Association Between Sleep Quality and Cognitive Symptoms in Patients with Major Depressive Disorder
Summary
Here, we present a protocol that investigates the relationship between cognitive symptoms and sleep quality in patients with major depressive disorder. The THINC-integrated tool and polysomnography were used to evaluate cognitive symptoms and sleep quality.
Abstract
Cognitive symptoms and sleep disturbance (SD) are common non-mood-related symptoms of major depressive disorder (MDD). In clinical practice, both cognitive symptoms and SD are related to MDD progression. However, there are only a few studies investigating the connection between cognitive symptoms and SD in patients with MDD, and only preliminary evidence suggests a significant association between cognitive symptoms and SD in patients with mood disorders. This study investigates the relationship between cognitive symptoms and sleep quality in patients with major depressive disorder. Patients (n = 20) with MDD were enrolled; their mean Hamilton Depression Scale-17 score was 21.95 (±2.76). Gold standard polysomnography (PSG) was used to assess sleep quality, and the validated THINC-integrated tool (the cognitive screening tool) was used to evaluate cognitive function in MDD patients. Overall, the results showed significant correlations between the cognitive screening tool’s total score and sleep latency, wake-after-sleep onset, and sleep efficiency. These findings indicate that cognitive symptoms are associated with poor sleep quality among patients with MDD.
Introduction
Major depressive disorder (MDD), a serious mood disorder that causes significant morbidity and mortality, affects over 300 million people worldwide1. MDD is characterized by low mood, reduced drive, loss of interest, and thoughts of self-harm or suicide2. Cognitive symptoms and sleep disturbance (SD) are two non-mood-related symptoms of MDD3,4. Previous studies have shown that the prevalence of SD is 60-90% in patients with MDD, while the prevalence of sleep disorders among these patients in the Chinese population is 85.22%5.
Although few studies have investigated the causal relationship between cognitive symptoms and SD, preliminary evidence suggests a significant association between sleep disorders and cognitive symptoms in individuals with MDD6. For example, Cha et al.3 found that subjective sleep quality predicted both subjective cognitive performance and objective cognitive impairments, which were partially mediated by depression severity. Biddle et al.7 also found that among older men with MDD, poor sleep efficiency (SE), measured by actigraphy, was associated with worse cognitive function independent of depression severity. The observed discrepancies may be caused by methodological differences.
Within this literature, researchers have used various tools to evaluate sleep and cognition. The Pittsburgh Sleep Quality Index (PSQI) was commonly selected as a subjective tool. Actigraphic devices have also been used. However, PSG, regarded as the gold standard for detecting sleep disorders8, is a sensitive, objective SD assessment9. To our knowledge, among the studies of the relationship between sleep and cognition in patients with MDD, few have used PSG to evaluate sleep quality.
Cognitive symptoms in MDD might be multidimensional and addressed in some cases by new pharmacological strategies10,11. Several cognitive assessment tools suitable for MDD have emerged in recent years12. THINC-integrated tool (THINC-it; henceforth referred to as the cognitive screening tool), a validated cognitive screening tool developed by Hou et al.13, is composed of a subjective questionnaire (perceived deficits questionnaire, PDQ-5) and four objective cognitive tests (choice reaction time, 1-back, digit symbol substitution, and trail making part-B). It is considered a time-efficient, acceptable method for detecting cognitive deficits in patients with MDD14. There are two other tools commonly used to assess cognitive function in patients with MDD: the Screen for Cognitive Impairments in Psychiatry-D (SCIP-D) and Cognitive Complaints in Bipolar Disorder Rating Assessment (COBRA)12. The former is an objective tool lacking self-instructed testing, and the latter is a subjective tool lacking an objective version. The constituent tests also differ between these tools. The SCIP-D is comprised of five objective tests: verbal learning, working memory, verbal fluency, delayed memory, and processing speed. The COBRA includes executive function, processing speed and attention, verbal learning and memory, and visual memory12. The recently developed Chinese version of the cognitive screening tool has been confirmed to be a good tool for evaluating cognitive deficits in Chinese patients with MDD15. Thus, using the cognitive screening tool software and PSG, the current study tried to reveal the relationship between cognitive symptoms and SD in MDD patients.
