Apoplast-Extraction Based Method to Improve the Purity of Plant Produced Recombinant Protein
Summary
The purification of recombinant proteins from plant systems is usually challenged by plant proteins. This work provides a method to effectively extract and purify a secreted recombinant protein from the apoplast of Nicotiana benthamiana.
Abstract
Plants are a newly developing eukaryotic expression system being explored to produce therapeutic proteins. Purification of recombinant proteins from plants is one of the most critical steps in the production process. Typically, proteins were purified from total soluble proteins (TSP), and the presence of miscellaneous intracellular proteins and cytochromes poses challenges for subsequent protein purification steps. Moreover, most therapeutic proteins like antigens and antibodies are secreted to obtain proper glycosylation, and the presence of incompletely modified proteins leads to inconsistent antigen or antibody structures. This work introduces a more effective method to obtain highly purified recombinant proteins from the plant apoplastic space. The recombinant Green fluorescent protein (GFP) is engineered to be secreted into the apoplast of Nicotiana benthamiana and is then extracted using an infiltration-centrifugation method. The GFP-His from the extracted apoplast is then purified by nickel affinity chromatography. In contrast to the traditional methods from TSP, purification from the apoplast produces highly purified recombinant proteins. This represents an important technological improvement for plant production systems.
Introduction
Nowadays, various plant-produced recombinant therapeutic proteins are under study, including antibodies, vaccines, bioactive proteins, enzymes, and small polypeptides1,2,3,4,5,6. Plants are becoming an increasingly utilized platform for producing therapeutic proteins due to their safety, low cost, and ability to rapidly scale production7,8. Protein expression and modification in plant systems, along with protein purification, are critical factors determining the productivity of plant bioreactors9,10. Enhancing plant productivity and obtaining high-purity target proteins are core challenges facing plant-based production systems11,12.
The subcellular localization and modification of recombinant proteins depends on the specific signal peptide encoded at the N-terminus, which targets the protein to its final organelle destination during gene expression. Recombinant proteins are designed to localize to the apoplast, endoplasmic reticulum (ER), chloroplast, vacuole, or cytoplasm, based on their unique properties13. For antigens and antibodies, secreted proteins are preferred. Before secretion, proteins progress through ER and Golgi for correct folding and post-translational modifications14,15,16.
After production in plants, recombinant proteins must be purified from plant extracts. Typically, proteins are purified from total soluble plant extracts, which contain abundant intracellular proteins, misfolded and unmodified products, and cytochromes17. To remove impurities, plant extracts undergo extensive fractionation, including ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO)-specific affinity chromatography, phytate precipitation, polyethylene glycol precipitation, and adjustment of temperature and pH18,19. Such impurity-removal steps are laborious and can compromise protein quality.
The plant cell compartment beyond the plasma membrane is defined as the apoplast, encompassing the cell wall, intercellular spaces, and xylem20. The aqueous phase is commonly called apoplast washing fluid (AWF). AWF contains molecules secreted by cells to regulate numerous biological progresses like transport, cell wall metabolism, and stress responses21,22. In contrast to TSP, the molecular constituents of AWF are less complex. Extracting recombinant proteins from AWF may avoid contamination by intracellular proteins, misfolded products, and cytoplasmic remnants. Briefly, extraction lipid enters leaves through the stomata under negative pressure, and the AWF is collected by gentle centrifugation23. While isolation of AWF is widely employed to study the biological progress inside apoplast24,25,26,27, it has not been exploited in plant-based production systems.
Improving plant productivity and obtaining high purity target proteins are central challenges for plant production systems28. Typically, proteins are purified from total soluble plant extracts containing large amounts of intracellular proteins, misfolded and unmodified products, and cytochromes29, which require great costs for subsequent purification30. The use of AWF protein extraction methods for the extraction of exogenous recombinant proteins secreted into apoplast can effectively improve the homogeneity of recombinant proteins and reduce the cost of protein purification. Here, we use a signal peptide PR1a to enable exogenous protein secretion into the apoplast space and introduce a detailed method for recombinant protein purification from the AWF of N. benthamiana.
