Field Collection and Laboratory Routine Identification of Rhodiola crenulata
Summary
Here, we describe the identification of Rhodiola Crenulata from habitat, plant morphology, medicinal properties, microscopic features, and thin-layer chromatography.
Abstract
The identification of medicinal materials is the premise and guarantee of drug safety. The majority of scientific researchers are bound to favor the simple, fast, effective, and inexpensive identification process of herbals. Rhodiola crenulata is a traditional Tibetan medicine grown at high altitudes, mainly distributed in Tibet, Yunnan, and Sichuan regions of China. Rhodiola crenulate possesses multiple bioactivities, such as anti-inflammatory, anti-hypoxia, and antioxidant properties, and has great potential for development. With the increasing market demand and a rapid decrease in resource content, a large number of confused products of Rhodiola crenulata have been troubling people. Therefore, this protocol introduces a standard process for the identification of Rhodiola crenulata in the field combined with routine laboratory testing. The combination of habitat, microscopic features, and thin-layer chromatography will undoubtedly identify Rhodiola crenulata quickly, efficiently, and economically, contributing to the continuous development of Tibetan medicine and the quality control of medicinal materials.
Introduction
Herbal medicine has a long history and rich application experience in China, and it was the first systematic recording in Shennong's herbal classic1. The discovery of artemisinin applied to malaria promoted the development of herbal medicine into a new stage1. The use of modern scientific technology to uncover the exact mechanism of herbal medicine increases the utilization rate and demand for herbal medicine, opening a new international market for it2,3,4. However, this has led to a series of negative effects. Non-professionals have a vague understanding of the characteristics of herbal medicine, which makes the use of herbal medicine face a huge safety risk5.
Rhodiola crenulata, one of the plants of the Rhodiola species, is mainly distributed in Tibet, northwest Yunnan, and western Sichuan of China (Figure 1)6,7. Rhodiola crenulata comprises salidroside, tyrosol, gallic acid, and other compounds for the treatment of hypoxic-related diseases through the function of "invigorating qi and promoting blood circulation, clearing pulse and calming asthma"8,9,10,11. Field investigation shows that Rhodiola crenulata can be found in the alpine talus zones, gully slopes, and rock crevices at an altitude of 4,000-5,600 m. Its growing environment is cold, full of sunshine and intense radiation, and it belongs to the alpine meadow ecosystem. Rhodiola crenulata can be distributed in lamellar and point-like populations according to the growth terrain, and gene flow can be carried out through cross-pollination.
The pollen abortion of the genus Rhodiola, illegality excavation, and degenerated ecological environment make Rhodiola crenulata an endangered species6,12. In view of the high medicinal value of Rhodiola crenulata, counterfeit products are expected to flow into the market. This article presents the habitat of Rhodiola crenulata and some convenient laboratory identification methods. Firstly, we observed the growth environment of Rhodiola crenulata and its medicinal properties. Secondly, the microstructure of medicinal powder was observed by microscope. The last step is the key point. The representative components of Rhodiola crenulata were separated and identified according to the different adsorption or dissolution properties of these components in a certain substance. DNA-based authentication or metabolomics analysis methods of medicinal plants are complicated and expensive13. These basic, convenient, and economical methods can quickly identify Rhodiola crenulata.
Protocol
Rhodiola crenulata is collected from Zhuoda Snow Mountain, Ganzi County, Ganzi Tibetan Autonomous Prefecture, Sichuan Province, China (N 31.44570°, E 99.96086°, 4892 m). The plants are authenticated as genuine by Professor Yi Zhang at the School of Ethnic Medicine, Chengdu University of Traditional Chinese Medicine.
1. Collection of Rhodiola crenulata
- Photograph the habitat map of Rhodiola crenulata.
- Photograph the whole plant, leaves, calyx, and rhizome of Rhodiola crenulata.
- Use a spade to clear the weeds and broken stones within 1 m of the Rhodiola crenulata to ensure the subsequent smooth mining.
