3D Modeling of Dendritic Spines with Synaptic Plasticity
PREPARAZIONE ISTRUTTORI
CONCETTI
Student Protocol
NOTE: Please see the Supplementary file 1 for the glossary of terms used in this protocol.
1. Install Blender, CellBlender, and MCell
NOTE: This protocol requires installation of MCell, Blender, and Cell Blender.
- Download and install the software on the MCell homepage (https://mcell.org/tutorials_iframe.html). Go to downloads on the top of the page and then follow the step-by-step instructions to download and install the software in the environment of choice (e.g., Linux, Mac OSX, or Windows).
NOTE: All computational models and simulations described in this protocol were tested on a CellBlender 1.1 bundle that includes Blender 2.78 with MCell 3.4 and CellBlender 1.1. It worked also on Blender 2.79b. All these software programs are open access and do not require reprint permission to be used. The instructions for the construction and simulation of the model can change slightly from one version to another. Parts of this protocol have been adapted from Czech et al.16.
2. Create a single dendritic spine
NOTE: This procedure creates a mesh of a single dendritic spine with a spine head and a spine neck using a modified sphere.
- Set up Blender 3D view at the Main Panel.
- Open Blender with CellBlender already installed. Press 5 on the keypad to change from Perspective a Orthogonal view and press 1 to change to front view. The perspective view has depth, but this is not needed now. Changing from Perspective a Orthogonal view allows better visualization of the mesh. Press Shift+C to center the cursor (Figure 1A).
- Create the spine head.
- Press Shift+A to open the mesh palette. Select the Mesh and then select UV Sphere. A UV sphere is a mesh mapped to the 3D surface of a sphere. The UV sphere represents the spherical head of a mushroom dendritic spine. The software assumes the units of the UV sphere are micrometers.
- Change the parameters on the Add UV Sphere panel. Change Size a 0.25 and Rings a 32 (Figure 1B). Press + or – on the keypad to respectively zoom in and zoom out of the visualization of the mesh. Alternatively, use the scroll button in the mouse to zoom in and out (Figure 1C).
NOTE: The parameter size scales the size of the original sphere and the parameter rings define the resolution of the mesh.
- Make the top of the head flat.
- Press Tab to switch Blender from the Object Mode, the standard object interaction mode, to Edit Mode. Work in Edit Mode for modifying the components of an existing mesh.
- Once the created mesh has been automatically selected, press a to deselect the created mesh. Press z to make the mesh transparent, which helps in visualizing the parts that will be edited. Zoom in on the mesh. Press b to select the top ¾ of the sphere with the mouse (Figure 2A). Press delete, select vertices, and enter to remove the vertices (Figure 2B).
- Press b and select the top. Press e, s, 0, and enter to seal the top with the vertices still selected. Move the blue arrow down to align to the top of the spine head (Figure 2C). Press z to change to solid view (Figure 3A). Press 7 to change to top view.
NOTE: The top of the sphere is made flat to model the PSD region of the spine head.
- To increase the mesh resolution at the top of the spine first select Tool and Knife. Cut a circle with the knife around the center of the top (Figure 3B). Select Tool and Loop Cut and Slide. Repeat this step four times to create four concentric circles around the center of the top (Figure 3C).
NOTE: The concentric circles are used to add new voxels that will increase the resolution of the PSD. - Create the spine neck.
- Press a to deselect the mesh. Press 1 to change to the front view. Press z to make the mesh transparent. Press b and then select the bottom of the mesh (Figure 4A). Press delete and vertices (Figure 4B). Press b and select the bottom of the mesh (Figure 4C). Press e and z, -0.45 to create an extrusion (Figure 4D).
NOTE: This creates an extrusion to the z axis position at -0.45 μm. Press a to deselect the whole mesh. - Press b and select the bottom of the neck. Press e, s, and 0 to seal the bottom (Figure 4E). Press a to select the whole mesh.
- Press a to deselect the mesh. Press 1 to change to the front view. Press z to make the mesh transparent. Press b and then select the bottom of the mesh (Figure 4A). Press delete and vertices (Figure 4B). Press b and select the bottom of the mesh (Figure 4C). Press e and z, -0.45 to create an extrusion (Figure 4D).
- Make the mesh compatible with MCell.
- Press Crtl+T to triangulate the mesh. The mesh is transformed into a set of interconnected triangles. This is a required procedure to make the mesh compatible with MCell. Select Tool and Remove Doubles. Use the Remove Doubles tools to remove duplicated vertices, if any, that have the same coordinates or are very close to each other, to make the mesh compatible with MCell.
NOTE: Double superposed vertices may have been accidentally created during the process of mesh creation and editing. - Select Model Objects on the CellBlender panel. Change the name of the Active Object a spine and press + to create the object spine. On the CellBlender panel, select Mesh Analysis and then click on Analyze Mesh (Figure 4F). This procedure will analyze the properties of the created mesh, including the number of vertices, edges, faces, surface area, volume, and mesh topology.