Protocol
1. Preparation of N. benthamiana plants
- Place seedling blocks in a tray, add 1 L of water and soak for approximately 2 h until fully saturated.
- Evenly sprinkle wild-type N. benthamiana seeds on the seedling blocks, cover with a lid, and germinate for 1 week. Grow N. benthamiana in a greenhouse with 18 h photoperiod, 24 °C, 45% relative humidity, and fertilize every 2 weeks.
- Once the seeds have sprouted, use tweezers to gently remove a seedling, taking care not to cause damage to its roots. Dig a small pit in the center of a new tray, place the seedling in it, and cover the roots lightly with soil. Cover with a lid and continue to grow.
- Grow till the seedlings have three leaves, with the lid on. Remove the lid when the 4th true leaf emerges.
- Allow plants to continue growing for two more weeks before agroinfiltration.
2. Construction of the recombinant GFP-His
- Perform N. benthamiana codon optimization and synthesis of GFP and PR1a (MGFVLFSQLPSFLLVSTLLLFLVISHSCRAG) based on the gene sequences.
- Carry out double digestion of pCAMBIA1300 vector using Xba I and Sac I enzymes (see Table of Materials).
- Perform amplification of PR1a using upstream primers (TTGGAGAGAACACGGGGGACTCTAGAATGGGATT
TGTTCTCTTTTC) and downstream primers (TCCTCG
CCCTTGCTCACCATGTCGACCCCGGCACGGCAAGA
GTGGG), amplification of GFP using upstream primers (CCCACTCTTGCCGTGCCGGGGTCGACATGGTGAG
CAAGGGCGAGGA) and downstream primers (GGGGAAATTCGAGCTCTCAGTGGTGATGGTGGTG
GTGAGATCTTCCGGACTTGT). - Using homologous recombination, clone PR1a, and GFP into the pCAMBIA1300 plant expression vector. After transferring into E. coli, select single clones and send them to the company for sequencing. Comparison of sequencing results and the right plasmid is the recombinant plasmid pCAMBIA1300-PR1a-GFP-His, referred to as p35s-PR1a-GFP-His.
3. Production of GFP-His in N. benthamiana
- Add 1 µL of GFP plasmid into 50 µL of Agrobacterium receptor cells GV3101, mix gently and add to the electrode cup, remove after electrocution on the electroshocker.
- Spread transformed Agrobacterium tumefaciens on an LB solid medium plate containing 50 µg/mL kanamycin (Kan) and 50 µg/mL rifampicin (Rif). Incubate inverted at 28 °C for 48 h. The pCAMBIA1300 vector is kan-resistant, and Agrobacterium is rif-resistant; only the correct Agrobacterium can grow on plates with kan and rif. The monoclonal clones were the positive transformants, named A. tumefaciens GV3101-p35s-PR1a-GFP-His (AtGPG).
- Use a sterilized toothpick to pick up an appropriate amount of AtGPG and inoculate it into 4 mL of liquid LB with Kan and Rif. Culture at 28 °C, 220-300 rpm for 24 h.
- Subculture bacteria 1:50 into fresh LB (50 µg/mL Kan, 10 mM 2-(N-Morpholino) ethanesulfonic acid (MES), 20 µM acetosyringone (AS)) and incubate at 28 °C, 220-300 rpm for 10-12 h.
- Harvest bacteria by centrifugation at 1500 x g for 10 min and resuspend the pellet with 1 mL of MMA (10 mM MgCl2, 10 mM MES pH 5.6, 100 µM AS).
- Measure and adjust the optical density (OD) of AtGPG to OD600=0.8 using MMA. Incubate the bacteria for another 2-4 h at room temperature. Draw up the bacterial solution with a needleless syringe, press it against the back of the leaves lightly, and give pressure to penetrate the solution into the leaves. Inject only the middle 3-4 leaves per plant.
- Continue to grow N. benthamiana in the greenhouse for 3-4 days before harvesting leaves for protein extraction.
4. Observing subcellular localization of GFP
- After 48 h of agroinfiltration, select a flat leaf and use a 6 mm hole punch to clamp the blade for punching.
- Drip 30% sucrose solution onto a glass slide, place the leaf sample with the back facing upwards and cover it with a cover glass.