Protocol
The study was approved by the Academic Ethics Committee of the Inner Mongolia Mental Health Center (The Third Hospital of Inner Mongolia Autonomous Region, Brain Hospital of Inner Mongolia Autonomous Region) in Hohhot, China. An informed consent form was signed by all study participants.
1. PSG procedure
- Data collection
- Include patients with MDD who were diagnosed according to the DSM-V.
NOTE: This study included 22 patients with MDD who were diagnosed according to the DSM-V. Due to the low educational level, 2 patients with MDD refused to participate. Because both of them found it was not easy to read characters while completing TMT-B in the cognitive screening tool. - Exclude participants who were diagnosed with severe dementia, anxiety, delirium, and other diseases, which made them unable to complete PSG for the whole night.
- Collect participants' demographics, including name, nationality, gender, age, date of birth, marriage, complaints, symptoms, main diagnosis, history, department, temperature, pulse, respiratory rate, blood pressure, height, weight, body mass index, abdominal circumference, and upper chest circumference, were collected.
- Each participant was instructed to complete the Epworth Sleeping Scale16.
- Include patients with MDD who were diagnosed according to the DSM-V.
- Connecting derivations
- After collecting the participants' information, lead the participant to the sleep monitoring room.
- Collect all the derivations of electroencephalogram (EEG), electrooculogram (EOG), chin electromyogram (EMG), and electrocardiogram (ECG) step-by-step.
NOTE: The participant should keep his/her face, skin, and hair clean and dry. - Connect 15 electrodes of EEG according to the international 10-20 system of electrode placement (Figure 1)17.
- Connect two derivations of EOG (E1, E2) as shown in Figure 2.
- Connect three derivations of chin EMG (Chin1, Chin2, ChinZ) as shown in Figure 3.
- Connect two derivations of ECG (+, −) as shown in Figure 4.
- Starting the data acquisition system
- Double-click Device Manager to start the software.
- Double-click the Room option and add the personal information of the participant.
NOTE: The personal information includes name, gender, date of birth, file name, file path, etc. More details can be added as needed (for example, height, body weight, neck circumference, doctor, technician, hospital, address, marital status, profession, and guardian [if the participant is a child]). - Click the OK button.
- Click the Start Recording button.
- Click the Calibration button and instruct the participant to perform according to the step-by-step calibration procedure (see Supplementary Figure 1).
NOTE: The calibration step-by-step procedure is listed in Table 1. During the calibration, the remote call system should be turned on so the participant can be guided to perform according to the doctors' instructions.
- Collecting data all night long
- Keep the derivations in the correct position all night long.
NOTE: This will be done by the duty technician. The duty technician should have emergency management training for conditions like asthma, unstable angina, etc.
- Keep the derivations in the correct position all night long.
- Post calibration
- Awaken the participant and turn on the light of the sleep monitoring room.
- The wake-up time is usually at 6:00 am. Stop the monitoring if the participant wakes up before 6:00 am and does not want to continue.
- If the participant is in a rapid eye movement (REM) sleep period at 6:00 am, continue monitoring until the REM sleep period ends.
- Repeat step 1.3.5 once again for post-calibration.
- Click the Close button to stop recording.
- Remove all the derivations gently to avoid skin damage.
- Scoring
- Double-click the ProFusion PSG4 button to start the scoring system.
- Double-click the name of the participant to select the participant.
- Click the Scoring button and select the Scoring Automatically option to score sleep phases and arousals.
NOTE: Sleep phases and arousals were scored according to The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology, and Technical Specifications18. Scoring can be manually modified by a technician, too. - Click the Create button and layout the results (Figure 5).
- Definitions and equations
- Get variables including time in bed (TIB), total sleep time (TST), sleep latency (SL), wake after sleep onset (WASO), sleep efficiency (SE, equals TST/TB), and REM latency according to the PSG data and waves (as shown in Table 2).
2. Using the cognitive screening tool
- Double-click the THINC-it button to start the software on the computer, enter the personal information, and instruct the participant to complete each subtest step by step.
- Click the Chinese button to select the simplified Chinese version.
- Click the Plus button to add a new participant.
- Type in name, age, gender, dominant hand, educational level, and location at the corresponding blanks.