- Dig up the soil with a hoe until the whole rhizosphere is seen and collect the taproot.
NOTE:The roots and rhizomes of Rhodiola crenulata used in medicinal parts should be collected in autumn, when the flower stems wither.
2. Characteristic identification
- Observe the appearance traits of Rhodiola crenulata with the unaided eye: cylindrical and short taproots and rhizomes, brown surface, membranous yellow epidermis with a pink pattern, and orange-red or burgundy slices.
- Identify it by smell: It gives a fragrant smell when near the nose.
- Identify it by taste: Take a small piece of root into the mouth, first sip and then chew, taste slightly bitter, then sweet.
3. Microscopic identification of starch granules in medicinal powder
- Remove the soil on the surface of the Rhodiola crenulata with a brush, put it in the oven at 40 °C, and turn the herbs every 24 h.
NOTE: The ease of breaking of medicinal materials is considered the standard for drying moisture. - Powder the dried medicinal materials using a powder machine and filter the medicinal powder using the medicated No. 3 sieve (see Table of Materials).
- Take a clean slide (see Table of Materials), dig the powder with a dissecting needle (see Table of Materials), and place it evenly at one-third of the slide within 2 mm.
- Use a glass dropper (see Table of Materials) to add a drop of deionized water to the powder. Use a tweezer (see Table of Materials) to hold one end of the cover glass (see Table of Materials) to touch the liquid level quickly and cover the powder.
NOTE: Use an anatomical needle to gently mix water and medicinal powder to ensure uniform sample mixing. There should be no air bubbles between the slide, powder, and cover glass. - Open the microscope (see Table of Materials) and place the slide in step 3.4 onto the platform to secure it. Adjust the light source and the coarse focus spiral to see the powder. Adjust the fine parafocal spiral until the tissues are seen clearly. Switch to a 40x objective and observe the starch granules.
NOTE: Starch the grains presenting as single or multiple grains, and the umbilical point appears herringbone or crack-shaped.
4. Microscopic identification of catheters, cork cells, fibers, wood parenchyma cells, and pigment masses in medicinal powder
- Take a clean slide (see Table of Materials), dig the powder with a dissecting needle (see Table of Materials), and place it at one-third of the slide.
- Use a glass dropper (see Table of Materials) to add a drop of chloral hydrate (see Table of Materials) to the powder. Take the slide with a tweezer (see Table of Materials) and heat it in the alcohol lamp three times, each time for 1 s.
NOTE: Bubbles should be avoided during heating. The liquid remains non-flowing, indicating that the penetration is complete. - Use a glass dropper to add a drop of glycerin (see Table of Materials). Use a tweezer to hold one end of the cover glass to touch the liquid level quickly.
- Open the microscope and place the slide onto the platform to secure it. Adjust the light source and the coarse focus spiral to see the powder. Adjust the fine parafocal spiral until the tissues are seen clearly. Observe the catheter, cork cells, fibers, wood parenchyma cells, and pigment block by switching to a 40x objective lens.
NOTE: Polygonal or long polygonal of cork cells, spiral vessel with obvious helical structure, xylem parenchyma containing calcium oxalate sand crystals, and red or brownish-red pigment block.
5. Preparation of thin layer chromatographic (TLC) samples of Rhodiola crenulata and its reference
- Place the weighing paper on the balance (see Table of Materials) and weigh 3 g powder of Rhodiola crenulata.
- Take the powder into a 100 mL conical bottle (see Table of Materials), and add 25 mL of methanol with a big belly pipette (see Table of Materials). Put the conical bottle into the ultrasonic instrument. Set the power to 250 W, frequency to 40 kHz, and time to 30 min (see Table of Materials), and switch on the instrument.
NOTE: The purpose of the ultrasound instrument is to ensure that the powder of Rhodiola crenulata is completely dissolved without affecting the results of subsequent thin-layer chromatographic experiments. - Remove the conical bottle and rinse the outer bottle with running water to room temperature (RT).
