment43. We hypothesize that fluctuations in pH within any one organoid culture may be due to local ion gradients at the microscale and the heterogeneous distribution of luminal mucus as determined by immunofluorescence and immunohistochemical staining (unpublished data). The observed variations in pH between individual organoid lines and passages may be due to individual differences in cell composition and secretory activity of the organoids. We also found that some organoid lines were inherently more difficult to penetrate than others. Similarly, some may lose their structural integrity and collapse upon electrode entry (Supplemental Figure S2 and Supplemental Video S1). This does not seem to be associated with organoid passage but has been observed most commonly in organoids over ~1.5 mm in diameter. In a previous study, we showed that the mucus inside the organoids was heterogeneously distributed29. Because of this heterogeneity, it becomes difficult to determine whether (or the extent to which) mucus distribution has impacted a measurement.
Microsensors are minimally invasive tools that can profile organoid intraluminal physiology with high speed and high spatial resolution. Such sensors also do not disturb chemical gradients and are minimally sensitive to turbulence in the microenvironment of interest. The small design is made possible by separating the sensor electrode from the reference electrode-standard laboratory pH probes combine these two electrodes, leading to a larger size. Optical pH and O2 nanosensors for intracellular measurements have been developed by the Cash group at the Colorado School of Mines and have been successful in measuring metabolites in both mouse and biofilm models. Because of the heterogeneity in the organoid lumen, however, such sensors may lead to uninterpretable results17. McCracken et al. analyzed luminal pH in embryonic stem cell-derived gastric organoids by injecting SNARF-5F and visualizing the pH-sensitive dye with real-time confocal microscopy37. Notably, measurements reported by McCracken were highly similar to the pH values obtained in our current study (Figure 2B,C). Our technique could perhaps be applied in a monolayer architecture, with more of a vertical setup as in biofilm profiling, though this may pose a risk of damaging the electrodes44. Future studies also could involve sequential measurements of pH over multiple days to assess changes associated with cell development. Since medium-sized organoids generally recover and heal after probing, one organoid could theoretically be probed multiple times as long as the sterility of the cultures can be maintained. As we refine our understanding of pH dynamics in organoid models, spatial resolution must be a priority for the precise mapping of the complex and historically inaccessible 3D microenvironment within.
In summary, this protocol provides a detailed method for accurate measurements of pH with high spatiotemporal resolution using pH microelectrodes mounted on a micromanipulator and a stereomicroscope. This method was validated with human adult stem cell-derived organoids and is suitable for epithelial organoids of at least 200 µm in diameter that have a distinct lumen. We anticipate that by selecting alternative microelectrodes, this method can be easily adapted to other types of organoids and the measurement of alternative compounds, such as NO or O2.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
The authors would like to acknowledge Dr. Ellen Lauchnor, Dr. Phil Stewart, and Bengisu Kilic for their previous work and assistance with the O2 microsensors; Andy Sebrell for training in organoid culture and micromanipulation; Lexi Burcham for assistance in organoid culture, media preparation, data recording, and organization; and Dr. Susy Kohout for general advice in electrophysiology. We would like to thank Dr. Heidi Smith for her assistance with imaging and acknowledge the Center for Biofilm Engineering Bioimaging Facility at Montana State University, which is supported by funding from the National Science Foundation MRI Program (2018562), the M.J. Murdock Charitable Trust (202016116), the US Department of Defense (77369LSRIP & W911NF1910288), and by the Montana Nanotechnology Facility (an NNCI member supported by NSF Grant ECCS-2025391).
Special thanks to the entire Unisense team who made this work possible, especially Dr. Andrew Cerskus, Dr. Laura Woods, Dr. Lars Larsen, Dr. Tage Dalsgaard, Dr. Line Daugaard, Dr. Karen Maegaard, and Mette Gammelgaard. Funding for our study was provided by the National Institutes of Health grants R01 GM13140801 (D.B., R.B.) and UL1 TR002319 (K.N.L), and a Research Expansion Award from the Montana State University Office for Research and Economic Development (D.B.). Figure 1A was created with BioRender.