- Click the BEGIN TEST button to begin the subtests step by step (see Supplementary Figure 2).
- Complete the perception deficits questionnaire (PDQ-5, subjective cognitive deficits).
- Read aloud, "During the past 7 days, how often did you have trouble getting things organized?" and instruct the participant to choose one of the 5 response options: Never in the past 7 days, Rarely (once or twice), Sometimes (3 – 5 times), Often (about once a day), and Very often (more than once a day).
- Read aloud, "During the past 7 days, how often did you have trouble concentrating on what you were reading?" and instruct the participant to choose one of the 5 response options described in step 2.2.1.
- Read aloud, "During the past 7 days, how often did you forget the date unless you looked it up?" and instruct the participant to choose one of the 5 response options described in step 2.2.1.
- Read aloud, "During the past 7 days, how often did you forget what you talked about after a telephone conversation?" and instruct the participant to choose one of the 5 response options described in step 2.2.1.
- Read aloud, "During the past 7 days, how often did you feel like your mind went totally blank?" and and instruct the participant to choose one of the 5 response options described in step 2.2.1.
- Click the OK button to start other subtests.
- Complete the spotter test (SPO, reflection of attention).
- Click the Tutorial button and the Next button to read the step-by-step instructions.
- Click the Start button to begin the SPO test.
NOTE: The SPO test is used to assess attention; the participant needs to touch the suddenly appearing symbol as quickly as possible.
NOTE: This software will automatically show the Best Reaction Time (milliseconds), Correct Responses, Longest Streak, and Index on screen. - Click the OK button to begin the next subtest.
- Complete the symbol check test (SC, reflection of working memory).
- Click the Tutorial and Next buttons to read the step-by-step instructions.
- Click the Start button to begin the SC test.
NOTE: The SC test is used to assess working memory. The participant needs to identify the symbol hidden before the timer runs out and recall the next hidden symbol. This software will automatically show the Fastest Reaction Time (milliseconds), Correct Answers, Longest Streak, and Index on screen. - Click the OK button to begin the next subtest.
- Complete the code breaker test (CB, reflection of information processing speed).
- Click the Tutorial and Next buttons to read the step-by-step instructions.
- Click the Start button to begin the CB test.
NOTE: The CB test is used to assess information processing speed. The participant needs to click the correct symbol to fit the indicated circle (each symbol is identified into a certain number inside the circle). This software will show the Fastest Reaction Time (milliseconds), Correct Nodes, Longest Streak, as well as Index automatically on the screen. - Click the OK button to enter the next subtest.
- Complete the TMT-B (trails, reflection of executive function).
- Click the Tutorial and Next buttons to read the step-by-step instructions.
- Click the Start button to begin the CB test.
NOTE: Trails test is used to assess executive function. The participant needs to trace a line beginning from a character to number 1 without lifting his/her finger, then continue from the number 1 to another character, then continue from this character to number 2, and continue it till 9, as fast as possible. This software automatically shows the Completion Time (milliseconds), Errors, Longest Streak, and Index on screen. - Click the OK button to complete the last subtest.
NOTE: The software will automatically show participant performance on each test; download these results.
3. Statistical analysis
- Use the Students' t-tests to assess group differences. Perform the Pearson correlation analysis to evaluate the relation between sleep quality and cognitive function. Consider P < 0.05 as statistically significant.
NOTE: All statistical analyses were performed using SPSS version 25.0.
Representative Results
The male/female ratio was 11/9 for the participants. For all the individuals, the age reached 35.35 ± 6.83 years, educational level was 14.80 ± 3.29 years, Hamilton depression scale-17 (HAMD-17) score was 21.95 ± 2.76 points, SE was 76.71 ± 8.57%. Regarding the cognitive screening tool scores, PDQ-5, SPO, SC, CB, TMT-B, and total scores were 1756.35 ± 395.49, 2046.75 ± 684.45, 852.15 ± 651.81, 845.75 ± 641.15, 911.45 ± 619.13 and 4140.80 ± 1298.14 points, respectively (Table 2).
