Single Step Isolation of Extracellular Vesicles from Large-Volume Samples with a Bifurcated A4F Microfluidic Device
PREPARAZIONE ISTRUTTORI
CONCETTI
Student Protocol
Sample collection was approved by the Latvian University Life and Medical Science Research Ethics Committee (decision N0-71-35/54)
NOTE: The materials used in this study are included in the Table of Materials file.
1. Three-dimensional (3D) printed mold fabrication
- Design a serpentine-shaped double negative mold in CAD software with the following dimensions for the top channel: height of 0.5 mm, width of 1 mm, and length of 210 mm. For the bottom channel, use the same design with a width of 1.5 mm, height of 0.6 mm, and length of 210 mm. Dimensions such as mold width, length, height, and design isometric illustration are shown in Figure 1. Save the design as .stl file.
- Prepare the file in Z-Suite; no support needs to be added. Print the molds using an ultraviolet liquid crystal display (UV LCD) 3D printer with tough black resin.
- After printing, carefully remove the molds from the printer surface. Wash the molds in isopropanol (IPA) in sonication for 20 min.
- Blow-dry the molds with N2.
- Expose the molds to flood exposure using an ND33 filter for 810 s, then place them into an oven at 60 °C for 48 h.
2. Preparation of the PDMS molds
- Weight PDMS at a 10:1 w/w ratio (for each mold, use 16 g of elastomer base and 1.6 g of cross-linking agent). Mix the components in a planetary centrifugal mixer, mixing for 1 min at 500 RPM.
- Cast the mixed PDMS into the 3D-printed molds and degas in a vacuum desiccator at -800 mbar for 30 min or until no bubbles are observed.
- Carefully place a 100 µm thick polyvinyl chloride liner on top of the liquid PDMS.
NOTE: Apply the liner slowly from one side to another to avoid bubble formation. - Insert the mold with PDMS into a Mold-in-Place Jig and tighten the jig by using a torque wrench set to 0.3 Nm.
- Place the jig into a 60 °C oven for 3 h to cure PDMS.
- Disassemble the jig using a hex key and remove the 3D-printed mold from the jig.
- Carefully de-mold PDMS molds from the 3D-printed master molds, running around the edges of the mold with a scalpel, then removing the mold with tweezers.
NOTE: For easier mold removal, IPA can be used; however, the molds need to be heated at 60 °C for 10 min afterward to evaporate excess IPA.
3. Preparation of OSTE-COC top channel
- Weight OSTE at 1.09:1 w/w ratio (for 5 devices, 1.56 g of A part and 1.44 g of B part are needed). Mix the components in a planetary centrifugal mixer: mix for 5 min at 750 RPM, then degas for 5 min at 750 RPM.
- Transfer the mixed OSTE to a centrifuge tube and degas at -800 mbar in a vacuum desiccator for 30 min.
- Clean COC slides with 2 x 16 mini Luer connectors by sonication in IPA for 10 min. Carefully dry the slides with N2.
- Place the clean COC slides into the plasma asher so that the flat surface is placed upwards, and treat the surface with 800 SCCM oxygen plasma for 2 min at 700 W power with 0.133 mbar pressure.
- Place the oxidized COC slide on the PDMS mold for the top channel so that the treated surface is in contact with the structured surface of PDMS. Place the assembly into a Mold-in-Place jig, tightening the jig with a torque wrench set to 0.3 Nm.
- Connect the pressure system to the compressed air line at 6 bar. Take the centrifuge tube with degassed OSTE and assemble it per pressure system instructions using polytetrafluorethylene (PTFE) tubing with 0.8 mm inner diameter (ID).
- Connect the PTFE tube to the inlet of the PDMS mold and, using 1000 mbar pressure maintained by a piezoelectric pump located in the pressure system, fill the device. Place the jig with the glass side up into the mask aligner and expose with 850 mJ dose using ND33 filter.
- After exposure, disassemble the jig using a hex key and carefully remove the COC slide with the structured OSTE layer from the PDMS mold.
- Bring the structured OSTE in contact with a 25 mm x 75 mm membrane with 50 nm pores and 11.8% pore density so that the channels are fully covered with the membrane.
NOTE: It is particularly important to keep the membrane as straight as possible during the process of application. - Place the assembly onto a hot plate set to 60°C with the membrane side up, place a clean glass slide on top of the membrane, and apply a 1.6 kPa pressure from the top to ensure even bonding. Heat the assembly overnight.
NOTE: After this step, it is especially important to evaluate bonding performance and channel deformation.
