An Intact Pericardium Ischemic Rodent Model
Summary
This protocol outlines the steps for inducing myocardial infarction in mice while preserving the pericardium and its contents.
Abstract
This protocol has shown that the pericardium and its contents play an essential anti-fibrotic role in the ischemic rodent model (coronary ligation to induce myocardial injury). The majority of pre-clinical myocardial infarction models require the disruption of pericardial integrity with loss of the homeostatic cellular milieu. However, recently a methodology has been developed by us to induce myocardial infarction, which minimizes pericardial damage and retains the heart's resident immune cell population. An improved cardiac functional recovery in mice with an intact pericardial space following coronary ligation has been observed. This method provides an opportunity to study inflammatory responses in the pericardial space following myocardial infarction. Further development of the labeling techniques can be combined with this model to understand the fate and function of pericardial immune cells in regulating the inflammatory mechanisms that drive remodeling in the heart, including fibrosis.
Introduction
To this day, cardiovascular disease (CVD) is recognized as the leading cause of death globally, resulting in a significant financial burden and reduction in patient quality of life1. Coronary artery disease (CAD) is a sub-type of CVD and plays an essential role in the development of myocardial infarction (MI), which is a chief contributor to mortality. By definition, MI results from irreversible injury to the myocardial tissue due to prolonged conditions of ischemia and hypoxia. Myocardial tissue lacks regeneration capacity, so injuries are permanent and result in the replacement of heart muscle with a fibrotic scar that can be initially protective but ultimately contributes to adverse cardiac remodeling and eventual heart failure2.
Although the management of patients with CAD has dramatically improved over the past few decades, chronic heart failure (CHF) secondary to ischemia affects many patients worldwide. For preventing and managing this epidemic, it is necessary to understand the underlying mechanisms more extensively and develop new therapeutic approaches. Moreover, past findings highlight the limitations of systemic therapy and the necessity of developing precise alternatives. Given investigating the molecular sequelae of MI in humans is affected by the ability to access infarcted tissue, animal models that recapitulate the characteristics and development of human MI and CHF related to CVD are indispensable.
As ideal animal models closely resemble a human disorder for structural and functional characteristics, disease etiology should guide their conception. In CAD, it is the chronic atherosclerotic stenosis of coronary arteries or acute thrombotic occlusion. Different methods have been developed and applied in various species of laboratory animals to induce coronary artery narrowing or occlusion. Such strategies can be broadly classified into two groups: (1) mechanical manipulation of a coronary artery to induce an MI and (2) expediting atherosclerosis to facilitate coronary narrowing leading to an MI. The first strategy usually involves either the ligation of a coronary artery or the placement of a stent within the artery. The second approach tends to rely on modifying the animal's diet to include high fat/cholesterol food. Some of the limitations of this latter approach include the lack of control on the timing and site of coronary occlusions.
In contrast, the surgical induction of MI or ischemia in an animal model has several advantages, such as location, precise timing, and extent of the coronary event, leading to more reproducible results. The most widely used method is surgical ligation of the left anterior descending coronary artery (LAD). Such models recapitulate human responses to acute ischemic injury, as well as the progression to CHF3. Initially developed in larger animals, LAD surgery on small animals such as rodents has become more feasible with advancements in technology4. In establishing such models, mice have been favored for various reasons, including their relative availability, low expense in housing, and their capacity for genetic manipulation.
Contemporary surgical models of ischemic heart disease using LAD occlusion require the researcher to open the pericardium to temporarily or permanently ligate the artery5. Such strategies result in the disruption of the pericardial space, which plays an essentially mechanical and lubricating function to ensure proper cardiac function. Another disadvantage of opening the pericardium is losing the animal's native pericardial fluid with its various cellular and protein components6,7. In response, a method to induce MI while keeping the pericardium intact was developed by us. In addition to minimizing the perturbation of this homeostatic environment, this approach allows for tagging and tracing specific cells after causing an MI. In addition, this approach better represents myocardial ischemic injury in the human setting.