Figure 5 illustrates two representative participants. A female patient with MDD (Figure 5A), aged 49 years, had a lower THNIC-it score, including SPO score = 0, SC score = 0, CB score = 0, and TMT-B score = 680. She had higher SL (40.5 min), WASO (60.5 min), and lower SE (82.2%). A male patient with MDD (Figure 5B), aged 48 years, had a higher score, including SPO score = 1856, SC score = 1250, CB score = 163, and TMT-B score = 1484. He had higher SL (7.0 min), WASO (5.5 min), and lower SE (88.1%). The total score was significantly correlated with SL (r = −0.0856, p < 0.001), WASO (r = -0.687, p = 0.001), and SE (r = 0.081, p < 0.001).
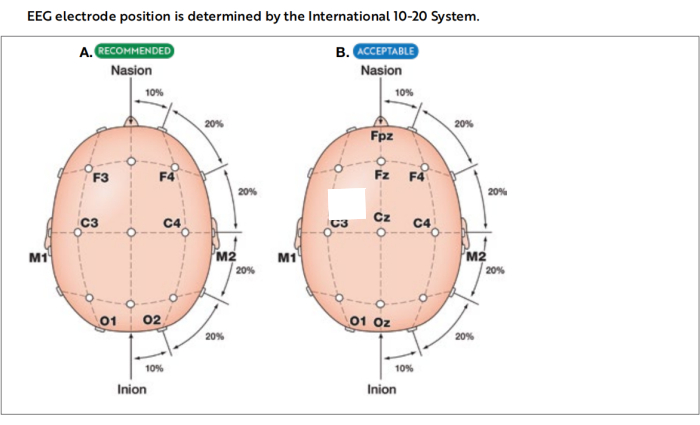
Figure 1: International 10-20 system of electrode placement. Images illustrating the placement of electrodes utilized in recommended (A) and acceptable (B) derivations for electroencephalography (EEG) during polysomnography. This figure has been adapted with permission from The AASM Manual for the Scoring of Sleep and Associated Events18. Please click here to view a larger version of this figure.
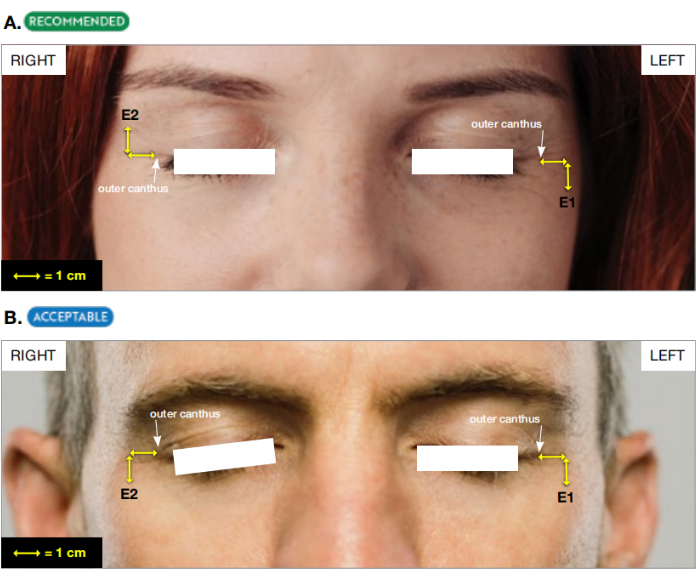
Figure 2: The placement of two derivations of EOG. (A) Recommended and (B) acceptable derivations for electrooculogram (EOG). This figure has been adapted with permission from The AASM Manual for the Scoring of Sleep and Associated Events18. Please click here to view a larger version of this figure.
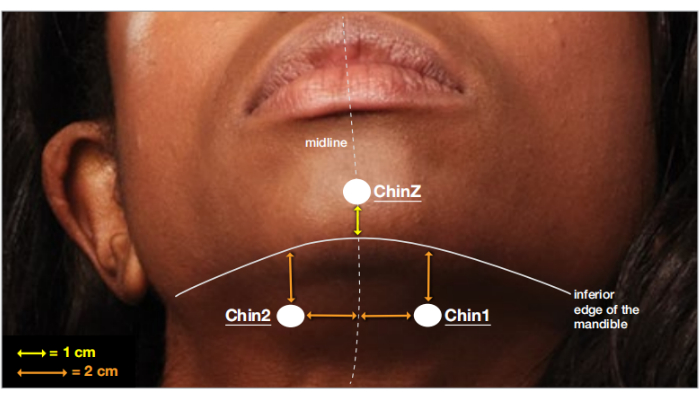
Figure 3: The placement ofthree derivations of chin EMG. Placement of electrodes on the chin for electromyogram (EMG) recording. This figure has been adapted with permission from The AASM Manual for the Scoring of Sleep and Associated Events18. Please click here to view a larger version of this figure.