4. Preparation of OSTE-COC bottom channel and device assembly
- Mix OSTE at 1.09:1 w/w ratio (for 5 devices 1.56 g of A part and 1.44 g of B part are needed). Mix the components in a planetary centrifugal mixer: mix for 5 minutes at 750 RPM, then degas for 5 minutes at 750 RPM.
- Transfer the mixed OSTE to a falcon tube and degas at -800 mbar in a vacuum desiccator for 30 min.
- Clean COC slides by sonication in IPA for 10 minutes. Carefully dry the slides with N2.
- Place the clean COC slides into plasma asher and treat the surface with 800 SCCM oxygen plasma for 2 min at 700 W power with 0.133 mbar pressure.
- Place the oxidized COC slide on the PDMS mold for the bottom channel so that the treated surface is in contact with the structured surface of PDMS. Place the assembly into a Mold-in-Place jig, tightening the jig with a torque wrench set to 0.3 Nm.
- Take the falcon tube with degassed OSTE and assemble it as per pressure system instructions using PTFE tubing with 0.8 mm ID.
- Connect the PTFE tube to the inlet of the PDMS mold and using 1000 mbar pressure fill the device. Place the jig with the glass side up into mask aligner and expose with 1100 mJ dose using ND33 filter.
- After exposure, disassemble the jig using a hex key and carefully remove the COC slide with the structured OSTE layer from the PDMS mold.
- Combine the structured OSTE layer with the top channel assembly so that the design of the top and bottom layer is aligned with one another and that the OSTE bottom layer is in contact with the membrane.
- Insert the design into a Mold-in-Place jig and, using bulldog clips, apply a 1.6 kPa pressure to the device evenly. Place the jig into a 60°C oven overnight.
NOTE: Uneven placement of the clips can lead to uneven bonding.
5. Device evaluation
- After bonding, visually evaluate the devices with a microscope for visual defects, for example, deformation of the channel or unsuccessful bonding.
- Test the devices with no defects by passing 0.02 µm filtered deionized water through the channels with 30 mbar pressure. Observe for flow channel and Luer port leaks and water flow.
NOTE: If no leakage can be detected and channel flow is uninterrupted, devices are considered usable for further experiments.
6. Device setup
- Fill two 5 mL syringes with 2 mL of 20 nm filtered 3% H2O2 and fit them on a syringe pump (use the movement arrows for the needed distance, insert syringes, and fix them in place with the top bar).
CAUTION! Always be careful when handling H2O2. Wear a lab coat, protective rubber gloves, and eye protection. - Connect 14 cm long 800 µm ID PTFE tubing to the syringes using the syringe needles and to the inlets of the device using Luer connectors.
- Connect 20 cm long 200 µm ID polyether ether ketone (PEEK) tubing to the outlets.
- Connect the other end of the PEEK tubing to a waste container. The setup can be seen in Figure 3.
- Start the syringe pump at the flow speed of 500 µL/min for each inlet (on the instrument screen: Settings > System Set > Syringe (manufacturer: medical syringe, syringe: 5 mL) > Indietro button > Parameters (Select: infuse, Repeat: 1, Volume: 1.5 mL, Flow: 500 µL/min) > Indietro button > Indietro button). Collect the flow-through in a waste container.
- Fill two new 5 mL syringes with 2 mL of 20 nm filtered 70% ethanol.
- Replace the previous syringes with the ethanol-filled syringes and start the syringe pump using the same parameters described in step 6.5.
- Use new 5 mL syringes and fill them with 5 mL of 20 nm filtered phosphate-buffered saline (PBS).
- Replace the previous syringes with the PBS-filled syringes and flush the device with 4 mL of PBS by repeating steps 6.1. and 6.5.
NOTE: Make sure to change the system setup parameters to Volume: 4 mL (on the instrument screen: Settings > Parameters > (Select: Volume: 4 mL). - Unplug the tubing from the device and close each inlet and outlet with Luer plugs.
NOTE: Leave PBS in the device and avoid trapping any air by adding additional PBS on each inlet and outlet using a micropipette before closing. - Leave the device overnight in a dark place at room temperature (RT).
- The following day, flush the PBS-filled device with 4 mL of 20 nm filtered PBS (as described in step 6.8).
- Collect the last 1 mL of the flow-through from both outlets in a 2 mL protein low-binding tube and store at 4 °C as a blank for particles introduced by the device.
- Remove the PBS-filled syringes and replace them with new 5 mL syringes filled with air.
- Purge the device with 4 mL of air until all the liquid has exited the device and tubing.