- Aspirate 800 µL of liquid prepared in step 5.3 with a 1 mL syringe. Filter with 0.22 µm microporous filter membrane (see Table of Materials) and collect 400 µL of midstream sample solution of Rhodiola crenulata in a chromatographic sample bottle.
- Weigh and add 2 mg of salidroside, tyrosol, and gallic acid (see Table of Materials) into 3 separate 100 mL conical bottles, respectively. Add 25 mL of methanol with a big belly pipette into every conical bottle.
- Put the conical bottle into the ultrasonic instrument, set the power to 250 W, frequency to 40 kHz, and time to 30 min, and repeat step 5.3 (see Table of Materials).
- Aspirate 800 µL of liquid prepared in step 5.6 with a 1 mL syringe. Filter with 0.22 µm microporous filter membrane (see Table of Materials) and collect 400 µL of midstream salidroside, tyrosol, and gallic acid standard solution in corresponding chromatographic sample bottles.
6. TLC identification
- Pipette trichloromethane (5 mL), ethyl acetate (4 mL), methanol (2 mL), and formic acid (0.5 mL) (see Table of Materials). Add to one side of the double-tank chromatography cylinder (see Table of Materials), shake, and mix evenly. Cover the upper cylinder head.
- Place the gallic acid, salidroside, tyrosol standard solution, and Rhodiola crenulata solution on the sample rack in positions A1-A4.
- Place the 5 cm x 10 cm silicone sheet (see Table of Materials) on the sampling table. Start the automatic sampling machine (see Table of Materials), and open the air control valve.
- Open the visionCATS software (see Table of Materials). Click New > New Folder (named Rhodiola crenulata sample test) > OK. Click New Method (name Rhodiola crenulata sample test) > OK > ATS 4.
- Click Finish Step Definition. Click Track Assignment to edit the description (gallic acid, salidroside, tyrosol, and Rhodiola crenulata sample).
- Click HPTLC Steps. Set the thin layer parameters (5 cm x 10 cm). Select Application Type (band), set the parameters (Table 1), and click the OK button.
- Open Filling/Rinsing Quality. Check Fill only Programmed Volume. Set the vial bottom level (mm) to 0.5, and click the OK button.
- Click Execute Method.
- Click the Track Assignment button, check Center, and set the parameters (Table 2).
- Click the Continue button for automatic sampling.
- Turn off the automatic sampling machine and air valve. Remove the silicone sheet from the automatic sampling machine.
- Put the silicone sheet into the other side of the double tank chromatography cylinder in step 6.1, cover the upper cylinder head, and pre-saturate for 20 min.
- Gently clamp the upper end of the thin layer plate with a tweezer, quickly put the silicone sheet into the development agent, and cover the upper cylinder head.
NOTE: Take out the silicone sheet when the unfolding front edge is 0.5-1.0 cm away from the upper end of the thin layer plate. - After the organic solvent evaporates, spray the chromogenic solution onto the surface of the silicone sheet at room temperature to obtain chromogenic results.
NOTE: The chromogenic solution is an aqueous solution containing 2% FeCl3 and 1% K3[Fe(CN)6].
Representative Results
This experimental protocol describes identifying and collecting Rhodiola crenulate in the field. Rhodiola crenulate tends to live in the alpine talus zones, gully slopes, and rock crevices at high altitudes. The habitat, whole plant, flower, and leaves of Rhodiola crenulate can be shown in Figure 2. Rhodiola crenulate has a reddish-brown rhizome (Figure 3A). A representative image of the medicinal powder is shown in Figure 3B. According to the above experimental protocol, the microscopic results can be listed as follows: 1) brownish yellow or colorless cork cells, large appearance, polygonal or long polygonal, with slightly thicker walls (Figure 3C); 2) starch grains presenting as single or multiple grains, and umbilical point appearing herringbone or crack shaped (Figure 3D); 3) mainly spiral vessels that are closely arranged (Figure 3E); 4) achromatous and oval xylem parenchyma presenting in sheets and containing calcium oxalate sand crystals (Figure 3F); 5) irregular shaped, brown-red pigment block (Figure 3G). The thin layer separation results showed that the Rhodiola crenulata sample (A4) appeared as spots of the same color in the position corresponding to the chromatogram of the gallic acid (A1), salidroside (A2), tyrosol standard solution (A3) (Figure 3H). Gallic acid, salidroside, and tyrosol are the prime and representative components of Rhodiola crenulate. These results show that a preliminary identification of Rhodiola crenulata is possible with the tests discussed in the protocol.