NOTE: The analysis will print the information in the Mesh Analysis Panel and it should be Watertight, Manifold, and Outward-Facing Normals. This step is required to ensure that the mesh will work on MCell. Otherwise, a step was probably missed. In this case, delete the mesh and start from step 2.1 again. - Press z to visualize the solid view of the spine. Press File and Save to have a copy of your blender file with the spine on the disk.
NOTE: The dimensions (i.e., length, diameter, size) of the meshes are in micrometers. See the glossary for the meaning of each keyboard shortcut.
- Press Crtl+T to triangulate the mesh. The mesh is transformed into a set of interconnected triangles. This is a required procedure to make the mesh compatible with MCell. Select Tool and Remove Doubles. Use the Remove Doubles tools to remove duplicated vertices, if any, that have the same coordinates or are very close to each other, to make the mesh compatible with MCell.
3. Creating a dendrite with multiple spines
- Generate a spine as described previously in sections 2.1–2.6. Press a to deselect the spine. Type Shift+C to center the cursor.
- Create a dendrite. Press Shift+A to open the mesh palette. Select Mesh and then Cylinder. Change the parameters on the Add Cylinder menu: Radius = 0.3 μm, Depth = 2 μm. Press Enter.
NOTE: The parameters radius and depth are defined according to the geometric characteristics of the dendrite. - Insert a spine in the dendrite.
- Press r and type 90 to rotate the cylinder 90° (Figure 5A). Use the blue arrow to drag the cylinder down to the bottom of the spine. Press 3 on the keypad to have a front view of the cylinder.
- Press z to make the mesh transparent. Use the mouse to move the blue normal arrow of the cylinder downwards to move the base of the spine to the interior of the cylinder (Figure 5B). Press a to deselect all objects.
- Use the right button of the mouse to select the dendrite (Figure 5C). Select Modifier on Blender panel (Figure 5D), select Add Modifier. Then select Boolean, select Operation Union, and select Object spine. Press Apply to create a joint mesh of the dendrite and the spine (Figure 5E). This operation creates a new mesh merging two meshes into a single one.
NOTE: The new mesh will be the combined dendrite and spine. The isolated dendrite disappears when the different meshes are combined, but the isolated spine mesh remains overlapped with the new mesh and is used to generate multiple copies of the same spine. Delete all the isolated spines after finishing the mesh. It is critical to have a complete overlap between the spine neck and the dendrite, otherwise, the mesh will not be watertight.
- Set the dendrite object into the CellBlender environment.
- Press a to deselect the meshes. Right-click in the dendrite with the mouse to select the dendrite only. Select CellBlender, Model Objects, and change Active Object a Dendrite and press + to create the Dendrite object.
- Insert new spines in the dendrite.
- Press 1 to change to the side view of the cylinder. Use the mouse to select the mesh of the isolated spine. To insert more spines, follow step 3.3, changing the position and the angle to insert each one to obtain a physiological distribution.
- Make the mesh compatible with MCell. To do so, press Tab to go to edit mode. Press a to select the whole mesh. Press Crtl+T to triangulate the mesh. Select Tool on the Blender panel and select Remove Doubles.
- Stylize the meshes.
- Smooth the mesh. Press Tab to change to object mode. Select Tool on the Blender panel and select Smooth. Select CellBlender, Model Objects, and select Add a Material.
- Make the mesh transparent by selecting Object Transparent and Material Transparent. Change alpha a 0.5 and enter to make the mesh partially transparent. Press z to change to solid view.
- Confirm whether the mesh is still compatible with MCell. To do so, select Mesh Analysis on the CellBlender panel to make sure that the mesh is still watertight, manifold mesh, and outward-facing normal.
- Save the blender file as dendrite_with_spines.blend.
4. Define surface regions
NOTE: This procedure creates the surface regions of the mesh that later will be used to set up how the regions interact with the molecules.
- Open the file dendrite_with_spines in the Blender environment. To do so, select File, Open, dendrite_with_spines.blend, and Open Blender File.
- Prepare the mesh for defining the surface regions. To do so, press Tab to change to edit mode. Press z to change to transparent view (Viewport shading, wireframe). Press a to select the whole mesh of the dendrite with spines. Select Model Objects. Select Dendrite. Press t to hide the CellBlender panel and better visualize the whole mesh in the main panel.
- Use + and – on the keypad to zoom in and out or scroll with the mouse. This is required for better visualization of the top of the spines to select and define the surface regions. Press a to deselect the object. Press Tab to change to Edit Mode. Press t to show the CellBlender panel again.
- Define the PSD surface region. To do so, press b and select the top of a dendritic spine with the mouse (Figure 6A,6B). Press + on Defined Surface Regions. Change the Region Name a PSD1 and click Assign (Figure 6C). Press a to deselect the object.