7. Device testing with standardized latex beads
- Prepare a standardized bead sample using 1.0 µm polystyrene and 0.1 µm carboxylate beads. To a 15 mL tube, add 500 µL of 1.0 µm polystyrene bead standards, 1 µL of 0.1 µm carboxylate bead standards, and 9499 µL of 20 nm filtered PBS (final concentration of 1.0 and 0.1 µm bead mix will be 5 × 108 beads/mL and 3.6 × 1010 beads/mL respectfully).
- Vortex the bead sample for 30 s and store it at +4 °C while not in use.
- Fill one new 5 mL syringe with 1.5 mL of the standardized bead mixture and another one with 1.5 mL of 20 nm filtered PBS.
- Remove the previously attached syringes and attach the bead mixture syringe to the first inlet tubing and the other to the second inlet.
- Remove the waste container and prepare at least five 0.5 mL microcentrifuge tubes for each outlet tubing.
- Change the syringe pump system start-up parameters to Volume: 1 mL; Flow: 250 µL/min.
NOTE: Depending on the user's needs, different flow speeds such as 250 µL/min, 200 µL/min, 150 µL/min, 100 µL/min, and 50 µL/min can be set. A preliminary study to choose the most favorable speed for the wanted outcome is recommended. - Start the syringe pump and collect the outflow – 5 to 6 droplets in each microcentrifuge tube.
NOTE: If the same device is used multiple times, flush the device as described in step 6.8 and change all connectors, plugs, and needles. Wash the used connectors, plugs, and needles by leaving them in 20 nm filtered 70% ethanol for at least 30 min. Then, move all parts to a tube containing 20 nm filtered deionized water, shake the tube thoroughly, and transfer them to a Petri dish containing 20 mL of 20 nm filtered deionized water. Leave the Petri dish on the shaker for at least 30 min at 100 rpm, then leave the things to dry in a dry Petri dish (for at least 12 h) before reusing.
8. Device testing with urine samples
- Prepare the device as described in steps 6.1-6.15.
- Collect 100 mL of morning urine per donor using urine sterile containers and deliver it to the laboratory for processing within 2 hours of collection.
- Separate each urine sample into two 50 mL tubes and centrifuge at 2'000 x g for 15 min at RT.
- Collect the supernatant and discard the pellet.
- Centrifuge the supernatant at 10'000 x g for 30 minutes at RT to get rid of large particles and cell debris.
- Collect the supernatant and discard the pellet.
- Wash the device as described in 6.6.
- Change the 5 mL syringes to a 20 mL syringe filled with the prepared urine sample for the sample inlet and another 20 mL syringe filled with 20 mL of 20 nm filtered DEPC-PBS for the buffer inlet. Change the syringe pump settings to BDPlastipak; Syringe: cc; Volume: 20 mL; Flow: 250 µL/min (optimal flow rate), similarly as described in step 6.2. Start the syringe pump and collect the flow-through from each outlet in separate 50 mL tubes.
- Concentrate each flow-through separately using 100,000 molecular weight cut-off concentration tubes to 100 µL each at 3,000 x g in 4 °C.
- Transfer the concentrates to 0.5 mL microcentrifuge tubes.
- Vortex the concentrated samples for 30 seconds, then spin for 10 seconds.
- Aliquot the samples by transferring 25 µL of sample per tube, label them, and freeze at -80 °C for long-term storage.
9. Device testing with conditioned media
- Prepare the device as described in steps 6.1- 6.15.
- Culture cells in a 5% CO2 humidified environment at 37 °C until the total count reaches 100 million according to Bajo-Santos et al.20 Passage the cells to a single T175 suspension flask in 100 mL of serum-free media supplemented with 2% B-27 serum substitute supplement and culture in a 5% CO2 humidified environment at 37 °C for additional 48 h.
- Collect the cell suspension and centrifuge it at 300 x g for 5 min at RT to get rid of cells.
- Collect and centrifuge the supernatant again at 3,000 x g for 30 min at +4 °C to get rid of large particles and cell debris.
- Collect the supernatant and store it at +4°C until the separation can take place, but no longer than 3 h.
- Change the 5 mL syringes with 20 mL syringes filled with the prepared conditioned media for the sample inlet and filled with 20 mL of 20 nm filtered PBS for the buffer inlet.
- Set the settings in the pump station as described in step 7.8.
- Start the syringe pump and collect the flow-through from each outlet in a separate 50 mL tube.
- Concentrate each flow-through separately using 100,000 molecular weight cut-off concentration tubes to 150 µL each at 3,000 x g at +4 °C.
- Transfer the concentrates to 0.5 mL microcentrifuge tubes.
- Vortex concentrated samples for 30 s.
- Spin concentrated samples for 10 s.
- Aliquot the samples by transferring 25 µL of sample per tube, label them, and freeze them at -80 °C for long-term storage.