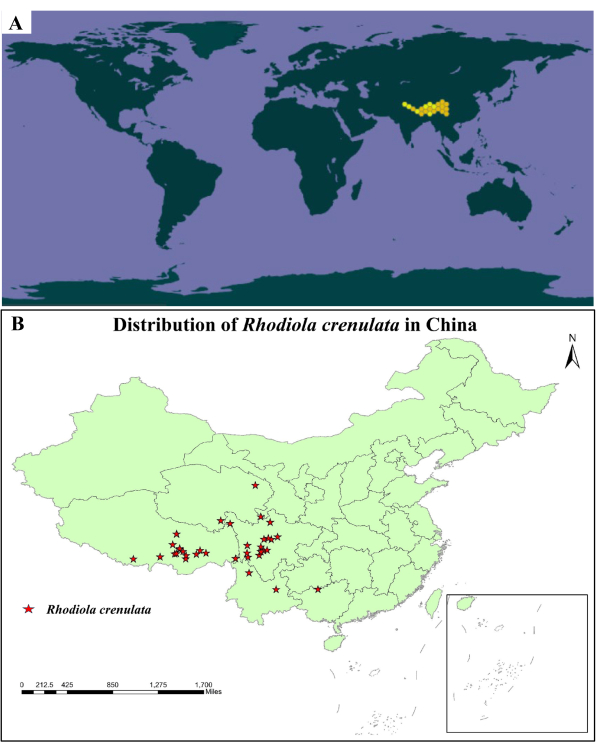
Figure 1: Distribution map of Rhodiola crenulata. (A) Rhodiola crenulata is mainly distributed in China, India, Nepal, and Bhutan14. (B) Rhodiola crenulata is mainly distributed in Tibet, Qinghai, Sichuan, Yunnan, and Guizhou of China (produced by soft ArcGis 10.6). Statistical data comes from the websites of the Institute of Botany15,16. Please click here to view a larger version of this figure.

Figure 2: Picture of Rhodiola crenulata plant. (A) Biotope of Rhodiola crenulate. (B) Close shot of Rhodiola crenulate. (C) Whole plant of Rhodiola crenulate. (D) Flower of Rhodiola crenulata. (E) Leaves of Rhodiola crenulate. Please click here to view a larger version of this figure.

Figure 3: Microscopic characteristics and thin layer chromatogram of Rhodiola crenulata. (A) Medicinal root of Rhodiola crenulata. (B) Medicinal material power of Rhodiola crenulata. (C) Cork cell. (D) Starch grain. (E) Spiral vessel. (F) Xylem parenchyma (calcium oxalate crystal). (G) Pigment. (H) The thin layer chromatography separation of salidroside (A1), gallic acid (A2), tyrosol (A3), and Rhodiola crenulata (A4). Please click here to view a larger version of this figure.
| Parameters | Option |
| Application position Y (mm) | 10 |
| First track position (mm) | 10 |
| Track distance (mm) | 10 |
| Application length (mm) | 5 |
| Application width (mm) | 0.5 |
Table 1: Parameter settings of automatic sampling position.
| Vial ID | Description | Volume (μL) | Position | Type |
| 1 | salidroside | 3 | A1 | Reference |
| 11 | gallic acid | 1 | A2 | Reference |
| 12 | tyrosol | 2 | A3 | Reference |
| 13 | Rhodiola crenulate sample | 2 | A4 | Sample |
Table 2: Parameter settings of automatic sampling order.