- Set the excitation wavelength to 488 nm and detect the green fluorescence at 490-550 nm after magnification of 400x. Take the first photo once the slide is prepared and another one 10 min later.
5. Extracting GFP from plant
- AWF extraction
- Collect the intact fresh N. benthamiana leaves expressing recombinant GFP at 3 days post agroinfiltration. Treat the leaf gently and maintain leaf integrity to avoid intracellular protein efflux. Use collected leaves for the next step immediately to avoid protein degradation.
- Rinse leaf blade with pre-cooled sterilized ddH2O to remove debris.
- Weigh fresh leaves and submerge 20 g leaves (abaxial side down) in 100 mL of pre-cooled 100 mM phosphate buffer (PB 100 mM Na2HPO4 and 100 mM NaH2PO4 were separately configured and mixed at a ratio of 2.2:1 pH 7.0) in a 200 mL wide-mouth beaker. Cover leaves with 100 mesh nylon fine mesh yarn (see Table of Materials) and a plastic plate.
NOTE: Pre-cooling prevents protein degradation. Buffer volumes can be adjusted based on the experimental scale. Ensure all leaves are immersed. - Place the beaker into a vacuum pump. Apply 0.8 MPa vacuum for 1 min, then quickly restore to atmospheric pressure.
- Remove leaves from the buffer and gently blot dry with absorbent paper. Roll up leaves, wrap with mesh yarn, and place 5 g of leaves into each 50 mL centrifuge tube. To collect AWF, position rolled leaves tip-side up in tubes with mesh yarn fastened by the tube caps.
- Centrifuge at 4 °C, 500 x g for 5 min. Remove leaves and collect the AWF at the bottom of the tube using a pipette. About 5 mL of AWF can be obtained from each tube.
NOTE: In order to minimize interference from intracellular proteins and to extract the maximum amount of AWF proteins, the extraction can be repeated 2x-3x, but not more than 3x.
- Total soluble protein extraction
- Grind 20 g leaves into fine powder in liquid nitrogen. Add extraction buffer (50 mM Tris-HCl pH 7.4, 150 mM NaCl, 10% glycerol, 1% Triton X-100, 0.034% β-mercaptoethanol (see Table of Materials)) at a 1: 2 ratio.
- Shake homogenate in a vertical mixer for 30 min at 4 °C. Centrifuge homogenate at 13,000 x g for 15 min at 4 °C.
- Transfer the supernatant to a new tube and repeat centrifugation for 15 min. Transfer the cleared supernatant to another tube.
- Filter the supernatant through a 0.22 µm filter (see Table of Materials) to obtain total soluble protein extract.
6. Purification of GFP-His with nickel (Ni) affinity chromatography
NOTE: Perform all steps at 4 °C.
- Add Ni sepharose excel resin (see Table of Materials) to a tube in a 1:1000 ratio relative to the protein extract volume from step 5.1.6. Resin volume equals 1/1000th of the protein extract volume.
- Wash Ni resin 3x with 10x resin volume of ddH2O and PB (pH 7.0) separately in turn and remove the supernatant.
- Incubate protein extract with Ni resin for 2 h to allow full binding. Centrifuge at 500 x g for 5 min to remove unbound proteins in the supernatant. Wash 3x with 20x resin volume of PB. Centrifuge at 500 x g for 5 min and remove the supernatant.
- Elute GFP-His by adding 10x resin volume of 250 mM imidazole (50 mM Tris-HCl pH 7.4, 150 mM NaCl, 10% glycerol, 250 mM imidazole) and collect the supernatant.
7. Coomassie blue staining of proteins
- Add 10 µL of 5x SDS loading buffer to 40 µL of protein samples, boil for 10 min, then run on 15% SDS-PAGE at 110 V for 60 min.
- Stain protein gels with Coomassie blue solution (0.1% R-250, 25% isopropanol, 10% Glacial acetic acid) for 2 h.
- Pour off the solution and wash with decolorizing solution for 2 h. Observe the gel; proteins on the gel will be colored blue.
8. Western blotting of proteins
- Add 10 µL of 5x SDS loading buffer to 40 µL of protein samples, boil for 10 min, then run on 15% SDS-PAGE at 110 V for 60 min.