- Define the extrasynaptic surface region. To do so, press b and select the region around the top of the dendritic spine with the mouse (Figure 6D). Repeat step 4.3 for the Region Name a Extra_syn1. Repeat the step 4.3 for the other spines to define the other regions of the mesh (PSD2, PSD3, PSD4, Extra_syn2, Extra_syn3, and Extra_syn4) (Figure 6F). Press a to deselect the object.
- Define the surface regions of the ends of the dendrite. To do so, press b and select the left end of the dendrite. Change the Region Name a Left_end and click Assign. Press a to deselect the object. Press b and select the right end of the dendrite (Figure 6E). Change the Region Name a Right_end and click Assign.
NOTE: Move the mesh to find the best position to select each Defined Region.
5. Create molecules
- Create AMPARs. To do so, select Molecules on the CellBlender Panel. Select + on Defined Molecules to insert a new molecule and change Name a AMPAR. Change Molecule Type a Surface Molecule and Diffusion Constant a 0.05e-8 cm2/s14 to define the diffusion constant of AMPARs in the membrane (Figure 7A).
- Create Anchors. To do so, select Molecules on the CellBlender Panel. Select + on Defined Molecules to insert a new molecule and change Name a anchor. Change Molecule Type a Surface Molecule and change Diffusion Constant a 0.001e-8 cm2/s14 to define the diffusion constant of anchors in the membrane (Figure 7A).
- To create Anchors Bound to AMPARs, select Molecules on the CellBlender Panel. Select + on Defined Molecules to insert a new molecule. Change Name a anchor_AMPAR. Change Molecule Type a Surface Molecule. Change Diffusion Constant a 0.001e-8 cm2/s14.
- Create the anchor_LTP and anchor_AMPAR_LTP. To do so, repeat step 5.2. Name the molecule anchor_LTP. Repeat step 5.3. Name the molecule anchor_AMPAR_LTP.
NOTE: The anchor_LTP has a high affinity for AMPAR; thus, AMPARs increase in the synaptic regions. - Create the anchor_LTD and anchor_AMPAR_LTD. To create an anchor_LTD, repeat step 5.2. Name the molecule anchor_LTD. Repeat step 5.3. Name the molecule anchor_AMPAR_LTD.
NOTE: The anchor_LTD has a low affinity for AMPAR; thus, AMPARs decrease in the synaptic region.
6. Define surface classes
NOTE: This procedure defines the classes with the properties that are associated with the surface regions. The extrasynaptic regions reflect the free anchors and anchors bound to AMPAR. The lateral ends of the dendrite reflect all the molecules.
- Define the properties of the extrasynaptic regions.
- Press Tab to change to object mode. Select Surface Classes on the CellBlender Panel. Press + on the Surface Class to define a new surface class.
- Make the extrasynaptic region reflect the AMPAR bound to the anchor molecules.
NOTE: This procedure will trap the anchors and everything bound to them within the synaptic region.- Change Surface Class Name a reflective_extra_syn. Press + on reflective_extra_syn Properties to associate it with a molecule. Select Molecules | Single Molecule. Select anchor_AMPAR. Select Orientation = Ignore. Select Tipo = Reflective to make the region show the anchor_AMPAR molecules.
- Repeat step 6.1.2.1 for anchor_AMPAR_LTP and anchor_AMPAR_LTD.
- Make the extrasynaptic region reflect the anchors.
- Press + on reflective_extra_syn Properties to associate it with a molecule. Select Molecules | Single Molecule. Select anchor. Select Orientation = Ignore. Select Tipo = Reflective to make the region reflect the anchor molecules.
- Repeat step 6.1.3.1 for anchor_LTP and anchor_LTD.
- Define the properties of the dendrite ends. To do so, press + on Surface Class to define a new surface class. Change Surface Class Name a reflective_ends. Press + on Properties to associate it with a molecule. Select Molecules | All Surface Molecules. Select Orientation | Ignore. Select Tipo | Reflective to make it reflect all surface molecules.
7. Assign the created classes to each surface region
NOTE: This step assigns the surface classes to the surface regions.
- Assign the properties of the ends of the dendrite.
- Press + a assign a surface class with a region. Select reflective_ends for Surface Class Name (Figure 7C). Select Dendrite for Object Name. Select Specified Region for Region Selection. Select Left_end for Region Name.
- Repeat step 7.1.1 for the Right_end (Figure 7D).
- Assign the properties of the extrasynaptic regions.
- Press + to assign a surface class with a region. Select reflective_extra_syn for Surface Class Name. Select Dendrite for Object Name. Select Specified Region for Region Selection. Select Extra_syn1 for Region Name.
- Repeat step 7.2.1 for Extra_syn2, Extra_syn3, and Extra_syn4.
8. Place molecules on the mesh
NOTE: This step places the AMPARs, anchors, and AMPAR bound to anchors on the mesh.