Protocol
Male and female C57BL/6J mice between 8-14 weeks of age were used for these experiments. This protocol has received ethical approval from the Animal Care Committee at the University of Calgary and follows all animal care guidelines.
1. Mouse preparation and surgery
- Sterilize surgical tools (via bead sterilizer or autoclave).
- Weigh mouse for presurgical weight and analgesic dose.
- Place the mouse in an induction box with 4% isoflurane and 800 mL/min of oxygen. Confirm the anesthetic plane by pinching the toes and observing the animal for lack of reflex.
- Inject analgesic subcutaneously (0.1 mg/kg of Buprenorphine) (see Table of Materials).
- Place the mouse on a heated surgical pad during surgery and maintain anesthesia with 3% isoflurane delivered via nose cone. Apply ophthalmic ointment to avoid dry eyes to each eye.
- Shave the hair from the chest and neck surgical areas.
- Restrain the paws of the mouse and position them on the surgical table.
- Intubate the mouse by inserting a smooth tipped 23 G catheter into the trachea through the mouth and the pharynx.
- Ventilate the mouse following intubation with 2% isoflurane and 100% oxygen as a carrier gas using a commercial Ventilator (see Table of Materials) set at a rate of 110 breaths/min, a tidal volume of 250 µL, and positive end-expiratory pressure (PEEP) of 4 mmHg.
- Roll the mouse 30% on its right side to position the left side of the chest for surgery.
- Clean the surgical area with 3 alternating scrubs of 70% ethanol and betadine in a circular motion(seeTable of Materials). Place sterile surgical drapes around the surgical area.
- Make a 2-3 cm lateral incision in the skin of the chest to visualize the pectoralis muscles on the left side. Cut the pectoralis major and minor using a 1 cm incision from the midline outward to visualize the intercostal muscles between the third and fourth ribs.
NOTE: Care should be taken to avoid excess bleeding from the pectoralis muscle through cauterization of bleeding vessels. - Make a 2 cm incision in the left intercostal muscle to introduce air (by passive air movement) into the chest cavity to allow the heart and lungs to fall away from the surgical incision. Further, expand the opening with the help of a cautery device (see Table of Materials) to incise the intercostal and prevent bleeding
NOTE: The ventilator should be temporarily stopped during the period of cauterization to avoid explosive reactions with oxygen. Care is taken not to damage the pericardial sac. - Using retractors, retract the ribs to expose the heart.
- Observe the pericardium and the underlying heart under a stereomicroscope.
NOTE: The mouse pericardium is thin enough to visualize the vasculature of the heart. - Gently place forceps on the surface of the pericardium to reduce its movement and that of the underlying heart.
- Visually landmark the left anterior descending (LAD) coronary artery by tracing its emergence from under the left appendage.
- Using the micro-needle driver, guide an appropriate suture (see Table of Materials) through the pericardium, under the LAD, with the suture emerging on the other side of the LAD and pericardium. Tie the suture to restrict the blood flow through the coronary artery and trim the excess suture with the help of scissors (Figure 1A).
NOTE: Upon restricting blood flow to the coronary artery, blanching of the anterior portion of the left ventricle should be visible. This procedure represents a permanent ligation model. However, a transient ligation approach with different ischemia periods could also be applied at this stage. - Below the incision site within the sterile area, introduce a 24 G catheter percutaneously into the chest (remove the guide needle after entering the chest cavity). Then, close the ribs followed by muscle layers and skin using an appropriate suture (a taper needle for muscle, conventional cutting needle for skin).
- Once the chest is closed, evacuate the remaining air from the chest cavity via the 24 G catheter using gentle suction with a 3 mL syringe and chest compressions. Once the air is removed, withdraw the 24 G catheter.
- Reduce the isoflurane to 1%.
- Turn off the isoflurane while maintaining ventilation with oxygen to allow the mouse to recover from anesthesia. Once the animal shows signs of breathing independently, remove the 23 G tracheal tube from the mouth and place the mouse in a recovery cage to be monitored for resumption of normal breathing.
- Allow the mouse to recover in the cage with a portion of the cage placed on a warming pad to provide an external heat source.