- Transfer proteins to a 0.45 µm nitrocellulose membrane at 280 mA. Block the membrane with 5% skimmed milk for 1 h.
- Dilute the anti-His-tag antibodies at 1:5000 and incubate the membrane with the antibodies for 1 h. Use the tris-buffered saline and tween 20 (TBST) to wash the membrane 4x for 5 min.
- Dilute the anti-mouse antibodies at 1:10000 and incubate the membrane with the antibodies for 1 h. Use TBST to wash the membrane 4x for 5 min.
- Mix the developer (see Table of Materials) in the ratio of 1:1. Add 400 µL to the membrane and allow to react for 1 min. Take photographs after 1 min exposure with a visualizer.
9. Protein concentration assay
- Add 5 µL of protein to 1 mL of Bradford 1x dye reagent and react for 5 min.
- Determine the OD595 value after zeroing and calculate the protein concentration from the standard curve.
10. Protein quantification 31
- Perform Western blotting of protein samples together with standards.
- Perform quantification of GFP by ratio to standards using ImageJ 1.5.0 greyscale analysis.
Representative Results
GFP-His is targeted for secretion into the apoplastic space of N. benthamiana
GFP was cloned into the pCAMBIA1300 plasmid with an N-terminal PR1a signal peptide for secretion and a C-terminal His-tag for purification, generating p35s-PR1a-GFP-His (Figure 1A). The recombinant protein was transiently expressed in N. benthamiana and a 30 kDa band was detected by western blot using anti-His antibody at 3 days post agroinfiltration (Figure 1B). Plasmolysis is the treatment of leaves using a hypertonic solution to separate the cell wall from the cell membrane32. To determine whether GFP is located in the apoplast, we used 30% sucrose to make the leaves plasmolysis. Fluorescence microscopy illustrated the presence of GFP-His within the apoplast (Figure 1C). AWF proteins extracted by vacuum infiltration-centrifugation also contained GFP-His (Figure 1D). These results demonstrate the secretion of GFP-His into the AWF of N. benthamiana leaves, from which it can be extracted for downstream purification.
Protein purification from AWF
The efficacy of AWF extraction strongly influences the final GFP yield. Building on published AWF extraction protocols24,25,26,27, we tested various solutions to optimize the recovery of secreted GFP-His. Here, eight extraction solutions were examined: 25 mM Tris-HCl (pH 7.4), 100 mM NaH2PO4 (pH 4.5), 50 mM NaCl (pH 7.1), 40 mM KCl (pH 7.0), 20 mM CaCl2 (pH 5.7), 100 mM PB (pH 7.0), 10 mM MES (pH 5.6) and ddH2O (pH 7.0). PB extracted the greatest amount of AWF and GFP-His, exceeding other solutions by over 4x (Figure 2A,B). Comparing PB at varying pH revealed improved AWF and GFP-His extraction at neutral to mildly alkaline conditions (pH 6.0, 6.5, 7.0, 7.5, and 8.0; Figure 2C,D). Additionally, PB with concentrations of 50 mM, 100 mM, and 150 mM were compared for the extraction efficacy. Results indicated that 100 mM PB showed maximal efficacy for recovering both AWF and GFP-His (Figure 2E,F).
Based on these optimization experiments, 100 mM PB, pH 7.0, was selected for large-scale AWF extraction from N. benthamiana. Using this buffer, 495 µg of total AWF proteins per gram of fresh leaf material were recovered from the apoplast (Figure 2G). The extracted AWF contained 10 µg of GFP-His (Figure 2H), which corresponded to approximately 18% of the total GFP-His in the TSP (Figure 2I). Following Ni-affinity chromatography, GFP-His purity reached 84.3% from AWF extraction, substantially higher than the 44.9% purity achieved through total soluble protein extraction (Figure 2J).