Discussion
There are more than 90 species of Rhodiola plants in the world, and more than 60% of all species are found in China, common ones including Rhodiola crenulata, Rhodiola rosea, Rhodiolas achalinensis, and Rhodiola algida17. Rhodiola crenulata, recorded in the first part of the Chinese Pharmacopoeia (2020), is a traditional Tibetan medicine grown at high altitudes. The market demand for Rhodiola crenulata is increasing yearly, so ensuring the correct use of the source is the key to ensuring safe use. Particularly, the standardization from field picking to simple and rapid laboratory routine identification can not be ignored. It has been reported that high-performance liquid chromatography, mass spectrometry, and inter simple sequence repeats can accurately identify Rhodiola crenulate from other Rhodiola species, which is time-consuming, complex, and expensive18,19,20. Meanwhile, we have established a multidimensional evaluation method of sensory recognition (E-nose and color analysis) and HPLC method to distinguish Rhodiola crenulate21,22.
Each medicinal material has its unique growing environment, microscopic structure characteristics, and index components. This protocol provides a comprehensive method for identifying Rhodiola crenulata, from field identification to laboratory microscopy and validation using thin-layer chromatography. Rhodiola crenulata mainly grows at altitudes greater than 3000 m, low temperature, low oxygen, and high ultraviolet radiation areas23. Rhodiola crenulata is a succulent herb, which is its intuitive visual feature. Its powder appears reddish brown with a fragrant smell. Based on its habits, plant morphology, flowers, and leaves, Rhodiola crenulata is distinguished from other Rhodiola species in the field. The microscopic results of medicinal materials showed the existence of starch granules, wood parenchyma cells (including calcium oxalate sand crystals), cork cells, conduit (mainly threaded conduit), and large amounts of red pigment. TLC is a chromatographic separation technique for the separation of multicomponent samples, commonly used in the identification of Chinese medicinal materials. Gallic acid, tyrosol, and salidroside are often identified as the index components of Rhodiola crenulate24. The results of thin-layer chromatography showed that the solution of Rhodiola crenulata showed the same color spots at the corresponding position as the control (gallic acid, tyrosol, and salidroside). It shows that Rhodiola crenulata contains gallic acid, tyrosol, and salidroside. Combined with the knowledge of the growing environment and microscopic results, Rhodiola crenulata can be preliminarily identified.
It is worth noting that the unique growing environment determines that it is almost impossible to have wild Rhodiola crenulata below the altitude of 3000 m. In addition, it is not recommended to harvest the roots and rhizome of Rhodiola crenulata during the flowering period. For laboratory microscopic identification, drying the roots and sifting the powder are prerequisites for the successful preparation of microscopic samples. Ensuring that there is no air bubble between the slide, the medicinal powder, and the cover glass is also key to observing the characteristic composition under the microscope. For thin layer chromatography, pre-saturation of silica gel plate, reasonable development agent, and sample concentration are important factors for successfully separating different components in the test sample. Compared with traditional manual sampling, the automatic sampling process of this protocol undoubtedly increases the accuracy of the results and the repeatability of the experiment. The subjectivity of aroma and taste identification is too strong and may lead to misjudgments. Compared with high-performance liquid chromatography, H-nuclear magnetic resonance, and mass spectrometry, thin-layer chromatography cannot quantitatively analyze the content of compounds in medicinal materials25. Although DNA barcoding has superior accuracy in herbal identification, its high price determines that it is not universal in the identification of medicinal materials26. Furthermore, the field-to-laboratory identification and microscopy combined with the thin-layer chromatography technique provided in this protocol is applicable to almost all medicinal materials. This is a cheap, simple, and fast process for the identification of any medicinal herbal materials.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81973569, 82274207 and 82104533), the Xinglin Scholar Research Promotion Project of Chengdu University of TCM (XKTD2022013), and the Key Research and Development Program of Ningxia (2023BEG02012).