10. Isolation of EVs using ultracentrifugation
- Centrifuge 20 mL of urine or conditioned cell media at 100,000 x g for 70 min at +4 °C using a centrifuge with a fixed-angle rotor.
- Discard the supernatant and resuspend the pellet in 20 mL of 20 nm filtered PBS.
- Centrifuge again, as explained in 10.1.
- Discard the supernatant and resuspend the pellet in 12 mL of 20 nm filtered PBS.
- Centrifuge at 100,000 x g for 70 min at +4 °C using a swing rotor.
- Discard the supernatant.
NOTE: Be careful when discarding the supernatant this time. It is recommended to do it with a pipette. - Resuspend the pellet in 100 µL of 20 nm filtered PBS.
- Aliquot the sample and proceed to setion 12 for EV characterization. Otherwise, store at -80 °C until further use.
11. Isolation of EVs using size exclusion chromatography (SEC)
- Concentrate 20 mL of urine or conditioned cell media to 500 µL using 100 kDa molecular weight cut-off (MWCO) centrifugal filter units at 3,000 x g at +4 °C for 15 min.
- Transfer the concentrate onto the SEC column (35 nm load range).
- Pour the 20 nm filtered PBS onto the column and collect 15 fractions of 0.5 mL.
- Analyze fractions with a dynamic light scattering (DLS) system following the manufacturer's instructions.
- Pool together the selected fractions. Choose fractions with an attenuator lower than 11 and at least 30% of total particles being bigger than 40 nm.
- Concentrate selected fractions to 100 µL using 3 kDa molecular weight cut-off (MWCO) centrifugal filter units at 14,000 x g at +4 °C for 15 min.
- Aliquot the sample and proceed to section 12 for EV characterization. Otherwise, store at -80 °C until further use.
12. EV characterization
- Take the sample to be characterized from the freezer and defrost it slowly (on an ice block or at +4 °C).
- Perform nanoparticle tracking analysis (NTA) to determine particle size distribution and amount13. Perform high-affinity T-cell membrane protein 4 (TIM-4) double sandwich enzyme-linked immunosorbent assay (dsELISA)14 to confirm EV presence in the samples.
13. NTA
- Vortex the thawed EV sample for 30 s and spin for 10 s, then transfer 1 µL to 999 µL of 20 nm filtered PBS in a 2 mL microcentrifuge tube to dilute it 1000-fold. Save 1 mL of extra PBS used for dilution to check that it contains no particles. Store everything at +4 °C until measurement.
NOTE: Filter PBS through a 0.02 nm filter prior to use. - Vortex the EV sample or blank for 30 s and insert it into the module using a syringe. Fill the module with approximately 0.7 mL of sample and place it in the instrument.
- Measure the sample using the nanoparticle analyzer instrument following the manufacturer's instructions.
14. dsELISA for EV markers
- Prepare a solution of 1 µg/mL TIM4-Fc protein. Coat the wells of a 96-well ELISA plate by transferring 100 µL of solution to each necessary well and incubate overnight at +4 °C shaking at 200 RPM.
- The following day, prepare washing solution (1x TBS + 0.05% Tween20 + 2 mM CaCl2).
- Discard all the liquid in each well. Wash each well by adding 200 µL of wash solution, shaking at 200 RPM for 5 min at RT, and discarding. Repeat this step twice.
- Prepare blocking solution (1x TBS + 0.05% Tween20 + 1% bovine serum albumin. Add 200 µL of blocking solution to each well and incubate at RT for 1 h while shaking at 200 RPM.
- Wash each well as described in step 14.3.
- Prepare 220 µL of each sample dilution in washing buffer (optimal EV concentration is 2.7 × 105 EVs/µL). Add 100 µL of the diluted sample per well and perform two replicates. Incubate at RT for 90 min while shaking at 200 RPM.
- Wash each well as described in step 14.3.
- Add 100 µL of corresponding antibodies to each well. Use proper EV markers for confirming EVs and contaminants4. Incubate at RT for 2 h while shaking at 200 RPM. NOTE: If secondary antibodies are used, repeat steps 14.7-14.8. However, reduce the incubation period of secondary antibodies to 1 h.
- Wash each well as described in step 14.3.
- Add 100 µL of horseradish peroxidase substrate to each well, mix the plate for 1 min, and incubate at RT for 30 min.
- Add 1 M H2SO4 to each well to stop the reaction.
- Using a microplate reader, measure the absorbance at 450 nm.