- To place AMPAR Molecules on the Mesh, select Molecule Placement on the CellBlender Panel. Press + on the Release/Placement Sites to create a new release site. Change Site Name a relAMPAR (Figure 7B). Select Molecule = AMPAR. Object/Region = Dendrite[ALL]-(Dendrite[Left_end]+Dendrite[Right_end]). Quantity to Release = 1,000.
- Place anchor molecules on the Mesh.
- Select Molecule Placement on the CellBlender Panel. Press + on the Release/Placement Sites to create a new release site. Change Site Name a rel_anchor_PSD1. Select Molecule anchor. Object/Region = Dendrite[PSD1]. Quantity to Release = 200.
- Repeat step 8.2.1 for PSD2, PSD3, and PSD4.
- Place anchor_LTP Molecules on the Mesh. To do so, select Molecule Placement on the CellBlender Panel. Press + on the Release/Placement Sites to create a new release site. Change Site Name a rel_anchor_LTP_PSD1. Select Molecule = anchor_LTP. Object/Region = Dendrite[PSD1]. Quantity to Release = 0.
NOTE: anchor_LTP is an anchor with high binding affinity for AMPARs. - Place anchor_LTD Molecules on the Mesh by repeating step 8.3 for anchor_LTD.
NOTE: anchor_LTD is an anchor with low binding affinity for AMPARs.
9. Create the chemical reactions
- Creating the reaction between anchor and AMPARs.
- Select Reactions (Figure 7D) to create the reactions. Press + to include a new reaction. Reactants = anchor’ + AMPAR’. Reaction Type = <->. This defines a bidirectional reaction. Products = anchor_AMPAR’. Forward Rate = 0.03. Backward Rate = 0.05.
- Create the reaction between ANCHOR_LTP and AMPARs. To do so, repeat step 9.1, but replace anchor with anchor_LTP, and use a Backward Rate = 0.005 to increase the affinity between the reactants.
- Create the reaction between anchor_LTD and AMPARs and save the file. To do so, repeat step 9.2, but replace anchor with anchor_LTD, and use a Backward Rate = 0.5 to decrease the affinity between the reactants. Then Save the file.
10. Plot the output of the model
- Plot anchors bound to AMPARs at the PSD1 during the basal condition. To do so, select Plot Output Settings. Press + to define the molecules. Select anchor_AMPAR on Molecule. Select dendrite on Object. Select PSD1 on Region. Repeat step 10.1 for all PSD regions.
NOTE: It is useful to observe the basal number of trapped AMPARs to the PSD of each dendritic spine. The number of anchors bound to AMPARs can increase or decrease in comparison to the basal conditions during LTP and LTD. - Plot anchors bound to AMPARs at the PSD1 during LTP. Do this by repeating step 10.1. Replace anchor_AMPAR with anchor_AMPAR_LTP, then plot anchors bound to AMPARs at the PSD1 during LTD and finally repeat step 10.1, but replace anchor_AMPAR_LTP with anchor_AMPAR_LTD.
11. Run the simulations
- To run the basal condition, select Run Simulation. Select Iterations = 30,000. Set Time Step = 1e-3 s. Press Export & Run. Wait until the simulation ends. It may take from minutes to hours.
NOTE: In the basal condition, there is no release of anchor_LTP and rel_anchor_LTD molecules. Regarding the parameters of the simulation, the number of iterations needs to be long enough to be able to observe the diffusion of AMPARs from the dendrites and their anchoring at PSD. Small time-steps are more precise but slower to complete the simulation. - Select Reload Visualization Data. Select play animation to visualize the spatiotemporal results (Figure 8). Select Plot Output Settings. Press Plot.
NOTE: The graphs generated by CellBlender are isolated time series of the selected chemical species. Third-party programs can be used to import the data saved from multiple simulations to create overlaid plots of several conditions (e.g., basal, LTP, LTD; see Figure 8). - Run the homosynaptic potentiation condition (i.e., LTP; see Figure 8). To do so, select Molecule Placement on the CellBlender Panel. Select rel_anchor_LTP_PSD1 on the Release/Placement Sites.
- Change quantity to release = 200. Select rel_anchor_LTD_PSD1 on the Release/Placement Sites. Change quantity to release = 0. Select rel_anchor _PSD1 on the Release/Placement Sites. Change quantity to release = 0. Repeat steps 11.1–11.2.
- Run the homosynaptic depression condition (i.e., LTD; see Figure 8). To do so, Release 200 rel_anchor_LTD_PSD1 instead of rel_ANCHOR_LTP_PSD1. Set rel_anchor and rel_anchor_LTP_PSD1 a zero. Repeat steps 11.1–11.2.
3D Modeling of Dendritic Spines with Synaptic Plasticity
Learning Objectives
These results provide the steps for the construction of a 3D mesh that simulates a dendritic spine with a spine head and spine neck (Figure 1 a Figure 4). In addition, multiple dendritic spines can be inserted in a single dendritic segment (Figure 5) to study heterosynaptic plasticity of AMPARs14. The PSD on the top of the spine head (Figure 6) is the place where synaptic anchors bind to AMPARs and trap them temporarily at the synapse (Figure 7, Figure 8).