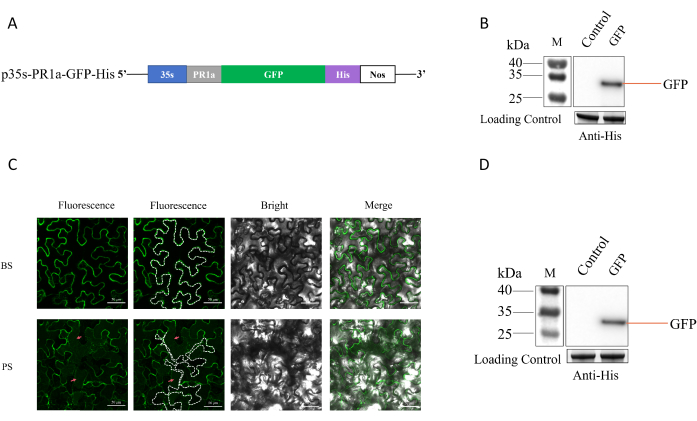
Figure 1: GFP-His is targeted for secretion into the apoplast of N. benthamiana. (A) Schematic of PR1a-GFP-His construction in the pCAMBIA1300 plant expression vector. (B) Western blot detecting GFP-His production in N. benthamiana. Here,10 µL of protein samples were loaded and detected. The N. benthamiana leaves simultaneously injected with MMA were used as a negative control. The same volume of samples was examined and stained using plant large subunits as loading control (C) Confocal microscopy showing the subcellular localization of GFP-His (green). N. benthamiana leaves were plasmolyzed with 30% sucrose. White dashed lines indicate the plasma membrane. Red arrows denote GFP-His accumulation in the apoplast. (D) Western blot demonstrating the presence of GFP-His in the extracted apoplastic fluid. Here,10 µL of protein samples were loaded and detected. The N. benthamiana leaves simultaneously injected with MMA were used as a negative control. Abbreviations: 35s = 2x Cauliflower mosaic virus 35s promoter; PR1a = tobacco pathogenesis-related protein 1a signal peptide; His = 6x His tag; Nos = nopaline synthase terminator. BS = before plasmolysis; PS = post-plasmolysis. Please click here to view a larger version of this figure.
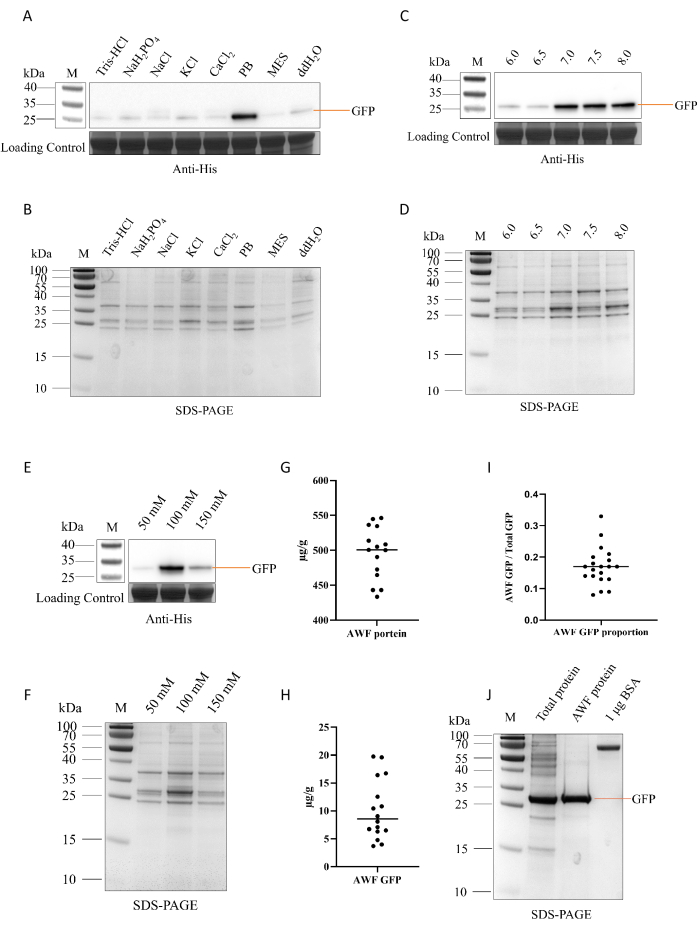
Figure 2: Optimization of the protein extraction method from AWF. (A-B) Extraction of proteins from AWF using different buffers. Eight solutions were tested for protein extraction from AWF: 25 mM Tris-HCl (pH 7.4), 100 mM NaH2PO4 (pH 4.5), 50 mM NaCl (pH 7.0), 40 mM KCl (pH 7.0), 20 mM CaCl2 (pH 5.7), 100 mM PB (pH 7.0), 10 mM MES (pH 5.6), and ddH2O (pH 7.0). The extraction efficiency was analyzed by SDS-PAGE (A), and GFP-His was detected by western blotting using anti-His antibody (B). (C-D) Extraction using PB at different pH values. PB with pH 6.0, 6.5, 7.0, 7.5, and 8.0 was used for extraction from AWF. The extraction efficiency was detected by SDS-PAGE (C), and GFP-His was detected by western blotting with anti-His antibody (D). (E-F) Extraction using PB at different concentrations. PB at 50 mM, 100 mM, and 150 mM were used for extraction from AWF. The extraction efficiency was analyzed by SDS-PAGE (E), and GFP-His was detected by western blotting with anti-His antibody (F). (G-H) Quantitative analysis of extracted proteins. Proteins were extracted using 100 mM PB (pH 7.0). The total soluble protein (G) and GFP-His (H) were quantified for statistical analysis. (I) Statistical analysis of the proportion of AWF GFP to total GFP. (J) Purification of GFP-His from total soluble proteins of N. benthamiana or AWF. Proteins were extracted using 100 mM PB (pH 7.0), and GFP-His was purified by Ni affinity chromatography. Purification was analyzed by SDS-PAGE. The rightmost lane is the 1 µg BSA standard. Please click here to view a larger version of this figure.
Discussion