Materials
| 0.22 μm millipore filter | Millipore | SLGP033RB | |
| Automatic sampling machine | CAMAG | ATS 4 | |
| Chloral hydrate | Fuzhou Brunei Technology Co., Ltd | ST1002 | |
| Chromatographic sample bottles | Zhejiang ALWSCI Technology Co., Ltd | C0000008 | |
| Conical flask | Sichuan Shubo Co., Ltd | 1121 | |
| Cover glass | Citotest Labware Manufacturing Co., Ltd | 10211818c | |
| Dissecting needle | Shanghai Bingyu Fluid Technology Co., Ltd | BY-5026 | |
| Electronic balance | SHIMADZU | ATX124 | |
| Ethyl acetate | Chengdu Kelong Chemical Co., Ltd | 2022120901 | |
| Formic acid | Chengdu Kelong Chemical Co., Ltd | 2021110801 | |
| Gallic acid | Chengdu Herbpurify Co., Ltd | M-017 | |
| Glycerol | Sinopharm Chemical Reagent Co., Ltd | 10010618 | |
| High speed crusher | Beijing Zhongxingweiye Instrument Co., Ltd | FW-100 | |
| Methanol | Chengdu Kelong Chemical Co., Ltd | 20230108 | |
| Microscope | Chongqing Oprec Nistrument Co., Ltd | B203 | |
| Microscope slide | Citotest Labware Manufacturing Co., Ltd | 7105P-G | |
| Oven | Shanghai Yuejin Medical Equipment Co., Ltd | DHG-8145 | |
| Pharmacopoeia sieve | Hangzhou Xingrun sieve factory | 572423281330 | |
| Pipette | Changde BKMAM Biotechnology Co., Ltd | 120302008 | |
| Salidroside | Chengdu Herbpurify Co., Ltd | H-040 | |
| Saturate tank | Yancheng Liegu Technology Co., Ltd | 10*20 P-1 | |
| Silica gel plate | Yantai Jiangyou Silica Gel Development Co., Ltd | HSG20211227 | |
| Trichloromethane | Chengdu Kelong Chemical Co., Ltd | 20221013-1 | |
| Tweezer | Changde BKMAM Biotechnology Co., Ltd | 130302027 | |
| Tyrosol | Chengdu Herbpurify Co., Ltd | L-042 | |
| Ultrasound equipment | Ningbo Xinyi Ultrasonic Equipment Co., Ltd | SB-8200DTS | |
| Volumetric pipet | Changde BKMAM Biotechnology Co., Ltd | 120301006 |
References
- Zhang, J., et al. Traditional herbal medicine and nanomedicine: Converging disciplines to improve therapeutic efficacy and human health. Advanced Drug Delivery Reviews. 178, 113964 (2021).
- Yin, R., et al. The positive role and mechanism of herbal medicine in Parkinson’s Disease. Oxidative Medicine and Cellular Longevity. 2021, 9923331 (2021).
- Li, F. S., Weng, J. K. Demystifying traditional herbal medicine with modern approach. Nature Plants. 3, 17109 (2017).
- da Fonseca, L. R., et al. Herbal medicinal products from Passiflora for anxiety: An unexploited potential. The Scientific World Journal. 2020, 6598434 (2020).
- Aziato, L., Antwi, H. O. Facilitators and barriers of herbal medicine use in Accra, Ghana: an inductive exploratory study. BMC Complementary and Alternative Medicine. 16, 142 (2016).
- Tao, H., et al. Rhodiola species: A comprehensive review of traditional use, phytochemistry, pharmacology, toxicity, and clinical study. Medicinal Research Reviews. 39 (5), 1779-1850 (2019).
- Wang, Y., et al. The dietary supplement Rhodiola crenulata extract alleviates dextran sulfate sodium-induced colitis in mice through anti-inflammation, mediating gut barrier integrity and reshaping the gut microbiome. Food & Function. 12 (7), 3142-3158 (2021).
- Xie, N. Rhodiola crenulate alleviates hypobaric hypoxia-induced brain injury via adjusting NF-κB/NLRP3-mediated inflammation. Phytomedicine. 103, 154240 (2022).