Synaptic plasticity could be verified roughly through changes in the number of species of anchor_AMPAR, anchor_AMPAR_LTP, and anchor_AMPAR_LTD at each spine. For the exact calculation of the occurrence of synaptic plasticity, it is recommended to calculate the variation in the total number of anchored and free AMPARs at the synapse. This can be performed using third-party programs to open the saved data of the simulation to summate the time series of the free AMPARs and the anchored AMPARs at each PSD (Figure 8).
The release of AMPARs on the mesh allowed the observation of their diffusion by a stochastic random walk along the dendrite and dendritic spines. Factors that modify the affinity of AMPARs for the anchors, such as posttranslational modifications and alterations of the rates of endocytosis and exocytosis, can trap the AMPARs at the PSD24,25,26. The binding of AMPARs with the anchors located at the PSD trapped a high density of AMPARs at the synapse. Homosynaptic potentiation (Figure 9) and depression (Figure 10) could be verified respectively through increases and decreases in the number of anchored AMPARs caused by changes in the affinity of AMPARs by anchors in comparison to the basal condition (Figure 11). Factors that reduced the affinity of AMPARs with the anchors released multiple AMPARs from one dendritic spine (i.e., homosynaptic depression) and induced heterosynaptic potentiation at the neighboring spines. Also, factors that increased the affinity of AMPARs for the anchors at one spine induced homosynaptic potentiation at that spine and heterosynaptic depression at the neighboring spines14. In this way, heterosynaptic plasticity was observed as the opposite effect at the neighboring spines of the homosynaptic plasticity induced at a given spine. For instance, homosynaptic LTP induction at a single spine created a heterosynaptic LTD effect at the neighboring spines (Figure 8E,F,G).
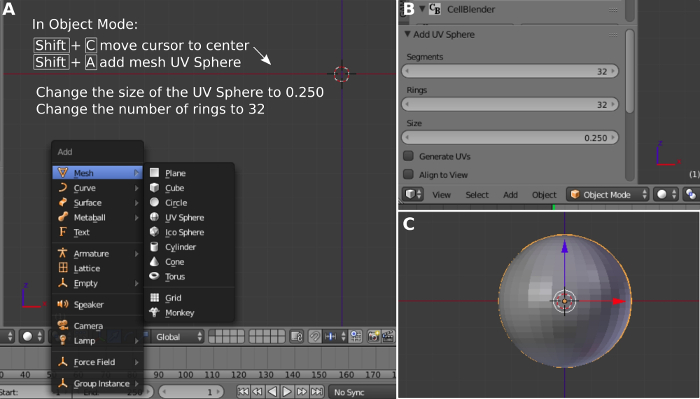
Figure 1: Creation of the dendritic spine head using a spherical mesh. (A) Adding the UV sphere. (B) Setting up the sphere dimensions. (C) Observing the created sphere. Please click here to view a larger version of this figure.
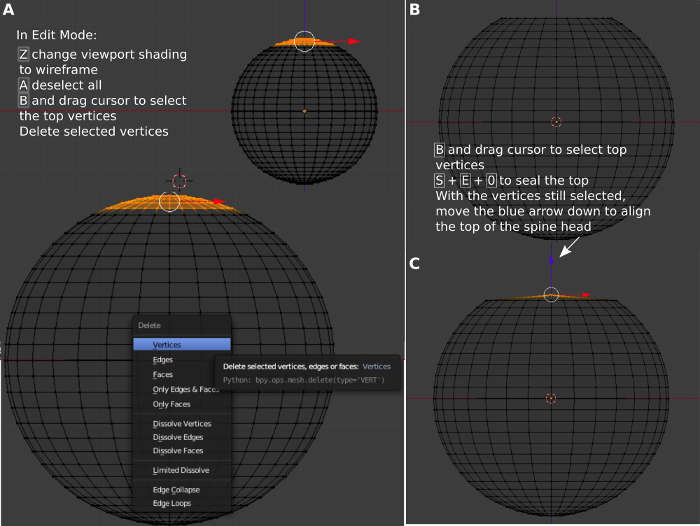
Figure 2: Construction of the top region. (A) Selecting the top region of the sphere. (B) Removing the selected region to make it flat. (C) Sealing the flat top. Please click here to view a larger version of this figure.
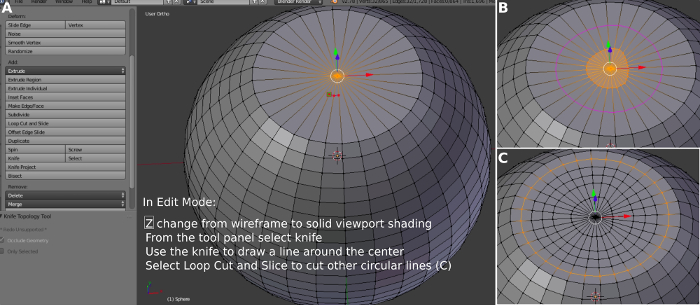
Figure 3: Creating concentric areas on the top of the spine. (A) Visualizing the top. (B) Using a knife to define a concentric region. (C) Creating multiple concentric regions. Please click here to view a larger version of this figure.