- Provide maintenance injections of analgesic (Buprenorphine 0.1 mg/kg, subcutaneously) every 12 h for 72 h post-surgery.
- Monitor the health status of mice daily for 7 days, which includes evaluating incisions and animal discomfort.
NOTE: Due to the invasiveness of this procedure (thoracotomy), the administration of antibiotics may be necessary.
2. Functional assessment of cardiac function by echocardiography (ECG)
- Induce and maintain the mouse under general anesthesia with 1.5-2% isoflurane and 800 mL/min of oxygen.
- Place mouse in a supine position on a heated stage platform and attach the paws to the ECG leads.
- Shave the chest of the mouse.
- Acquire echocardiography images using a 40 MHz linear transducer probe and contact gel and analyze with the appropriate software (see Table of Materials).
- Turn off the isoflurane and allow the mouse to recover on the heating platform before returning the cage to an active state.
NOTE: Echocardiography assessment is non-invasive and thus can be performed longitudinally throughout the experiment to determine changes before and after coronary ligation.
3. Heart tissue collection for fibrosis staining
- Sacrifice the mice with inhalation of 100% CO2 and carefully dissect out the heart.
NOTE: Using scissors and forceps, this is achieved by cutting through the large vessels entering (vena cava, pulmonary vein) and exiting (pulmonary artery, aorta) the heart to release it from the circulatory system in the thoracic cavity. - Fix the heart in 10% formalin for at least 24 h.
- Cut samples using a straight razor blade through the right ventricle, interventricular septum, and left ventricle, ensuring that the incision went through the center of the infarct zone. Samples are then sent to the core facility for paraffin embedding.
- Cut tissue sections of 5 µm thickness with a microtome and place them on glass slides for staining.
- Deparaffinize using commercial xylene and graded alcohol washes (2x 99%, 1x 95%, 1x 70%) with deionized water, then rehydrate.
- Stain with 0.1% Sirius red in picric acid for 2 h at room temperature.
- Wash sections with 0.5% acetic acid for 3 min and rinse with 70% ethanol for 1 min.
- Dehydrate the sections using the reverse order of washes outlined in 3.4, with increasing and graded ethanol concentrations then xylene.
- Mount tissue sections with a mounting solution (see Table of Materials) for microscopic evaluation.
4. Flow cytometry of heart and pericardial cavity lavage
- Sacrifice the mice with inhalation of 100% CO2 to effect.
- Place the mouse on its back and fix the arms and legs to a surgical board using tape.
- Carefully open the left side (right side from the experimenter view) of the thoracic cavity, starting with cutting the diaphragm to about the midpoint and subsequently cutting through the outside ribs towards the sternum.
NOTE: Avoid nicking large blood vessels, particularly those that run parallel to the sternum. - Retract the ribs using a hemostat to expose the underlying heart and pericardium.
NOTE: The pericardium is very fragile, so be sure not to catch it with scissors during the cutting. - Using a PE-10 tubing (see Table of Materials) catheter inserted into the pericardial space near the junction of the left atrium and left ventricle, inject 100 µL of sterile saline into the pericardial cavity.
- Allow saline to pool and collect from the posterior side of the heart, being careful not to puncture or tear the pericardium in the process. Repeat this step twice and place lavage fluid on ice while processing the heart.
- Excise the heart by cutting the major vessels (aorta, pulmonary artery, vein, and vena cava) entering and exiting the heart. Remove the right and left atria and weigh the ventricular heart tissue.
- Mince the tissue in small 1 mm2 pieces using scissors and place in 10 mL of digestion buffer containing 450 U/mL of collagenase I, 125 U/mL of collagenase XI, 60 U/mL of DNase I, and 60 U/mL of hyaluronidase in PBS for 1 h at 37 °C on an orbital shaker.
- Pass heart tissue homogenates through a 70 µm cell strainer (see Table of Materials) and spin down at 60 x g for 5 min at 4 °C to remove cardiac parenchymal cells.
- Collect the supernatant, pass through a 40 µm cell strainer (see Table of Materials) for a single cell suspension, and spin down at 400 x g for 5 min at 4 °C to pellet the cells.