Using plants to produce therapeutic proteins has expanded quickly in recent years1,2,3,4,5,6. Protein-encoding genes are cloned into expression vectors and delivered into plant tissues via agroinfiltration. After production by plant cells, proteins are purified for downstream applications. Typically, recombinant proteins are purified from TSP extracts. However, abundant plant proteins and cytochromes released from plant cells during extraction can hinder purification33. On the one hand, plant cytochromes can clog separation columns, slowing processing17; on the other hand, highly abundant proteins like RuBisCO are difficult to remove, reducing purity18,19. These issues become more problematic when the target proteins are expressed at low levels. Purifying from AWF bypasses these issues, enabling higher purity (Figure 2J).
While AWF extraction has been widely used in plant biology24,25,26,27, it is rarely exploited in molecular farming pipelines. Here, we use a plant-made GFP model to demonstrate AWF-based protein purification. Extraction of GFP from the AWF was performed by adding a signal peptide to enable GFP secretion into the apoplast space. This method extracts proteins with higher homogeneity and reduces downstream protein processing costs. Several considerations merit future investigation for implementing AWF purification in plant bioreactors. First, efficient secretion requires the fusion of specific N-terminal signal peptides. Second, to preserve AWF quality, intact leaves must be processed rapidly after harvest without freezing, which releases intracellular contents. Third, PB effectively extracts AWF proteins34 but buffer pH and salt concentration should be tailored for individual recombinant proteins (Figure 2C-D). Fourth, we achieved purified protein from 200 g of N. benthamiana many times in laboratory conditions with stable purity and yield. To be scaled up for use in the factory, larger vessels and vacuum pumps and a more effective purification method are asked, and conditions such as vacuum duration, number of recoveries, etc., may need to be optimized.
This method is limited by the fact that exogenous proteins are secreted into the AWF, and proteins inside the plasma membrane are deserted (Figure 2H-I). It brings the benefit of getting higher purified proteins with correct modification but at a higher cost of yield. The major technical bottleneck for AWF-based purification is the improvement of the target protein level inside AWF. Coupling with strong promoters and optimized secretion systems will improve plant expression platforms for both high purity and greater yields. To overcome this bottleneck, a higher production system and a more effective secretion system are asked for in future work.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work is supported by the Youth Innovation Promotion Association CAS (2021084) and the Key Deployment Project Support Fund of the Three-Year Action Plan of the Institute of Microbiology, Chinese Academy of Sciences.