Materials
| 3 M KCl | Unisense | ||
| 5 mL Wobble-not Serological Pipet, Individually Wrapped, Paper/Plastic, Bag, Sterile | CellTreat | 229091B | |
| 10 mL Wobble-not Serological Pipet, Individually Wrapped, Paper/Plastic, Bag, Sterile | CellTreat | 229092B | |
| 15 mL Centrifuge Tube – Foam Rack, Sterile | CellTreat | 229412 | |
| 24 Well Tissue Culture Plate, Sterile | CellTreat | 229124 | |
| 25 mL Wobble-not Serological Pipet, Individually Wrapped, Paper/Plastic, Bag, Sterile | CellTreat | 229093B | |
| 35 mm Dish | No. 1.5 Coverslip | 20 mm Glass Diameter | Uncoated | MatTek | P35G-1.5-20-C | |
| 50 mL Centrifuge Tube – Foam Rack, Sterile | CellTreat | 229422 | |
| 70% Ethanol | BP82031GAL | BP82031GAL | |
| 70 μm Cell Strainer, Individually Wrapped, Sterile | CellTreat | 229483 | |
| 1,000 µL Extended Length Low Retention Pipette Tips, Racked, Sterile | CellTreat | 229037 | |
| Amphotericin B (Fungizone) Solution | HyClone Laboratories, Inc | SV30078.01 | |
| Biosafety Cabinet | Nuaire | NU-425-600 | Class II Type A/B3 |
| Bovine Serum Albumin | Fisher Bioreagents | BP1605-100 | |
| Cell recovery solution | Corning | 354253 | Cell dissociation solution |
| DMEM/F-12 (Advanced DMEM) | Gibco | 12-491-015 | |
| Dulbecco's Modification of Eagles Medium (DMEM) | Fisher Scientific | 15017CV | |
| Fetal Bovine Serum | HyClone Laboratories, Inc | SH30088 | |
| G418 Sulfate | Corning | 30-234-CR | |
| Gentamycin sulfate | IBI Scientific | IB02030 | |
| HEPES, Free Acid | Cytiva | SH30237.01 | |
| HP Pavillion 2-in-1 14" Laptop Intel Core i3 | HP | M03840-001 | |
| Hydrochloric acid | Fisher Scientific | A144C-212 | |
| Incubator | Fisher Scientific | 11676604 | |
| iPhone 12 camera | Apple | ||
| L-glutamine | Cytiva | SH3003401 | |
| Large Kimberly-Clark Professional Kimtech Science Kimwipes Delicate Task Wipers, 1-Ply | Fisher Scientific | 34133 | |
| M 205 FA Stereomicroscope | Leica | ||
| Matrigel Membrane Matrix 354234 | Corning | CB-40234 | |
| MC-1 UniMotor Controller | Unisense | ||
| Methyl red | |||
| MM33 Micromanipulator | Marzhauser Wetzlar | 61-42-113-0000 | Right handed |
| MS-15 Motorized Stage | Unisense | ||
| Nanoject-II | Drummond | 3-000-204 | nanoliter autoinjector |
| Penicillin/Streptomycin (10,000 U/mL) | Gibco | 15-140-148 | |
| pH Microelectrodes | Unisense | 50-109158, 25-203452, 25-205272, 25-111626, 25-109160 | SensorTrace software is not compatible with Apple computers |
| Reference Electrode | Unisense | REF-RM-001652 | SensorTrace software is not compatible with Apple computers |
| SB 431542 | Tocris Bioscience | 16-141-0 | |
| Smartphone Camera Adapter | Gosky | ||
| Specifications Laboratory Stand LS | Unisense | LS-009238 | |
| Trypsin-EDTA 0.025%, phenol red | Gibco | 25-200-056 | |
| UniAmp | Unisense | 11632 | |
| United Biosystems Inc MINI CELL SCRAPERS 200/PK | Fisher | MCS-200 | |
| Y-27632 dihydrochloride | Tocris Bioscience | 12-541-0 | |
| µSensor Calibration Kit | Unisense/ Mettler Toledo | 51-305-070, 51-302-069 | pH 4.01 and 9.21, 20 mL packets |
Riferimenti
- Zhang, N., et al. Tissue spatial omics dissects organoid biomimicry. GEN Biotechnology. 2 (5), 372-383 (2023).
- Sato, T., et al. Single lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. 459 (7244), 262-265 (2009).