Single Step Isolation of Extracellular Vesicles from Large-Volume Samples with a Bifurcated A4F Microfluidic Device
Learning Objectives
We fabricated a microfluidic device using a 3D printed double negative mold (Figure 1) via soft-lithography (Figure 2A) for high throughput EV separation based on the bifurcated A4F principle (Figure 2B,C). The setup requires a pump and a flow-through station, as can be seen in Figure 3, for the isolation of EVs in an automated manner. Firstly, to evaluate the proof of concept of the devices, a mixture of polystyrene beads with diameters of 100 nm and 1000 nm was prepared to represent vesicles and fine cell debris, respectively10. Experiments were conducted with varying flow rates for the bead mix, both with and without bifurcating flow, to investigate the effect of linear velocity on separation efficiency. Across all experiments, the recovery of small beads remained consistent and above 90%10,showing the potential of the device to recover EVs.
Then, we assessed and compared the potential of the COC-OSTE device in isolating EVs from large volumes (>1 mL) from complex biofluids with minimal pre-processing. As such, urine from 10 healthy donors (Figure 4) and cell media from two different prostate cell lines (Figure 5) were used as a template to simultaneously isolate EVs following three different procedures: ultracentrifugation (UC), size exclusion chromatography (SEC), and the A4F microfluidics-based OSTE-COC device. After isolation, the total number of particles and their size distribution were assessed using nanoparticle tracking analysis (NTA). On average, the OSTE-COC device showed better total particle recovery from biofluids compared to UC, but the statistical significance was only achieved when combining the particle numbers from both ports (Figure 4A). In order to compare the device performance with other systems, R & L outlet together should be taken into consideration. As shown in Figure 4, L & R outlet together performance on recovering EVs outperforms UC and SEC. Separately, the L-port was designed to capture the small EV fraction, while the R-port was designed to collect the bigger EV fraction with other molecules of similar size. Interestingly, the recovery using the R-PORT of the OSTE-COC device was slightly higher than SEC and UC alone (Figure 4A). CD63 expression showed a similar pattern (Figure 4C). This finding indicates that the OSTE-COC device was more effective in total EV recovery. Equivalent size distribution was found between the different methodologies, except for UC, which shows a bigger particle size distribution (Figure 4B).
Comparable results were observed in cell media cultures. In both scenarios, the total particle recovery from both device ports exhibited superior performance compared to SEC or UC methodologies (as depicted in Figure 5A,B). Notably, EVs derived from PC3 cells demonstrated a distinct size distribution, with greater homogeneity in the L-PORT distribution when contrasted with other experimental groups (Figure 5C,D). Furthermore, the analysis of CD63 expression confirmed the higher EV recovery rates achieved using the COC-OSTE device (as illustrated in Figure 5E,F). A summary comparing the isolation characteristics of the different methodologies examined in this study can be found in Table 1.
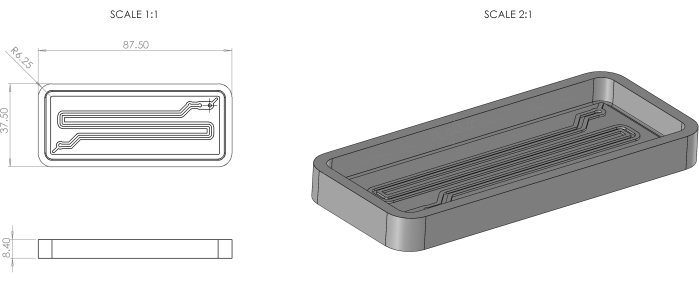
Figure 1: 3DP serpentine-shaped double negative mold dimensions and isometric view. Please click here to view a larger version of this figure.
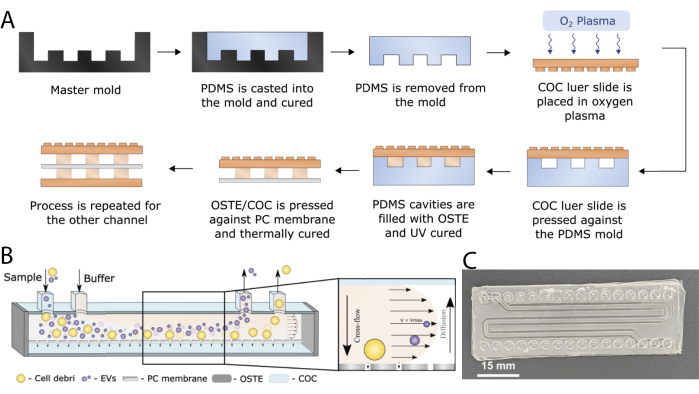
Figure 2: COC-OSTE microfluidic device. (A) Scheme of the different main steps of fabricating the OSTE-COC device. (B) Device working principle. (C) Image of the finished device. Scale bar: 15 mm. This figure has been modified with permission from Priedols et al.10 and Bajo-Santos et al.20. Please click here to view a larger version of this figure.