- Perform fragment crystallizable (Fc) receptor blocking and staining of cellular markers on pericardial and cardiac cells as previously described8.
- Run samples on a flow cytometer.
5. Labeling pericardial macrophage using the Intercostal Approach to the Pleural Space (ICAPS) method9
- Sterilize surgical tools (via bead sterilizer or autoclave) and spray 70% ethanol before commencing.
- Induce and maintain the mouse under general anesthesia with 1.5-2% isoflurane and 800 mL/min of oxygen. Confirm the anesthetic plane by pinching the toes and observing the animal for lack of reflex.
- Inject analgesic subcutaneously (Buprenorphine 0.1 mg/Kg).
- Place the mouse on a heated surgical pad during surgery.
- Shave the right anterolateral thoracic area.
- Clean the surgical area with ethanol and betadine.
- Make a 3 cm long incision in the skin, and with forceps expose the rib cage.
- Load 5 µL of fluorescent beads (commercially available fluorescent microspheres, 1 µm, see Table of Materials) and 45 µL of saline into a PE-10 tubing syringe catheter with a beveled tip.
- Guide the catheter into the intercostal space as previously described9, slowly inject the bead solution and remove the catheter in one motion.
- Close the skin using staples.
NOTE: Staples are used in place of sutures to minimize potential re-opening of the incision. - Turn off the isoflurane, place the mouse in the recovery cage and monitor for complications over the first 24 h.
Representative Results
This modified coronary ligation model has been optimized to achieve reproducibility and animal survival. However, due to the significant injury induced in the heart, some expected intra-operative and post-operative mortality are associated with the procedure. The standard mortality is typically higher in males (~25-35%) than in females (~ 10-15%).
Successful induction of an MI with the modified coronary ligation should be evident by changes in the heart's functional parameters and structural features. For function, decreases in parameters such as left ventricle (LV) ejection fraction as assessed by echocardiography will be noticeable within 3-4 weeks post-MI (Figure 1A). These functional changes should be accompanied by significant fibrosis of the free wall of the LV as assessed by histological staining such as picrosirius red (PR) (Figure 1B). For this analysis, the use of longitudinal cross-sections through the infarcted should allow representation of the infarcted area, peri-infarct and remote zones of the heart.
Maintaining an intact pericardium throughout the procedure provides an opportunity to study the concurrent inflammatory response in the pericardial cavity. It also allows for determining how immune cells within this compartment can contribute to ongoing remodeling processes. Combining the fluorescent bead labeling method with flow cytometry analysis provides one approach to track with high selectivity resident Gata6+ pericardial macrophages (GPCMs). This procedure involves injecting beads directly into the pleural space. These are equally taken up by resident Gata6 macrophages in both the pleural and pericardial cavities (Figure 2A) due to communication between this two cavities10. Importantly, little to no labeling should be detected in the heart or blood (Figure 2A). Once labeled, the relocation of the cells following inflammatory challenges such as MI can be tracked by flow cytometry (Figure 2B) and/or imaging. For avoiding any potential inflammatory effects from the ICAPS procedure, this labeling should be performed one week before subsequent interventions.