- Lancaster, M. A., et al. Cerebral organoids model human brain development and microcephaly. Nature. 501 (7467), 373-379 (2013).
- Dutta, D., Heo, I., Clevers, H. Disease modeling in stem cell-derived 3d organoid systems. Trends Mol Med. 23 (5), 393-410 (2017).
- Achberger, K., et al. Merging organoid and organ-on-a-chip technology to generate complex multi-layer tissue models in a human retina-on-a-chip platform. Elife. 8, e46188 (2019).
- Bartfeld, S., et al. In vitro expansion of human gastric epithelial stem cells and their responses to bacterial infection. Gastroenterology. 148 (1), 126-136 (2015).
- Fatehullah, A., Tan, S. H., Barker, N. Organoids as an in vitro model of human development and disease. Nat Cell Biol. 18 (3), 246-254 (2016).
- Clevers, H. Modeling development and disease with organoids. Cell. 165 (7), 1586-1597 (2016).
- Davies, J. A., Davies, J. A., Lawrence, M. L. . Organoids and Mini-organs. Ch. 1-2, 3-40 (2018).
- Ambrosini, Y. M., et al. Recapitulation of the accessible interface of biopsy-derived canine intestinal organoids to study epithelial-luminal interactions. PLoS One. 15 (4), e0231423 (2020).
- Williams, S. E., Turnberg, L. A. Demonstration of a pH gradient across mucus adherent to rabbit gastric mucosa: Evidence for a ‘mucus-bicarbonate’ barrier. Gut. 22 (2), 94-96 (1981).
- Schubert, M. L. Gastric secretion. Curr Opin Gastroenterol. 20 (6), 519-525 (2004).
- Celli, J. P., et al. Rheology of gastric mucin exhibits a pH-dependent sol−gel transition. Biomacromolecules. 8 (5), 1580-1586 (2007).
- Takeshita, Y., et al. Assessment of pH-dependent errors in spectrophotometric pH measurements of seawater. Marine Chemistry. 223, 103801 (2020).
- Mccracken, K. W., et al. Wnt/β-catenin promotes gastric fundus specification in mice and humans. Nature. 541 (7636), 182-187 (2017).
- Larsen, M., Borisov, S. M., Grunwald, B., Klimant, I., Glud, R. N. A simple and inexpensive high resolution color ratiometric planar optode imaging approach: Application to oxygen and ph sensing. Limnology and Oceanography: Methods. 9 (9), 348-360 (2011).
- Jewell, M. P., Galyean, A. A., Kirk Harris, J., Zemanick, E. T., Cash, K. J. Luminescent nanosensors for ratiometric monitoring of three-dimensional oxygen gradients in laboratory and clinical pseudomonas aeruginosa biofilms. Appl Environ Microbiol. 85 (20), e01116-e01119 (2019).
- Brooks, E. L., Hussain, K. K., Kotecha, K., Abdalla, A., Patel, B. A. Three-dimensional-printed electrochemical multiwell plates for monitoring food intolerance from intestinal organoids. ACS Sens. 8 (2), 712-720 (2023).
- . pH and reference electrode manual Available from: https://unisense.com/wp-content/uploads/2023/05/2023.05-pH-and-ref-sensor-manual.pdf (2023)
- Villahermosa, D., Corzo, A., Garcia-Robledo, E., Gonzalez, J. M., Papaspyrou, S. Kinetics of indigenous nitrate reducing sulfide oxidizing activity in microaerophilic wastewater biofilms. PLoS One. 11 (2), e0149096 (2016).
- Pabst, B., Pitts, B., Lauchnor, E., Stewart, P. S. Gel-entrapped staphylococcus aureus bacteria as models of biofilm infection exhibit growth in dense aggregates, oxygen limitation, antibiotic tolerance, and heterogeneous gene expression. Antimicrob Agents Chemother. 60 (10), 6294-6301 (2016).
- Ploug, H., Stolte, W., Epping, E. H. G., Jørgensen, B. B. Diffusive boundary layers, photosynthesis, and respiration of the colony-forming plankton algae, phaeocystis sp. Limnology and Oceanography. 44 (8), 1949-1958 (1999).