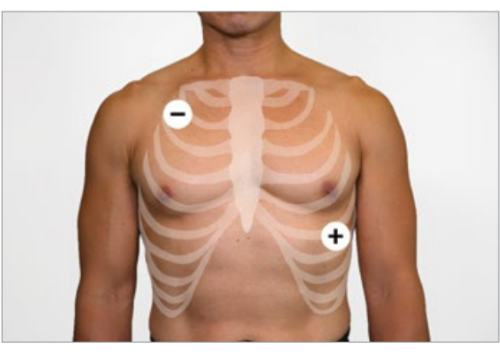
Figure 4: The placement of two derivations of ECG. The diagram shows Lead II placement on the torso during cardiac recording. This figure has been adapted with permission from The AASM Manual for the Scoring of Sleep and Associated Events18. Please click here to view a larger version of this figure.
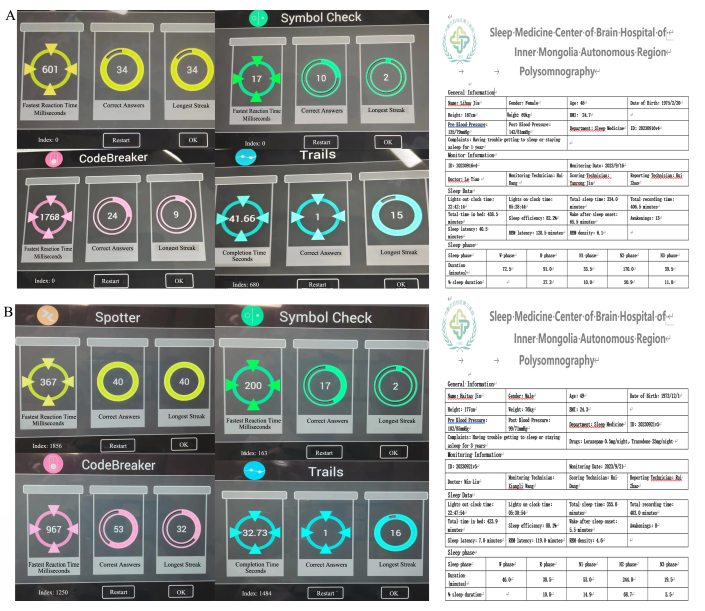
Figure 5. Representative participants. (A) A 49-year-old participant with a lower score also had higher SL (40.5 min), WASO (60.5 min), and lower SE (82.2%). (B) A 49-year-old participant with a higher THNIC-it score also had lower SL (7.0 min), WASO (5.5 min), and higher SE (88.1%). Please click here to view a larger version of this figure.
| Calibration steps to document appropriate system response. |
| 1. Perform and document an impedance check of the EEG and EMG electrodes. |
| 2. Record a minimum of 30 s of EEG with patients awake lying quietly with eyes open. |
| 3. Record a minimum of 30 s of EEG with patients awake lying quietly with eyes closed. |
| 4. Ask the patient to look up and down without moving head (5x). |
| 5. Ask the patient to look left and right without moving head (5x). |
| 6. Ask the patient to blink (5x). |
| 7. Ask the patient to grit teeth and/ or chew (5 s). |
| 8. Ask the patient to simulate a snore or hum (5 s). |
| 9. Ask the patient to breathe normally and assure that an airflow and effort channel signals are synchronized. |
| 10. Ask the patient to perform a breath hold (10 s). |
| 11. Ask the patient to breathe through the nose only (10 s). |
| 12. Ask the patient to breathe through the mouth only (10 s). |
| 13. Ask the patient to flex the left foot /rase toes on left foot (5x). |
| 14. Ask the patient to flex the right foot /rase toes on right foot (5x). |
| 15. Adjust ECG signal to provide a clear waveform-the R wave should deflect upward. |
| 16. Perform and document a repeat impedance check of the EEG, EOG and EMG electrodes at the end of the PSG recording. |
| 17. Repeat impedance measures and physiological calibrations at the end the PSG recording. |
Table 1: PSG calibration procedure. Abbreviations: EEG: Electroencephalogram; EMG: Electromyography; EOG: Electrooculogram; PSG: Polysomnography.