Figure 3: Experimental configuration of the device. The syringe pump is on the left, the OSTE-COC device is in the middle, and the recovery station is on the right. This figure has been modified with permission from Priedols et al.10 and Bajo-Santos et al.20. Please click here to view a larger version of this figure.
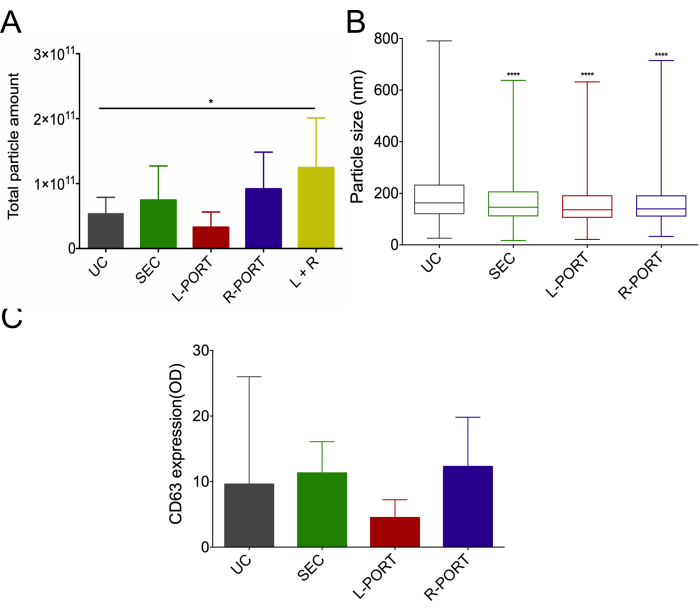
Figure 4: Urinary EV size distribution and particle recovery from 10 donors using ultracentrifugation (UC), size-exclusion chromatography (SEC), and the COC-OSTE device. (A) Particle amount recovered from each isolation method by nanoparticle tracking analysis (NTA). Data represented as mean +/- standard deviation. Statistical significance denoted by * (p<0.05). (B) Boxplots display the average particle size distribution among all urine samples assessed by NTA, with whiskers indicating the minimum and maximum values. P-values were derived from comparisons to UC, with **** indicating high statistical significance (p<0.0001). (C) The median and range of the average CD63 amount, assessed using double-sandwich enzyme-linked immunosorbent assay (dsELISA), for each isolation method, were calculated across all samples. L-port: Left port; R-port: Right port. This figure has been modified with permission from Bajo-Santos et al.20. Please click here to view a larger version of this figure.
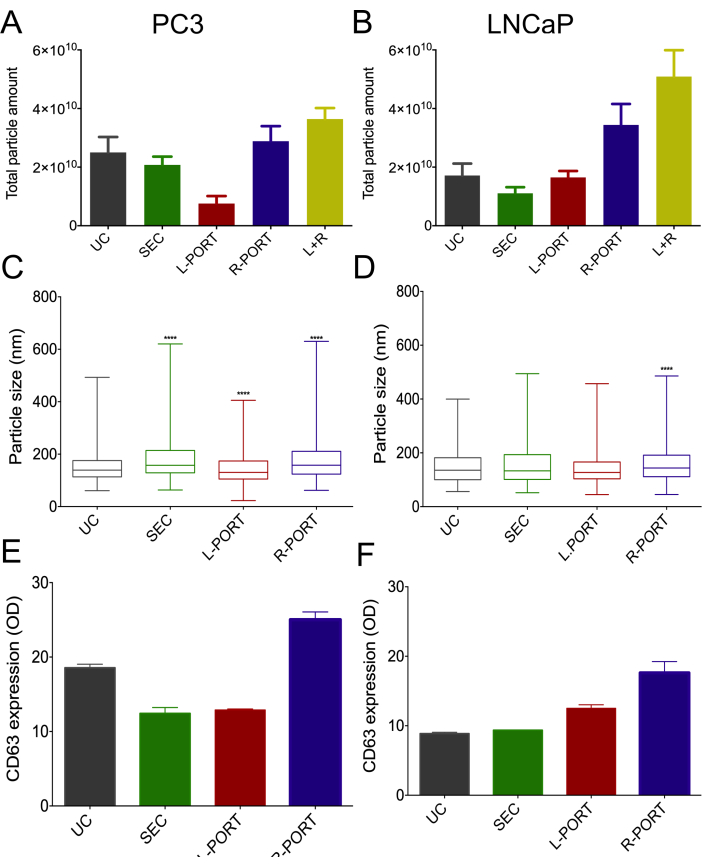
Figure 5: Characterization of EVs isolated from PC3 and LNCaP cells using different methods. (A) Particle amount recovered from PC3 media using nanoparticle tracking analysis (NTA) represented by mean+/- standard deviation. (B) Particle amount recovered from LNCaP cell media using NTA represented as mean +/- standard deviation. (C) Median particle size distribution of EVs from PC3 cultures, along with the range, was determined by NTA. (D) Median size distribution of EVs isolated from LNCaP cultures with range. (E) Median and range of CD63 expression for each isolation method for PC3 cell line-derived EV, using double-sandwich enzyme-linked immunosorbent assay (dsELISA). (F) Median and range of LNCaP-derived EV CD63 expression by dsELISA for each isolation method. UC: Ultracentrifugation; SEC: Size-exclusion chromatography; L-port: Left port; R-port: Right port. This figure has been modified with permission from Bajo-Santos et al.20. Please click here to view a larger version of this figure.