Materials
| 100 mesh nylon fine mesh yarn | Guangzhou Huayu Trading limited company | hy-230724-2 | For AWF protein extraction |
| 50 ml centrifuge tubes | Vazyme | TCF00150 | For centrifuge |
| ClonExpress II one step cloning kit | Vazyme | C112-02 | For clone |
| FastDigest Sac1 | NEB | R3156S | For clone |
| FastDigest XbaI | NEB | FD0685 | For clone |
| ImageJ 1.5.0 | / | / | For greyscale analysis |
| Large filter | Thermo Fisher | NEST | For filtering |
| Laser scanning confocal microscopy | Leica SP8 | For subcellular localization | |
| Ni sepharose excel | Cytiva | 10339806 | For protein purification |
| Precast protein plus gel | YEASEN | 36246ES10 | For SDS-PAGE |
| Small filter | Nalgene | 1660045 | For filtering |
| SuperSignal West Femto Substrate Trial Kit | Thermo Fisher | 34076 | For western blotting |
| Triton X-100 | SCR | 30188928 | To configure the extraction buffer |
| Type rotaryvang vacuum pump | Zhejiang taizhouqiujing vacuum pump limited company | 2XZ-2 | For vacuum infiltration |
| Vertical mixer | Haimen Qilinbel Instrument Manufacturing limited company | BE-1200 | For mixing |
| β-mercaptoethanol | SIGMA | BCBK8223V | To configure the extraction buffer |
References
- Zahmanova, G., et al. Green biologics: Harnessing the power of plants to produce pharmaceuticals. Int J Mol Sci. 24 (24), 1757 (2023).
- PREVAIL Writing Group. A randomized, controlled trial of ZMapp for Ebola virus infection. N Engl J Med. 375 (15), 1448-1456 (2016).
- Ma, J. K. C., et al. Regulatory approval and a first-in-human phase I clinical trial of a monoclonal antibody produced in transgenic tobacco plants. Plant Biotechnol J. 13 (8), 1106-1120 (2015).
- Shanmugaraj, B., Rattanapisit, K., Manopwisedjaroen, S., Thitithanyanont, A., Phoolcharoen, W. Monoclonal antibodies B38 and H4 produced in Nicotiana benthamiana neutralize SARS-CoV-2 in vitro. Front Plant Sci. 11, 589995 (2020).
- Takeyama, N., Kiyono, H., Yuki, Y. Plant-based vaccines for animals and humans: Recent advances in technology and clinical trials. Ther Adv Vaccines. 3 (5-6), 139-154 (2015).
- Ward, B. J., Séguin, A., Couillard, J., Trépanier, S., Landry, N. Phase III: Randomized observer-blind trial to evaluate lot-to-lot consistency of a new plant-derived quadrivalent virus like particle influenza vaccine in adults 18-49 years of age. Vaccine. 39 (10), 1528-1533 (2021).
- Siriwattananon, K., et al. Plant-produced receptor-binding domain of SARS-CoV-2 elicits potent neutralizing responses in mice and non-human primates. Front Plant Sci. 12, 682953 (2021).
- Leblanc, Z., Waterhouse, P., Bally, J. Plant-based vaccines: The way ahead. Viruses. 13 (1), 5 (2020).
- Sethi, L., Kumari, K., Dey, N. Engineering of plants for efficient production of therapeutics. Mol Biotechnol. 63 (12), 1125-1137 (2021).
- Webster, D. E., Thomas, M. C. Post-translational modification of plant-made foreign proteins; glycosylation and beyond. Biotechnol Adv. 30 (2), 410-418 (2012).
- Streatfield, S. J. Approaches to achieve high-level heterologous protein production in plants. Plant Biotechnol J. 5 (1), 2-15 (2007).
- Nandi, S., et al. Process development and economic evaluation of recombinant human lactoferrin expressed in rice grain. Transgenic Res. 14 (3), 237-249 (2005).
- Ullrich, K. K., Hiss, M., Rensing, S. A. Means to optimize protein expression in transgenic plants. Curr Opin Biotechnol. 32, 61-67 (2015).
- Su, H., et al. Plant-made vaccines against viral diseases in humans and farm animals. Front Plant Sci. 14, 1170815 (2023).