- Kolpen, M., et al. Nitrous oxide production in sputum from cystic fibrosis patients with chronic pseudomonas aeruginosa lung infection. PLoS One. 9 (1), e84353 (2014).
- Murphy, K. C., et al. Measurement of oxygen tension within mesenchymal stem cell spheroids. J R Soc Interface. 14 (127), 20160851 (2017).
- Sebrell, T. A., et al. A novel gastric spheroid co-culture model reveals chemokine-dependent recruitment of human dendritic cells to the gastric epithelium. Cell Mol Gastroenterol Hepatol. 8 (1), 157-171.e153 (2019).
- Miyoshi, H., Stappenbeck, T. S. In vitro expansion and genetic modification of gastrointestinal stem cells in spheroid culture. Nat Protoc. 8 (12), 2471-2482 (2013).
- Takase, Y., Fujishima, K., Takahashi, T. The 3d culturing of organoids from murine intestinal crypts and a single stem cell for organoid research. J Vis Exp. (194), (2023).
- Cherne, M. D., et al. A synthetic hydrogel, vitrogel((r)) organoid-3, improves immune cell-epithelial interactions in a tissue chip co-culture model of human gastric organoids and dendritic cells. Front Pharmacol. 12, 707891 (2021).
- Sebrell, T. A., et al. Live imaging analysis of human gastric epithelial spheroids reveals spontaneous rupture, rotation and fusion events. Cell Tissue Res. 371 (2), 293-307 (2018).
- . Sensortrace suite user manual. 3.3.000 Available from: https://unisense.com/wp-content/uploads/2021/10/SensorTrace-Suite-Manual.pdf (2023)
- . Microprofiling system user manual Available from: https://unisense.com/wp-content/uploads/2021/09/2023.11-MicroProfiling-System-2.pdf (2023)
- Wolffling, S., et al. Egf and bmps govern differentiation and patterning in human gastric glands. Gastroenterology. 161 (2), 623-636 (2021).
- Boccellato, F., et al. Polarised epithelial monolayers of the gastric mucosa reveal insights into mucosal homeostasis and defence against infection. Gut. 68 (3), 400-413 (2019).
- Mccracken, K. W., et al. Modelling human development and disease in pluripotent stem-cell-derived gastric organoids. Nature. 516 (7531), 400-404 (2014).
- Schumacher, M. A., et al. The use of murine-derived fundic organoids in studies of gastric physiology. J Physiol. 593 (8), 1809-1827 (2015).
- . Unisense Available from: https://unisense.com/products/ph-microelectrode/ (2024)
- Mccracken, K. W., et al. Wnt/beta-catenin promotes gastric fundus specification in mice and humans. Nature. 541 (7636), 182-187 (2017).
- Schreiber, S., et al. In situ measurement of ph in the secreting canaliculus of the gastric parietal cell and adjacent structures. Cell Tissue Res. 329 (2), 313-320 (2007).
- Xu, H., Li, J., Chen, H., Wang, C., Ghishan, F. K. Nhe8 plays important roles in gastric mucosal protection. Am J Physiol Gastrointest Liver Physiol. 304 (3), G257-G261 (2013).
- Gawenis, L. R., et al. Impaired gastric acid secretion in mice with a targeted disruption of the nhe4 na+/h+ exchanger. J Biol Chem. 280 (13), 12781-12789 (2005).
- Lewis, O. L., Keener, J. P., Fogelson, A. L. A physics-based model for maintenance of the ph gradient in the gastric mucus layer. Am J Physiol-Gastrointest Liver Physiol. 313 (6), G599-G612 (2017).
- Okkelman, I. A., Neto, N., Papkovsky, D. B., Monaghan, M. G., Dmitriev, R. I. A deeper understanding of intestinal organoid metabolism revealed by combining fluorescence lifetime imaging microscopy (flim) and extracellular flux analyses. Redox Biol. 30, 101420 (2020).
- Pleguezuelos-Manzano, C., et al. Establishment and culture of human intestinal organoids derived from adult stem cells. Curr Protoc Immunol. 130 (1), e106 (2020).
- Guimera, X., et al. A minimally invasive microsensor specially designed for simultaneous dissolved oxygen and ph biofilm profiling. Sensors (Basel). 19 (21), 4747 (2019).

.