| Gender (male/female) | 11/9 |
| Age (years) | 35.35 ± 6.83 |
| Educational level (years) | 14.80 ± 3.29 |
| HAMD-17 (score) | 21.95 ± 2.76 |
| SE (%) | 76.71 ± 8.57 |
| PDQ-5 (score) | 1756.35 ± 395.49 |
| SPO (score) | 2046.75 ± 684.45 |
| SC (score) | 852.15 ± 651.81 |
| CB (score) | 845.75 ± 641.15 |
| TMT-B (score) | 911.45 ± 619.13 |
| THINC-it total score | 4140.80 ± 1298.14 |
Table 2: Patient demographics and scores. HAMD: Hamilton depression scale; SE: sleep efficiency; PDQ-5: perceived deficits questionnaire; SPO: spotter test; SC: symbol check test; CB: code breaker test; TMT-B: trail making test-B; THINC-it: THINC-integrated tool.
Supplementary Figure 1: Screenshot showing the calibration procedure. Please click here to download this File.
Supplementary Figure 2: Screenshot showing the cognitive screening tool subtests. Please click here to download this File.
Discussion
This two-part protocol has critical steps that demand attention. For PSG, scoring is the most important step; thus, PSG technicians need to be certified and experienced. In addition, before PSG, communication between technicians and participants is necessary because patients might feel uncomfortable and quit during PSG. For the cognitive screening tool, the critical step is evaluating the cognitive function with digitized equipment. Doctors need to introduce the procedures step by step to ensure the participant understands them. In addition, if a participant's poor performance is due to unfamiliarity with the instructions, repeating the task is allowed. Last but not least, as has been mentioned by Hou et al.13, the cognitive screening tool was not suitable for individuals with low intelligence quotient (IQ). Doctors should collect the history of the patients to exclude congenital low-intelligence individuals.
PSG is limited by the 'first night effect', which is induced by the novel laboratory environment. This usually causes increased PSG-measured sleep onset latency, increased REM latency, a lower percentage of REM sleep, and lower SE, and may lead to clinical misinterpretation19,20. Subjective sleep questionnaires like the PSQI, home-based PSG, and actigraphic devices are alternative methods for evaluating sleep quality. The main limitation of the Chinese version of the cognitive screening tool is the TMT-B1 that some less-educated Chinese participants are unfamiliar with traditional Chinese characters, which may be one reason for the non-significant correlation with this measure. Accordingly, it is recommended that a combination of the cognitive screening tool elements with stronger validity (i.e., PDQ-5, Spotter, and Codebreaker) should be used with Chinese patients with MDD.
In the current study, the recruited patients with MDD were first episode and drug naïve. In clinical practice, selective serotonin reuptake inhibitors (e.g., paroxetine) and serotonin and norepinephrine reuptake inhibitors (e.g., duloxetine) can impact sleep structure by reducing sleep contuinity21. Thus, more studies should be launched to elucidate the relation between cognitive symptoms and SD in patients with MDD receiving antidepressant treatment. Circadian rhythm is another point that should not be neglected in clinical practice. Circadian rhythmicity has been found to be associated with cognitive function and sleep quality22. It is recommended to use actigraphic devices to detect features of the MDD patients' circadian rhythm for 1 week before or after PSG.
The shortcomings of the present study need to be mentioned. First, the sample size was small. Second, circadian rhythm, which was reported to influence sleep quality, was not assessed. Because the cognitive screening tool includes executive function assessment, it can be used with those who have neurocognitive disorders characterized by this type of dysfunctions, such as cerebral small vessel disease and schizophrenia.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We want to thank Ms Wanting Li for the digital technology support. This work was supported by Inner Mongolia Medical Fund (2022QNWN0010, 2023SGGZ047, 2023SGGZ0010).