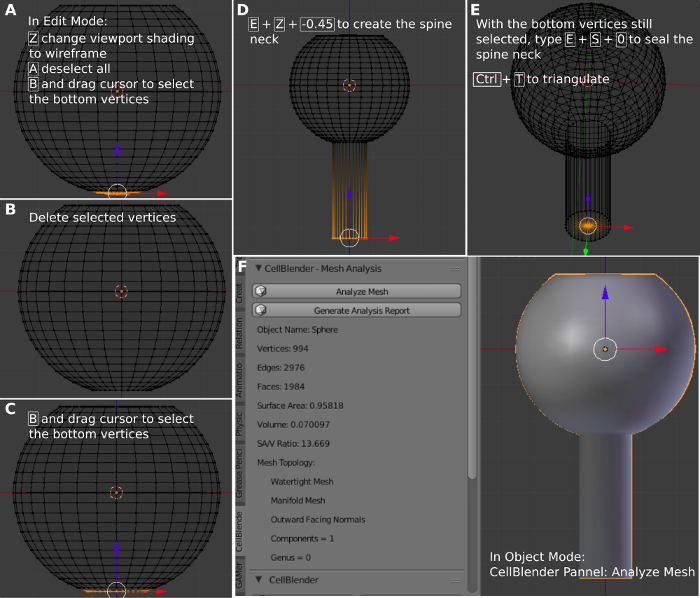
Figure 4: Creating the dendritic spine neck. (A) Selecting the bottom of the modified sphere. (B) Deleting the selected vertices. (C) Selecting the bottom. (D) Extrusion of the bottom to create the spine neck. (E) Sealing the bottom of the spine neck. (F) Analyzing the created spine. Please click here to view a larger version of this figure.
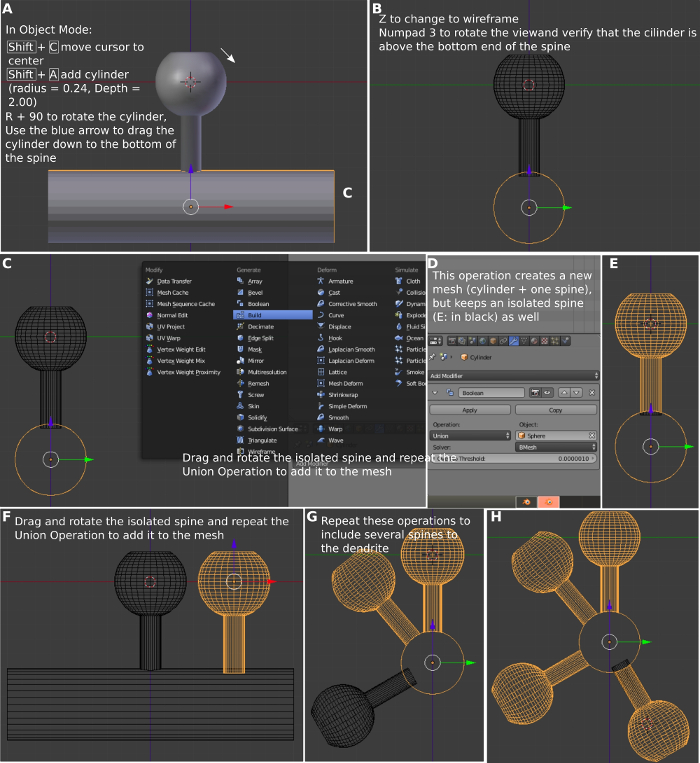
Figure 5: Creation of the dendrite with multiple spines. (A) Using the cylindrical mesh to create a dendrite. (B) Aligning the dendritic spine with the cylinder. (C) Joining the cylinder with the spine. (D) The Boolean operation to join the meshes. (E) The new combined mesh. (F) Adding the second spine. (G) Adding the third spine. (H) Adding the fourth spine. Please click here to view a larger version of this figure.
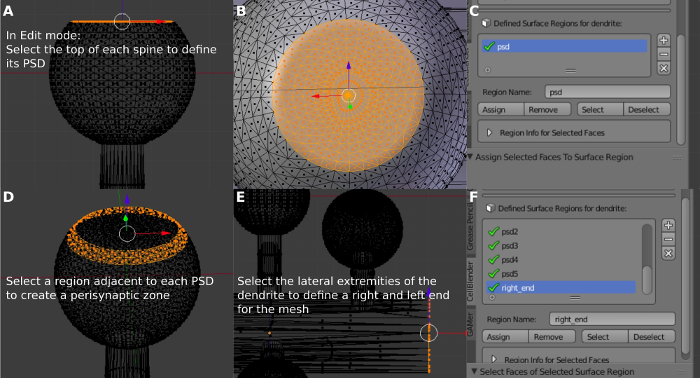
Figure 6: Defining the PSD region and the perisynaptic zone. (A) Selecting the PSD region. (B) Detailed view of the created PSD. (C) Defining the PSD surface region. (D) Selecting and defining the perisynaptic zone around the PSD. (E) Selecting and defining the lateral surface of the dendrite. (F) Defined surface regions. Please click here to view a larger version of this figure.