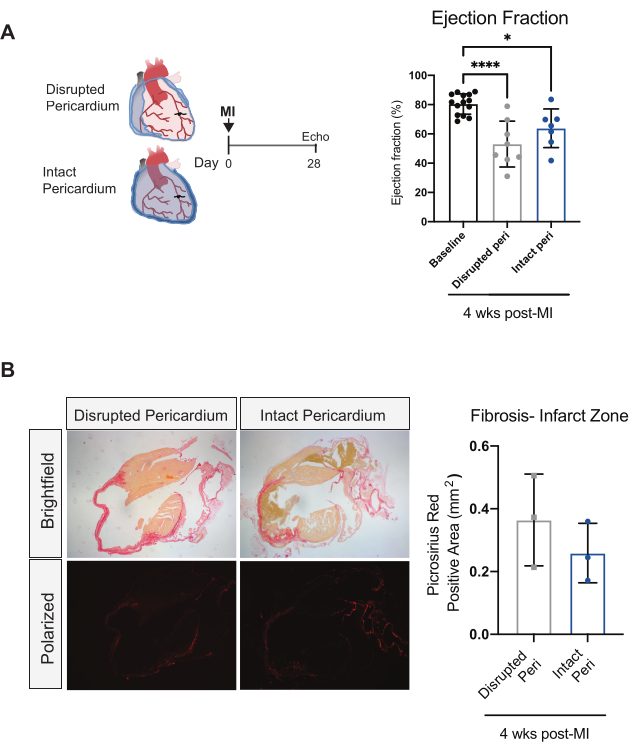
Figure 1: Intact pericardial coronary ligation model induces functional and structural alterations in the heart. (A) Schematic timeline and LV ejection fraction quantification at baseline or 4 weeks post-coronary ligation for animals with disrupted or intact pericardium. Data are represented as mean ± SD. ***= p < 0.001, *= p < 0.05 vs baseline, one-way ANOVA. Adapted from Deniset JF, et al., with permission from Elsevier8. (B) Representative images and quantification of picrosirius red fibrosis staining in mouse heart cross-sections at 4 weeks post-infarct with the disrupted or intact pericardial coronary ligation models. Please click here to view a larger version of this figure.
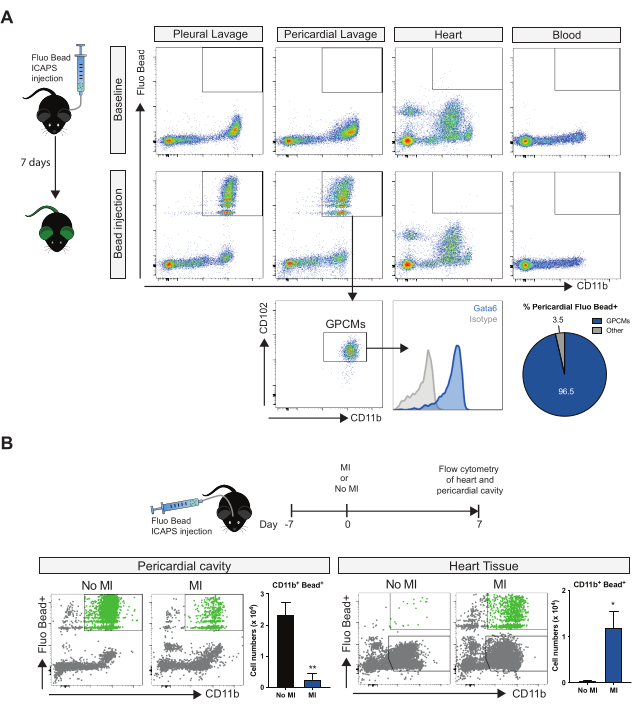
Figure 2: Labeling and tracking of pericardial cavity macrophages following MI. (A) Representative flow cytometry plots of fluorescent bead containing myeloid cells from the pleural cavity, pericardial cavity, cardiac tissue, and blood at baseline or 7 days following local injection of fluorescent beads using the ICAPS method. Bottom panels- Characterization of bead and cells in the pericardial cavity as predominantly Gata6+ pericardial macrophages (GPCMs). (B) Flow cytometry analysis and quantification of fluorescent bead labeled pericardial myeloid cells in pericardial cavity and heart with or without MI. *= p < 0.05, ** = p < 0.01. Adapted from Deniset JF, et al., with permission from Elsevier8. Please click here to view a larger version of this figure.
Discussion
Inducing an MI in a closed pericardium in rodents is unique and can have potentially significant applications. The procedure relies heavily on the surgeon's familiarity with the rodent model and rodent cardiac anatomy. Success is also dependent on the care given during three critical steps: intercostal muscle incision and rib retraction (Steps 1.11-1.13), creating the infarct (Step 1.17 ), and animal recovery (Steps 1.22-1.24).