| UC | SEC | COC-OSTE | |
| Processing time/sample | ++/+++ | +++ | + |
| Throughput | ++ | + | +++ |
| Cost/sample | + | +++ | ++ |
| Purity | + | +++ | ++ |
| Automatization | ++ | + | +++ |
| EV Yield | ++ | ++ | +++ |
| Size selection | + | ++ | ++ |
Table 1. Comparison of isolation characteristics of EVs with the three methods UC SEC, and the COC-OSTE device. UC: Ultracentrifugation; SEC: Size-exclusion chromatography. COC-OSTE: cyclic olefin copolymer-off-stoichiometry thiol-ene. +: low; ++: medium; +++: high. * Device dependent.
List of Materials
| 0.1 µm carboxylate FluoSpheres | Invitrogen | #F8803 | Stock concentration: 3.6 x 1013 beads/mL (LOT dependent) |
| 0.5 mL microcentrifuge tubes | Starstedt | 72.704 | |
| 1 mL Luer cone syringe single use without needle | RAYS | TUB1ML | |
| 1.0 µm polystyrene FluoSpheres | Invitrogen | #F13083 | Stock concentration: 1 x 1010 beads/mL (LOT dependent) |
| 10 mL Serological pipettes | Sarstedt | 86.1254.001 | |
| 15 mL (100k) Amicon Ultra centrifugal filters | Merck Millipore | UFC910024 | |
| 2.0 mL Protein LoBind tubes | Eppendorf | 30108132 | |
| 20 mL syringes | BD PlastikPak | 10569215 | |
| 250 µm ID polyether ether ketone tubing | Darwin Microfluidics | CIL-1581 | |
| 3 kDa MWCO centrifugal filter units | Merck Millipore, | UFC200324 | |
| 5 mL Medical Syringe without Needle | Anhui Hongyu Wuzhou Medical | 159646 | |
| 50 mL conical tubes | Sarstedt | 62.547.254 | |
| 70 Ti fixed angle ultracentrifuge rotor | Beckman Coulter | 337922 | |
| 800 µm ID polytetrafluoroethylene tubing | Darwin Microfluidics | LVF-KTU-15 | |
| 96 well microplate, f-bottom, med. binding | Greiner Bio-One | 655001 | ELISA plate |
| B-27 Supplement (50x), serum free | Thermo Fisher Scientific | 17504044 | |
| Bovine serum albumin | SigmaAldrich | A7906-100G | |
| COC Topas microscopy slide platform | Microfluidic Chipshop | 10000002 | |
| COC Topas microscopy slide platform 2 x 16 Mini Luer | Microfluidic Chipshop | 10000387 | |
| Elveflow OB1 pressure controller | Elvesys Group | ||
| Luer connectors | Darwin Microfluidics | CS-10000095 | |
| Mask aligner Suss MA/BA6 | SUSS MicroTec Group | ||
| Mixer Thinky ARE-250 | Thinky Corporation | ||
| NanoSight NS300 | Malvern Panalytical | NS300 | nanoparticle analyzer |
| Optical microscope Nikon Eclipse LV150N | Nikon Metrology NV | ||
| OSTE 322 Crystal Clear | Mercene Labs | ||
| PBS TABLETS.Ca/Mg free. Fisher Bioreagents. 100 g | Fisher Scientific | BP2944-100 | |
| PC membrane (50 nm pore diameter, 11.8% density) | it4ip S.A., Louvain-La Neuve, Belgium | ||
| Petri dishes, sterile | Sarstedt | 82.1472.001 | |
| Plasma Asher GIGAbatch 360 M | PVA TePla America, LLC | ||
| qEVoriginal/35 nm column | Izon | SP5 | SEC column |
| QSIL 216 Silicone Elastomer Kit | PP&S | ||
| Resin Tough Black | Zortrax | ||
| SW40 Ti swing ultracentrifuge rotor | Beckman Coulter | 331301 | |
| Syringe pump | DK Infusetek | ISPLab002 | |
| T175 suspension flask | Sarstedt | 83.3912.502 | |
| TIM4-Fc protein | Adipogen LifeSciences | AG-40B-0180B-3010 | |
| TMB (3,3',5,5'-tetramethylbenzidine) | SigmaAldrich | T0440-100ML | Horseradish peroxidase substrate |
| Tween20 | SigmaAldrich | P1379-100ML | |
| Ultracentrifuge Optima L100XP | Beckman Coulter | ||
| Ultrasonic cleaning unit P 60 H | Elma Schmidbauer GmbH | ||
| Universal Microplate Spectrophotometer | Bio-Tek instruments | 71777-1 | |
| Urine collection cup, 150mL, sterile | APTACA | 2120_SG | |
| Whatman Anotop 25 Syringe Filter | SigmaAldrich | 68092002 | |
| Zetasizer Nano ZS | Malvern Panalytical | dynamic light scattering (DLS) system | |
| Zortrax Inkspire | Zortrax |
Lab Prep