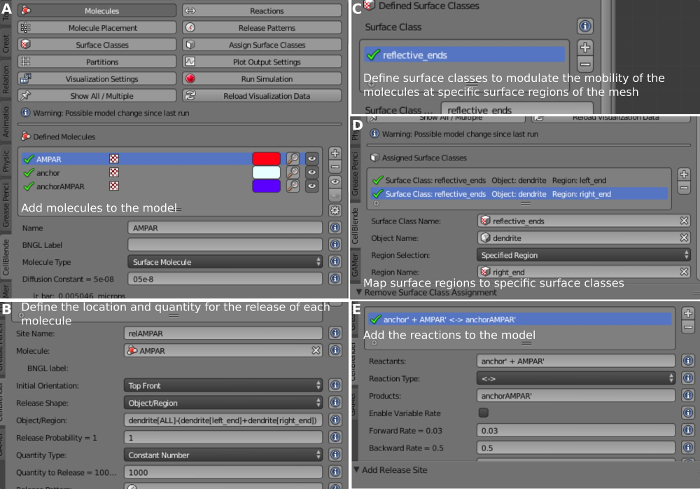
Figure 7: Defining the surface molecules. (A) Defining AMPAR, anchor, and AMPAR bound to anchor. (B) Defining the location and quantity of AMPAR copies. (C) Defining the Surface Classes. (D) Assigning the Surface Classes. (E) Creating the chemical reactions between the molecules. Please click here to view a larger version of this figure.
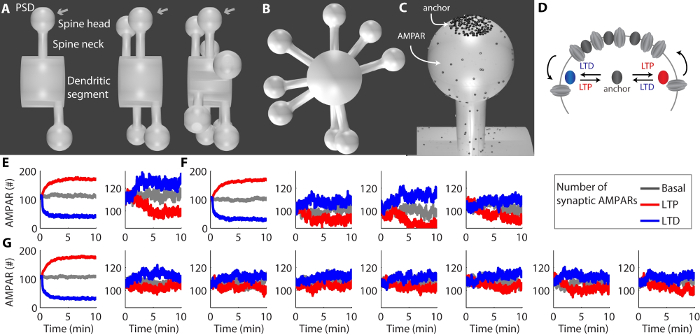
Figure 8: Representative results of synaptic plasticity. (A) Different meshes of a dendritic segment with two, four, or eight spines. (B) A different view of the dendritic segment with eight spines. (C) Detailed view of a dendritic spine with AMPARs and anchors at the PSD. (D) Diagram of the trafficking of AMPARs in and out of the PSD through their interactions with the anchors. (E-G) The curves show the number of synaptic AMPARs at each PSD for the basal condition and during LTP and LTD. The induction of homosynaptic LTP or LTD at a single spine created a heterosynaptic effect in the nearby spines for the mesh with two spines (E), four spines (F), and eight spines (G). Please click here to view a larger version of this figure.
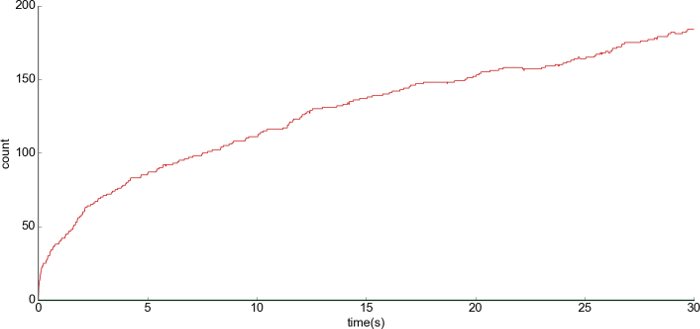
Figure 9: Representative result of the LTP condition. (A) The x-axis is the time and the y-axis is the number of the complex anchor_LTP_AMPAR at PSD1. There was a release of 200 free anchor_LTP at the beginning of the simulation. A higher number of bonds with anchors was formed in comparison to the basal condition (Figure 11) Please click here to view a larger version of this figure.