- Agrawal, G. K., Jwa, N. S., Lebrun, M. H., Job, D., Rakwal, R. Plant secretome: Unlocking secrets of the secreted proteins. Proteomics. 10 (4), 799-827 (2010).
- Kim, T., Gondré-Lewis, M. C., Arnaoutova, I., Loh, Y. P. Dense-core secretory granule biogenesis. Physiology. 21, 124-133 (2006).
- Menkhaus, T. J., Bai, Y., Zhang, C., Nikolov, Z. L., Glatz, C. E. Considerations for the recovery of recombinant proteins from plants. Biotechnol Prog. 20 (4), 1001-1014 (2004).
- Kim, S. T., Cho, K. S., Jang, Y. S., Kang, K. Y. Two-dimensional electrophoretic analysis of rice proteins by polyethylene glycol fractionation for protein arrays. Electrophoresis. 22 (10), 2103-2109 (2001).
- Xi, J., et al. Polyethylene glycol fractionation improved detection of low-abundant proteins by two-dimensional electrophoresis analysis of plant proteome. Phytochemistry. 67 (21), 2341-2348 (2006).
- Dani, V., Simon, W. J., Duranti, M., Croy, R. R. D. Changes in the tobacco leaf apoplast proteome in response to salt stress. Proteomics. 5 (3), 737-745 (2005).
- Delaunois, B., et al. Uncovering plant-pathogen crosstalk through apoplastic proteomic studies. Front Plant Sci. 5, 249 (2014).
- Andreasson, E., Abreha, K. B., Resjö, S. Isolation of apoplast. Methods Mol. Biol. 1511, 233-240 (2017).
- Lohaus, G., Pennewiss, K., Sattelmacher, B., Hussmann, M., Hermann, M. K. Is the infiltration-centrifugation technique appropriate for the isolation of apoplastic fluid? A critical evaluation with different plant species. Physiol Plant. 111 (4), 457-465 (2001).
- Nouchi, I., et al. Overcoming the difficulties in collecting apoplastic fluid from rice leaves by the infiltration-centrifugation method. Plant Cell Physiol. 53 (9), 1659-1668 (2012).
- Rumyantseva, N. I., Valieva, A. I., Kostyukova, Y. A., Ageeva, M. V. The effect of leaf plasticity on the isolation of apoplastic fluid from leaves of tartary buckwheat plants grown in vivo and in vitro. Plants. 12 (23), 4048 (2023).
- Ekanayake, G., Gohmann, R., Mackey, D. A method for quantitation of apoplast hydration in Arabidopsis leaves reveals water-soaking activity of effectors of Pseudomonas syringae during biotrophy. Sci Rep. 12 (1), 18363 (2022).
- Preston, G. M., Fones, H. N., Mccraw, S., Rico, A., O’leary, B. M. The infiltration-centrifugation technique for extraction of apoplastic fluid from plant leaves using phaseolus vulgaris as an example. J Vis Exp. (94), e52113 (2014).
- Nandi, S., et al. Process development and economic evaluation of recombinant human lactoferrin expressed in rice grain. Transgenic Res. 14 (3), 237-249 (2005).
- Menkhaus, T. J., et al. Considerations for the recovery of recombinant proteins from plants. Biotechnol Prog. 20 (4), 1001-1014 (2004).
- Streatfield, S. J. Approaches to achieve high-level heterologous protein production in plants. Plant Biotechnol J. 5 (1), 2-15 (2007).
- Yamamoto, T., et al. Improvement of the transient expression system for production of recombinant proteins in plants. Sci Rep. 8 (1), 4755 (2018).
- Zhao, J., et al. Wheat Apoplast-localized lipid transfer protein TaLTP3 enhances defense responses against Puccinia triticina. Front Plant Sci. 12, 771806 (2021).
- Venkataraman, S., et al. Recent advances in expression and purification strategies for plant made vaccines. Front Plant Sci. 14, 1273958 (2023).
- Witzel, K., Shahzad, M., Matros, A., Mock, H. P., Mühling, K. H. Comparative evaluation of extraction methods for apoplastic proteins from maize leaves. Plant Meth. 7, 48 (2011).

.