Extracellular vesicles (EVs) hold immense potential for various biomedical applications, including diagnostics, drug delivery, and regenerative medicine. Nevertheless, the current methodologies for isolating EVs present significant challenges, such as complexity, time consumption, and the need for bulky equipment, which hinders their clinical translation. To address these limitations, we aimed to develop an innovative microfluidic system based on cyclic olefin copolymer-off-stoichiometry thiol-ene (COC-OSTE) for the efficient isolation of EVs from large-volume samples in a continuous manner. By utilizing size and buoyancy-based separation, the technology used in this study achieved a significantly narrower size distribution compared to existing approaches from urine and cell media samples, enabling the targeting of specific EV size fractions in future applications. Our innovative COC-OSTE microfluidic device design, utilizing bifurcated asymmetric flow field-flow fractionation technology, offers a straightforward and continuous EV isolation approach for large-volume samples. Furthermore, the potential for mass manufacturing of this microfluidic device offers scalability and consistency, making it feasible to integrate EV isolation into routine clinical diagnostics and industrial processes, where high consistency and throughput are essential requirements.
Extracellular vesicles (EVs) hold immense potential for various biomedical applications, including diagnostics, drug delivery, and regenerative medicine. Nevertheless, the current methodologies for isolating EVs present significant challenges, such as complexity, time consumption, and the need for bulky equipment, which hinders their clinical translation. To address these limitations, we aimed to develop an innovative microfluidic system based on cyclic olefin copolymer-off-stoichiometry thiol-ene (COC-OSTE) for the efficient isolation of EVs from large-volume samples in a continuous manner. By utilizing size and buoyancy-based separation, the technology used in this study achieved a significantly narrower size distribution compared to existing approaches from urine and cell media samples, enabling the targeting of specific EV size fractions in future applications. Our innovative COC-OSTE microfluidic device design, utilizing bifurcated asymmetric flow field-flow fractionation technology, offers a straightforward and continuous EV isolation approach for large-volume samples. Furthermore, the potential for mass manufacturing of this microfluidic device offers scalability and consistency, making it feasible to integrate EV isolation into routine clinical diagnostics and industrial processes, where high consistency and throughput are essential requirements.
Procedura
Extracellular vesicles (EVs) hold immense potential for various biomedical applications, including diagnostics, drug delivery, and regenerative medicine. Nevertheless, the current methodologies for isolating EVs present significant challenges, such as complexity, time consumption, and the need for bulky equipment, which hinders their clinical translation. To address these limitations, we aimed to develop an innovative microfluidic system based on cyclic olefin copolymer-off-stoichiometry thiol-ene (COC-OSTE) for the efficient isolation of EVs from large-volume samples in a continuous manner. By utilizing size and buoyancy-based separation, the technology used in this study achieved a significantly narrower size distribution compared to existing approaches from urine and cell media samples, enabling the targeting of specific EV size fractions in future applications. Our innovative COC-OSTE microfluidic device design, utilizing bifurcated asymmetric flow field-flow fractionation technology, offers a straightforward and continuous EV isolation approach for large-volume samples. Furthermore, the potential for mass manufacturing of this microfluidic device offers scalability and consistency, making it feasible to integrate EV isolation into routine clinical diagnostics and industrial processes, where high consistency and throughput are essential requirements.