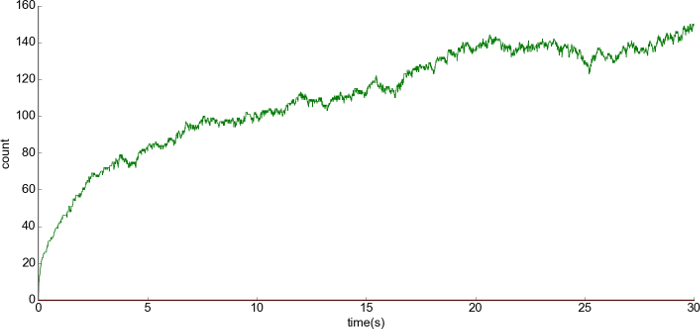
Figure 10: Representative result of the LTD condition. (A) The x-axis is the time and the y-axis is the number of the complex anchor_LTD_AMPAR at PSD1. There was a release of 200 free anchor_LTD at the beginning of the simulation. A lower number of bonds with anchors was formed in comparison to the basal condition (Figure 11). Please click here to view a larger version of this figure.
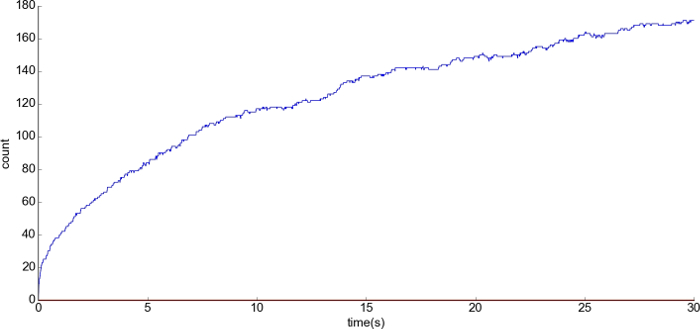
Figure 11: Representative result during basal condition. (A) The x-axis is the time and the y-axis is the number of the complex anchor_AMPAR at PSD1. There was a release of 200 free anchors at the beginning of the simulation. Please click here to view a larger version of this figure.
Supplementary File 1. Please click here to download this file.
List of Materials
| Blender | Blender Foundation | https://www.blender.org/ | |
| CellBlender | University of Pittsburgh | https://mcell.org/ | |
| Mcell | University of Pittsburgh | https://mcell.org/ |
Lab Prep
Computational modeling of diffusion and reaction of chemical species in a three-dimensional (3D) geometry is a fundamental method to understand the mechanisms of synaptic plasticity in dendritic spines. In this protocol, the detailed 3D structure of the dendrites and dendritic spines is modeled with meshes on the software Blender with CellBlender. The synaptic and extrasynaptic regions are defined on the mesh. Next, the synaptic receptor and synaptic anchor molecules are defined with their diffusion constants. Finally, the chemical reactions between synaptic receptors and synaptic anchors are included and the computational model is solved numerically with the software MCell. This method describes the spatiotemporal path of every single molecule in a 3D geometrical structure. Thus, it is very useful to study the trafficking of synaptic receptors in and out of the dendritic spines during the occurrence of synaptic plasticity. A limitation of this method is that the high number of molecules slows the speed of the simulations. Modeling of dendritic spines with this method allows the study of homosynaptic potentiation and depression within single spines and heterosynaptic plasticity between neighbor dendritic spines.
Computational modeling of diffusion and reaction of chemical species in a three-dimensional (3D) geometry is a fundamental method to understand the mechanisms of synaptic plasticity in dendritic spines. In this protocol, the detailed 3D structure of the dendrites and dendritic spines is modeled with meshes on the software Blender with CellBlender. The synaptic and extrasynaptic regions are defined on the mesh. Next, the synaptic receptor and synaptic anchor molecules are defined with their diffusion constants. Finally, the chemical reactions between synaptic receptors and synaptic anchors are included and the computational model is solved numerically with the software MCell. This method describes the spatiotemporal path of every single molecule in a 3D geometrical structure. Thus, it is very useful to study the trafficking of synaptic receptors in and out of the dendritic spines during the occurrence of synaptic plasticity. A limitation of this method is that the high number of molecules slows the speed of the simulations. Modeling of dendritic spines with this method allows the study of homosynaptic potentiation and depression within single spines and heterosynaptic plasticity between neighbor dendritic spines.
Procedura
Computational modeling of diffusion and reaction of chemical species in a three-dimensional (3D) geometry is a fundamental method to understand the mechanisms of synaptic plasticity in dendritic spines. In this protocol, the detailed 3D structure of the dendrites and dendritic spines is modeled with meshes on the software Blender with CellBlender. The synaptic and extrasynaptic regions are defined on the mesh. Next, the synaptic receptor and synaptic anchor molecules are defined with their diffusion constants. Finally, the chemical reactions between synaptic receptors and synaptic anchors are included and the computational model is solved numerically with the software MCell. This method describes the spatiotemporal path of every single molecule in a 3D geometrical structure. Thus, it is very useful to study the trafficking of synaptic receptors in and out of the dendritic spines during the occurrence of synaptic plasticity. A limitation of this method is that the high number of molecules slows the speed of the simulations. Modeling of dendritic spines with this method allows the study of homosynaptic potentiation and depression within single spines and heterosynaptic plasticity between neighbor dendritic spines.