Materials
| Computer | Surface | Surface pro2 | |
| Derivation | Compumedics | Grael | |
| Electrode | Compumedics | Grael | |
| Polysomnography | Compumedics | Grael | |
| Software | Compumedics | Compumedics Profusion PSG4 |
References
- Liu, X., et al. Altered gamma oscillations and beta-gamma coupling in drug-naive first-episode major depressive disorder: Association with sleep and cognitive disturbance. J Affect Disord. 316, 99-108 (2022).
- Cha, D. S., et al. Perceived sleep quality predicts cognitive function in adults with major depressive disorder independent of depression severity. Ann Clin Psychiatry. 31 (1), 17-26 (2019).
- Waters, K. The clinical utility of newer antidepressant agents: Understanding the role in management of MDD. Ment Health Clin. 12 (5), 309-319 (2022).
- Chen, R. F., et al. Sleep disorder as a clinical risk factor of major depression: associated with cognitive impairment. Asian J Psychiatr. 76, 103228 (2022).
- Pearson, O., et al. The relationship between sleep disturbance and cognitive impairment in mood disorders: A systematic review. J Affect Disord. 327, 207-216 (2023).
- Biddle, D. J., Naismith, S. L., Griffiths, K. M., Christensen, H., Hickie, I. B., Glozier, N. S. Associations of objective and subjective sleep disturbance with cognitive function in older men with comorbid depression and insomnia. Sleep Health. 3 (3), 178-183 (2017).
- Li, Y., et al. Sleep 1uality evaluation based on Single-Lead wearable cardiac cycle acquisition device. Sensors. 23 (1), 328 (2022).
- Withrow, D., Roth, T., Koshorek, G., Roehrs, T. Relation between ambulatory actigraphy and laboratory polysomnography in insomnia practice and research. J Sleep Res. 28 (4), e12854 (2019).
- Orsolini, L., et al. Current and future perspectives on the major depressive disorder: Focus on the new multimodal antidepressant vortioxetine. CNS Neurol Disord Drug Targets. 16 (1), 65-92 (2017).
- De Berardis, D., et al. Adjunctive vortioxetine for SSRI-resistant major depressive disorder: a "real-world" chart review study. Braz J Psychiatry. 42 (3), 317-321 (2020).
- Fiorillo, A., et al. Assessment and management of cognitive and psychosocial dysfunctions in patients with major depressive disorder: A clinical review. Front Psychiatry. 9, 493 (2018).
- Hou, Y., et al. PSYCHOMETRIC properties of the Chinese version of the THINC-it tool for cognitive symptoms in patients with major depressive disorder. J Affect Disord. 273, 586-591 (2020).
- McIntyre, R. S., et al. The THINC-Integrated Tool (THINC-it) screening assessment for cognitive dysfunction: validation in patients with major depressive disorder. J Clin Psychiatry. 78 (7), 873-881 (2017).
- Han, H., et al. The relationship between cognitive dysfunction through THINC-Integrated Tool (THINC-it) and psychosocial function in Chinese patients with major depressive disorder. Front Psychiatry. 12, 763603 (2021).
- Chen, N. H., et al. Validation of a Chinese version of the Epworth sleepiness scale. Qual Life Res. 11 (8), 817-821 (2002).
- Swarnkar, V., Abeyratne, U. R., Hukins, C. Inter-hemispheric asynchrony of the brain during events of apnoea and EEG arousals. Physiol Meas. 28 (8), 869-880 (2007).
- Berry, R. B., et al. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications. Version 2.5. American Academy of Sleep Medicine. , (2018).
- McCall, C., McCall, W. V. Objective vs. subjective measurements of sleep in depressed insomniacs: first night effect or reverse first night effect. J Clin Sleep Med. 8 (1), 59-65 (2012).
- Tamaki, M., Nittono, H., Hayashi, M., Hori, T. Examination of the first-night effect during the sleep-onset period. Sleep. 28 (2), 195-202 (2005).
- Wichniak, A., et al. Effects of antidepressants on sleep. Curr Psychiatry Rep. 19 (9), 63 (2017).
- Zuurbier, L. A., et al. Cerebral small vessel disease is related to disturbed 24-h activity rhythms: a population-based study. Eur J Neurol. 22 (11), 1482-1487 (2015).

.