The thoracotomy must be done diligently to avoid puncturing or lacerating the pericardium. The most crucial step of this protocol is the suturing of the LAD to induce an infarction. As is the case with all LAD ligation models, appropriate placement of the ligation suture on the LAD is critical: proximal ligation may result in a fatal MI, whereas distal ligation may not cause a functionally relevant MI. Landmarking the LAD in the approximate center of the heart avoids these issues. As LAD ligation is performed with the heart beating, gently stabilizing the heart with forceps can help minimize movement, allowing the LAD to be sutured without damaging it. Small vessel laceration on the epicardium can occur during this procedure. Minor bleeds will resolve over 2-3 days and will not contaminate the pericardial fluid. The rodent pericardium, especially in mice, is very thin and can be easily torn if the surgeon does not exercise caution. Lastly, the operator must pay close attention to the animal in the post-procedure (i.e., recovery) phase. The timing of stopping isoflurane and removing the endotracheal tube must be done methodically to ensure the rodent can self-ventilate. The mouse should also be monitored after recovery to ensure no post-operative complications necessitate immediate intervention before being put into animal housing facilities. Examples of these complications include hemothorax, pneumothorax, and the inability to regain consciousness after anesthesia.
Most current mouse models of MI require opening the pericardium to ligate the LAD, resulting in a non-intact pericardium. The present model is unique as it preserves the homeostatic aspect of the pericardial space during infarction, hence providing a more clinically relevant representation of an MI. Maintaining the pericardial space results in improved functional characteristics of the mouse heart compared to procedures that divide the pericardium. Preservation of the native pericardial fluid also provides significant benefits to research possibilities as well as infarct healing. Intra-pericardial pressure is significant11,12, while pericardial fluid contains proteins that promote non-fibrotic healing pathways13. Recent research has revealed that macrophages that reside in the pericardial fluid also play an essential role in cardiac tissue repair and healing8. The current protocol provides a specific labeling method to track the fate of these macrophages following MI. Other cells within the pericardial space can be similarly labeled to assess their role in cardiac remodeling. Animal models that maintain the pericardium may better preserve these crucial pathways, making them a more accurate representation of patients' pathophysiology and the healing processes.
This model allows the user to study and manipulate the entire pericardial space, potentiating research that explores the complex healing and inflammatory pathways mediated by pericardial cells. This model also provides an improved rodent infarct model for research not focused on the pericardial space. The preserved pericardial injury pathways allow for infarcts to have more human relevancy. The significant limitations of this model lay in user skill due to its technical nature. If the surgeon is not proficient in tissue handling and surgical techniques, errors can result in a torn pericardium or mortality. Finally, to leverage the advantages of this protocol, users should be able to use established and advanced imaging modalities, such as echocardiography and microscopy.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
None.
Materials
| Steri-350 Bead Sterilizer | Inotech | NC9449759 | |
| 10% Formalin | Millipore Sigma | HT501128-4L | |
| 40 µm Cell strainer | VWR | CA21008-949 | Falcon, 352340 |
| 70 µm Cell strainer | VWR | CA21008-952 | Falcon, 352350 |
| ACK Lysis Buffer | Thermo Fisher | A1049201 | |
| BD Insyte-W Catheter Needle 24 G X 3/4" | CDMV Inc | 108778 | |
| Betadine (10% povidone-iodine topical solution) | CDMV Inc | 104826 | |