- Wang, X., et al. Salidroside, a phenyl ethanol glycoside from Rhodiola crenulata, orchestrates hypoxic mitochondrial dynamics homeostasis by stimulating Sirt1/p53/Drp1 signaling. Journal of Ethnopharmacology. 293, 115278 (2022).
- Ma, D., et al. Chemical characteristics of Rhodiola Crenulata and its mechanism in acute mountain sickness using UHPLC-Q-TOF-MS/MS combined with network pharmacology analysis. Journal of Ethnopharmacology. 294, 115345 (2022).
- Wang, X., et al. Rhodiola crenulata attenuates apoptosis and mitochondrial energy metabolism disorder in rats with hypobaric hypoxia-induced brain injury by regulating the HIF-1α/microRNA 210/ISCU1/2(COX10) signaling pathway. Journal of Ethnopharmacology. 241, 111801 (2019).
- d’Avigdor, E., Wohlmuth, H., Asfaw, Z., Awas, T. The current status of knowledge of herbal medicine and medicinal plants in Fiche, Ethiopia. Journal of Ethnobiology and Ethnomedicine. 10, 38 (2014).
- Sánchez, M., González-Burgos, E., Divakar, P. K., Gómez-Serranillos, M. P. DNA-based authentication and metabolomics analysis of medicinal plants samples by DNA barcoding and ultra-high-performance liquid chromatography/triple quadrupole mass spectrometry (UHPLC-MS). Plants (Basel, Switzerland). 9 (11), 1601 (2020).
- . Rhodiola renulata (Hook.fil & Thomson) H.Ohba Available from: https://www.gbif.org/species/4200529 (2023)
- . Plant + data center Available from: https://www.iplant.cn/ (2023)
- . Chinese virtual herbarium Available from: https://www.cvh.ac.cn/ (2023)
- Cunningham, A. B. There "ain’t no mountain high enough"?: The drivers, diversity and sustainability of China’s Rhodiola trade. Journal of Ethnopharmacology. 252, 112379 (2020).
- Wang, Q., Ruan, X., Jin, Z. H., Yan, Q. C., Tu, S. Identification of Rhodiola species by using RP-HPLC. Journal of Zhejiang University. Science. B. 6 (6), 477-482 (2005).
- Dong, X., Guo, Y., Xiong, C., Sun, L. Evaluation of two major Rhodiola species and the systemic changing characteristics of metabolites of Rhodiola crenulata in different altitudes by chemical methods combined with UPLC-QqQ-MS-based metabolomics. Molecules. 25 (18), 4062 (2020).
- Zhao, W., et al. HPLC fingerprint differentiation between natural and ex situ populations of Rhodiola sachalinensis from Changbai Mountain, China. PloS One. 9 (11), 112869 (2014).
- Yu, Y. Y. Quality evaluation of Tibetan medicine Rhodiolae crenulatae radix et rhizome based on sensory recognition and near infrared spectrum analysis. Chengdu University of Traditional Chinese Medicine. , (2020).
- Lv, X. M. Simultaneous determination of 5 chemical components in 3 kinds of Rhodiola rosea by HPLC. China Pharmacy. 29 (18), 2515-2519 (2018).
- Dong, T., Sha, Y., Liu, H., Sun, L. Altitudinal variation of metabolites, mineral elements and antioxidant activities of Rhodiola crenulata (Hook.f. & Thomson) H.Ohba. Molecules. 26 (23), 7383 (2021).
- Ma, D., et al. Application of UHPLC fingerprints combined with chemical pattern recognition analysis in the differentiation of six Rhodiola species. Molecules. 26 (22), 6855 (2021).
- Li, X. H., et al. Metabolic discrimination of different Rhodiola species using 1H-NMR and GEP combinational chemometrics. Chemical & Pharmaceutical Bulletin. 67 (2), 81-87 (2019).
- Liu, Y., et al. Identification of Hippophae species (Shaji) through DNA barcodes. Chinese Medicine. 10, 28 (2015).