| Blunt Forceps | Fine Science Tools | FST 11000-12 | |
| BNP Ophthalmic Ointment | CDMV Inc | 17909 | |
| Castroviejo Needle Driver | Fine Science Tools | FST 12061-01 | |
| Centrifuge 5810R | Eppendorf | 22625101 | |
| Collagenase I | Millipore Sigma | SCR103 | |
| Collagenase XI | Millipore Sigma | C7657 | |
| Covidien 5-0 Polysorb Suture – CV-11 taper needle | Medtronic Canada | GL-890 | |
| Covidien 5-0 Polysorb Suture – PC-13 cutting needle | Medtronic Canada | SL-1659 | |
| Curved Blunt Forceps | Fine Science Tools | FST 11009-13 | |
| Dako Mounting Medium | Agilen | CS70330-2 | |
| DNase I | Millipore Sigma | 11284932001 | |
| Ethanol, 100% | Millipore Sigma | MFCD00003568 | |
| Ethicon 8-0 Ethilon Suture – BV-130-4 taper needle | Johnson & Johnson Inc. | 2815G | |
| Fiber-Optic Light | Nikon | 2208502 | |
| Fine Forceps | Fine Science Tools | FST 11150-10 | |
| Fluoresbrite® YG Carboxylate Microspheres 1.00 µm | Polysciences, Inc. | 15702 | |
| Geiger Thermal Cautery Unit | World Precision Instruments | 501293 | Model 150-ST |
| Hyaluronidase | Millipore Sigma | H4272 | |
| Isofluorane Vaporizer | Harvard Apparatus | 75-0951 | |
| Isoflurane USP, 250 mL | CDMV Inc | 108737 | |
| Magnetic Fixator Retraction System | Fine Science Tools | 18200-20 | |
| MX550D- 40 MHz probe | Fujifilm- Visual Sonics | ||
| Needle Driver | Fine Science Tools | FST 12002-12 | |
| PE-10 Tubing | Braintree Scienctific, Inc. | PE10 50 FT | |
| Scissors | Fine Science Tools | FST 14184-09 | |
| SMZ-1B Stereo Microscope | Nikon | SMZ1-PS | |
| VentElite Small Animal Ventilator | Harvard Apparatus | 55-7040 | |
| Vetergesic (10 mL, 0.3mg/mL buprenorphine)) | CDMV Inc | 124918 | controlled drug |
| Vevo 2100 Software | Fujifilm-Visual Sonics |
Riferimenti
- Virani, S. S., et al. Heart disease and stroke statistics-2020 update: A report from the American Heart Association. Circulation. 141, 139 (2020).
- Iismaa, S. E., et al. Comparative regenerative mechanisms across different mammalian tissues. NPJ Regenerative Medicine. 3 (6), (2018).
- Bayat, H., et al. Progressive heart failure after myocardial infarction in mice. Basic Research in Cardiology. 97 (3), 206-213 (2002).
- Virag, J. A., Lust, R. M. Coronary artery ligation and intramyocardial injection in a murine model of infarction. Journal of Visualized Experiments. 52, 2581 (2011).
- De Villiers, C., Riley, P. R. Mouse models of myocardial infarction: comparing permanent ligation and ischaemia-reperfusion. Disease Models & Mechanisms. 13 (11), (2020).
- Borlaug, B. A., Reddy, Y. N. V. The role of the pericardium in heart failure: Implications for pathophysiology and treatment. JACC Heart Failure. 7 (7), 574-585 (2019).
- Pfaller, M. R., et al. The importance of the pericardium for cardiac biomechanics: from physiology to computational modeling. Biomechanics and Modeling in Mechanobiology. 18 (2), 503-529 (2019).
- Deniset, J. F., et al. Gata6(+) Pericardial Cavity Macrophages Relocate to the Injured Heart and Prevent Cardiac Fibrosis. Immunity. 51 (1), 131-140 (2019).
- Weber, G. F. Immune targeting of the pleural space by intercostal approach. BMC Pulmonary Medicine. 15, 14 (2015).
- Nakatani, T., Shinohara, H., Fukuo, Y., Morisawa, S., Matsuda, T. Pericardium of rodents: pores connect the pericardial and pleural cavities. The Anatomical Record. 220, 132-137 (1988).
- Tyberg, J. V., et al. The relationship between pericardial pressure and right atrial pressure: an intraoperative study. Circulation. 73, 428-432 (1986).
- Hamilton, D. R., Sas, R., Semlacher, R. A., Kieser Prieur, T. M., Tyberg, J. V. The relationship between left and right pericardial pressures in humans: an intraoperative study. The Canadian Journal of Cardiology. 27, 346-350 (2011).
- Park, D. S. J., et al. Human pericardial proteoglycan 4 (lubricin): Implications for postcardiotomy intrathoracic adhesion formation. The Journal of Thoracic and Cardiovascular Surgery. 156 (4), 1598-1608 (2018).

